What is Methyl Red (MR) Test?
The Methyl Red (MR) test is a biochemical test used in microbiology to identify and differentiate bacteria mainly among Enterobacteriaceae family. It is done to check the glucose fermentation pathway. It is used for detecting mixed-acid fermentation which produces stable acidic end products.
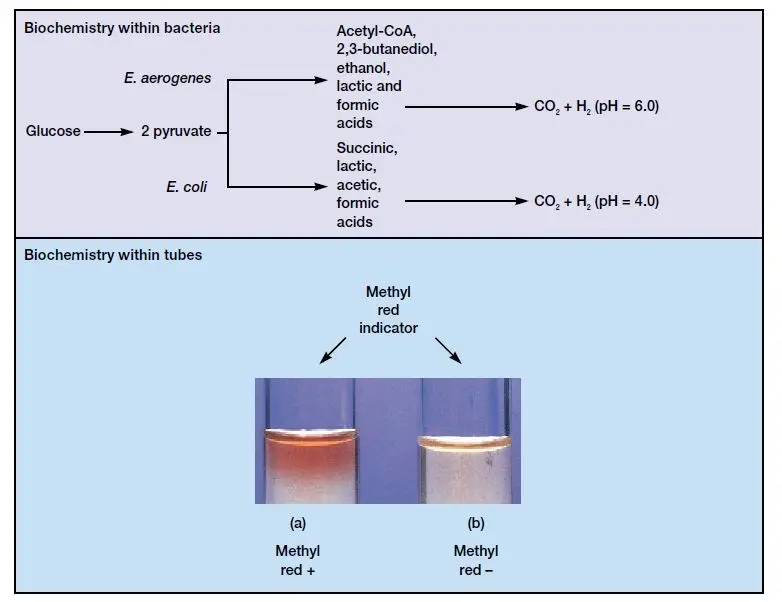
In this test the organism is grown in glucose rich MR-VP broth for minimum 48 hours. After incubation methyl red indicator is added in culture. Methyl red is red in pH 4.4 or below. It is turned to yellow at pH 6.0 or above.
If the broth is changed to distinct red colour then MR test is positive. It is indicated that large amount of stable acids are produced (lactic acid acetic acid formic acid succinic acid). These acids is enough to overcome buffer of medium and pH is lowered. This is commonly seen in Escherichia coli.
If the broth is changed to yellow colour then MR test is negative. It is indicated that stable acids are not produced in large amount and pH is not lowered much. In this case glucose is fermented by other pathway and neutral end products is formed. This is commonly seen in Enterobacter aerogenes and Klebsiella pneumoniae.
Objectives of Methyl Red (MR) Test
Objectives of Methyl Red (MR) test are–
- It is used to determine that bacterial organism has ability to produce and maintain stable acid end products by mixed-acid fermentation of glucose.
- It is used to differentiate major types of facultative anaerobic enteric bacteria among Enterobacteriaceae family based on acid production level.
- It is used to distinguish members of coli-aerogenes group such as Escherichia coli from Enterobacter species and Klebsiella species.
- It is used for characterization and identification of unknown bacterial species by analyzing metabolic pathway and fermentation pathway.
Principle of Methyl Red test
Principle of Methyl Red (MR) test is based on detection of mixed-acid fermentation of glucose by bacteria. In this test glucose is fermented and stable acidic end products are produced. It is used to differentiate organisms which produces large amount of stable acids from those which produces neutral end products.
In this process bacteria are grown in a glucose containing medium known as MR-VP broth. The glucose is metabolized to pyruvic acid. After this pyruvic acid is converted to stable organic acids such as lactic acid acetic acid formic acid and succinic acid. Due to formation of these acids the phosphate buffer of medium is overcome and pH is lowered to 4.4 or below.
After incubation methyl red indicator is added in broth. Methyl red changes colour depending on pH. If the pH is 4.4 or below it remains red and it is referred to as positive MR test. If the pH is higher and acids are not produced in large amount it is turned to yellow and it is referred to as negative MR test.
Requirements for Methyl Red (MR) test
Media and reagents required for Methyl Red (MR) test are–
- MR-VP broth (Methyl Red-Voges Proskauer broth). It is also known as buffered glucose broth or glucose phosphate broth. It is used as growth medium and it contains glucose peptone and dipotassium phosphate buffer.
- Methyl red indicator solution. It is used as pH indicator reagent and it is added after incubation for detection of stable acidic end products.
- Quality control organisms. These are known bacterial cultures used for checking media and reagents are working properly. Escherichia coli is used as positive control. Enterobacter aerogenes or Klebsiella pneumoniae is used as negative control.
Equipments required are–
- Inoculating loop or wire. It is used to transfer light inoculum aseptically from pure culture to broth.
- Loop sterilization device (incinerator or Bunsen burner). It is used for sterilizing the inoculating loop before and after use.
- Incubator. It is used to maintain temperature for growth (35°C–37°C) during incubation. The incubation period is minimum 48 hours and it may be extended up to 5 days.
- Clean test tubes. It is used to take small portion of incubated broth (about 1 mL or 2.5 mL) so that methyl red reagent can be added without destroying original culture.
- Transfer pipettes. It is used to transfer incubated broth from original tube to clean test tube.
- Test tube rack. It is used to hold test tubes upright and undisturbed during incubation and testing process.
Procedure of Methyl Red test
Procedure of Methyl Red (MR) test are–
- MR-VP broth (glucose phosphate broth) is allowed to equilibrate to room temperature before inoculation.
- A light inoculum is taken from pure 18–24 hours bacterial culture by sterile inoculating loop. The MR-VP broth is inoculated aseptically. Heavy inoculum is avoided because it can inhibit growth and result may be invalid.
- The inoculated MR-VP broth is incubated aerobically at 35°C to 37°C for minimum 48 hours. Testing before 48 hours is avoided because false positive result may be obtained and MR negative organisms may not converted initial acidic products to neutral end products.
- After 48 hours incubation a portion of broth (about 1 mL to 2.5 mL or half of the tube content) is transferred to a clean test tube by transfer pipette. The remaining broth is kept for VP test or for prolonged incubation if result is not clear.
- Methyl red indicator reagent is added directly to the aliquoted broth. Usually 2 to 6 drops are added (it may be up to 10 drops depending on protocol and volume).
- The tube is shaken gently to mix the contents and colour change is observed immediately on surface of medium.
- If the result is equivocal or weak positive (orange colour) then remaining original broth is incubated further up to total 5 days and test is repeated.
Result of Methyl Red test
Result of Methyl Red (MR) test are–
- Positive result (red or pink colour)– A distinct stable red colour is obtained. It is indicated that bacteria has used mixed-acid fermentation pathway and large amount of stable acidic end products are produced. Due to this pH of medium is dropped to 4.4 or below.
- Negative result (yellow colour)– Yellow colour is obtained. It is indicated that stable mixed acids are not produced and neutral end products are formed (often by butanediol pathway). In this case pH of medium remains higher (about 6.0 to 6.2 or above).
- Weak positive or equivocal result (orange or red-orange colour)– Orange colour is obtained and it is considered as inconclusive or weak positive. It can occur when organism is slow acid producer or test is done early before fermentation is completed. In this case broth is incubated further for 2 to 3 days and test is repeated.

List of Methyl Red Positive and negative Organisms
List of Methyl Red (MR) positive and negative organisms are–
Methyl Red positive organisms– These organisms produces stable acidic end products by mixed-acid fermentation and pH is lowered to 4.4 or below. The methyl red indicator is turned to red.
- Escherichia coli
- Proteus species (Proteus mirabilis, Proteus vulgaris)
- Salmonella species (Salmonella typhi)
- Shigella species (Shigella flexneri)
- Citrobacter species (Citrobacter freundii)
- Yersinia species (Yersinia enterocolitica)
- Morganella species
- Providencia species
- Edwardsiella species
Methyl Red negative organisms– These organisms usually uses 2,3-butanediol fermentation pathway and neutral end products are formed. The pH is maintained 6.0 or above and methyl red indicator remains yellow.
- Enterobacter species (Enterobacter aerogenes)
- Klebsiella species (Klebsiella pneumoniae, Klebsiella aerogenes)
- Serratia species (Serratia marcescens)
- Hafnia alvei (when it is grown at 30°C)
Exceptions (MR and VP both positive)– In general MR and VP test shows inverse relation but some organisms can show both positive.
- Proteus myxofaciens
- Enterobacter intermedium
- Klebsiella planticola
- Serratia liquefaciens
Limitation of Methyl Red test
Limitation of Methyl Red (MR) test are–
- Incubation time is required minimum 48 hours and sometimes it may be extended up to 5 days. If the result is read too early then false positive may be obtained because MR negative organisms may not converted initial acidic products to neutral end products.
- Inoculum density affects the result. Heavy inoculum can inhibit bacterial growth and invalid result may be obtained.
- MR test alone cannot identify bacteria definitively. It is required to be used with other biochemical tests for accurate identification.
- Broth volume and aeration can affect the test. If large volume broth is used in narrow test tubes then oxygen is restricted. MR negative organisms requires oxygen for shifting to butanediol pathway so poor aeration can give orange colour or false result.
- The test depends on proper glucose and phosphate buffer concentration (usually 0.5% each). High glucose can give false positive by producing excess acids and low glucose may not produce enough acids for overcoming buffer so true positive may not be obtained.
- MR and VP test generally shows inverse relation but some organisms can show both positive which makes interpretation difficult (Proteus mirabilis Enterobacter hafnia Serratia liquefaciens Klebsiella planticola).
Quality Control of Methyl Red (MR) Test
Quality control organisms for Methyl Red (MR) test are–
- Positive quality control organism (expected result– red colour)
- Escherichia coli (ATCC 25922)
- Negative quality control organisms (expected result– yellow colour)
- Klebsiella pneumoniae (ATCC 13883 or ATCC 27736)
- Enterobacter aerogenes or Klebsiella aerogenes (ATCC 13048)
- Enterobacter cloacae (ATCC 23355)
Uses of Methyl Red (MR) Test
Uses of Methyl Red (MR) test are–
- It is used for detecting mixed-acid fermentation. It is done to determine that organism can ferment glucose and stable acidic end products are produced.
- It is used to differentiate Enterobacteriaceae family organisms. It is also used for characterization of other bacterial groups such as Actinobacteria.
- It is used to distinguish Escherichia coli from other enteric bacteria. Escherichia coli is MR positive and Klebsiella-Enterobacter group is MR negative.
- It is used in water and food safety testing. It is used for coliform analysis in water raw materials and processed foods for detection of E. coli which indicates food spoilage or recent fecal contamination.
- It is used for identification of human pathogens. It helps in detection of organisms causing intestinal infections (Salmonella Shigella Yersinia) and it is also used in oral infections and some kidney diseases.
References
- Alfa Chemistry. (n.d.). CAS 493-52-7 Methyl red.
- American Society for Microbiology. (2009). Methyl Red and Voges-Proskauer Tests. Image Gallery.
- Aryal, S. (2022). API (Analytical Profile Index) 20E Test – Procedure, Uses and Interpretation. MicrobiologyInfo.com.
- Aryal, S. (2022). Methyl Red (MR) Test- Principle, Procedure and Result Interpretation. MicrobiologyInfo.com.
- Aryal, S. (2022). Methyl Red (MR) Test- Principle, Procedure, Results, Uses. Microbe Notes.
- Barry, A. L., Bernsohn, K. L., Adams, A. P., & Thrupp, L. D. (1970). Improved 18-hour methyl red test. Applied Microbiology, 20(6), 866–870. https://doi.org/10.1128/am.20.6.866-870.1970
- bioMérieux. (2002). API 20 E: Identification system for Enterobacteriaceae and other non-fastidious Gram-negative rods.
- bioMérieux. (2026). VITEK® 2 | Pioneering Diagnostics.
- Catalanotti, C., Yang, W., Posewitz, M. C., & Grossman, A. R. (2013). Fermentation metabolism and its evolution in algae. Frontiers in Plant Science, 4(150). https://doi.org/10.3389/fpls.2013.00150
- Creative Enzymes. (n.d.). Biochemical-Based Identification System.
- Dahal, P. (2026). Enterobacteriaceae: Characteristics, Identification. Microbe Notes.
- Dalynn Biologicals. (2014). Methyl Red Reagent.
- DrChika. (2023). Methyl red test. Microbiology Class.
- Funke, G., Monnet, D., deBernardis, C., von Graevenitz, A., & Freney, J. (1998). Evaluation of the VITEK 2 System for Rapid Identification of Medically Relevant Gram-Negative Rods. Journal of Clinical Microbiology, 36(7), 1948–1952. https://doi.org/10.1128/jcm.36.7.1948-1952.1998
- Hardy Diagnostics. (2020). Methyl Red-Voges Proskauer (MR-VP) Broth.
- Hartline, R. (2023). 1.27: MR-VP Tests. Biology LibreTexts.
- HiMedia Laboratories. (n.d.). MR-VP Broth (Glucose Phosphate Broth) LQ082.
- Hinsu, M. (n.d.). Methyl red test(mr test). SlideShare.
- Lenahan, M., & Salas, M. (2014). API 20E Microbial Identification SOP. Montgomery County Community College. Biomanufacturing.org.
- MacsChem. (n.d.). Methyl-red | 493-52-7.
- Martinez-Urtaza, J., Lozano-Leon, A., Viña-Feas, A., de Novoa, J., & Garcia-Martin, O. (2006). Differences in the API 20E biochemical patterns of clinical and environmental Vibrio parahaemolyticus isolates. FEMS Microbiology Letters, 255(1), 75–81. https://doi.org/10.1111/j.1574-6968.2005.00052.x
- McDevitt, S. (2009). Methyl Red and Voges-Proskauer Test Protocols. American Society for Microbiology.
- MedChemExpress. (n.d.). Methyl red | Azo Dye.
- Merck KGaA. (2018). 39484 Methyl Red Voges Proskauer Broth (MR VP Broth; Buffered Glucose Broth).
- Merck. (n.d.). MR-VP Broth (Methyl-red VOGES-PROSKAUER Broth). Merck Microbiology Manual 12th Edition.
- Microxpress. (n.d.). Methyl Red Indicator. Tulip Diagnostics.
- O’Hara, C. M., & Miller, J. M. (2003). Evaluation of the Vitek 2 ID-GNB Assay for Identification of Members of the Family Enterobacteriaceae and Other Nonenteric Gram-Negative Bacilli and Comparison with the Vitek GNI+ Card. Journal of Clinical Microbiology, 41(5), 2096–2101. https://doi.org/10.1128/JCM.41.5.2096-2101.2003
- Randomknow99. (n.d.). Methyl Red Test: Principle and Results. Scribd.
- Rao, S. P. N. (n.d.). MR-VP tests. Microrao.
- Raymond, J., Boorse, G., & Mason, A. (2022). Identification of Enterobacteriaceae. In Red Mountain Microbiology. Maricopa Open Digital Press.
- Study.com. (n.d.). Could an organism be both MR and VP positive?
- ThermoFisher (Remel). (2012). Methyl Red.
- University of Windsor. (n.d.). Methyl Red.
- Unknown Author. (n.d.). The Methyl Red Test: Principles, Biochemical Mechanisms, and Diagnostic Utility in Microbiology.
- Vivijs, B., Haberbeck, L. U., Baiye Mfortaw Mbong, V., Bernaerts, K., Geeraerd, A. H., Aertsen, A., & Michiels, C. W. (2015). Formate hydrogen lyase mediates stationary-phase deacidification and increases survival during sugar fermentation in acetoin-producing enterobacteria. Frontiers in Microbiology, 6(150). https://doi.org/10.3389/fmicb.2015.00150
- Wikipedia contributors. (2025). Analytical profile index. In Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (2025). Methyl red. In Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (2025). Mixed acid fermentation. In Wikipedia, The Free Encyclopedia.
- Williams, P. M., & Foist, L. (n.d.). Methyl Red Test Definition, Purpose & Results. Study.com.
Thank you very much, Sourav.
Most Welcome,…stay with us.