X-Ray Crystallography is an experimental technique used to know the three dimensional arrangement of atoms and molecules inside a crystal.
It is used for studying the exact position of atoms. It also shows the chemical bonds and the total molecular structure present in the crystal.
In this method, a focused beam of X-rays is passed on a crystalline sample. The X-rays interact with electron clouds of atoms present inside the crystal.
As the atoms in crystal are arranged in a highly ordered and repeating pattern, the X-rays are scattered in some fixed directions. This scattering of X-rays is known as diffraction.
The diffracted X-ray beams are measured by their angles and intensities. These measured values are used for mathematical calculation to make a three dimensional electron density map.
This map shows the position of individual atoms in the crystal. It also helps to know the bonding pattern and overall molecular architecture of the molecule.
X-Ray crystallography is important in structural biology, drug design and materials engineering. It gives atomic level information of the crystal structure.
Principles and Workings of X-Ray Crystallography
X-Ray crystallography is based on the scattering of X-rays by the electron clouds of atoms present inside a crystalline sample.
The wavelength of X-rays is almost similar to the distance between atoms. Due to this, the regular and repeating arrangement of atoms in a crystal behaves like a three dimensional grating.
When an X-ray beam falls on the crystal, the X-rays are scattered by the atoms. Most of the scattered waves cancel each other by destructive interference.
But in some specific directions, the scattered waves combine together. This is called constructive interference. It produces definite diffraction spots.
These diffraction spots are like a unique pattern of the crystal. The position of these spots depends on the arrangement of atoms in the crystal lattice.
This condition of diffraction is explained by Bragg’s Law. It relates the wavelength of X-rays, angle of incidence and the distance between the parallel atomic planes of the crystal.
Working of X-Ray Crystallography
The working of X-Ray crystallography starts with crystallization of the target molecule.
In this step, the molecule is converted from disordered solution into a highly ordered solid crystal. This is the most difficult part of the process because a proper crystal is needed for good diffraction.
After crystallization, the crystal is mounted on a precise mechanical stage called goniometer. Then the crystal is exposed to a strong and focused beam of X-rays.
The goniometer rotates the crystal in different directions. During this process, the X-rays are diffracted from the crystal lattice.
An electronic detector records the angles and intensities of the diffracted X-rays. Many two dimensional diffraction images are collected from different positions of the crystal.
After data collection, computational data processing is done. The diffraction pattern is related to the actual structure of the crystal by Fourier transform.
The detector can record the intensity of scattered waves but it cannot record the phase information. This is called phase problem.
The phase information is recovered by using mathematical methods or experimental chemical methods. After this, the phases are used to prepare a three dimensional electron density map.
This electron density map shows where atoms are present in the molecule. The atomic model is then built, fitted and refined again and again to get the correct structure of the molecule.
What is Diffraction?
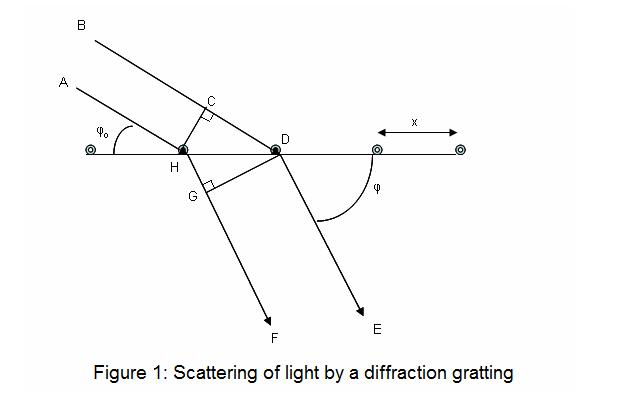
Diffraction is a physical phenomenon which occurs when waves interact with a regularly spaced structure or grating.
It takes place when the spacing of the structure is nearly similar to the wavelength of the incident radiation. The waves may be visible light, X-rays or other type of radiation.
When the waves strike such structure, they are scattered. These scattered waves then interfere with one another.
In X-Ray crystallography, the crystal itself acts as a three dimensional optical grating. This is because the distance between atoms in a crystal is almost similar to the wavelength of X-rays.
When the X-ray beam falls on the atoms, the electron clouds of atoms start to oscillate. These electron clouds then re-emit X-rays in all directions. This process is called elastic scattering.
The final result of scattering depends on the exact arrangement of atoms inside the crystal. In most directions, the scattered waves are out of phase and cancel each other. This is called destructive interference.
But in some specific directions, the scattered waves come in same phase. These waves combine with each other and become stronger. This is called constructive interference.
Constructive interference produces a concentrated and detectable beam of radiation. This beam is known as diffraction spot or reflection.
The condition for formation of diffraction spot is explained by Bragg’s Law. It relates the wavelength of X-rays, the angle of incidence and the spacing between atomic planes.
The arrangement and intensity of diffraction spots forms a unique pattern for the crystal. By measuring these spots, the position of atoms can be calculated.
Fourier transform and other mathematical methods are used for this calculation. Finally, a three dimensional electron density map is prepared, which shows the exact position of atoms inside the material.
What is Bragg’s Law?
Bragg’s Law is a mathematical equation that explains the condition needed for diffraction of X-rays by atomic planes of a crystal.
It was developed by Lawrence Bragg in 1912. It is used to relate the X-ray wavelength, angle of incidence and the distance between parallel planes of atoms.
The equation of Bragg’s Law is as follows-
nλ = 2d sinθ
Where,
n = order of reflection.
λ = wavelength of incident X-rays.
d = distance between the parallel atomic planes.
θ = angle of incidence.
Bragg’s Law is based on constructive interference of scattered X-ray waves. When X-rays strike the crystal, they are scattered from different atomic planes.
For diffraction to occur, the scattered waves must reinforce each other. This happens only when the path difference between the waves is equal to whole number multiple of wavelength.
If this condition is not fulfilled, the scattered waves cancel each other. So diffraction does not occur in all directions.
Diffraction occurs only at some fixed angles. These angles are known as Bragg angles.
The diffraction spots produced at these angles form a unique pattern of the crystal. This pattern is used to calculate the distance between atoms and to determine the three dimensional structure of the material.
Components of X-Ray Crystallography
The following are the important components of X-Ray crystallography-
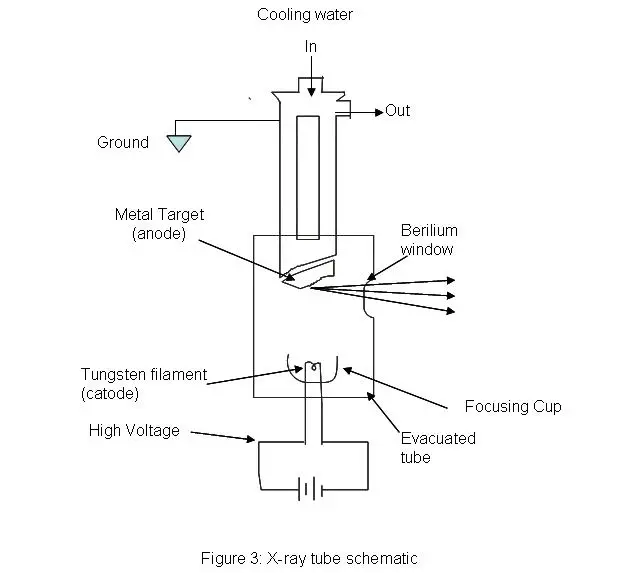
- X-ray Source– X-ray source is used to generate the X-rays which are used to illuminate the sample. It may be a laboratory vacuum tube, high-brilliance synchrotron or X-ray free-electron laser (XFEL). In laboratory vacuum tube, cathode and metal anode are present, and copper or molybdenum is commonly used as anode.
- X-ray Optics and Beam Conditioning– X-ray optics and beam conditioning system is used to refine the raw X-ray beam before it reaches the sample. It contains different parts such as monochromator, collimator, Soller slits, divergence and receiving slits, beta-filters, beam attenuator and beamstop.
- a. Monochromator is used to select a single constant wavelength of X-rays. It may use curved graphite or silicon crystal.
- b. Collimator is used to focus the X-rays into a narrow and parallel beam by using lead slits or polycapillary optics.
- c. Soller slits are parallel metal plates used to limit axial divergence of the beam and improve the shape of diffraction peaks.
- d. Divergence and receiving slits are used to control equatorial divergence of the beam and improve resolution of the diffractometer.
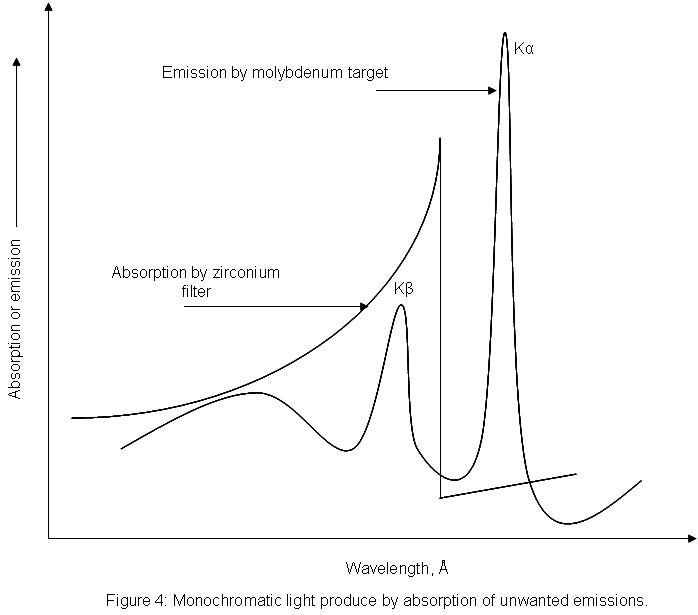
- e. Beta-filters are used to suppress unwanted Kβ radiation from the X-ray tube.
- f. Beam attenuator contains absorbing foils such as copper or nickel which reduce the intensity of strong primary beams.
- g. Beamstop is a small lead or tungsten disk used to block the primary beam and prevent over-saturation or damage of detector.
- Sample Handling and Positioning– Sample handling and positioning system is used to hold the crystal sample and place it properly in the X-ray beam. It includes goniometer, sample holders and cryosystem.
- a. Goniometer is a high precision mechanical stage which holds the sample exactly in the centre of X-ray beam and rotates it through precise angles.
- b. Sample holders are used to hold the crystal. Nylon loops are used for cryo-crystallography, glass or quartz capillaries are used for air-sensitive samples and flat plate holders are used for powder diffraction.
- c. Cryosystem is a cooling device which maintains biological or sensitive crystals at cryogenic temperature, generally about 100 K, by using a constant stream of cold nitrogen gas. It helps to reduce radiation damage.
- Detector– Detector is used to count the photons and measure the position and intensity of diffracted X-rays. Modern systems use Charge-Coupled Devices (CCDs) or Hybrid Photon Counting (HPC) detectors such as PILATUS and EIGER. Other detectors include scintillation counters, proportional counters and older photographic film.
Step by Step procedure of X-Ray Crystallography
The following are the steps of X-Ray crystallography-
- Crystallization– The target molecule is first converted from solution into a pure and highly ordered single crystal. This step is very important because diffraction depends on the proper crystal formation. It is also the most difficult and time taking step of the whole process.
- Sample mounting– The crystal formed is collected and fixed on a precise mechanical stage called goniometer. It is usually mounted by using a small nylon loop or glass capillary. The crystal is often flash frozen and kept at cryogenic temperature to protect it from damage by X-rays.
- Data collection– The mounted crystal is placed in the path of a focused and monochromatic X-ray beam. The goniometer rotates the crystal in small steps. During this process, the detector records many two dimensional diffraction patterns from different orientations.
- Data processing– The raw diffraction images are analysed by computational software. In this step, the reflections are indexed to find the unit cell dimensions and symmetry of the crystal. The intensity of each diffracted spot is also measured.
- Solving the phase problem– The detector records only the intensity of scattered X-rays but phase angles are lost. This missing phase information is recovered by computational or experimental methods. Direct methods, molecular replacement and anomalous dispersion are used for this purpose.
- Electron density map generation– After the phases are estimated, they are combined with the measured intensities. Then Fourier transform is applied. This produces a three dimensional electron density map of the crystal.
- Model building and refinement– A preliminary atomic model is prepared by fitting atoms, chemical groups or amino acids into the electron density map. Then the model is refined by computer. In this step, atomic positions are adjusted again and again to reduce the difference between observed data and calculated data.
- Validation and deposition– The final structure is checked properly. It is validated to see whether bond lengths, bond angles and other chemical rules are correct or not. After verification, the three dimensional structure is deposited into public database such as Protein Data Bank for scientific use.
What is Fourier Transform?
Fourier Transform is a mathematical process which connects two functions having reciprocal dimensions.
It is a reversible mathematical process. When Fourier transform is done two times in succession, the original function can be obtained again.
In X-Ray crystallography, Fourier transform gives the relation between the physical structure of the crystal and the diffraction pattern produced by it.
The physical structure of crystal means the electron density present inside the crystal. The diffraction pattern is produced when X-rays are scattered by this electron density.
Forward Fourier transform represents the actual diffraction experiment. It shows how electron density of the crystal gives rise to the diffraction pattern.
Reverse Fourier transform is used as a computational calculation. It recombines the diffracted waves and reconstructs a three dimensional electron density map of the molecule.
For reverse Fourier transform, amplitude and phase of the diffracted X-ray waves are needed. The amplitude is obtained from the intensity of diffraction spots, but phase angles are missing and must be determined.
Fourier transform is also used in Nuclear Magnetic Resonance (NMR) spectroscopy. In NMR, it converts recorded radio frequency signals into readable NMR spectra.
Applications of X-Ray Crystallography
The following are the applications of X-Ray crystallography-
- Biological and macromolecular studies– X-Ray crystallography is used to determine the three dimensional atomic structure of biological molecules such as DNA, RNA, proteins and viruses. It is also used to study large molecular machines like ribosome.
- Pharmaceutical research and drug design– It is used in structure based drug design (SBDD). It helps to see the active binding pockets of target proteins. It is also used to improve lead compounds and to know how drugs inhibit enzymes in diseases like cancer and sickle-cell disease.
- Battery technology and energy storage– It is used for real time study of battery materials. It helps to observe phase transition, lattice expansion or contraction and degradation of electrode materials during charging and discharging process. It is commonly used in lithium-ion battery studies.
- Materials science and engineering– It is used to determine atomic arrangement of different materials. It also helps to study grain orientation, crystallite size and film thickness of alloys, semiconductors, thin films and newly synthesized inorganic compounds.
- Enzyme mechanisms and dynamic studies– It is used to study fast enzyme reactions and their intermediate structural states. Ultra-fast X-ray free-electron lasers (XFELs) are used for this purpose. It helps to capture molecular changes during the reaction.
- Metal-Organic Frameworks (MOFs)– It is used to verify the complex structure and pore environment of Metal-Organic Frameworks. These porous materials are used in carbon capture, gas storage and low-dose medical imaging detectors.
- Mineralogy and earth sciences– It is used to identify and study minerals, crystals and rocks. It helps to know the atomic structure of naturally occurring minerals. It is also useful in soil property study and mineral morphology analysis for oil and gas extraction.
Advantages of X-Ray Crystallography
The following are the advantages of X-Ray crystallography-
- High atomic level resolution– X-Ray crystallography gives highly precise atomic level resolution. It helps to see the exact position and arrangement of atoms present inside a molecule.
- No upper size limit– This technique has no upper molecular weight limit. It can be used for small inorganic compounds and also for large biological molecules such as membrane proteins, water soluble proteins and ribosomes.
- Gives structural and functional information– It gives detailed information about molecular interactions, binding sites and conformational changes. These informations are useful for understanding biological mechanisms and structure based drug design.
- Fast analysis after crystal formation– After a good quality crystal is formed, the data collection and computational processing can be done quickly. The structural analysis also becomes faster after proper crystal growth.
- Non-destructive and accurate method– It is a non-destructive analytical technique. It is also highly accurate and precise for finding unknown molecular structures.
- Well established technique– X-Ray crystallography is a mature and developed technique. Atomic models can be built and refined from electron density maps with the help of computational tools.
Limitations of X-Ray Crystallography
The following are the limitations of X-Ray crystallography-
- Crystallization problem– X-Ray crystallography needs a high quality and well ordered single crystal. This step is difficult and takes long time. It has high chance of failure, mainly for large, flexible or membrane associated proteins.
- Static structure– This technique usually gives a fixed snapshot of a molecule inside the crystal lattice. The structure may be affected by crystal packing forces. So it may not always show the natural movement and flexibility of the molecule in fluid condition.
- Phase problem– The detector records only the intensity or amplitude of diffracted X-rays. The phase angles are lost during detection. So the missing phase information must be obtained by mathematical or experimental methods before making electron density map.
- Radiation damage– The strong X-ray beam can damage sensitive biological samples. It may produce free radicals from water and destroy the crystal. Sometimes the crystal is damaged before enough high resolution data is collected.
- Crystal imperfections– Real crystals may contain defects which make structure determination difficult. Twinning and disorder are common problems. In twinning, different crystal domains overlap with each other. In disorder, atoms may vibrate more or occupy different positions, which affects accurate data processing.
References
- The transformative impact of AI-enabled AlphaFold 3: evolution, current status, and future prospects in structural biology. (n.d.). ResearchGate.
- A brief history of macromolecular crystallography, illustrated by a family tree and its Nobel fruits. (n.d.). PubMed Central (PMC).
- A bright future for serial femtosecond crystallography with XFELs. (n.d.). PubMed Central (PMC).
- Shimabukuro, J. (2025, December 13). AI Scientific Research Innovations (Dec. 2025). Educational Technology and Change Journal.
- Advancements in X-ray Crystallography: An Exhaustive Analysis of Structural Elucidation from Classical Diffraction to Femtosecond Laser Science. (n.d.).
- Advances in cryo-electron microscopy (cryoEM) for structure-based drug discovery. (n.d.). PubMed Central (PMC).
- Iowa State University Chemical Instrumentation Facility. (n.d.). Basics of X-Ray Crystallography.
- CCD vs CMOS: A Review of Sensor Technology. (n.d.).
- Parkin, S. (2019). Combined Twinning and Disorder. University of Kentucky X-Ray Crystallography Facility.
- Creative Biostructure. (n.d.). Comparison of X-ray Crystallography, NMR and EM.
- Wikipedia. (2026, March 23). Crystal twinning.
- Crystallography of metal–organic frameworks. (n.d.). PubMed Central (PMC).
- Martín García, J. M. (n.d.). Crystallography. The revolution of XFELs.
- Martín García, J. M. (n.d.). Crystallography. The revolution of XFELs.
- Förster, A., Brandstetter, S., Müller, M., & Schulze-Briese, C. (2016). EIGER detectors in biological crystallography [White paper]. DECTRIS Ltd..
- Calvo, C. (2025, September 22). FIU acquires new tool to study life and death struggles of batteries. FIU News.
- High-resolution protein modeling through Cryo-EM and AI: current trends and future perspectives – a review. (n.d.). PubMed Central (PMC).
- Nobel Prize Outreach. (2026). How X-rays and crystals revealed the true nature of things. NobelPrize.org.
- Brok, E. S. (n.d.). Identification of crystalline materials with X-Ray Diffraction (XRD). FORCE Technology.
- Identifying Structure and Texture of Metal–Organic Framework Cu2(bdc)2(dabco) Thin Films by Combining X‑ray Diffraction and Quantum Mechanical Modeling. (n.d.). PubMed Central (PMC).
- Wallen, J. R. (2020). Integrative Structural Biology: Using X-ray Crystallography, Small-Angle X-ray Scattering, and Cryogenic Electron Microscopy to Determine Protein Structures. ACS Symposium Series, 1357, 31-56.
- Bétard, A., & Fischer, R. A. (2012). Metal–Organic Framework Thin Films: From Fundamentals to Applications. Chemical Reviews, 112(2), 1055-1083.
- Salway, H., Chua, X. W., & Anaya, M. (2025). Metal–organic frameworks as potential materials for X-ray detectors: recent progress and unique opportunities. Materials Horizons, 12, 8989-9008.
- Overview of X-Ray Diffraction Systems. (n.d.). Labcompare.com.
- Watanabe, N. (2010). Practical crystal mounting method for the longer wavelength SAD phasing [Presentation]. Synchrotron Radiation Research Center, Nagoya University.
- Properties of X-rays. (n.d.).
- Yin, T., Chen, Y., Wang, Y., Su, H., Duan, C., & Liu, J. (2026). Protein structure prediction powered by artificial intelligence: from biochemical foundations to practical applications. Frontiers in Molecular Biosciences, 13, 1767821.
- Spectrum Logic. (n.d.). Remembering Lawrence Bragg and his contribution to the development of X-ray Crystallography.
- Bocarsly, J. (2025, August 6). Revealing Atomic-Level Battery Material Structures with Electron Diffraction [Webinar]. Rigaku.
- Cockcroft, J. K. (2006). Sample Holders. Birkbeck College, University of London.
- Serial Femtosecond Crystallography (SFX) combined with an X-ray Free Electron Laser (XFEL) for Determination of Protein Structure with Reduced Sample Volume. (n.d.). Spectroscopy Online.
- Sygnature Discovery. (n.d.). Structural Biology Techniques Compared: X-ray, NMR & Cryo-EM.
- Robas, R. (n.d.). Synchrotrons and X-ray free-electron lasers: How they work, what they do, and where they’re headed [Video]. YouTube.
- Ball, P. (n.d.). THE BIRTH OF CRYSTALLOGRAPHY. The Astbury Centre.
- Critchley, L. (2020, October 16). The Applications & Principles of X-Ray Crystallography. AZoM.
- The transformative impact of AI-enabled AlphaFold 3: evolution, current status, and future prospects in structural biology. (n.d.). PubMed Central (PMC).
- OlexSys Ltd. (2026). Twinned Structures.
- Watkin, D. (1997, November 12). Twins. Chemical Crystallography.
- Rigaku. (n.d.). Webinar Summary: X-ray Diffraction Measurements for Battery Research.
- Nuhfer, E. B. (2023). X-Ray Crystallography Is Developed by the Braggs. EBSCO Research Starters.
- X-Ray Crystallography of Chemical Compounds. (n.d.). PubMed Central (PMC).
- X-Ray Diffraction Systems. (n.d.).
- Clegg, W. (2015). X-ray Crystallography (2nd ed.). Oxford University Press.
- Technology Networks. (n.d.). X-ray Crystallography.
- Malvern Panalytical. (n.d.). X-ray Diffraction (XRD) – Overview.
- Wang, N., Zhang, X., Tan, S., Lee, S., & Hu, E. (2025). X-ray Diffraction Studies of Single-Crystal Materials for Broad Battery Applications. Chemical Reviews, 125(20).
- Wikipedia. (2026, April 16). X-ray crystallography.
- X-ray imaging detectors for synchrotron and XFEL sources. (n.d.). PubMed Central (PMC).
- Liang, Y., Zhu, J.-Z., Jin, S.-Y., Meng, Y.-R., Li, S.-F., Zuo, J.-L., Zhang, G., & Su, J. (2025). X-ray luminescent metal–organic frameworks: design strategies and functional applications. Journal of Materials Chemistry C, 13, 4836-4860.
- x Ray crystallography. (n.d.). PubMed Central (PMC).