OF (Oxidation-Fermentation) test is a biochemical test used to find out how a bacterium utilize carbohydrate (mainly glucose). It was developed by Hugh and Leifson (1953). It differentiates organisms into fermentative type and oxidative type based on acid production in anaerobic and aerobic condition.
It is done in a semi-solid OF medium having high carbohydrate to peptone ratio, with a pH indicator (bromothymol blue). Two tubes are inoculated with same organism. One tube is kept open (aerobic) and the second tube is covered with sterile mineral oil or liquid paraffin (anaerobic) to block oxygen.
Fermentative bacteria produce acid in both tubes, so both tubes change colour from green to yellow. Oxidative bacteria produce acid only in open tube, so only open tube becomes yellow and the oil covered tube remains green. Non-saccharolytic bacteria do not use the sugar, so no yellow colour is seen and the open tube may turn blue due to alkaline products from protein breakdown.
Objectives of OF Test
- To determine the mode of carbohydrate utilization (glucose) by the test organism, either oxidative or fermentative.
- To detect acid production from carbohydrate in aerobic condition and anaerobic condition and interpret the pattern.
- To differentiate bacteria into oxidative group, fermentative group and non-saccharolytic group (no sugar utilization).
- To differentiate glucose fermenting Enterobacteriaceae from non-fermenting aerobic gram negative bacilli (Pseudomonas, Acinetobacter, Stenotrophomonas).
- To differentiate Staphylococcus species (fermentative) from Micrococcus species (oxidative or non-reactive) by using OF medium modification.
- To study general metabolic pathway of carbohydrate utilization in clinical and environmental isolates.
Principle of OF (Oxidation–Fermentation) Test
OF (Oxidation–Fermentation) test principle is based on the ability of bacteria to break down glucose either by oxidation (in presence of oxygen) or by fermentation (in absence of oxygen). The organism is inoculated in Hugh-Leifson medium in two conditions, aerobic and anaerobic. Acid formed during carbohydrate utilization is detected by pH indicator.
It is based on a special medium having high carbohydrate (about 1%) and low peptone (about 0.2%). Oxidative organisms produce only small amount of weak acids, so peptone content is kept low to prevent alkaline amines from protein breakdown which can neutralize the acid. The medium is semi-solid because agar helps to hold and concentrate acid near the site of reaction.
The pH indicator (bromothymol blue) is used. When acid is produced the colour changes from green to yellow. One tube is kept open for oxygen supply and one tube is covered with sterile mineral oil to make anaerobic condition. According to yellow colour in open tube or both tubes, oxidative and fermentative type is identified, and if no acid then it remains green or open tube may turn blue due to alkaline products.
Requirements for OF (Oxidation–Fermentation) Test
- Hugh and Leifson OF (basal) medium
- Peptone/tryptone (low concentration)
- Sodium chloride
- Dipotassium hydrogen phosphate (buffer)
- Agar (semi-solid)
- Bromothymol blue (pH indicator)
- Carbohydrate (glucose mostly. lactose/sucrose/maltose also used). final about 1%
- Sterile test tubes (two tubes per isolate)
- Sterile inoculating wire/needle (for stab)
- Sterile mineral oil/liquid paraffin (for anaerobic tube overlay). oil is sterilized by dry heat
- Incubator (35–37°C)
- Autoclave (for basal medium)
- Basic lab supplies (burner, balance, pipette, marker, PPE)
- Fresh pure culture of test organism
- Control strains
- Fermentative control. Escherichia coli
- Oxidative control. Pseudomonas aeruginosa
- Negative (non-saccharolytic) control. Alcaligenes faecalis
Hugh and Leifson’s OF basal medium (H-L medium)
Composition (per litre)
- Carbohydrate (glucose/dextrose) 10.0 g
- Peptone (peptic digest of animal tissue) 2.0 g
- Sodium chloride (NaCl) 5.0 g
- Dipotassium hydrogen phosphate (K2HPO4) 0.3 g
- Agar 2.0 to 3.0 g
- Bromothymol blue 0.03 to 0.05 g
- Final pH 6.8 ± 0.2 (at 25°C)
Preparation
- Weigh the ingredients and suspend in 1 litre distilled water.
- Heat with mixing and boil to dissolve agar and medium properly.
- If carbohydrate is heat stable (glucose), dispense about 5 ml in test tubes (duplicate tubes).
- Sterilize by autoclave at 121°C (15 psi) for 15 minutes.
- If carbohydrate is heat labile, autoclave basal medium without sugar first. Cool to about 40–45°C. Add filter sterilized 10% sugar solution aseptically to make final 1% carbohydrate. Then dispense into sterile tubes.
- Keep tubes upright and allow to cool and solidify in vertical position.
Procedure of OF (Oxidation–Fermentation) Test
- Take a fresh pure culture of the test organism.
- Label two OF medium tubes for the same organism. One is aerobic tube and one is anaerobic tube.
- Using sterile inoculating wire/needle, stab inoculate the organism straight down into the medium (deep stab, near bottom but not touching, around 0.5 cm to 3 cm from bottom). Do same for both tubes.
- Add sterile mineral oil/liquid paraffin in one tube to form about 1 cm to 2.5 cm layer over the medium. This tube becomes anaerobic (oil sealed).
- Keep the second tube without oil. Cap is kept loose or tube is left open to air for aerobic condition.
- Incubate both tubes at 35–37°C.
- Observe after 18–48 hours for colour change (green to yellow).
- If no change, continue incubation and check daily. Negative tubes are kept up to 14 days for slow growers or late fermenters.
Results of OF (Oxidation-Fermentation) Test

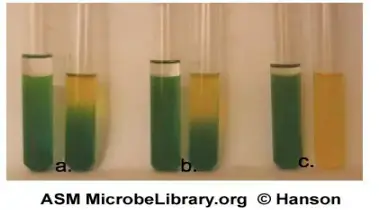

Fermentative (F)
Both tubes show acid production. The open tube turns yellow and the oil sealed tube also turns yellow. This indicates fermentation of carbohydrate in presence and absence of oxygen.
Oxidative (O)
Only the open tube turns yellow. The oil sealed tube remains green. This indicates carbohydrate is utilized only aerobically and acid is formed only in oxygen.
Non-saccharolytic (Negative / Non-reactive)
No acid is produced from carbohydrate. The oil sealed tube remains green and the open tube also remains green or it may turn blue due to alkaline products from peptone utilization.
Baird-Parker modification (for gram positive cocci)
- Fermentative.
- Both tubes change from purple to yellow.
- Oxidative.
- Only open tube turns yellow and sealed tube remains purple.
- Negative.
- Both tubes remain purple (no acid).
List of organisms in OF (Oxidation–Fermentation) test (positive and negative)
Fermentative (positive in both tubes)
- Escherichia coli
- Klebsiella pneumoniae
- Enterobacter aerogenes (Klebsiella aerogenes)
- Enterobacter cloacae
- Salmonella Typhi
- Salmonella Enteritidis
- Shigella sonnei
- Shigella flexneri
- Vibrio cholerae
- Staphylococcus aureus (Baird-Parker modification)
Oxidative (positive only in open tube. sealed tube negative)
- Pseudomonas aeruginosa
- Burkholderia cepacia
- Acinetobacter baumannii
- Acinetobacter anitratus
- Stenotrophomonas maltophilia (often weak/slow for glucose)
- Flavobacterium meningosepticum
- Micrococcus luteus (Baird-Parker modification)
Non-saccharolytic / Non-reactive (negative in both tubes)
- Alcaligenes faecalis
- Acinetobacter lwoffii
- Moraxella spp.
- Branhamella (Moraxella) catarrhalis
Quality control strains of OF (Oxidation–Fermentation) test
Gram-Negative Bacilli:
- Escherichia coli (ATCC 25922 / NCTC 10418 / NCTC 12241). Fermentative control. Both open tube and oil sealed tube turns yellow.
- Pseudomonas aeruginosa (ATCC 27853 / NCTC 10662 / NCTC 12903). Oxidative control. Open tube turns yellow and oil sealed tube remains green.
- Alcaligenes faecalis. Negative (non-saccharolytic) control. Sealed tube remains green and open tube remains green or may turn blue (alkaline).
- Acinetobacter lwoffii (NCTC 5866). Negative control. No reaction in both tubes (green).
- Branhamella (Moraxella) catarrhalis (ATCC 25240). Negative control for different sugars. No oxidation or fermentation, both tubes shows no yellow.
Gram-Positive Cocci (when using the Baird-Parker modification):
- Staphylococcus aureus (NCTC 6571 / NCTC 12973). Fermentative control. Both tubes turn yellow (from purple to yellow).
- Micrococcus luteus (NCTC 2665). Oxidative control. Only open tube turns yellow and sealed tube remains purple.
Precautions of OF (Oxidation–Fermentation) Test
- Carbohydrate solution should be sterile before adding in basal medium. For heat labile sugars, basal medium is autoclaved first, cooled and then filter sterilized sugar is added aseptically.
- Mineral oil/liquid paraffin should be sterilized by dry heat only (about 160°C to 170°C for 2 hours). Do not autoclave oil because steam do not penetrate properly and moisture may spoil the anaerobic seal.
- Use non-acidic mineral oil. Acidic oil can give false yellow colour in sealed tube.
- Inoculation should be done by straight stab with sterile wire/needle. Stab is taken deep (near bottom but not touching, about 0.5 cm to 3 cm from bottom).
- Oil layer should be sufficient (about 1 cm to 2.5 cm) to make strict anaerobic condition. The second tube must be kept open/loose cap for oxygen.
- Do not discard negative tubes early. Some organisms are late fermenters and may need prolonged incubation up to 14 days.
- Starting pH of medium should be proper (around 6.8). If medium becomes too alkaline, weak acid of oxidative reaction may be masked and false negative result can come.
Uses of OF (Oxidation–Fermentation) Test
- It is used to determine the carbohydrate utilizing process of bacteria, oxidative or fermentative or non-saccharolytic.
- It is used for identification and classification of gram negative bacteria by biochemical character.
- It is used to differentiate Enterobacteriaceae (fermenters) from non-fermenting aerobic gram negative bacilli like Pseudomonas, Acinetobacter and Stenotrophomonas.
- It is used to differentiate gram positive cocci, mainly Staphylococcus (fermentative) from Micrococcus (oxidative).
- It is used in clinical lab to detect non-fermenters which are often hospital associated and drug resistant, so it helps in selecting proper antibiotic treatment.
Advantages of OF (Oxidation–Fermentation) Test
- It is highly sensitive for detecting weak acids of oxidative metabolism because medium has high carbohydrate and low peptone ratio, so alkaline products do not mask the acid.
- It gives easy visual reading because semi-solid agar holds the acid near the site of reaction and colour change is seen clearly.
- Bromothymol blue indicator is used which shows early acid production, so small pH shift can be detected.
- Motility can also be seen in same tube because medium is semi-solid, hazy growth spreading away from stab line indicates motility.
- It helps in proper classification and identification of bacteria, like separating Enterobacteriaceae (fermenters) from non-fermenting gram negative rods, and also Staphylococcus from Micrococcus.
Limitations of OF (Oxidation–Fermentation) Test
- OF test alone cannot confirm the exact organism. It is only a screening biochemical test. Other tests are needed for final identification.
- Standard OF basal medium may not support fastidious organisms properly. Some organisms need extra supplements like sterile serum or yeast extract for good growth.
- Some bacteria are late fermenters. They produce acid very slowly and may need prolonged incubation up to 14 days. If tubes are discarded early it gives false negative.
- Mineral oil problem can occur. If oil is acidic it can lower pH and sealed tube may become yellow giving false positive fermentative result.
- False negative can occur by alkaline masking. If initial pH is too alkaline, or peptone is more, or organism grows slow, weak oxidative acid can be neutralized and colour change is not seen.
- Single sugar may not be enough. Some organisms oxidize only specific carbohydrates, so glucose OF may be negative but other sugar may be positive. So different sugars may be required.
References
- Advanced analysis of the oxidative-fermentative (OF) test: Biochemical principles, taxonomic utility, and clinical diagnostic application. (n.d.)..
- Alcantara, S. Y. (n.d.). Nonfermenting gram-negative bacilli overview [PDF document]. Scribd..
- Antimicrobial susceptibility pattern of Burkholderia cepacia complex & Stenotrophomonas maltophilia from North India: Trend over a decade (2007-2016). (n.d.). PubMed Central (PMC)..
- Central Drug House (P) Ltd. (n.d.). Technical information – Hugh Leifson medium (DM 1826)..
- Chawla, K., Vishwanath, S., & Munim, F. C. (2013). Nonfermenting gram-negative bacilli other than Pseudomonas aeruginosa and Acinetobacter spp. causing respiratory tract infections in a tertiary care center. Journal of Global Infectious Diseases, 5(4), 144–148. https://doi.org/10.4103/0974-777X.121996.
- Conway, T. (1992). The Entner-Doudoroff pathway: History, physiology and molecular biology. FEMS Microbiology Reviews, 103, 1–28..
- Dahal, P. (2023, March 6). OF test- Oxidation/oxidative-fermentation/fermentative test. Microbe Notes..
- Epidemiology and outcomes of Stenotrophomonas maltophilia and Burkholderia cepacia infections among trauma patients of India: A five year experience. (n.d.). PubMed Central (PMC)..
- Gautam, V., Ray, P., Vandamme, P., Chatterjee, S. S., Das, A., Sharma, K., Rana, S., Garg, R. K., Madhup, S. K., Mahajan, M., & Sharma, M. (2009). Identification of lysine positive non-fermenting gram negative bacilli (Stenotrophomonas maltophilia and Burkholderia cepacia complex). Indian Journal of Medical Microbiology, 27(2), 128–133. https://doi.org/10.4103/0255-0857.49425.
- Glycolytic strategy as a tradeoff between energy yield and protein cost. (n.d.). PubMed Central (PMC)..
- Hanson, A. (2008, September 1). Oxidative-fermentative test. American Society for Microbiology..
- Hanson, A. (2008, September 8). Oxidative-fermentative test protocol. American Society for Microbiology..
- Hardy Diagnostics. (2020). Oxidative-fermentative medium (OF basal medium) [Instructions for use]..
- HiMedia Laboratories. (2017). Hugh Leifson medium (M826) [Technical data sheet]..
- HiMedia Laboratories. (2025). Hugh Leifson medium..
- HiMedia Laboratories. (2026). Hugh Leifson glucose medium (M871) [Technical data sheet]..
- HiMedia Laboratories. (2026). OF basal medium (M395) [Technical data sheet]..
- Nonfermenting gram-negative bacilli other than Pseudomonas aeruginosa and Acinetobacter spp. causing respiratory tract infections in a tertiary care center. (n.d.). PubMed Central (PMC)..
- One Health assessment of antimicrobial-resistant Enterobacterales and ESKAPE pathogens in little stints (Calidris minuta) and aquatic ecosystems of the Kenyan Rift Valley. (n.d.). bioRxiv..
- PatSnap Eureka. (2025, September 2). Autoclave vs. dry heat: Best sterilization method for lab equipment..
- Public Health England. (2014, March 11). Identification of glucose non-fermenting gram negative rods (UK Standards for Microbiology Investigations ID 17, Issue 2.2)..
- Public Health England. (2019, January 16). Oxidation/fermentation of glucose test (UK Standards for Microbiology Investigations TP 27, Issue 4)..
- Sandle, T. (2015, March 2). Oxidation-fermentation test explained. Pharmaceutical Microbiology Resources..
- Sigma-Aldrich. (n.d.). Hugh Leifson medium (H8282) [Datasheet]..
- The difference between a dry heat sterilizer and a steam sterilizer autoclave. (n.d.). Duraline Systems..
- The Entner-Doudoroff pathway is an essential metabolic route for Methylotuvimicrobium buryatense 5GB1C. (n.d.). PubMed Central (PMC)..
- Thong, M. L. (1977). Differentiation of nonfermentative gram-negative bacilli in the clinical laboratory. Southeast Asian Journal of Tropical Medicine and Public Health, 8(1), 7–12..
- TM Media. (2019, November 8). TM 125 – Hugh Leifson medium [Product data sheet]..
- Varun, C. N. (2016, October 24). Lab series# 15: Biochemical tests for identification of bacterial isolates. Microboids..
- Vegge, C. S., Jansen van Rensburg, M. J., Rasmussen, J. J., Maiden, M. C. J., Johnsen, L. G., Danielsen, M., MacIntyre, S., Ingmer, H., & Kelly, D. J. (2016). Glucose metabolism via the Entner-Doudoroff pathway in Campylobacter: A rare trait that enhances survival and promotes biofilm formation in some isolates. Frontiers in Microbiology, 7, Article 1877. https://doi.org/10.3389/fmicb.2016.01877.
- Wikipedia contributors. (2026, April 8). Citrobacter freundii. In Wikipedia, The Free Encyclopedia..