Hopkins-Cole test is a biochemical test used to detect tryptophan amino acid in proteins. It is also known as glyoxylic acid reaction. This test was discovered in 1901 by Hopkins and Cole.
It is based on the reaction of glyoxylic acid with the indole ring of tryptophan. In this test the protein solution is mixed with Hopkins-Cole reagent (contains glyoxylic acid). Concentrated sulphuric acid is then added carefully along the side of the test tube to form a separate layer.
If tryptophan is present, a purple or violet red ring is formed at the junction of two layers. If the protein does not contain tryptophan (gelatin), no purple ring is seen and it is negative test.
Objectives of Hopkin’s Cole Test (Adamkiewicz–Hopkins)
- To detect the presence of indole ring in protein sample.
- To identify tryptophan or proteins containing tryptophan.
Principle of Hopkin’s Cole Test (Adamkiewicz–Hopkins)
Principle of Hopkin’s Cole Test (Adamkiewicz–Hopkins) is based on the reaction of glyoxylic acid with the indole ring of tryptophan in strong acidic condition. The protein sample is mixed with Hopkins-Cole reagent (glyoxylic acid) and concentrated sulphuric acid is layered below it carefully.
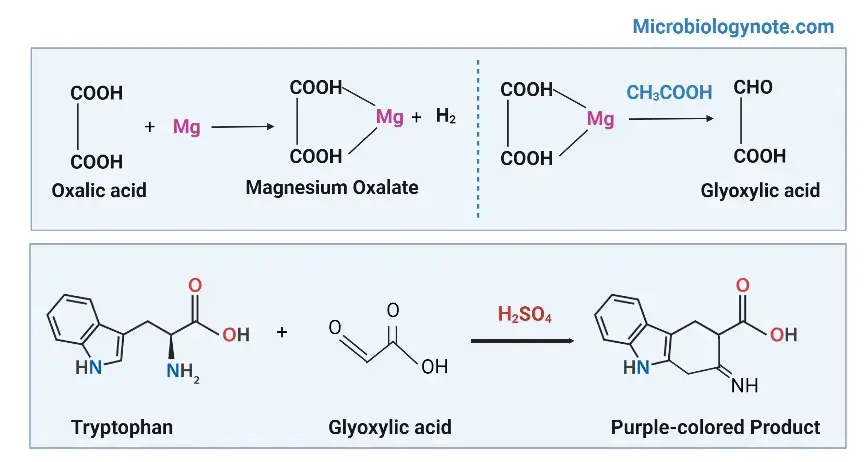
The concentrated sulphuric acid acts as oxidizing and dehydrating agent. In this condition the aldehyde group of glyoxylic acid condenses with the indole ring of tryptophan and an intermediate complex is formed. This complex undergoes dehydration and a coloured product is produced.
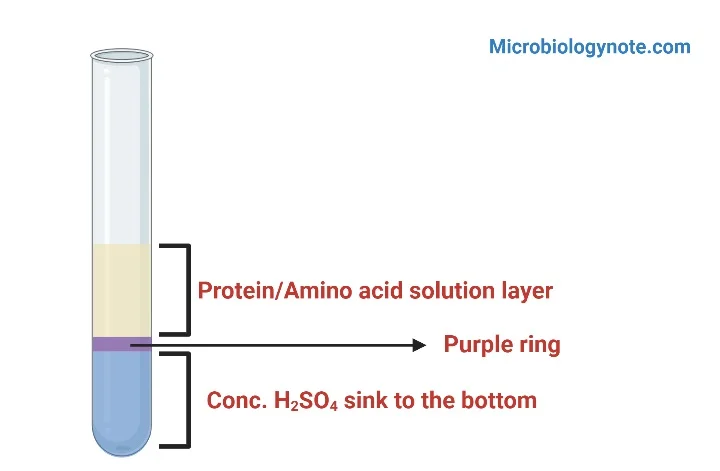
A distinct violet or purple coloured ring is formed at the junction of the two layers. This indicates the presence of tryptophan in the protein sample.
Reaction
Requirements for Hopkin’s Cole Test
- Hopkins-Cole reagent (glyoxylic acid solution). It is prepared by magnesium reduction method or by keeping glacial acetic acid in sunlight for few days.
- Concentrated sulphuric acid (H2SO4) (95 to 98%). It should be analytical grade and free from nitrous contamination.
- Test sample. 0.1% to 1% solution of amino acid or protein sample is used.
- Clean dry test tubes (borosilicate glass). It is preferred because it can tolerate the heat produced during addition of concentrated sulphuric acid.
- Pipette or dropper. It is used for transferring reagents and for layering the sulphuric acid along the side of the tube.
- Test tube stand. It is used to keep the tube in vertical position for ring formation.
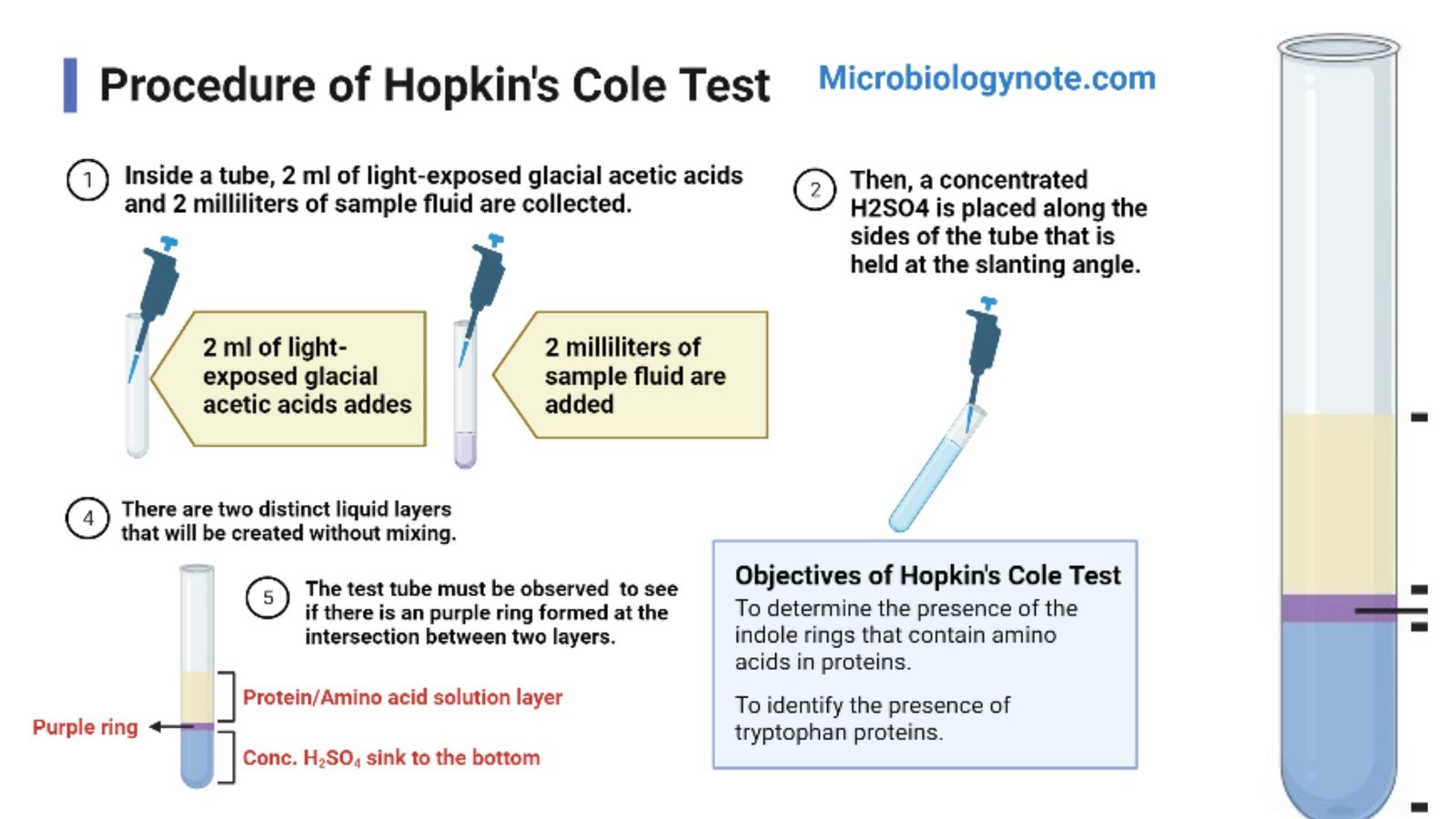
Procedure of Hopkin’s Cole Test
- Take a clean and dry test tube. Add 1 to 2 ml of the protein sample solution in the tube.
- Add equal volume (1 to 2 ml) of Hopkins-Cole reagent (glyoxylic acid) to the sample. Mix gently.
- Tilt the test tube at about 45° to 60° angle.
- Take concentrated sulphuric acid in a pipette. Add 1 to 2 ml concentrated sulphuric acid slowly along the side of the slant tube. Mouth of the tube should be away from face.
- Allow the sulphuric acid to settle at the bottom and form a separate layer. Do not shake and do not mix the layers.
- Keep the tube in vertical position in a stand. Leave it undisturbed for few minutes. Observe the junction of two layers.
- Violet or purple ring at the interface indicates positive Hopkin’s Cole test for tryptophan.
Result and Interpretation of Hopkin’s Cole Test
Positive result
A distinct violet or purple coloured ring is formed at the junction of two layers. It indicates presence of tryptophan amino acid or protein containing tryptophan. In microbiology, positive result can also indicate indole producing bacteria (E. coli, Proteus mirabilis) which breaks down tryptophan.
Negative result
No purple ring is formed at the interface. It indicates tryptophan is absent in the sample or present in very low amount not detected by the test.
Trace result
A faint violet ring or slight pink/red tint is seen at the junction. It indicates trace amount or low concentration of tryptophan. Sometimes it can be due to secondary decomposition of sample.
Interfered result
Yellow or brown ring is seen instead of purple ring. It indicates interference in the test. It can be due to strong oxidizing contaminants or other interfering substances like tyrosine.
False result
False positive may occur when interfering substances mimic the reaction. False negative may occur with old sample where tryptophan is degraded or wrong reagent concentration is used. Nitrites, nitrates or excess chlorides can inhibit the reaction and give false negative.
Precautions of Hopkin’s Cole Test
- Wear proper protective equipment. Use acid resistant gloves and safety goggles, because concentrated sulphuric acid is highly corrosive and can cause severe irritation.
- Use clean and dry borosilicate test tubes. It is required because heat is produced during addition of concentrated sulphuric acid and normal glass can crack.
- Mouth of the test tube should be away from face and others. While adding sulphuric acid this precaution is followed.
- Add the concentrated sulphuric acid carefully. Tilt the tube (45° to 60°) and add acid slowly along the side of the tube so it forms a separate layer at bottom.
- Do not shake or mix the contents after adding acid. Two distinct layers should be maintained, mixing will produce excess heat and colour development can be destroyed.
- If acid spill occurs, neutralize immediately with weak alkali like sodium bicarbonate and wash the area with plenty of water.
Uses of Hopkin’s Cole Test
- It is used to detect tryptophan in proteins. It is a qualitative test for indole ring of tryptophan in the protein sample.
- It is used in food science for protein quality checking. It helps in assessing nutritional completeness of proteins. It is used to differentiate tryptophan rich proteins from tryptophan lacking proteins (whey and gelatin). It is also used to check freshness of meat and protein changes during processing.
- It is used in microbiology for bacterial identification. It is used to differentiate indole producing bacteria like E. coli and Proteus mirabilis by checking breakdown of tryptophan into indole.
- It is used in clinical diagnosis. It can be used for screening biological fluids like urine and plasma. It helps in diagnosis of disorders related to tryptophan absorption (Hartnup disease). It also helps in UTI cases by indole producing bacteria.
- It is used in biochemical research. It is used to study amino acid metabolism, protein degradation and bacterial processes. It supports in development studies of new antibiotics.
- It is used in environmental monitoring. Its principle is used to detect pollutants having indole derivatives, mainly in waste water treatment.
Advantages of Hopkin’s Cole Test
- It is highly specific for tryptophan. It detects the indole group and gives specific reaction. False positive with other aromatic amino acids like tyrosine and phenylalanine is not seen like in xanthoproteic test.
- It is simple and quick test. It is easy to perform in test tube and result is obtained quickly. Distinct violet or purple ring is formed so it is easy to observe.
- It is cost effective. Reagents are cheap and easily available. It can be used in basic lab and resource limited settings, without using costly methods like chromatography.
- It has teaching value. It shows chemical specificity and amino acid side chain reaction clearly, so it is used in practical classes and biochemistry teaching labs.
- It has wide application. Principle is used in microbiology (indole test) and in food science for protein quality and freshness checking.
Limitations of Hopkin’s Cole Test
- It is mainly a qualitative test. It gives only yes or no result for tryptophan. Colour intensity gives only rough idea and it is not accurate like HPLC.
- Inhibitory compounds can affect the test. Nitrites, nitrates, chlorates and excess chlorides can inhibit the condensation reaction and violet ring may not form.
- Impure sulphuric acid can interfere. If sulphuric acid is contaminated with nitrous acid, wrong colour can be produced (ivy green) instead of violet ring.
- False positive may occur. Strong oxidizing agents or other interfering substances can mimic the reaction and give false positive.
- False negative can occur due to degradation. Tryptophan is acid labile. If reaction time is too long or heat is produced due to fast addition of sulphuric acid, tryptophan may degrade and ring will not form.
- Hydrolysis requirement is there. Tryptophan residues are present inside protein, so hydrolysis by sulphuric acid is needed to free tryptophan. Sometimes this step becomes problematic.
- Result reading is subjective. It depends on visual observation of colour at the interface, so interpretation can vary and it is not precise.
References
- Abdulwahed, M., Mamoly, L., & Bosnali, W. (2020). A simple spectrophotometric method for determination of glyoxylic acid in its synthesis mixture. International Journal of Analytical Chemistry, 2020, Article 5417549. https://doi.org/10.1155/2020/5417549
- Al-Garawi, Z. S., & Al-Mossawi, A. H. (2018). Qualitative tests of amino acids and proteins and enzyme kinetics. Mustansiriyah University.
- Amrita Vishwa Vidyapeetham. (2011). Qualitative analysis of amino acid. Biochemistry Virtual Lab I.
- Chainhit222 et al. (2010). Glyoxylic acid synthesis [Online forum post]. Sciencemadness Discussion Board.
- Color tests for proteins and amino acids. (n.d.).
- Comprehensive analysis of the Hopkins-Cole reaction: Analytical principles, chemical mechanisms, and applications in protein biochemistry. (n.d.).
- Core Zone. (2026, January 6). Hopkins Cole test: Understanding a positive result. Core Zone.
- delos Santos, C. J. (n.d.). Hopkins Cole test for tryptophan detection. Scribd.
- Free Minds. (2026, January 6). Hopkins Cole test: Understanding a positive result. Free Minds.
- Free Minds. (2026, January 6). Hopkins Cole test: Understanding positive results. Free Minds.
- Haravata, A. (n.d.). Hopkins-Cole test in biochemistry lab. Scribd.
- Hosokawa, S., Morinishi, T., Ohara, K., Yamaguchi, K., Tada, S., & Tokuhara, Y. (2023). A spectrophotometric method for the determination of tryptophan following oxidation by the addition of sodium hypochlorite pentahydrate. PLoS ONE, 18(1), e0279547. https://doi.org/10.1371/journal.pone.0279547
- King Saud University. (n.d.). Color tests for proteins and amino acids (Test for specific amino acids).
- kulangkatun. (n.d.). Protein identification tests overview. Scribd.
- Kumar, P. (n.d.). Qualitative and quantitative tests for amino acids and proteins. Biology Discussion.
- Michelet, D. (1975). Preparation of glyoxylic acid (U.S. Patent No. 3,929,613). U.S. Patent and Trademark Office.
- monters, h. (n.d.). Urinalysis reagent strip principles. Scribd.
- Rathod, Z. R., Shah, D., Shukla, P., Meghani, S., Patel, C., & Saraf, M. S. (2022). Scientific tools and techniques for qualitative and quantitative analysis of bacterial proteins. Acta Scientific Microbiology, 5(5), 152-160.
- Sapkota, A. (2022, September 5). Hopkin’s Cole test- Definition, principle, procedure, result, uses. Microbe Notes.
- Subroto, E., Lembong, E., Filianty, F., Indiarto, R., Primalia, G., Putri, M. S. K. Z., Theodora, H. C., & Junar, S. (2020). The analysis techniques of amino acid and protein in food and agricultural products. International Journal of Scientific & Technology Research.
- Takarkhede, R. C., Tembhurne, P., & Agarwal, M. K. (n.d.). Laboratory manual: Practical veterinary biochemistry. Mahatma Jyotiba Fule College of Veterinary & Animal Science.
- Wikipedia contributors. (2024, March 20). Hopkins–Cole reaction. In Wikipedia, The Free Encyclopedia. https://en.wikipedia.org/w/index.php?title=Hopkins%E2%80%93Cole_reaction&oldid=1214752256
- Wikipedia contributors. (2025, December 8). Adamkiewicz reaction. In Wikipedia, The Free Encyclopedia. https://en.wikipedia.org/w/index.php?title=Adamkiewicz_reaction&oldid=1326432042
- Wikipedia contributors. (2025, December 14). Glyoxylic acid. In Wikipedia, The Free Encyclopedia. https://en.wikipedia.org/w/index.php?title=Glyoxylic_acid&oldid=1327534520