X and V factor test is a phenotypic diagnostic test used in clinical microbiology to identify and differentiate fastidious gram negative bacteria mainly Haemophilus species. These organisms require specific growth factors present in blood for their growth.
X factor is a heat stable hemin (iron containing porphyrin). V factor is a heat labile coenzyme nicotinamide adenine dinucleotide (NAD) or NADP. The test is done by inoculating the organism on a basal agar medium which lacks both factors and then placing paper disks containing X factor, V factor and XV factor on the agar surface.
After incubation, growth is observed around the disks. The organism will grow only near the disk which supplies the required factor. Growth pattern around X, V or XV disk is used to determine the nutritional requirement and helps in identification of the specific Haemophilus species.
Objectives of X and V factor Test
- To determine the growth requirement of the organism for X factor (hemin). It is checked whether X factor is required or not.
- To determine the growth requirement of the organism for V factor (NAD). It is checked whether V factor is required or not.
- To determine whether both X and V factors are required for growth. Growth around XV disk is observed for this.
- To differentiate different Haemophilus isolates and identify them up to species level based on their growth factor dependence.
- To use as a primary phenotypic test for presumptive identification of Haemophilus species.
- To differentiate Haemophilus organisms from other gram negative bacilli having similar morphology.
Principle of X and V factor Test
Principle of X and V factor Test is based on the nutritional requirement of Haemophilus species for specific growth factors. It is used to differentiate the organisms based on their requirement of X factor, V factor or both.
In this test paper disks containing X factor (hemin), V factor (NAD) and XV factor are placed on a basal agar medium which lacks these factors. The factors diffuse from the disk into the surrounding agar. A lawn culture of the test organism is inoculated on this factor free agar.
After incubation, growth occurs only around the disk which provides the required factor to the organism. Presence or absence of growth around X, V and XV disks indicates whether the organism depends on X factor, V factor, both factors or neither.
Requirements for X and V factor Test
- Factor free basal agar medium. Tryptic Soy Agar (TSA) or Mueller Hinton Agar (MHA) or Brain Heart Infusion (BHI) agar or nutrient agar is used.
- X factor disk (hemin) and V factor disk (NAD). XV factor disk containing both factors is also required.
- Sterile factor free suspension fluid. Normal saline (0.85%) or sterile distilled water or tryptic soy broth (TSB) is used to emulsify the colony.
- Test organism. Pure well isolated young culture (18 to 24 hour old) suspected Haemophilus isolate is used.
- Quality control strains. Haemophilus influenzae (requires X and V). Haemophilus parainfluenzae (requires V only). Haemophilus ducreyi or Haemophilus haemoglobinophilus (requires X only).
- 0.5 McFarland standard. It is used to adjust turbidity of the bacterial suspension.
- Sterile cotton swab for making lawn culture. Inoculating loop or straight wire. Sterile forceps for placing disks.
- Incubator at 35°C to 37°C with CO2 atmosphere (about 3% to 10% CO2).
Procedure of X and V factor Test

- Select well isolated colonies from 18 to 24 hour old culture. Take sterile factor free liquid (0.85% saline or TSB) in a tube (2 to 5 ml). Emulsify the colonies and prepare suspension.
- Adjust the turbidity of the suspension to 0.5 McFarland standard. Do not carry agar from primary isolation medium, trace nutrients can change the result.
- Take a sterile cotton swab and dip in the suspension. Press the swab on the inside wall of the tube to remove excess fluid.
- Inoculate the factor free basal agar plate (TSA or MHA or BHI agar). Streak the swab over the entire agar surface. For lawn culture streak in 3 directions, rotating the plate about 60° after each streaking.
- Allow the plate surface to dry for 3 to 5 minutes.
- Place the factor disks on agar using sterile forceps. Put X factor disk, V factor disk and XV factor disk. Disks should be kept well apart (about 30 to 50 mm) so diffusion zone does not overlap. Press the disks gently for proper contact with agar.
- Invert the plate and incubate at 35°C to 37°C in CO2 enriched atmosphere (about 5% to 10% CO2). Incubate for 18 to 24 hours. If growth is poor it can be extended up to 48 hours.
- After incubation observe the plate under strong light. Growth around each disk is noted and result is interpreted.
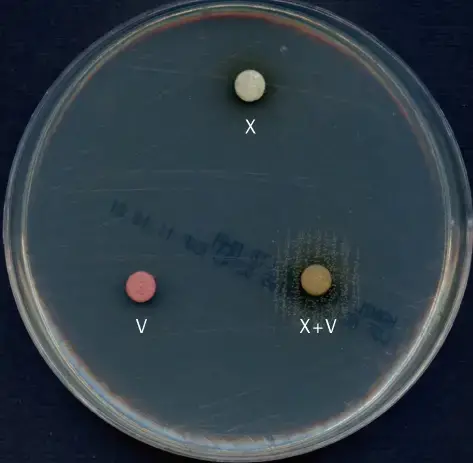
Expected Results of X and V factor Test
Requires both X and V factors
Growth is seen only around the XV disk. No growth around X disk alone and V disk alone. This indicates the organism requires both factors for growth (Haemophilus influenzae, Haemophilus haemolyticus).
Requires only X factor
Growth is seen around X disk and also around XV disk. No growth around V disk alone. This indicates X factor dependent organism (Haemophilus ducreyi).
Requires only V factor
Growth is seen around V disk and also around XV disk. No growth around X disk alone. This indicates V factor dependent organism (Haemophilus parainfluenzae, Haemophilus parahaemolyticus).
Requires neither factor (factor independent)
Growth is seen on entire agar surface around all area. Disk position does not matter. This indicates organism does not depend on exogenous X or V factor (Aggregatibacter aphrophilus).
![X and V factor Test - Principle, Purpose, Procedure, Result 4 X (hemin) and V (nicotinamide adenine dinucleotide [NAD]) factor test. A, Positive: growth
around XV disk only. B, Positive: growth around V disk.](https://biologynotesonline.com/wp-content/uploads/2024/05/image-78.png)
around XV disk only. B, Positive: growth around V disk.

Here is a list of organisms categorized by their positive (required) or negative (not required) dependencies in the X and V factor test:
Requires both X and V factors (Positive for X and V dependency)
- Haemophilus influenzae
- Haemophilus haemolyticus
- Haemophilus aegyptius
Requires X factor only (Positive for X, Negative for V dependency)
- Haemophilus ducreyi
- Haemophilus haemoglobinophilus
Requires V factor only (Negative for X, Positive for V dependency)
- Haemophilus parainfluenzae
- Haemophilus parahaemolyticus
- Haemophilus paraphrohaemolyticus
- Haemophilus sputorum
- Aggregatibacter segnis (formerly Haemophilus segnis)
Requires neither X nor V factor (Negative for both dependencies / Factor-independent)
- Aggregatibacter aphrophilus (formerly Haemophilus aphrophilus and Haemophilus paraphrophilus; note that some strains may have a variable requirement for the V factor)
Quality Control
Here are the quality control strains commonly used to validate the X and V factor test, categorized by their specific growth requirements:
- Requires both X and V factors (grows around XV disk only):
- Haemophilus influenzae ATCC 35056
- Haemophilus influenzae ATCC 10211
- Haemophilus influenzae ATCC 19418
- Haemophilus influenzae NCTC 11931 or NCTC 12975
- Requires V factor only (grows around V and XV disks):
- Haemophilus parainfluenzae (also referred to as Aggregatibacter aphrophilus) ATCC 7901
- Haemophilus parainfluenzae NCTC 10665
- Requires X factor only (grows around X and XV disks):
- Haemophilus ducreyi ATCC 33940
- Haemophilus haemoglobinophilus NCTC 8540

Uses of X and V factor Test
- It is used for presumptive identification of Haemophilus species. It helps in species level classification based on X and V factor requirement (H. influenzae etc.). It is also used for related organisms like Aggregatibacter aphrophilus.
- It is used to differentiate pathogens from commensals. It helps in distinguishing disease causing Haemophilus (H. influenzae, H. ducreyi) from normal flora like H. parainfluenzae.
- It is used to differentiate Haemophilus from other morphologically similar gram negative bacteria. It helps in separating from similar looking organisms like Brucella species.
- It is used to guide clinical diagnosis and treatment. Species level identification helps in selecting proper treatment for infections like meningitis or chancroid.
- It is used in public health management. It helps in rapid identification of invasive pathogens in sterile sites or genital ulcers, and it supports reporting, surveillance and contact tracing or prophylaxis.
Advantages of X and V factor Test
- It is used for species level identification of Haemophilus. It helps to classify the species based on X and V factor requirement.
- It helps to differentiate pathogens from commensals. Pathogenic Haemophilus (H. influenzae) can be separated from normal flora like H. parainfluenzae.
- It supports public health management. Identification of important pathogens like Hib helps in reporting and contact prophylaxis.
- It is useful in clinical diagnosis from different specimens. It helps in identifying organisms from blood, sputum and CSF samples.
- It helps to differentiate Haemophilus from related organisms. It also helps in separating from similar looking organisms like Aggregatibacter aphrophilus.
Limitations of X and V factor Test
- Nutrient carryover is a major problem. If trace X factor or V factor is carried from primary medium like chocolate agar, wrong growth pattern can be seen. It may misidentify H. influenzae as H. parainfluenzae.
- Some basal media may contain trace amounts of X or V factors. Commercial TSA or other basal agar can support growth even without disks. Result becomes confusing.
- Disk placement error can affect the result. V factor diffuses faster than X factor. If disks are kept too close, V factor can overlap into X zone and false pattern is obtained.
- Incubation atmosphere may change the dependency pattern. CO2 enriched atmosphere or anaerobic atmosphere can sometimes give false positive V factor dependency in some strains.
- It cannot differentiate some species. H. influenzae and H. aegyptius both require X and V, so this test cannot separate them.
- It is not a standalone identification test. Different Haemophilus species can share similar factor requirement, so other tests are required for confirmation (biochemical, immunological, molecular, MALDI-TOF etc.).
- Prior genus identification is needed. The isolate should be presumptively Haemophilus before doing this test. Other fastidious bacteria may give confusing growth around disks.
Precautions of X and V factor Test
- Avoid nutrient carryover from primary isolation medium. While picking colonies do not pick any agar. Even trace X or V factor in agar can give false growth. Do not cool the loop on primary medium before picking colonies.
- Use only factor free basal agar medium. TSA or MHA or other validated basal media is used. Do not use blood agar or chocolate agar for doing this test.
- Place the disks properly. X, V and XV disks should be kept at sufficient distance (about 30 to 50 mm). V factor diffuses faster than X factor, so if disks are close the zones overlap and result becomes wrong.
- Standardize the inoculum. Prepare suspension and match it with 0.5 McFarland. Lawn culture should be uniform. Avoid very heavy suspension because it may carry trace hemin or NAD like compounds and can change result.
- Incubation atmosphere should be considered. Usually 5% to 10% CO2 is used for Haemophilus growth. Very high CO2 tension or strict anaerobic condition can sometimes show false V factor growth in some strains.
- Do not use this test alone for final identification. Some species share same requirement (H. influenzae and H. aegyptius), so confirm with other biochemical or immunological or molecular tests.
References
- Abou Rayia, D. M. (2024). Haemophilus influenzae [PowerPoint presentation]. Medical Microbiology and Immunology Department.
- California Department of Public Health, Immunization Branch. (2024, June). Invasive Haemophilus influenzae quicksheet.
- Centers for Disease Control and Prevention. (2026, March 6). Best practices for diagnosis of Haemophilus influenzae and Neisseria meningitidis disease.
- Classification, identification, and clinical significance of Haemophilus and Aggregatibacter species with host specificity for humans. (n.d.). PMC.
- Comparative genomic analysis identifies X-factor (haemin)-independent Haemophilus haemolyticus. (n.d.). PMC.
- Condalab. (n.d.). V factor disc Cat. 7378. BioTrading.
- Dahal, P. (2023, October 6). Satellitism test: Principle, media, procedure, results, uses. Microbe Notes.
- Dalynn Biologicals. (2014, October). Porphyrin (ALA substrate).
- Diagnostic assessment of X and V growth factor requirements for the speciation of the genus Haemophilus. (n.d.).
- Differentiation of Haemophilus aegyptius and Haemophilus influenzae. (n.d.). PubMed.
- DrChika. (2023, January 2). Satellitism test. Microbiology Class.
- Hardy Diagnostics. (2020). ALA differentiation disks.
- Hardy Diagnostics. (2020). Haemophilus ID quadplate.
- Hardy Diagnostics. (2020). X- and V-factor disks.
- HiMedia Laboratories. (2026, May). X+V factor – Technical data.
- Is a NAD pyrophosphatase activity necessary for Haemophilus influenzae type b multiplication in the blood stream? (n.d.). PubMed.
- Kemmer, G., Reilly, T. J., Schmidt-Brauns, J., Zlotnik, G. W., Green, B. A., Fiske, M. J., Herbert, M., Kraiß, A., Schlör, S., Smith, A., & Reidl, J. (2001). NadN and e (P4) are essential for utilization of NAD and nicotinamide mononucleotide but not nicotinamide riboside in Haemophilus influenzae. Journal of Bacteriology, 183(13), 3974–3981. https://doi.org/10.1128/JB.183.13.3974-3981.2001
- Liofilchem. (2024, May 13). X factor test, V factor test, V+X factor test.
- Microbiology pictures. (2015). Satelliting of H.influenzae on blood agar around Staphylococcus aureus.
- Musher, D. M. (1996). Haemophilus species. In S. Baron (Ed.), Medical microbiology (4th ed.). University of Texas Medical Branch at Galveston.
- Public Health England. (2015, February 3). Identification of Haemophilus species and the HACEK group of organisms. UK Standards for Microbiology Investigations, ID 12(3).
- Remel. (2003, January 16). Porphyrin test agar.
- Remel. (2011, September 14). Porphyrin test agar.
- Saeed, E. A. (n.d.). Genus: Haemophillus morphology. Department of Clinical Laboratory Sciences.
- Sandle, T. (2015, March 27). Porphyrin synthesis test explained. Pharmaceutical Microbiology Resources.
- stupidlavendar. (2024). How do we differentiate Haemophilus influenzae from Haemophilus aegyptus? Reddit.
- UK Health Security Agency. (2025, March 6). Porphyrin synthesis (ALA) test. UK Standards for Microbiology Investigations, TP 29(4.1).
- UK Health Security Agency. (2025, March 12). X and V factor test. UK Standards for Microbiology Investigations, TP 38(4.2).
- Wikipedia contributors. (n.d.). Haemophilus influenzae biogroup aegyptius. Wikipedia, The Free Encyclopedia.
- Zhou, L., Xia, Y., Feng, Y., Du, B., Huang, X., Xu, W., Li, J., Fang, F., Meng, C., Yu, L., Ma, L., Xue, G., & Yuan, J. (2025). Rapid detection of Hemophilus influenzae and Streptococcus pneumoniae simultaneously using a duplex recombinase-aided amplification assay directly from invasive clinical samples. Frontiers in Cellular and Infection Microbiology, 15. https://doi.org/10.3389/fcimb.2025.1631633