What is Ames Test?
Ames test is a rapid biological assay which is used to determine whether a chemical compound can cause genetic mutations. It was developed by Bruce Ames in 1970s. It is used as an early screening test for carcinogenic property because chemicals causing DNA mutation are frequently carcinogenic.
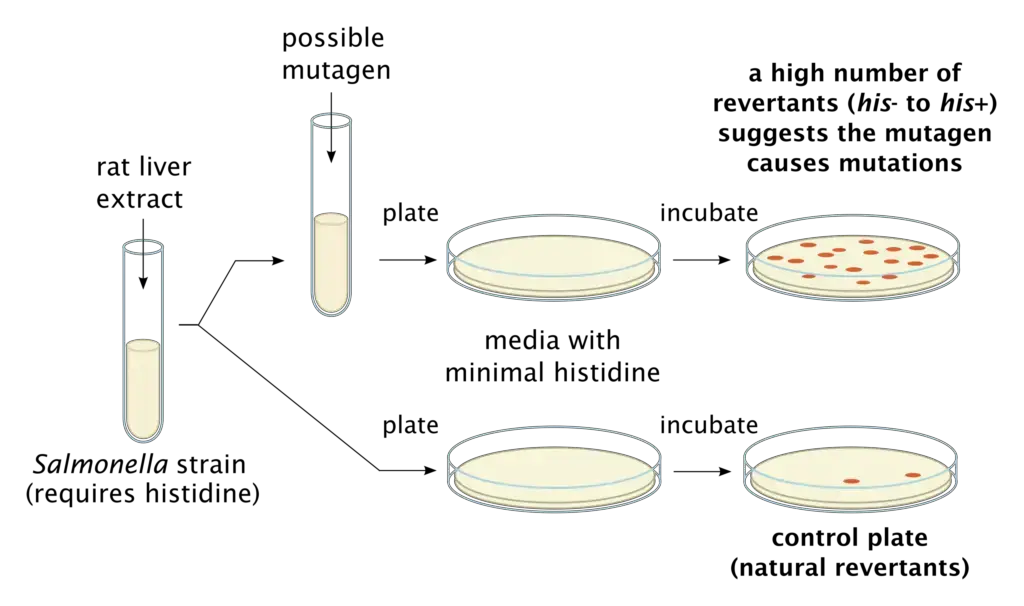
It is based on the fact that mutant strains of bacteria are used which carry a genetic defect and cannot synthesize essential amino acids like histidine or tryptophan. Due to this defect the bacteria cannot grow on a specialized culture medium lacking these nutrients. When bacteria are exposed to a mutagenic chemical the DNA is altered and a reverse mutation is produced which corrects the original defect. This is referred to as reverse mutation. The reverted bacteria can now survive and multiply into visible colonies and the number of colonies formed is directly proportional to the mutagenic strength of the tested chemical.
In this test mutant strains of Salmonella typhimurium or Escherichia coli are taken and plated on minimal medium without histidine or tryptophan. The test chemical is added and incubation is done. If the chemical is mutagenic revertant colonies are formed on the plate. For more accurate condition mammalian liver enzymes (rat liver S9 extract) is added to simulate human metabolism because some chemicals become mutagenic only after being processed by liver.
Definition of Ames Test
The Ames test is a biological assay developed by Bruce Ames, used to assess the mutagenic potential of chemical compounds by observing their ability to induce mutations in specific strains of bacteria, primarily Salmonella typhimurium. A positive result indicates that the chemical may be mutagenic and potentially carcinogenic.
Objectives of Ames test
Objectives of Ames test are as follows-
- It is used to determine whether a chemical compound can induce gene mutation in DNA of test organism.
- It is used as a rapid and inexpensive screening test for potential carcinogens because many mutagens are also carcinogens (approximately 90% mutagens are carcinogenic).
- It is used to evaluate safety of consumer products like cosmetics food additives flavours fragrances and agricultural chemicals so that genetic hazards to humans are not produced.
- It is used in drug discovery for screening new drug candidates in preclinical phase and harmful compounds are eliminated before human trials.
- It is used for fulfilling regulatory requirements and safety assessment protocols required by regulatory bodies (FDA EPA OECD) for marketing new drugs and consumer products.
Principle of Ames Test
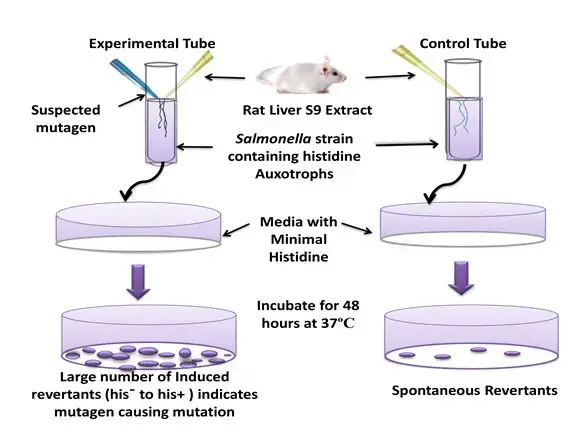
Principle of Ames test is based on the fact that specific mutant strains of bacteria are used which cannot synthesize essential amino acids due to a pre-existing mutation. Salmonella typhimurium mutants require histidine and Escherichia coli mutants require tryptophan. Due to this defect the bacteria cannot grow on minimal agar medium which is lacking histidine or tryptophan.
In this test the bacterial culture is exposed to suspected mutagenic chemical. If the chemical is mutagen it induces a reverse mutation in bacterial DNA and the original defect is corrected. This is referred to as reverse mutation. Due to this the reverted bacteria regain ability to synthesize the amino acid and they can survive and multiply on nutrient deficient medium. Visible colonies are formed on the plate.
Since bacteria do not show mammalian type metabolism liver enzyme extract (rat or hamster S9 fraction) is added to simulate mammalian metabolism. This helps in detecting those chemicals which become mutagenic only after liver processing. Mutagenic activity is determined by counting number of revertant colonies and significant increase in colonies compared to spontaneous background rate indicates the chemical is mutagen.
Strains recommended for use in Ames test
Materials and Reagents Required for Ames Test
- Salmonella typhimurium tester strains (TA98 TA100 TA102 TA1535 TA1537).
- Escherichia coli tester strain (WP2 uvrA).
- Nutrient broth (for overnight culture preparation).
- Vogel-Bonner (VB) medium E (50x salts).
- Minimal glucose agar (MGA) plates (bottom agar).
- Top agar (0.6% agar and 0.5% NaCl).
- 0.5 mM Histidine-Biotin solution (for trace supplementation in top agar).
- Mammalian liver extract (S9 fraction from induced rat or Syrian hamster).
- NADP (nicotinamide adenine dinucleotide phosphate sodium salt).
- D-glucose-6-phosphate.
- MgCl2 KCl and 0.2 M sodium phosphate buffer (pH 7.4) for S9 mix.
- L-histidine and D-biotin (for media preparation).
- Solvents for test chemicals (sterile distilled water DMSO methanol acetone).
- Agar-agar and D-glucose (dextrose).
- Ampicillin trihydrate and tetracycline (for confirming resistance plasmids like pKM101 or pAQ1).
- Crystal violet solution (0.1%) for confirming rfa cell wall mutation.
- 70% ethanol (for sterilization and cleaning).
- Negative control (sterile solvent like water or DMSO).
- Positive control without S9 (sodium azide 2-nitrofluorene 9-aminoacridine mitomycin C).
- Positive control with S9 (2-anthramine or 2-aminoanthracene).
- Sterile petri plates (100×15 mm) and test tubes (13×100 mm).
- Eppendorf tubes (1.5 ml and 2.0 ml).
- Micropipettes and sterile tips.
- L-shaped spreader and metal loop holder.
- Incubator (37°C) and orbital shaking incubator.
- Hot water bath heating block or microwave (to maintain top agar at 43°C–48°C).
- Vortex mixer.
- Laminar flow hood (biosafety cabinet).
- Colony counter (automatic or manual).
- pH meter and analytical balance.
Procedure of Ames Test
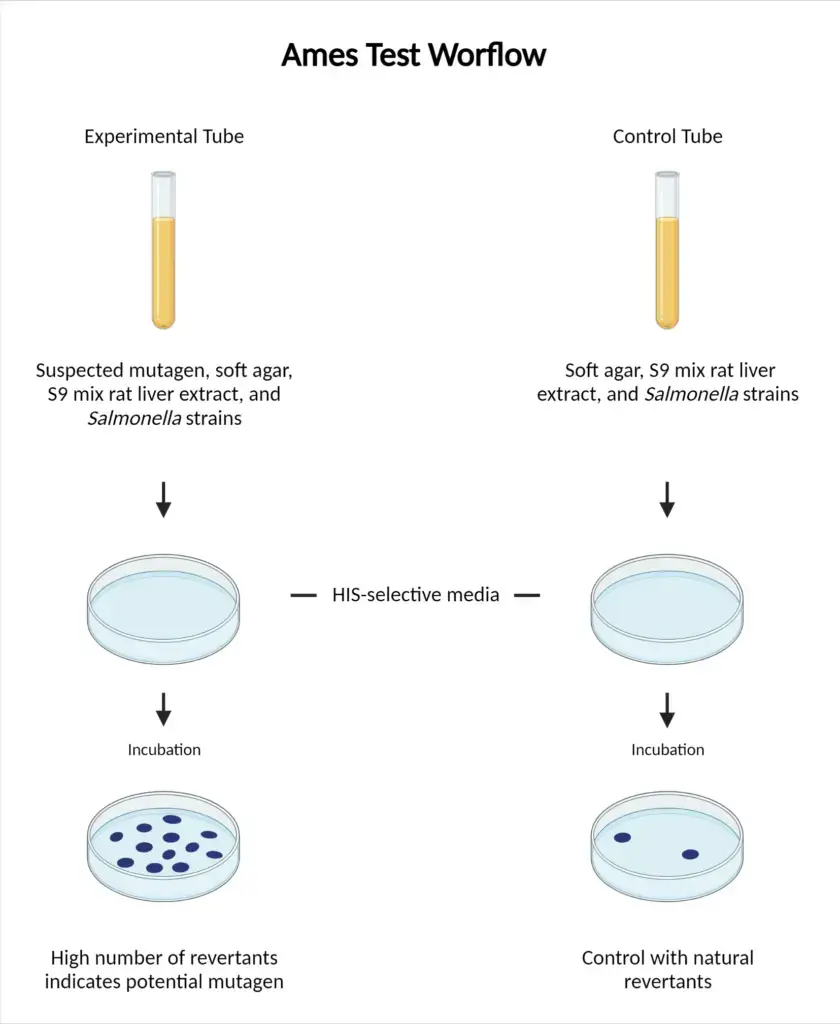
Procedure of Ames test is as follows-
- Tester strains of bacteria (Salmonella typhimurium or Escherichia coli) are grown overnight in nutrient broth to obtain high cell density.
- If metabolic activation is required S9 mixture is prepared using rodent liver extract (rat or hamster) along with enzymatic cofactors.
- In plate incorporation method bacterial culture is mixed with test chemical at different concentrations and S9 mix or buffer is added.
- This mixture is added into molten top agar which is maintained at 43°C–48°C and it contains trace histidine and biotin (or tryptophan for E. coli).
- In pre-incubation method bacteria test chemical and S9 mix (or buffer) are mixed first and incubated at 37°C for 20–30 minutes and then molten top agar is added.
- The top agar mixture is poured evenly on minimal glucose agar plate and it is allowed to solidify.
- Plates are inverted and incubation is done at 37°C for 48–72 hours.
- Negative control plates (solvent) are prepared to check spontaneous mutation background rate.
- Positive control plates are prepared using known mutagens to confirm strains and assay are working.
- After incubation revertant colonies are counted.
- Significant dose dependent increase in colonies as compared to negative control indicates the chemical is mutagenic.
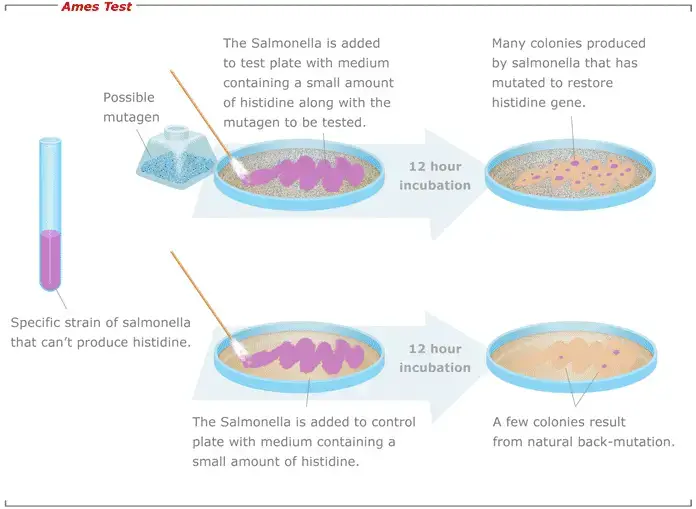
Result of Ames test
Result of Ames test are as follows-
- Positive result (mutagenic)
Significant reproducible and dose related increase in revertant colonies is observed as compared to negative control. In many cases 2 fold or 3 fold increase in colonies is taken as positive depending on strain used. Number of colonies formed is directly proportional to mutagenic strength of the chemical. - Negative result (non-mutagenic)
Very few colonies are present and colony count is almost equal to spontaneous mutation rate of negative control. No dose related increase in colonies is observed in at least two independent experiments. - Weakly positive result (weak mutagen)
Reproducible dose related increase in revertant colonies is obtained but the colony number does not double the background count of control plate. - Inconclusive result
Slightly elevated colony counts are obtained without clear dose response pattern or only a single elevated count is seen. Substance cannot be clearly identified as mutagen or non mutagen and test is repeated or modified. - Toxic result
When chemical concentration is highly toxic thinning or complete absence of background bacterial lawn is seen on plate. Sometimes tiny pinpoint non revertant colonies are also seen due to excess histidine left after bacterial death. It can give false negative result because bacteria are killed before mutation and test is repeated at lower doses.
Uses of Ames Test
Uses of Ames test are as follows-
- It is used in pharmaceutical drug discovery as an early preclinical screening test for new drug candidates active pharmaceutical ingredients and veterinary products and potential carcinogens are eliminated before human trials.
- It is used in pharmaceutical impurity testing to detect DNA reactive mutagenic impurities like N-nitrosamines which may be present due to synthesis process or degradation.
- It is used in consumer products and cosmetics for checking safety of cosmetics flavours fragrances dyes preservatives and UV filters so that genetic mutation are not introduced.
- It is used in food and agriculture for assessing agricultural chemicals pesticides biocides and food additives and genetic hazards to human health are checked. It has been used to identify harmful food colours and antibacterial additives.
- It is used in environmental monitoring to determine mutagenic load and genotoxicity of environmental samples like drinking water wastewater industrial effluents urban smog diesel particulates and hazardous soil leachates.
- It is used for regulatory compliance and it fulfills legal safety assessment requirements required by regulatory bodies like FDA EPA and OECD and it is used under frameworks like Toxic Substances Control Act and Pesticide Act.
- It is used in medical devices testing for determining biocompatibility and safety for consumer use.
- It is used in tobacco testing to measure mutagenic potential of mainstream tobacco smoke and tobacco smoke condensates.
Limitations of Ames test
Limitations of Ames test are as follows-
- It is based on prokaryotic bacterial model (Salmonella typhimurium) and it is not a perfect model for complex eukaryotic human cells.
- Bacteria do not have nuclear membrane and chromatin with histones is absent so mutagens do not cross same type barrier to reach DNA. DNA repair pathways in humans are more sophisticated and redundant as compared to these bacterial strains.
- It detects only DNA reactive mutagens and it cannot detect non genotoxic or epigenetic carcinogens. Some known carcinogens like dioxin benzene asbestos and certain heavy metals act by other mechanisms like chronic inflammation or hormonal imbalance so negative result is obtained in Ames test.
- S9 fraction is used for metabolic activation but rodent and human metabolism is different so assessment of mutagenicity may be altered. Also homogenized S9 mix does not show spatial organization and regulatory control like intact cells.
- False positive and false negative results can be obtained. Safe drugs containing nitrate moieties may generate nitric oxide giving positive result and false negative can occur due to missing extra hepatic metabolic pathways.
- Positive mutagen result in Ames test does not confirm it as carcinogen and further long term animal toxicology and outcome studies are required for proving safety or danger.
Precautions of Ames test
Precautions of Ames test are as follows-
- Gloves laboratory gown and protective eyeglasses should be worn during the test to avoid exposure to hazardous chemicals and bacteria. Safety glasses is preferred and contact lenses should be avoided because volatile chemicals may react with lenses.
- All work should be performed in biological safety cabinet or chemical fume hood because pathogenic bacteria and potential carcinogens are handled. Salmonella strains generally require Bio-Safety Level 2 (BSL-2) laboratory.
- Strict aseptic technique should be maintained to avoid contamination. Petri dishes and test tubes should be opened for minimum time and lids should be held facing downward. Mouth pipetting should never be done.
- Work surfaces and biosafety cabinet should be disinfected before and after experiment. Cleaning is done using 70% ethanol and UV exposure for about 15 minutes is used for proper sterilization. Glassware and plasticware should be cleaned and sterilized before assay.
- If tested chemical are photo reactive non UV lighting should be used or petri dishes should be covered with sterile aluminium foil to prevent photo degradation.
- Test chemicals should be treated as confirmed mutagens and carcinogens. Contaminated waste like pipette tips test tubes gloves and gowns should be collected in biohazard bags and autoclaving should be done before disposal. Unused chemical dilutions and stock solutions should be disposed by proper hazardous waste protocols.
References
- Ames, B. N., Magaw, R., & Gold, L. S. (1987). Ranking possible carcinogenic hazards. Science, 236(4799), 271-280.
- Aryal, S. (2022). Ames Test – Introduction, principle, procedure, uses and result interpretation. MicrobiologyInfo.com.
- Bienta. (2026). Ames Test. Bienta Contract Research Services.
- Bringezu, F., & Simon, S. (2022). Salmonella typhimurium TA100 and TA1535 and E. coli WP2 uvrA are highly sensitive to detect the mutagenicity of short Alkyl-N-Nitrosamines in the Bacterial Reverse Mutation Test. Toxicology Reports, 9, 250-255. https://doi.org/10.1016/j.toxrep.2022.02.005
- Cariello, N. F., & Piegorsch, W. W. (1996). The ames test: The two-fold rule revisited. Mutation Research – Genetic Toxicology, 369(1), 23-31. https://doi.org/10.1016/S0165-1218(96)90044-0
- Carnes, B. A., Dornfeld, S. S., & Peak, M. J. (1985). A quantitative comparison of a percentile rule with a 2-fold rule for assessing mutagenicity in the Ames assay. Mutation Research, 147(1-2), 15-21. https://doi.org/10.1016/0165-1161(85)90013-5
- Carr, S. M. (2026). Ames Test for mutagenicity.
- Creative Proteomics. (2026). What is Ames Test?
- Department of Biology, Davidson College. (2000). Pouring agar plates and spot-overlay Ames test.
- Dr.Oracle Medical Advisory Board & Editors. (2025). Why is rat liver extract, specifically the S9 (supernatant 9) fraction, used in the Ames test?
- Egorova, O. V., & Ilyushina, N. A. (2024). Standardization of criteria for interpreting the results of mutagenicity assessment in the Ames test. Toxicological Review, 32(5), 313-321. https://doi.org/10.47470/0869-7922-2024-32-5-313-321
- Gagwani, D., & Metzler, K. (n.d.). Ames Test | Explanation, purpose & limitations. Study.com.
- Gentronix. (2026). OECD 471 Ames Test (GLP) for reliable genotoxicity assessment.
- Goodson-Gregg, N., & De Stasio, E. A. (2009). Reinventing the Ames Test as a quantitative lab that connects classical and molecular genetics. Genetics, 181(1), 23-31. https://doi.org/10.1534/genetics.108.095588
- Hakura, A., Suzuki, S., & Satoh, T. (1999). Advantage of the use of human liver S9 in the Ames test. Mutation Research, 438(1), 29-36. https://doi.org/10.1016/s1383-5718(98)00159-4
- Health Canada. (2004). Bacterial reverse mutation assay for mainstream tobacco smoke.
- Honma, M. (2025). Guidelines for the assessment and control of mutagenic impurities in pharmaceuticals. Genes and Environment, 47, 26. https://doi.org/10.1186/s41021-025-00349-5
- Inotiv. (2026). Ames Assay.
- Kamber, M., Flückiger-Isler, S., Engelhardt, G., Jaeckh, R., & Zeiger, E. (2009). Comparison of the Ames II and traditional Ames test responses with respect to mutagenicity, strain specificities, need for metabolism and correlation with rodent carcinogenicity. Mutagenesis, 24(4), 359-366. https://doi.org/10.1093/mutage/gep017
- Mortelmans, K., & Zeiger, E. (2000). The Ames Salmonella/microsome mutagenicity assay. Mutation Research, 455(1-2), 29-60. https://doi.org/10.1016/S0027-5107(00)00064-6
- National Research Council (US) Committee on Chemical Environmental Mutagens. (1983). Quantitative relationship between mutagenic and carcinogenic potencies: A feasibility study. National Academies Press (US).
- Neely, C. (2023). 3.4: Ames Test. Biology LibreTexts.
- OECD. (2020). Test No. 471: Bacterial reverse mutation test. OECD Guidelines for the Testing of Chemicals. https://doi.org/10.1787/9789264071247-en
- Prival, M. J., & Zeiger, E. (1998). Chemicals mutagenic in Salmonella typhimurium strain TA1535 but not in TA100. Mutation Research, 412(3), 251-260. https://doi.org/10.1016/s1383-5718(97)00196-4
- Samoveanu, A. (2026). Ames Test. FUJIFILM Wako Pure Chemical Corporation.
- Shaffer, C. (2019). The Ames Test. News-Medical.Net.
- Sharma, D., Mishra, S., Jha, G., Tomar, B., & Rath, S. K. (2026). Bacterial reverse mutation test: Ames test. Methods in Molecular Biology, 2986, 3-15. https://doi.org/10.1007/978-1-0716-4976-3_1
- Sugiyama, K., Yamada, M., Awogi, T., & Hakura, A. (2016). The strains recommended for use in the bacterial reverse mutation test (OECD guideline 471) can be certified as non-genetically modified organisms. Genes and Environment, 38, 2. https://doi.org/10.1186/s41021-016-0030-3
- Thomas, D. N., Wills, J. W., Tracey, H., Baldwin, S. J., Burman, M., Williams, A. N., Harte, D. S. G., Buckley, R. A., & Lynch, A. M. (2023). Ames test study designs for nitrosamine mutagenicity testing: qualitative and quantitative analysis of key assay parameters. Mutagenesis, 39(2), 78-95. https://doi.org/10.1093/mutage/gead033
- Trinova Biochem. (2026). S9 liver extract and metabolic activation.
- Vijay, U., Gupta, S., Mathur, P., Suravajhala, P., & Bhatnagar, P. (2018). Microbial mutagenicity assay: Ames test. Bio-protocol, 8(6), e2763. https://doi.org/10.21769/BioProtoc.2763
- Weiss, C. (2026). Ames mutagenicity testing for safe products. Consumer Product Testing Company.
- Wikipedia contributors. (2025). Ames test. Wikipedia, The Free Encyclopedia.
- Williams, L. R., & Preston, J. E. (1983). Interim procedures for conducting the Salmonella/microsomal mutagenicity assay (Ames test). Environmental Protection Agency.
- Xenometrix. (n.d.). Ames Test – Scientific background.
- Zeiger, E., Mitchell, C. A., Pfuhler, S., Liao, Y., & Witt, K. L. (2024). Within-laboratory reproducibility of Ames test results: are repeat tests necessary?. Environmental and Molecular Mutagenesis, 65(3-4), 116-120. https://doi.org/10.1002/em.22597