Lysine decarboxylase test is a biochemical test used to differentiate mainly Gram negative bacteria (Enterobacteriaceae) on the basis of lysine decarboxylase enzyme production. It is done in a lysine broth medium (Moeller’s / Falkow’s / Taylor’s) containing L-lysine, small amount of glucose, pyridoxal (Vit B6) and a pH indicator like bromocresol purple. After inoculation, mineral oil layer is added to make anaerobic condition.
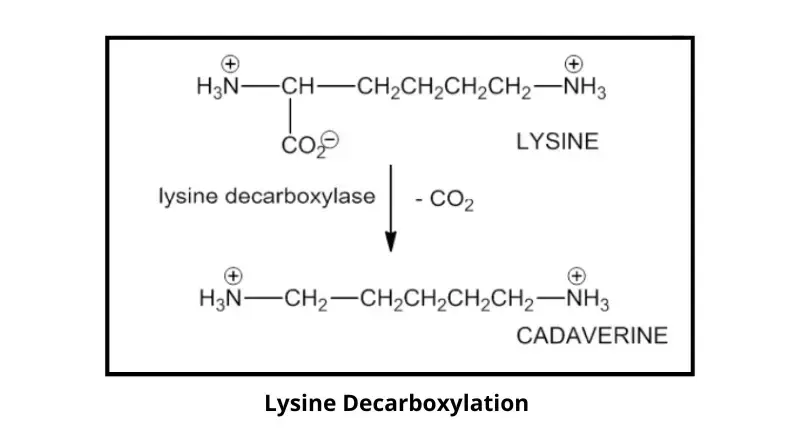
In this test, glucose is first fermented and acid is formed so the medium turns yellow. This acidic condition induces lysine decarboxylase in positive organism. Lysine is decarboxylated to cadaverine and CO2, cadaverine is alkaline and raises the pH, so the medium changes back to purple (positive). If enzyme is not produced, cadaverine is not formed and the medium remains yellow (negative).
Principle of Lysine decarboxylase test
Principle of Lysine decarboxylase test is based on the ability of the organism to produce lysine decarboxylase enzyme and act on L-lysine in the medium. The test medium contains glucose, L-lysine, pyridoxal (co-factor) and a pH indicator (bromocresol purple). Mineral oil is added over the broth to block oxygen, because this reaction is done in anaerobic condition.
In first stage, the organism ferment the glucose and acid is produced. Due to acid production the pH is lowered and the medium turns yellow from purple. This acidic pH is required to induce the decarboxylase enzyme in lysine positive organisms.
If enzyme is present, lysine is decarboxylated and cadaverine and CO2 is formed. Cadaverine is alkaline so it neutralize the acid and pH goes up. The indicator again turns purple and it is reported as positive. If enzyme is absent, lysine is not broken down and medium remains yellow (negative).
Objectives of Lysine decarboxylase test
- To detect whether the organism produce lysine decarboxylase enzyme or not.
- To determine the ability of organism to decarboxylate L-lysine and form cadaverine (alkaline amine).
- To study lysine utilization by the organism in the broth medium by change in pH reaction.
- To differentiate Gram negative enteric bacilli mainly Enterobacteriaceae by biochemical identification.
- To differentiate Salmonella (usually positive) from Shigella (usually negative).
- To distinguish Salmonella arizonae from Bethesda Ballerup group of Enterobacteriaceae.
- To help in classification of other related Gram negative bacteria like Vibrio, Aeromonas and Plesiomonas.
Requirements for Lysine decarboxylase test
- Decarboxylase broth base (Moeller’s / Falkow’s / Taylor’s).
- L-lysine (substrate) in the broth (0.5% or 1%).
- Glucose or dextrose (fermentable carbohydrate).
- Peptone / yeast extract / beef extract (nutritional ingredients).
- Pyridoxal (Vit B6) as co-factor.
- pH indicator (bromocresol purple, sometimes cresol red).
- Sterile mineral oil (liquid paraffin / vaspar / petroleum jelly) for anaerobic condition.
- Sterile test tubes (screw capped) with the medium.
- Sterile inoculating loop / needle / sterile stick.
- Incubator (35°C to 37°C).
- Confirmatory reagents if needed (ninhydrin reagent, KOH, chloroform).
- Test organism (fresh pure culture, 18 to 24 hours).
- Control organisms for QC (Salmonella typhimurium / Klebsiella pneumoniae as positive, Proteus vulgaris / Proteus mirabilis as negative).
Composition and preparation of decarboxylase broth medium
Composition (per 1 litre distilled water)
- Peptone 5.0 g.
- Beef extract 5.0 g. (Moeller’s base) (or yeast extract 3.0 g in Falkow’s / Taylor’s).
- Dextrose / glucose 0.5 g to 1.0 g.
- Bromocresol purple 0.01 g to 0.02 g.
- Cresol red 0.005 g. (Moeller’s base).
- Pyridoxal 0.005 g. (co-factor).
- L-amino acid (substrate) (L-lysine / L-arginine / L-ornithine) 10.0 g (1%). (If DL amino acid is used then 2% is added).
- Taylor’s modification (peptone free) may contain L-lysine HCl 5.0 g, yeast extract 3.0 g, glucose 1.0 g and bromocresol purple 0.015 g.
Preparation
- Take 1000 ml distilled water in flask.
- Suspend the dehydrated decarboxylase base powder in water. (or prepare base by adding peptone, extract, glucose, indicators and pyridoxal).
- Add the required L-amino acid to the base and mix.
- Heat with frequent shaking and boil about 1 minute till the medium dissolve properly.
- If ornithine is used, pH is checked and adjusted before dispensing.
- Dispense into screw capped test tubes (usually 5 ml per tube).
- Autoclave at 121°C (15 lbs) for 10 to 15 minutes.
- Cool the tubes in upright position.
- For anaerobic condition, add sterile mineral oil / liquid paraffin / vaspar overlay (2 to 5 mm layer). It is added before sterilization or it is added aseptically after inoculation.
Procedure of Lysine decarboxylase test
- Take lysine decarboxylase broth tube (test tube).
- Take one control tube containing basal medium without lysine.
- Pick 1 to 2 isolated colonies from fresh 18 to 24 hours pure culture with sterile loop or needle.
- Inoculate the test broth with the organism. (or add 1 to 4 drops of broth culture).
- Inoculate the control tube also with same organism.
- Mix both tubes gently so that inoculum is distributed in the medium.
- Add sterile mineral oil over the broth in both tubes (about 4 mm layer or around 1 ml).
- Tighten the caps and keep the tubes in incubator at 35°C to 37°C.
- Observe daily for colour change up to 4 days. Some slow organisms may take more days.
- In positive, the test tube first becomes yellow then turns purple again, control remains yellow.
- In negative, the test tube becomes yellow and remains yellow.
Lysine decarboxylase test results
Positive result
Test tube shows purple / violet or faded yellow-purple colour (turbid). The medium first turns yellow due to glucose fermentation and later again becomes purple due to cadaverine (alkaline) formation. Control tube turns yellow and remains yellow.
Negative result
Test tube turns bright yellow and remains yellow. It looks similar to control tube. It indicates glucose is fermented but lysine decarboxylase enzyme is not produced.
Invalid test
Control tube becomes purple or remains original colour or it does not turn yellow. It occurs when organism does not ferment glucose or anaerobic condition is not maintained (oil layer not proper), so false alkaline reaction can occur and result is not interpreted.

Organisms showing Lysine decarboxylase test result
Organisms showing a Positive Result:
- Salmonella species (including S. enterica, S. Typhi, S. Typhimurium, S. Enteritidis, S. Arizonae, S. Paratyphi B, and S. Paratyphi C)
- Klebsiella pneumoniae and Klebsiella oxytoca
- Enterobacter aerogenes (also known as Klebsiella aerogenes)
- Serratia marcescens
- Vibrio cholerae, Vibrio parahaemolyticus, Vibrio vulnificus, and Vibrio mimicus
- Plesiomonas shigelloides
- Most Aeromonas species (including A. hydrophila, A. sobria, and A. veronii)
- Edwardsiella species
Organisms showing a Negative Result:
- Shigella species (including S. dysenteriae, S. flexneri, and S. sonnei)
- Proteus species (including P. mirabilis, P. vulgaris, and P. hauseri)
- Citrobacter freundii
- Enterobacter cloacae
- Salmonella Paratyphi A
- Pseudomonas species (including P. aeruginosa, P. fluorescens, and P. putida)
- Yersinia enterocolitica
- Cronobacter sakazakii and Cronobacter muytjensii
- Alcaligenes species
- Flavobacterium species
- Xanthomonas species
Organisms showing a Variable Result:
- Escherichia coli (can test positive, negative, or display a delayed reaction depending on the specific strain)
Uses of Lysine decarboxylase test
- It is used to identify and differentiate Enterobacteriaceae and other related Gram negative bacilli by biochemical profile.
- It is used to differentiate Salmonella (lysine decarboxylase positive) from Shigella (lysine decarboxylase negative).
- It is used to differentiate Salmonella serotypes like Salmonella typhi (positive) and Salmonella paratyphi A (negative).
- It is used to differentiate Salmonella arizonae from Bethesda Ballerup group of Enterobacteriaceae.
- It is used for biochemical confirmation of Salmonella in food, water, animal feed and environmental samples (ISO methods).
- It is used for biochemical confirmation of Yersinia enterocolitica (usually lysine decarboxylase negative).
- It is used as confirmatory test for Vibrio species like Vibrio cholerae and Vibrio parahaemolyticus (ISO methods).
- It is used in biochemical profiling of Plesiomonas shigelloides and Aeromonas species along with other decarboxylase tests.
Advantages of Lysine decarboxylase test
- It is reliable test to classify and differentiate Gram negative bacilli especially Enterobacteriaceae.
- It is useful to separate Salmonella (usually positive) from Shigella (negative) in routine identification.
- It helps in differentiation of some Salmonella serotypes like Salmonella typhi (positive) and Salmonella paratyphi A (negative).
- It can distinguish Salmonella arizonae from Bethesda Ballerup group when proper medium like Taylor’s (peptone free) is used.
- It is used as a mandatory biochemical confirmation step for Salmonella detection in food, animal feed and water samples in standard protocols (ISO).
- It supports confirmation of Yersinia enterocolitica in standard method, as it is lysine decarboxylase negative.
- It is required confirmatory test for Vibrio parahaemolyticus and Vibrio cholerae in seafood and marine samples (ISO guideline).
- Along with other decarboxylase tests it helps to separate Plesiomonas shigelloides from Aeromonas species.
Limitations of Lysine decarboxylase test
- It is mainly for glucose fermenting organisms, because acid pH is needed first to induce the enzyme. Non fermenters may give unreliable or weak result.
- Early reading gives false negative. Before 18 to 24 hours result should not be taken, because glucose fermentation and acid production takes time.
- Some weak producers require long incubation, sometimes up to 10 days, so it becomes time taking.
- Mineral oil overlay is must. If oil layer is not given or tube is exposed to air, surface oxidation and peptone deamination can give alkaline reaction and false positive.
- Colour interpretation is sometimes difficult, yellow and purple layers can be seen, so tube need gentle shaking before reading.
- Sometimes grey colour can appear due to reduction of indicator, it is not true positive and it confuse the result.
- The test does not measure exact enzyme amount, it is only detected when pH change occurs. Medium composition like glucose, lysine concentration can affect the final colour.
- Some media formulation is not satisfactory for some organisms like Klebsiella and Enterobacter, so wrong result can occur.
- It is only one biochemical character, so final identification is not done by this alone, other biochemical tests and pure culture is required.
Precautions of Lysine decarboxylase test
- Use sterile mineral oil overlay (mineral oil / vaspar / paraffin) after inoculation and tighten the cap properly. Anaerobic condition is required, otherwise false alkaline reaction occurs at surface due to peptone oxidation and deamination.
- Do not read the result early. Minimum 18 to 24 hours incubation is needed. If read before 12 to 18 hours it may give false negative.
- Always keep a control tube (basal medium without lysine) along with test tube. If control tube becomes purple or it does not turn yellow, the test is invalid.
- Ensure glucose fermentation step. Non glucose fermenting organisms may show weak or delayed reaction and result becomes unreliable.
- Use fresh pure culture (18 to 24 hours) and isolated colonies for inoculation. Mixed culture should not be used. Inoculum should be proper, too less inoculum may delay the reaction.
- If yellow and purple layers are formed in tube, gently shake and mix the tube before final reading.
- Observe for grey colour. Grey colour indicates indicator reduction not true alkaline end product, so it can confuse. If needed add extra bromocresol purple indicator for proper reading.
References
- Abbott, S. L., Cheung, W. K. W., & Janda, J. M. (2003). The genus Aeromonas: Biochemical characteristics, atypical reactions, and phenotypic identification schemes. Journal of Clinical Microbiology, 41(6), 2348-2357. https://doi.org/10.1128/JCM.41.6.2348-2357.2003
- Biochemical characterization and phylogenetic analysis of the virulence factor lysine decarboxylase from Vibrio vulnificus. (n.d.). PubMed Central (PMC).
- Biolab Diagnostics Laboratory Inc. (2016). Culture media for amino acid decomposition studies (Version 5).
- Bio-Rad. (2011, August 5). LDC-ODC-ADH/Broth (Version 3).
- CBSAlife. (n.d.). Dexarboxylase medium.
- CJ CheilJedang Corp. (2016, August 18). Novel lysine decarboxylase, and method for producing cadaverine by using same (WIPO Patent No. WO2016129812A1).
- Condalab. (n.d.). Lysine decarboxylase medium ISO (Cat. 1176).
- Cooper, C. R., Jr. (2018). BIOL 3702 lab exercise – decarboxylation test. Youngstown State University.
- Current advances on structure-function relationships of pyridoxal 5′-phosphate-dependent enzymes. (n.d.). PubMed Central (PMC).
- Difco. (n.d.). Decarboxylase differential media. Manual Difco.
- Enhanced thermal and alkaline stability of L-lysine decarboxylase CadA by combining directed evolution and computation-guided virtual screening. (n.d.). PubMed Central (PMC).
- Hardy Diagnostics. (2020). Moellers decarboxylase media – for identification of gram negative enteric bacteria (IFU-10585[B]).
- HiMedia Laboratories Pvt. Ltd. (2015, February). Lysine decarboxylase broth – technical data (M376).
- HiMedia Laboratories Pvt. Ltd. (2024, March). Lysine decarboxylase broth w/o peptone – technical data (M376I).
- HiMedia Laboratories Pvt. Ltd. (2026, June). Decarboxylase broth base, Moeller (Moeller decarboxylase broth base) – technical data (M393).
- Key Scientific Products, Inc. (n.d.). Decarboxylase and dihydrolase tests ver. 1211.
- Lal, A., & Cheeptham, N. (2015, September 2). Decarboxylase broth protocol. American Society for Microbiology.
- Liofilchem s.r.l. (2023, May 15). Lysine decarboxylase test: Instructions for use (Ref. 88014).
- Lysine decarboxylase activity in broth and agar media. (n.d.). PubMed Central (PMC).
- Lysine decarboxylase with an enhanced affinity for pyridoxal 5-phosphate by disulfide bond-mediated spatial reconstitution. (n.d.). PubMed Central (PMC).
- Millership, S. E., & Chattopadhyay, B. (1984). Methods for the isolation of Aeromonas hydrophila and Plesiomonas shigelloides from faeces. Journal of Hygiene, 92(2), 145-152.
- Oxford Lab Fine Chem LLP. (n.d.). Technical data sheet: Moeller decarboxylase broth base (decarboxylase broth base, Moeller).
- Patsnap Synapse. (2024, July 17). What is the mechanism of pyridoxal phosphate hydrate?
- Plesiomonas shigelloides and Aeromonadaceae family pathogens isolated from marine mammals of Southern and Southeastern Brazilian coast. (n.d.). PubMed Central (PMC).
- Remel. (2014, June 30). Decarboxylase broth (Moeller) base control, arginine, lysine, and ornithine (IFU 60750).
- Sapkota, A. (2022, January 20). Decarboxylase test: Principle, procedure, results, uses. Microbe Notes.
- Sigma-Aldrich. (2018). 41932 L-Lysine decarboxylation medium (LD medium; lysine decarboxylase broth without peptone). Merck KGaA.
- Sigma-Aldrich. (2018). D2935 decarboxylase broth base, Moeller. Merck KGaA.
- Soderberg, T. (2024, August 24). 17.2: Pyridoxal phosphate (Vitamin B6). In Organic chemistry with a biological emphasis (v2.0). Chemistry LibreTexts.
- The comprehensive microbiology of lysine decarboxylation: Principles, clinical diagnostics, and industrial applications. (n.d.).
- Thermo Fisher Scientific. (2020, May 20). Product specification sheet – lysine decarboxylase broth (Taylor) (Version 06, PS-TV5028N-V6).
- TMMedia. (2019, November 8). TM 2173 – L-lysine decarboxylase saline broth (ISO 21872-1-2017).
- Virtual Microbiology Lab Simulator Software (VUMIE). (2022, November 1). Lysine decarboxylase test.
- Wong, T. Y., Tsui, H. Y., So, M. K., Lai, J. Y., Lai, S. T., Tse, C. W. S., & Ng, T. K. (2000). Plesiomonas shigelloides infection in Hong Kong: Retrospective study of 167 laboratory-confirmed cases. Hong Kong Medical Journal, 6(4), 375-380.