Starch Hydrolysis Test is a simple biochemical test used to determine whether a microorganism can hydrolyze starch into smaller sugars. It is done because starch is a large polysaccharide and cannot pass through the bacterial cell membrane, so some bacteria secrete extracellular amylase to break it down.
In this test, the organism is inoculated on starch agar plate and incubated for growth. After incubation, iodine solution is added on the plate, where iodine reacts with intact starch and produces blue-black or purple colour. If starch is hydrolyzed, a clear colourless zone (halo) is seen around the colony showing positive test. If starch is not hydrolyzed, the whole medium turns dark up to the colony margin showing negative test. It is used in diagnostic microbiology to differentiate bacteria like Bacillus, Clostridium and Streptococcus.
Objectives of Starch Hydrolysis Test
- To determine whether the given microorganism can hydrolyze starch into smaller usable sugars.
- To detect the production of extracellular starch hydrolyzing enzymes mainly amylase (α-amylase) and related enzymes.
- To differentiate and identify bacterial species based on positive and negative starch hydrolysis reaction.
- To correlate starch hydrolysis ability with presence of amylase gene in that organism.
- To understand the metabolic advantage of starch breakdown for getting carbon and energy source from polysaccharide.
Principle of Starch Hydrolysis test
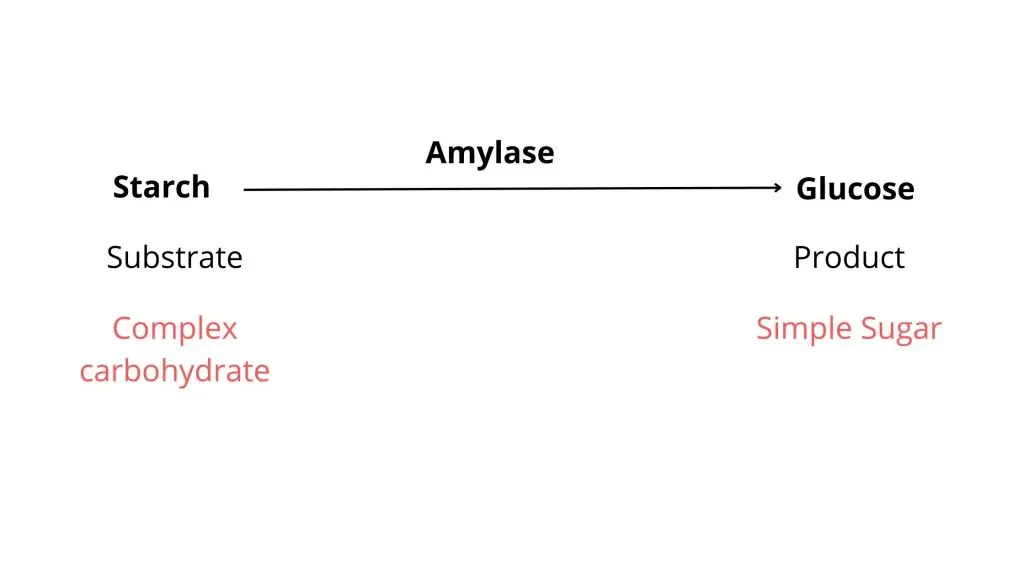
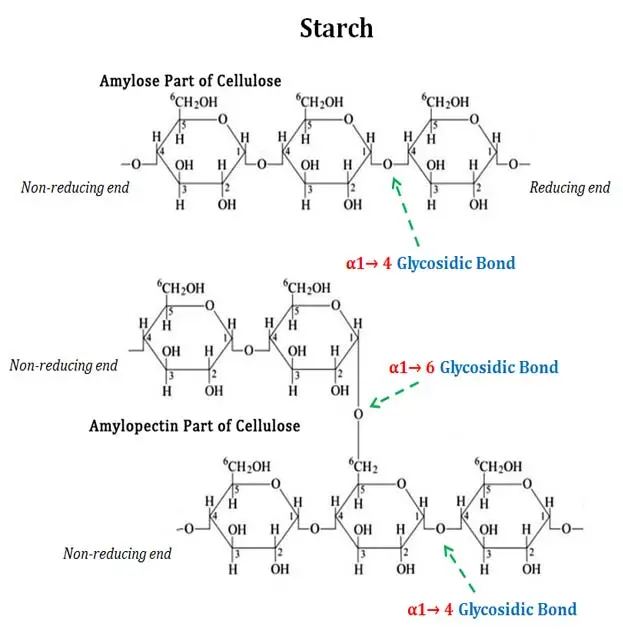
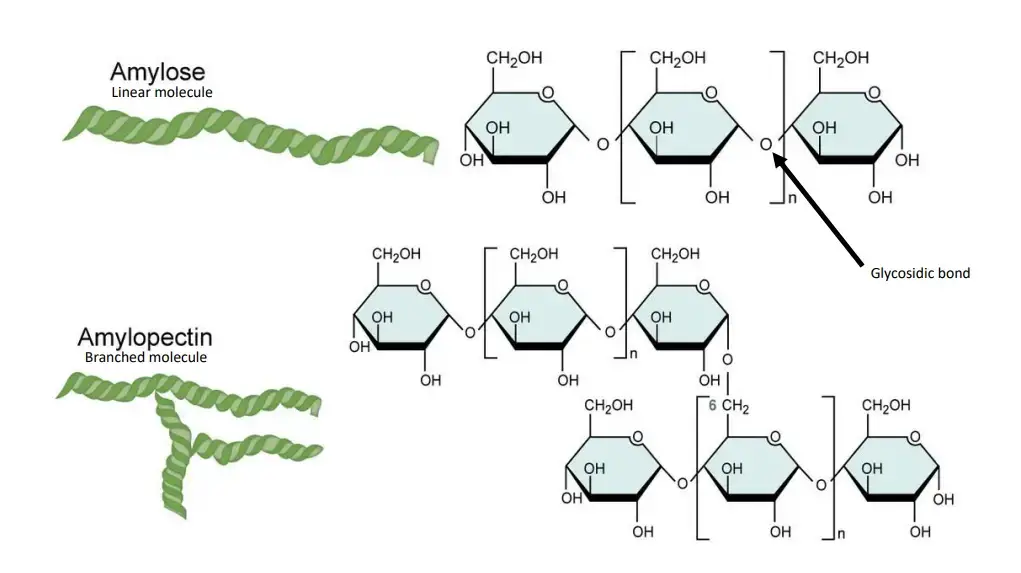
Principle of Starch Hydrolysis Test is based on the ability of some bacteria to produce extracellular amylase enzyme, which hydrolyzes starch present in the medium. Since starch is a large molecule, it cannot enter directly through bacterial cell membrane, so it is first broken outside the cell into smaller sugars (glucose and maltose).
In this test, organism is grown on starch agar and after incubation iodine is added as an indicator. Iodine reacts with intact starch and gives dark blue-black or purple colour. If starch is hydrolyzed by amylase, a clear zone appears around the colony due to absence of intact starch (positive). If there is no amylase production, the medium becomes uniformly dark around the growth showing negative result.
Requirement
- Culture media
- Starch agar plate (nutrient agar / Mueller-Hinton agar / heart infusion agar + soluble starch about 2%).
- Media contains peptone, beef extract, yeast extract, NaCl, soluble starch and agar.
- Reagent
- Gram’s iodine or Lugol’s iodine solution (indicator).
- It is prepared using iodine crystals, potassium iodide and distilled water (sometimes sodium bicarbonate is added).
- Equipments
- Petri plates.
- Incubator (35 ± 2°C).
- Inoculating loop / sterile swab / pipette tip.
- Autoclave.
- Bunsen burner, dropper and weighing balance.
- Organisms
- Test organism (fresh pure culture 18–24 hours).
- Positive control (Bacillus subtilis / Bacillus cereus / Streptococcus bovis).
- Negative control (Escherichia coli / Staphylococcus aureus).
Starch hydrolysis test Media (Compositions and Preparation)
Common media used
- Starch agar is used commonly for starch hydrolysis test.
- Mueller-Hinton agar (MHA) can also be used.
- Heart infusion agar can be used, but starch is supplemented in it.
Composition of Starch agar (per 1000 ml)
- Beef extract (meat extract)- 1.5 g to 3.0 g
- Peptone (peptic digest of animal tissue)- 5.0 g
- Yeast extract- 1.5 g to 5.0 g
- Soluble starch- 2.0 g to 10.0 g
- Sodium chloride- 5.0 g
- Agar- 15.0 g
- Distilled / demineralized water- 1000 ml
- Final pH- 7.2 to 7.4 ± 0.2 at 25°C
Composition of Mueller-Hinton agar (per 1000 ml)
- Beef infusion (HM infusion B)- 300.0 g
- Casein acid hydrolysate (Acicase / Casamino acid)- 17.5 g
- Starch- 1.5 g
- Agar- 17.0 g
- Distilled water- 1000 ml
- Final pH- 7.3 ± 0.1 at 25°C
Media preparation (general procedure)
- Required amount of dehydrated media powder is weighed. (Starch agar 25.0 to 30.0 g / L. MHA 38.0 g / L).
- Add the powder into 1000 ml distilled water in a flask or bottle.
- Mix well to make uniform suspension.
- Heat the media with stirring and boil till complete dissolving occurs.
- Over boiling is avoided, because starch may break down by heat and false positive may be produced.
- Sterilize by autoclaving at 121°C (15 lbs pressure) for 15 minutes.
- After autoclave, allow the media to cool to about 40°C to 50°C.
- Pour aseptically into sterile petri plates (about 20 to 25 ml in each 10 cm plate).
- Agar depth is maintained around 4 mm, too thick or too thin agar affects enzyme diffusion.
- Plates are kept undisturbed at room temperature to solidify completely.
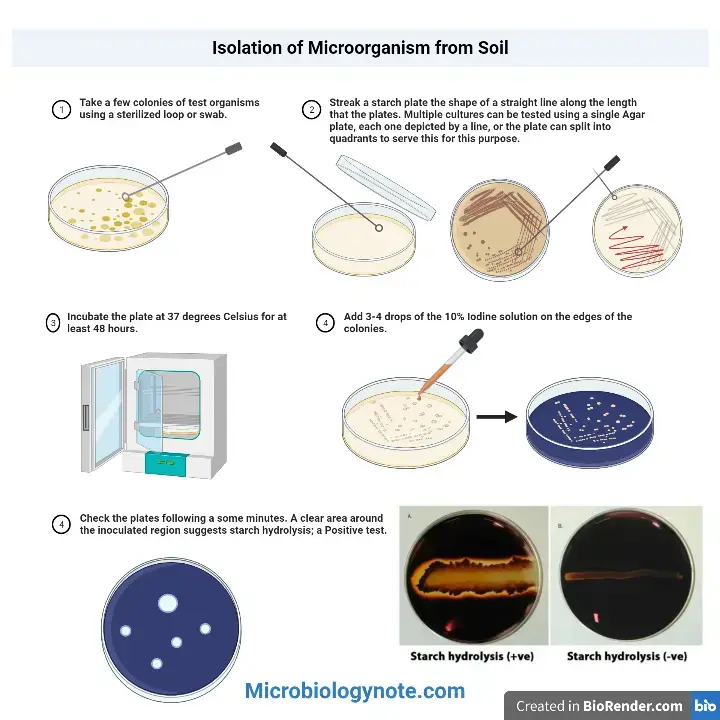
Procedure of Starch Hydrolysis Test
- Step 1
Prepared starch agar plates are kept and allowed to reach room temperature. - Step 2
A sterile inoculating loop or sterile swab is taken. Fresh bacterial culture is picked. - Step 3
Inoculate on starch agar surface. A single straight line streak is made (or spot inoculation is done). - Step 4
If more organisms are tested on same plate, streaks are kept at least 25 mm apart. This is done to avoid overlapping of enzyme diffusion. - Step 5
The plates are inverted. Incubate at 35 ± 2°C for 24 to 48 hours for growth. - Step 6
Slow growing organisms are incubated for longer time (7 to 10 days). - Step 7
After incubation, plate is placed on a white background for better contrast. - Step 8
Flood the agar surface (over the growth) with Gram’s iodine or Lugol’s iodine. - Step 9
Iodine is allowed to react for 30 seconds to few minutes. Then excess iodine is poured off carefully. - Step 10
Observation is done immediately because colour may fade with time. - Step 11
Clear or colourless halo around the growth is taken as positive (starch hydrolysis). - Step 12
Dark blue-black colour up to the edge of growth is taken as negative (no starch hydrolysis).
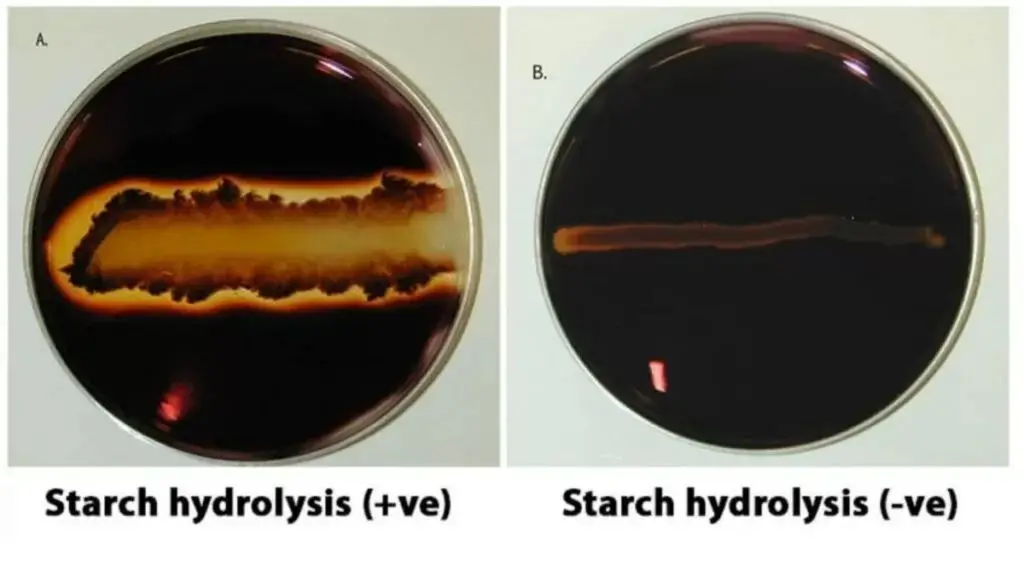
Starch Hydrolysis Test Results
Positive result
Appearance-
A clear zone is formed around the bacterial growth after adding iodine solution. The halo is colourless or light yellowish (zone of clearing). It is seen immediately surrounding the streak or colony.
Interpretation-
It indicates starch is hydrolysed in the medium. The organism produce and secrete amylase (exoenzyme). The starch around the growth is broken down, so iodine cannot produce blue-black colour in that area.
Examples-
Bacillus subtilis. Bacillus cereus. Streptococcus bovis.
Negative result
Appearance-
The whole agar becomes uniform dark blue, blue-black or purple after adding iodine. The dark colour is seen right up to the edge of the bacterial colonies. No clear zone is formed around the growth.
Interpretation-
It indicates starch is not hydrolysed. The organism does not possess amylase producing ability. Starch remain intact and reacts with iodine indicator giving dark colour.
Examples-
Escherichia coli. Staphylococcus aureus.
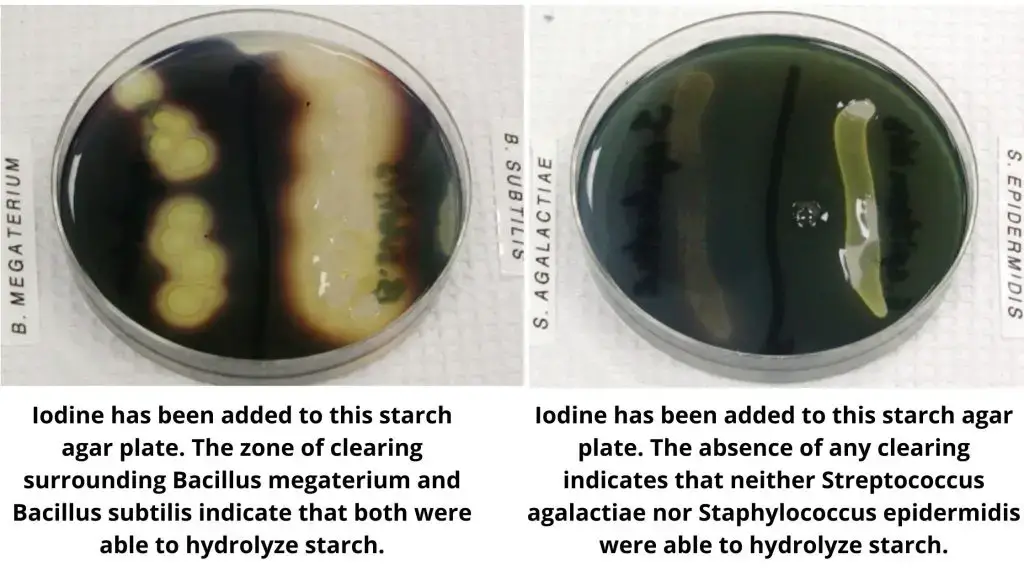
Organisms Showing Starch Hydrolysis Test Reaction
Here is a list of organisms categorized by their typical results in a starch hydrolysis test:
Organisms with a Positive Result (Starch Hydrolyzing / Amylase Producing)
- Bacillus subtilis
- Bacillus cereus
- Bacillus megaterium
- Bacillus anthracis
- Bacillus polymyxa
- Streptococcus bovis
- Streptococcus iniae
- Clostridium perfringens
- Chryseobacterium indologenes
- Streptomyces albus
Organisms with a Negative Result (Starch Non-hydrolyzing / Amylase Non-producing)
- Escherichia coli
- Staphylococcus aureus
- Pseudomonas aeruginosa
- Pseudomonas putida
- Corynebacterium diphtheriae
- Clostridium difficile
- Clostridium botulinum
- Clostridium sordellii
- Elizabethkingia meningoseptica
- Viridans streptococci (except S. bovis)
- Enterococcus species (including E. faecalis, E. durans, and E. seriolicida)
- Lactococcus species (including L. garvieae)
- Pediococcus dextrinicus
- Nocardia brasiliensis
Uses of Starch Hydrolysis Test
- It is used as a basic biochemical test to identify and differentiate bacteria based on amylase production.
- It is used to check the ability of organism to produce and secrete exoenzyme amylase and hydrolyse starch.
- It is used in clinical laboratory for identification of some pathogens. Example- Streptococcus bovis can be differentiated from other bile-esculin positive viridans streptococci.
- It is used to differentiate Clostridium perfringens (positive) from Clostridium difficile and Clostridium botulinum (negative).
- It is used to distinguish Chryseobacterium indologenes from Elizabethkingia meningoseptica.
- It is used in characterization of organisms of Bacillus, Corynebacterium, Fusobacterium, Enterococcus and Pseudomonas group.
- It is used in veterinary and aquaculture diagnosis. Streptococcus iniae in fish farming can be identified as it can hydrolyse starch.
- It is used as a primary screening test to isolate amylase producing bacteria from soil and sediment samples for industrial purpose.
- Selected strains are used in industry for production of glucose and maltose syrups and also in biofuel related processes.
- It is used in food and beverage quality control to detect starch hydrolysing bacterial contaminants in dairy, water and other food products.
- It is used in microbiology teaching lab to demonstrate exoenzyme secretion and breakdown of complex substrate (starch).
Advantages of Starch Hydrolysis Test
- It is used to differentiate bacterial species which look similar in morphology and routine biochemical tests.
- It is useful in genera like Bacillus, Clostridium, Corynebacterium, Streptococcus, Enterococcus and Pseudomonas for separation.
- It helps in targeted clinical identification. Streptococcus bovis is separated from other bile-esculin positive viridans streptococci by this test.
- It also helps to distinguish Chryseobacterium indologenes from Elizabethkingia meningoseptica.
- It is essential in aquatic and agricultural diagnosis. Streptococcus iniae in fish farming can be detected and outbreak monitoring is possible.
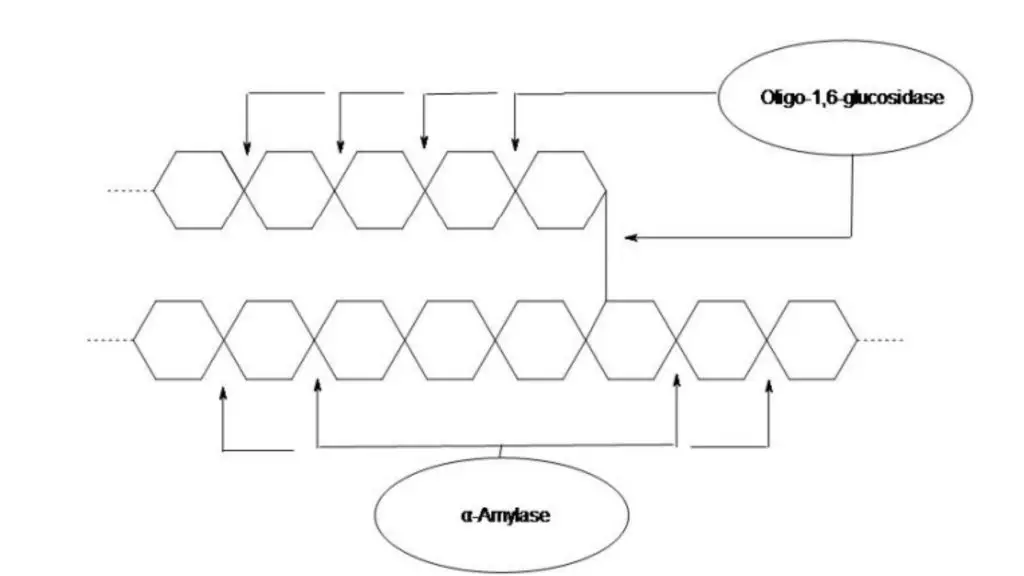
- It is used to evaluate exoenzyme production by organism. Mainly extracellular amylase (α-amylase) production is detected. Oligo-1,6-glucosidase activity can also be indicated in some organisms.
- It is used as primary screening method in industrial microbiology. New amylase producing strains can be isolated from soil and environmental samples.
- It is useful in food, beverage and biofuel industries for selecting efficient amylase producers.
- It is a simple and cost effective test. It is rapid and gives clear visible result after iodine addition.
- Many isolates can be tested on a single plate by keeping proper distance between streaks. So it is scalable.
- It is a good teaching laboratory model. Students can see enzyme action and substrate degradation directly on plate.
Limitations of Starch Hydrolysis Test
- It is not a confirmatory test. Alone it cannot confirm the exact identity of bacteria. Other biochemical tests, staining or molecular tests are needed with it.
- After iodine is added, plate cannot be used for further work. Iodine is lethal to bacteria, so colonies cannot be subcultured from that tested plate.
- Incubation time is more. Usually 24 to 48 hours are required for proper result. Slow growers may require 7 to 10 days, otherwise false negative can come.
- The blue-black colour is not stable for long time. Colour fades with time. If reading is delayed, fading can look like a false clear zone and false positive interpretation may occur.
- If media contains simple sugars like glucose in high amount, result can be affected. Organism will use glucose and amylase production may be suppressed, giving false negative.
- Swarming bacteria are not suitable for this test. Proteus vulgaris or Proteus mirabilis spreads on whole plate and localized zone cannot be identified properly.
- Agar depth affects result because enzyme diffusion is needed. Too deep agar (>5 mm) can dilute the enzyme and clearing zone may not be visible. Too shallow agar (<3 mm) can give oversized halo and result may be overestimated.
Quality Control Organisms of Starch Hydrolysis Test
Positive Control Organisms (Starch Hydrolysis Positive)
- Bacillus cereus
- Bacillus subtilis
- Streptococcus bovis
- Clostridium perfringens (used for anaerobic testing)
- Pseudomonas stutzeri
- Chryseobacterium indologenes
Negative Control Organisms (Starch Hydrolysis Negative)
- Escherichia coli
- Staphylococcus aureus
- Enterococcus faecalis
- Staphylococcus epidermidis
- Clostridium sordellii (used for anaerobic testing)
- Serratia marcescens
- Streptococcus agalactiae
Precautions of Starch Hydrolysis Test
- Media should not be over boiled before autoclaving. Excess heat can break down the starch without enzyme. It may give false positive result.
- Media with high simple sugar (glucose) is avoided. Bacteria will use glucose first and amylase production may get suppressed. It can give false negative result.
- Agar depth is maintained standard (around 4 mm). Too deep or too shallow agar affects diffusion of enzyme. Halo size can be disturbed.
- When multiple organisms are inoculated on same plate, streaks are kept at least 25 mm apart. Enzyme diffusion from one streak should not overlap with other streak.
- Iodine is added only after proper incubation is completed (usually 48 hours). Early addition may show no clear zone and false negative can come.
- After iodine addition, result is read immediately. Blue-black colour fades with time and false reading can occur.
- Subculture should not be done from iodine flooded plate. Iodine is toxic and kills the bacteria. Separate master plate is kept for further work.
- Swarming organisms are avoided (Proteus spp.). They spread on whole surface and zone cannot be localized properly.
References
- Analytical perspectives on the starch hydrolysis assay: Biochemical mechanisms, diagnostic methodologies, and clinical utility in microbial phenotyping. (n.d.).
- Chauhan, A. (2025, December 18). Hydrolysis test: Procedure and applications in microbiology. Flabs. https://flabslis.com/blogs/hydrolysis-test
- Evans, J. J., Klesius, P. H., & Shoemaker, C. A. (2004). Starch hydrolysis testing of multiple isolates for rapid differentiation of Streptococcus iniae. Bulletin of the European Association of Fish Pathologists, 24(5), 231-239.
- Evans, J. J., Klesius, P. H., & Shoemaker, C. A. (2004, October 1). Starch hydrolysis testing of multiple isolates for rapid differentiation of Streptococcus iniae [Publication No. 161219]. USDA Agricultural Research Service.
- Hardy Diagnostics. (2020). Instructions for use: Starch agar.
- Hartline, R. (2023, February 18). 1.17: Starch hydrolysis. Biology LibreTexts. https://bio.libretexts.org/Bookshelves/Microbiology/Microbiology_Laboratory_Manual_(Hartline)/01%3A_Labs/1.17%3A_Starch_Hydrolysis
- HiMedia Laboratories. (2018). Technical data: M107 Starch agar.
- Hudzicki, J. (2009, December 8). Kirby-Bauer disk diffusion susceptibility test protocol. American Society for Microbiology.
- InformationBoxTicket Lifestyles. (n.d.). Performing the starch hydrolysis test in the microbiology lab – Biochemical tests 101 [Video]. YouTube.
- Lal, A., & Cheeptham, N. (2012, November 1). Starch agar protocol. American Society for Microbiology.
- LaPierre, L., Cornejo, J., Asun, A., Vergara, C., & Varela, D. (2020, May). Laboratory guide: Methodologies for antimicrobial susceptibility testing. Asia-Pacific Economic Cooperation Secretariat. www.apec.org
- Megazyme. (2026). Oligo-α-1,6-glucosidase microbial enzyme.
- Pullulanase: Role in starch hydrolysis and potential industrial applications. (n.d.). PMC – NIH.
- Remel. (2009, November 25). Starch agar. Thermo Fisher Scientific.
- Remel. (2009, November 25). Starch hydrolysis agar. Thermo Fisher Scientific.
- ResearchTweet. (2025). Starch hydrolysis test: Result, principle, procedure, and reagents.
- Sigmon, J. (2008, September 1). The starch hydrolysis test. American Society for Microbiology.
- Starch agar [Image gallery]. (2012, November 1). American Society for Microbiology.
- Starch hydrolysis test. (n.d.). VUMIE.
- Starch hydrolysis test purpose. (n.d.).
- Tariq, A. L., Sudha, S., & Reyaz, A. L. (2016). Isolation and screening of Bacillus species from sediments and application in bioremediation. International Journal of Current Microbiology and Applied Sciences, 5(6), 916-924. http://dx.doi.org/10.20546/ijcmas.2016.506.099
- The impact of agar depth on antimicrobial susceptibility testing by disc diffusion. (n.d.). PMC – NIH.
- TMMedia. (2019, November 8). Product data sheet: TM 430 – Starch agar.
- Wikipedia contributors. (2026, April 6). Iodine–starch test. In Wikipedia, The Free Encyclopedia. https://en.wikipedia.org/w/index.php?title=Iodine–starch_test&oldid=1347386770
- Zerin, T., Ahmed, S., Badhan, R. A., Mostafa, S., Keya, A. S., Saqif, A. T., Alam, S. T., Thity, J. A., & Nesa, T. R. (2025). Characterization of amylase-producing Bacillus species and insight into its amylase activity. Journal of Microbiology, Biotechnology and Food Sciences. https://doi.org/10.55251/jmbfs.10694
- α-Amylase: An enzyme specificity found in various families of glycoside hydrolases. (n.d.). PMC – NIH.