Decarboxylase test is a biochemical test used in diagnostic microbiology to differentiate and identify bacteria. It is mainly used for gram negative rods like Enterobacteriaceae. This test is used to detect the ability of organism to decarboxylate amino acids.
In this test organism is inoculated in nutrient broth containing glucose, pH indicator (bromcresol purple) and a specific amino acid like lysine or ornithine or arginine. The broth is covered with mineral oil to create anaerobic condition. Initially glucose is fermented and acid is produced, so the medium becomes yellow.
If the organism has decarboxylase enzyme, it acts on the amino acid and forms alkaline amines. These amines increase the pH and the colour changes from yellow back to purple, so it indicates positive test. If enzyme is absent, medium remains yellow and it indicates negative test.
Principle of Decarboxylase Test
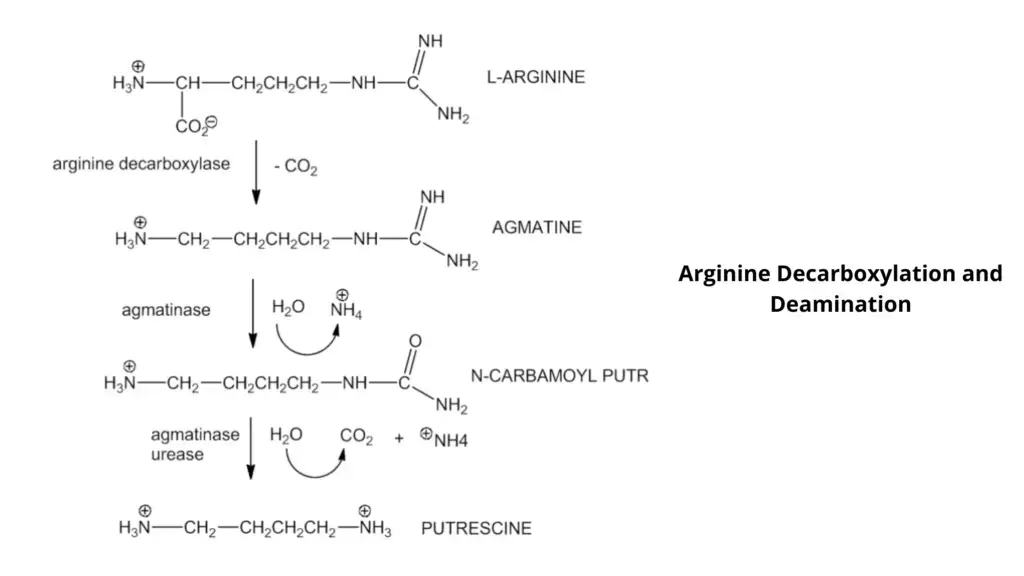
Principle of Decarboxylase Test is based on the ability of certain bacteria to remove the carboxyl group (COOH) from a specific amino acid (lysine, ornithine or arginine) and form alkaline amines. The medium contains the amino acid, small amount of glucose, pyridoxal (cofactor) and pH indicators like bromcresol purple or cresol red. After inoculation, sterile mineral oil layer is added to make anaerobic condition.
In the first phase, the organism ferments glucose and acidic products are produced. Due to acid formation, pH falls and the indicator changes the broth colour from purple to yellow. This acidic condition is required as a trigger for induction of decarboxylase enzyme. If the organism has the specific decarboxylase, the amino acid is decarboxylated and alkaline amines (cadaverine, putrescine) are formed.
Due to accumulation of these alkaline end products, pH rises again and the medium changes from yellow to purple (positive test). If the organism do not produce the decarboxylase enzyme, no amine is formed and the medium remains yellow (negative test).

Objective of Decarboxylase Test
- To determine whether a microorganism produce specific decarboxylase (or dihydrolase) enzyme and can metabolize amino acids like arginine, lysine or ornithine.
- To differentiate and identify closely related bacterial species of Enterobacteriaceae based on their amino acid metabolism.
- To distinguish decarboxylase producing Enterobacteriaceae from other Gram negative rods.
- To help in identification of some non fermentative Gram negative bacilli such as Pseudomonas spp.
Requirements for Decarboxylase Test
Media and Chemical Reagents
- Decarboxylase broth base (Moeller’s medium). It contains peptone, beef extract, glucose, pyridoxal (cofactor) and pH indicators (bromcresol purple, cresol red) to detect colour change.
- Amino acids. The specific amino acid is added in basal medium such as L-arginine, L-lysine or L-ornithine.
- Sterile mineral oil. It is used to overlay the inoculated broth (about 4-5 mm) to create anaerobic condition and to avoid false alkaline reaction on surface.
Microbiological Supplies
- Test organism. A pure fresh culture (18-24 hours) grown on solid medium like TSA or 5% sheep blood agar.
- Control medium. A tube of basal medium without amino acid is kept as control to compare colour reaction and to validate result.
Laboratory Equipment
- Test tubes. Sterile screw-cap tubes are used for holding the broth and cap can be tightened during incubation.
- Inoculation tools. Sterile inoculating loop/needle or plastic bulb pipette is used for aseptic inoculation.
- Incubator. It is maintained at 35°C to 37°C for incubation in ambient air.
Procedure of Decarboxylase Test
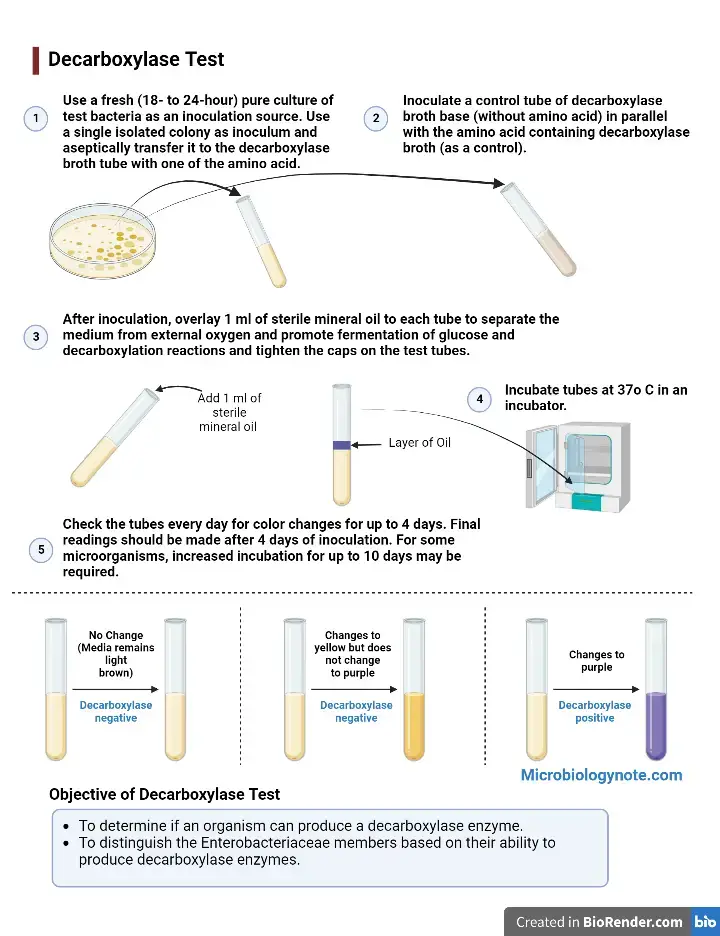
- Tubes of decarboxylase basal medium (control) and decarboxylase test broths containing arginine/lysine/ornithine are taken.
- A fresh pure bacterial culture (18 to 24 hours old) is selected. Culture from solid agar plate is preferred.
- The broths are inoculated. For glucose fermenting organism, a light inoculum is used (1-2 colonies with sterile loop/needle or 1 drop broth culture). For glucose non fermenting organism, heavy suspension is prepared (McFarland No. 5) and about 4 drops are added to each tube.
- Basal medium control tube (without amino acid) is always inoculated along with test tubes for comparison.
- Sterile mineral oil is added over the inoculated medium (4-5 mm layer or 1-2 mL) in each tube including control. This is done to create anaerobic condition and prevent false alkalinization.
- Caps of the tubes are tightened (snugly) to restrict entry of oxygen.
- Tubes are incubated in ambient air at 35°C to 37°C.
- Tubes are examined daily for colour change. Reading is usually done at 24, 48, 72 and 96 hours.
- Incubation can be extended up to 10 days for slow reacting organisms or non fermenters, as delayed reaction may occur.
- Final result is interpreted by comparing the colour of amino acid test tube directly with the inoculated control tube.
Result Interpretation of Decarboxylase Test
- Positive result. The medium first turns yellow and then changes back to turbid purple or pale yellow-purple colour. It indicates glucose is fermented first to produce acid and later amino acid is decarboxylated to alkaline amines. Any trace of purple or violet colour in a tube which was previously yellow is taken as positive.
- Negative result (glucose fermenting organism). The medium turns bright clear yellow and remains yellow throughout incubation. It shows glucose fermentation occurred but specific decarboxylase enzyme was absent, so no alkaline amine is formed.
- Negative result (non glucose fermenting organism). The medium shows little or no colour change and remains original light brown or amber colour. It indicates no glucose fermentation and no alkaline amine formation from amino acid.
- Invalid result. If the control tube (basal medium without amino acid) turns purple or alkaline, the test is invalid and result should not be interpreted.
| Media color | Bacterial reaction |
| No Change (Media remains light brown) | Decarboxylase negative (-) |
| Changes to yellow but does not change to purple | Decarboxylase negative (-) |
| Changes to purple | Decarboxylase positive (+) |




List of organisms showing positive and negative result in Decarboxylase Test
- Klebsiella pneumoniae: Lysine (+), Ornithine (-), Arginine (-)
- Enterobacter aerogenes: Lysine (+), Ornithine (+), Arginine (-)
- Enterobacter cloacae: Lysine (-), Ornithine (+), Arginine (+)
- Proteus mirabilis: Lysine (-), Ornithine (+), Arginine (-)
- Proteus vulgaris: Lysine (-), Ornithine (-), Arginine (-)
- Serratia marcescens: Lysine (+), Ornithine (+), Arginine (-)
- Salmonella Typhi: Lysine (+), Ornithine (-), Arginine (delayed + or -)
- Salmonella Typhimurium: Lysine (+), Ornithine (+), Arginine (+)
- Shigella dysenteriae: Lysine (-), Ornithine (-), Arginine (-)
- Shigella flexneri: Lysine (-), Ornithine (-), Arginine (-)
- Shigella sonnei: Lysine (-), Ornithine (+), Arginine (variable)
- Pseudomonas aeruginosa: Lysine (-), Ornithine (-), Arginine (+)
- Escherichia coli: Lysine (+), Ornithine (variable), Arginine (variable)
- Citrobacter freundii: Lysine (-), Ornithine (variable), Arginine (variable)
Precautions of Decarboxylase Test
- Sterile mineral oil overlay should be applied properly (4-5 mm) in all inoculated tubes and control tube. Caps should be tightened. If anaerobic condition is not maintained, oxygen enters and false alkaline reaction can occur due to surface oxidation and deamination of peptones.
- Inoculum should be taken from a pure fresh culture (18-24 hours) grown on solid agar plate. Broth culture is not preferred as it may carry acid or alkaline products and disturb the pH balance of Moeller medium.
- Control tube (basal medium without amino acid) must be inoculated along with test tubes. If control tube turns purple or alkaline, the whole test becomes invalid.
- Result should not be read early. Reading before 18-24 hours can give false negative as the initial yellow (acid) phase should occur first for enzyme induction.
- If two layers of colour is seen (yellow and purple layer), the tube should be shaken gently to mix before interpretation. Standing vertically may form separate layers.
- Mineral oil should not be autoclaved. Autoclaving may not sterilize properly and sometimes it can cause steam/oil explosion. Dry oven sterilization or filtration is used for sterilizing oil.
- Standard biohazard precautions should be followed. The medium contains animal components and live bacteria so it is treated as infectious. Gloves should be used and contact with skin or mucous membrane should be avoided. Pipettes should not be contaminated during overlay process.
Uses of Decarboxylase Test
- It is used to differentiate closely related members of Enterobacteriaceae and other Gram negative rods that show similar biochemical reactions.
- It is used to separate Klebsiella-Enterobacter-Serratia group. Ornithine decarboxylase test helps in differentiating non motile ornithine negative Klebsiella spp. from motile ornithine positive Enterobacter spp.
- It is used for identification of Salmonella and Shigella. Lysine decarboxylase test is used to differentiate Salmonella (usually lysine +) from Shigella (lysine -).
- It is used for speciation of Proteus. Ornithine test differentiates Proteus mirabilis (ornithine +) from Proteus vulgaris (ornithine -).
- It is used to identify some non fermenting Gram negative bacteria. Arginine dihydrolase test is useful for identification of Pseudomonas aeruginosa.
- It is used for Enterococcus species identification. Arginine decarboxylase test differentiates Enterococcus faecalis and Enterococcus faecium (arginine +) from Enterococcus avium (arginine -).
Advantages of Decarboxylase Test
- It is useful for effective differentiation and identification of closely related bacterial species of Enterobacteriaceae. It helps to separate Salmonella from Shigella and also Klebsiella-Enterobacter-Serratia group.
- It helps in identification of non fermenting Gram negative bacilli. Arginine dihydrolase test is used for Pseudomonas spp. identification.
- It is considered as a reliable reference method. Standard Moeller formulation is taken as clinical gold standard and interpretation is standardized.
- It gives clear visual interpretation. A biphasic colour change is seen (yellow to purple) giving a reliable positive reaction.
- It can be modified for rapid diagnosis. Some modified methods can reduce incubation time to 2-4 hours and still gives reliable result.
- It is a sensitive and specific test for differentiating a wide range of bacteria in routine laboratory.
Limitations of Decarboxylase Test
- It does not measure the quantity of enzyme. It is only a threshold test and shows positive only when alkaline amines are produced enough to raise pH and change colour.
- It is sensitive to growth condition. Decarboxylase activity can be suppressed by change in glucose concentration, amino acid concentration or presence of other amino acids in medium.
- Non fermenting bacteria gives problem in this test. As they do not ferment glucose to produce initial acid, the reaction may be weak or delayed and broth becomes less reliable.
- False positive can occur if mineral oil seal is not maintained. In presence of oxygen, peptones may be oxidatively deaminated and ammonia is released which raise pH giving false alkaline colour.
- Premature reading gives false negative. If result is read before 18-24 hours, yellow phase may not be completed and enzyme induction does not occur properly.
- Delayed reaction is seen in some slow reacting or fastidious organisms. Some strains may require 6 to 10 days incubation to show positive reaction.
- Colour change may be ambiguous. Sometimes yellowish-purple or grey colour is produced if indicator is reduced rather than true alkaline amine formation. Prolong incubation may destroy bromcresol purple and discolouration can occur, common in swarming Proteus.
- Stratified colour layers may be formed during incubation (yellow and purple). Tube should be shaken gently before final interpretation otherwise wrong reading can be done.
- It gives only presumptive identification. For final identification, other biochemical tests or morphological or molecular tests are also required.
References
- A comparative study between rapid and conventional Moeller… (n.d.). SciSpace. https://scispace.com/pdf/a-comparative-study-between-rapid-and-conventional-moeller-2h4q828tda.pdf
- Biolab Diagnostics Laboratory Inc. (2016, September 20). Culture media for amino acid decomposition studies. http://www.biolab.rs/wp-content/uploads/2018/01/Culture-Media-for-Amino-Acid-Decomposition-Studies.pdf
- Biolife Italiana S.r.l. (2025, May). Decarboxylase Moeller base broth. https://gest.joyadv.it/public/cartellina-allegati-schede-certificazioni/schede-tecniche-inglese/ts-4013662.pdf
- Comprehensive analysis of the decarboxylase test in diagnostic microbiology: Principles, methodologies, and clinical applications. (n.d.).
- Cooper, C. R., Jr. (2018). BIOL 3702 lab exercise – Decarboxylation test. Youngstown State University. http://crcooper01.people.ysu.edu/microlab/decarboxylation-test.pdf
- Crystal structures and biochemical analyses of the bacterial arginine dihydrolase ArgZ suggests a “bond rotation” catalytic mechanism. (n.d.). PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC7029115/
- Difco. (n.d.). Lysine decarboxylase broth. https://cdn.media.interlabdist.com.br/uploads/2021/01/Decarboxylase-Medium-Base.pdf
- Ewing, W. H. (1962). ID#: 27659 – Details. Public Health Image Library (PHIL), Centers for Disease Control and Prevention. https://wwwn.cdc.gov/phil/Details.aspx?pid=27659
- Filo. (2023, October 8). Why is the ornithine decarboxylation reaction indicated by a purple color and a negative is yellow?. https://askfilo.com/user-question-answers-chemistry/why-is-the-ornithine-decarboxylation-reaction-indicated-by-a-35363631383033
- Hardy Diagnostics. (2020). Moellers decarboxylase media. https://hardydiagnostics.com/media/assets/product/documents/MoellersDecarboxMed.pdf
- HiMedia Laboratories Pvt. Ltd. (2015, February). Decarboxylase test medium base (Falkow). https://exodocientifica.com.br/_technical-data/M912.pdf
- HiMedia Laboratories Pvt. Ltd. (2026, June). Decarboxylase broth base, Moeller (Moeller decarboxylase broth base). https://www.himedialabs.com/media/TD/M393.pdf
- HiMedia Laboratories Pvt. Ltd. (2026, May). Moeller decarboxylase broth with ornithine HCl. https://www.himedialabs.com/media/TD/M688.pdf
- HiMedia Laboratories. (n.d.). Decarboxylase broth base, Moeller. https://www.himedialabs.com/us/lq156-decarboxylase-broth-base-moeller.html
- Lal, A., & Cheeptham, N. (2015, September 2). Decarboxylase broth protocol. American Society for Microbiology. https://asm.org/asm/media/protocol-images/decarboxylase-broth-protocol.pdf?ext=.pdf
- Merck KGaA / Sigma-Aldrich. (2018). D2935 decarboxylase broth base, Moeller. https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/product/documents/299/486/d2935dat.pdf
- Oxford Lab Fine Chem LLP. (n.d.). Moeller decarboxylase broth base. https://www.oxfordlabfinechem.com/msds/OLFC-1170__Moeller_Decarboxylase_Broth_Base__Decarboxylase_Broth_Base__Moeller_-_TD_69070fdf68461.pdf
- Overview of changes in the Clinical and Laboratory Standards Institute performance standards for antimicrobial susceptibility testing: M100 32nd and 33rd editions. (n.d.). PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC12421849/
- Rapid identification of swarming Proteus using the PathoTec ornithine decarboxylase test strip. (n.d.). PubMed. https://pubmed.ncbi.nlm.nih.gov/1100667/
- Rapid ornithine decarboxylase test for the identification of Enterobacteriaceae. (n.d.). PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC380423/
- Regulation of the arginine dihydrolase pathway in Clostridium sporogenes. (n.d.). PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC235482/
- Remel. (2014, June 30). Decarboxylase broth (Moeller) base control, arginine, lysine, and ornithine. https://tools.thermofisher.com/content/sfs/manuals/IFU60750.pdf
- Sapkota, A. (2025, June 21). Decarboxylase test: Principle, procedure, results, uses. Microbe Notes. https://microbenotes.com/decarboxylase-test-principle-procedure-and-result-interpretation/
- TM Media. (2019, November 8). TM 085 – Decarboxylase broth base, Moeller. https://www.tmmedia.in/wp-content/uploads/TD/TD-TM-085.pdf
- TM Media. (2019, November 8). TM 1140 – Arginine dihydrolase broth. https://www.tmmedia.in/wp-content/uploads/TD/TD-TM-1140.pdf
- Virtual Microbiology Lab Simulator Software – VUMIE. (2022, November 1). Lysine decarboxylase test. https://vumicro.com/docs/lysine-decarboxylase-test/