Novobiocin Susceptibility Test is a laboratory test used to determine whether a bacterium is sensitive or resistant to novobiocin antibiotic. It is mainly used in clinical microbiology for identification of coagulase negative staphylococci (CoNS). It is used to differentiate Staphylococcus saprophyticus from other CoNS like Staphylococcus epidermidis.
In this test, a novobiocin impregnated paper disc is placed on an agar plate that is uniformly streaked with the isolated bacteria. The antibiotic diffuses into the medium. Susceptible bacteria do not grow near the disc and a clear zone of inhibition is formed around the disc.
Staphylococcus saprophyticus is intrinsically resistant and it continues to grow close to the disc. It shows a small zone of inhibition usually 16 mm or less. This is used as a quick presumptive test to identify S. saprophyticus which is a common cause of uncomplicated urinary tract infection in young sexually active women.
Objectives of Novobiocin Susceptibility Test
- To determine the susceptibility of bacterial isolate against novobiocin antibiotic.
- To identify Staphylococcus saprophyticus presumptively from urine culture samples, as this organism is resistant to novobiocin.
- To differentiate Staphylococcus saprophyticus from other coagulase negative staphylococci which are sensitive to novobiocin.
- To classify coagulase negative staphylococci into novobiocin sensitive group and novobiocin resistant group based on their reaction.
Principle of Novobiocin Susceptibility Test
Principle of Novobiocin Susceptibility Test is based on the difference in susceptibility and resistance shown by different bacteria to novobiocin. Novobiocin is an aminocoumarin antibiotic produced by Streptomyces niveus. It acts by binding to bacterial DNA gyrase and blocks the ATPase activity. DNA unpackaging and repackaging required for bacterial cell replication is inhibited.
In this test, a novobiocin impregnated paper disc is placed on an agar plate heavily seeded with the test organism. The antibiotic diffuses into the agar. Susceptible organism do not grow near the disc and a clear zone of inhibition is formed around the disc.
Resistant organism continues to grow and no significant clear zone is formed. Most coagulase negative staphylococci like Staphylococcus epidermidis are susceptible. Staphylococcus saprophyticus shows natural resistance due to structural modification in its DNA gyrase. This resistance pattern is used to differentiate S. saprophyticus from other CoNS and normal skin flora.
Requirements for Novobiocin Susceptibility Test
The following are the requirements for performing novobiocin susceptibility test-
- Culture media
Mueller-Hinton agar (MHA) / 5% sheep blood agar (BAP) / Tryptic soy agar (TSA).
Tryptic soy broth (TSB) / sterile distilled water / Brain heart infusion (BHI) broth for preparing bacterial suspension. - Reagents
Novobiocin antibiotic disc (5 µg).
McFarland turbidity standard (0.5 or 1.0).
Defibrinated sheep blood (if blood agar plates are prepared in the laboratory). - Equipment
Incubator (aerobic) at 35–37°C.
Sterile cotton swabs and inoculating loop for streaking.
Sterile forceps for placing the novobiocin disc.
Metric ruler or sliding calipers to measure zone of inhibition.
Other lab supplies like sterile test tubes, petri plates, loop sterilization device or Bunsen burner, autoclave, weighing machine, micropipette and PPE. - Test organisms
Pure culture of the isolated test organism (18–72 hours growth).
Quality control strains such as Staphylococcus aureus (ATCC 25923), Staphylococcus saprophyticus (ATCC 15305) and Staphylococcus epidermidis (ATCC 12228).
Procedure of Novobiocin Susceptibility Test
The test can be done by two methods.
A. Plate method (Disc diffusion method)
- Preparation of inoculum
Pure culture (18–72 hours) is taken. Few colonies are mixed in sterile TSB / sterile water / BHI broth. The turbidity is adjusted to 0.5 McFarland standard. - Inoculation of plate
A sterile cotton swab is dipped in the suspension. Excess fluid is removed by pressing on the tube wall. The agar plate (MHA / blood agar / TSA) is streaked evenly in three directions to make a lawn culture. - Drying
The plate is allowed to dry for few minutes (not more than 15 minutes). - Placement of disc
Novobiocin disc (5 µg) is placed on the agar surface using sterile forceps. The disc is pressed gently so it makes proper contact with agar. - Incubation
The plate is incubated aerobically at 35–37°C for 16–24 hours. - Reading of result
Zone of inhibition around the disc is observed. The diameter of the zone is measured in mm using ruler or caliper.
B. Rapid tube method (Broth disc method)
- Preparation of tubes
Two test tubes with 3 ml TSB are taken. Both tubes are lightly inoculated with well isolated colonies. Broth should look almost clear initially. - Addition of disc
One tube is added with a novobiocin disc (5 µg) and shaken for about 10 seconds. Second tube is kept without disc as growth control. - Incubation
Both tubes are incubated at 35–37°C up to 5 hours. - Reading of result
Incubation is stopped when control tube becomes turbid equal to 0.5 McFarland. Turbidity of disc tube is compared with control tube and growth is observed.
Result of Novobiocin Susceptibility Test
The following are the results of novobiocin susceptibility test.
Plate method (Mueller Hinton agar)
Sensitive- Zone of inhibition is more than 16 mm.
Resistant- Zone of inhibition is 16 mm or less.
Plate method (Blood agar)
Sensitive- Zone of inhibition is 12 mm or more.
Resistant- Zone of inhibition is less than 12 mm.
Tube method (Broth method)
Sensitive- Turbidity in tube containing novobiocin disc is less than the control tube.
Resistant- Turbidity in tube containing novobiocin disc is equal to or more than the control tube.
Interpretation
Resistant result indicates Staphylococcus saprophyticus (especially from urine culture).
Sensitive result indicates other coagulase negative staphylococci, commonly Staphylococcus epidermidis.
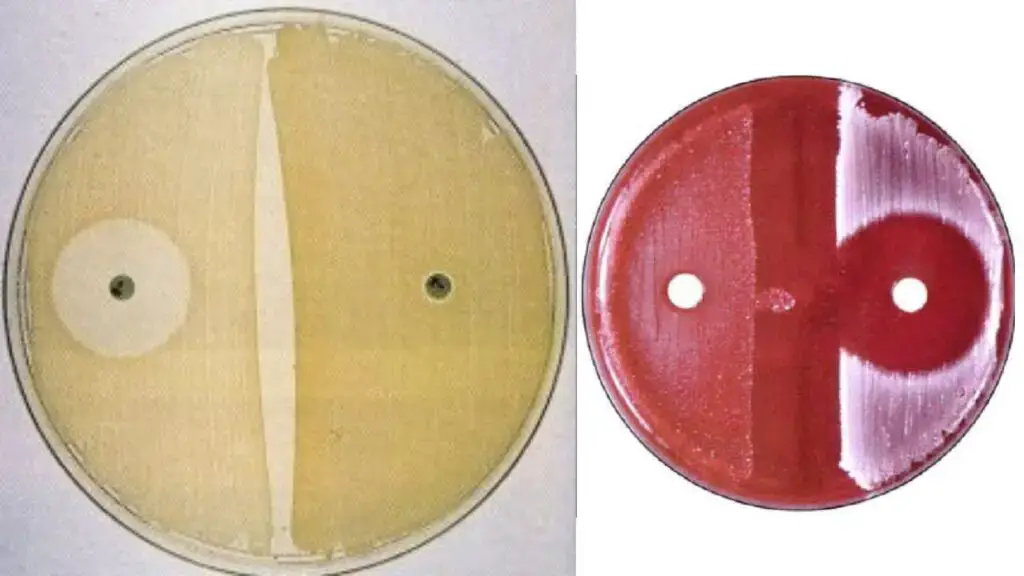
Organisms Showing Positive and Negative Result
In the novobiocin susceptibility test, a positive result indicates resistance to the antibiotic, while a negative result indicates sensitivity (susceptibility).
Here are the organisms categorized by their typical test results:
Positive Result (Novobiocin-Resistant Staphylococci)
- Staphylococcus saprophyticus (the primary human pathogen identified by this test)
- Staphylococcus cohnii
- Staphylococcus xylosus
- Staphylococcus kloosii
- Staphylococcus pulvereri
- Staphylococcus sciuri
- Staphylococcus arlettae
- Staphylococcus equorum
- Staphylococcus gallinarum
- Staphylococcus nepalensis
- Staphylococcus succinus
- Staphylococcus vitulinus
- Staphylococcus fleuretti
Negative Result (Novobiocin-Sensitive/Susceptible Staphylococci)
- Staphylococcus epidermidis
- Staphylococcus haemolyticus
- Staphylococcus hominis (including subsp. hominis)
- Staphylococcus capitis
- Staphylococcus warneri
- Staphylococcus lugdunensis
- Staphylococcus saccharolyticus
- Staphylococcus aureus (A coagulase-positive species that is sensitive to novobiocin and frequently used as a quality control organism for the test)
Uses of Novobiocin Susceptibility Test
The following are the uses of novobiocin susceptibility test.
- To presumptively identify Staphylococcus saprophyticus from other coagulase negative staphylococci (CoNS) in clinical specimen.
- It is used in urine culture to differentiate S. saprophyticus (uropathogen) from CoNS skin contaminants.
- It is used to divide CoNS into novobiocin susceptible group and novobiocin resistant group.
- It is used to rapidly differentiate Staphylococcus epidermidis (large clear zone) from Staphylococcus saprophyticus (growth near disc).
- It is used to categorize CoNS species like S. epidermidis, S. haemolyticus, S. hominis as susceptible and S. cohnii, S. xylosus, S. kloosii as resistant.
Advantages of Novobiocin Susceptibility Test
The following are the advantages of novobiocin susceptibility test.
- It is a simple test and easy to perform in routine laboratory.
- It is cost effective and does not require expensive instruments.
- It is highly useful for presumptive identification of Staphylococcus saprophyticus from urine culture.
- It helps to differentiate pathogen from skin flora like Staphylococcus epidermidis, so unnecessary broad spectrum antibiotic use can be avoided.
- Rapid tube method gives result within few hours (about 5 hours) as compared to overnight plate method.
- It can be used as a supportive confirmation test when other identification method gives unclear result.
- Result interpretation is simple and visual, zone of inhibition on plate or turbidity in tube.
Limitation of Novobiocin Susceptibility Test
The following are the limitations of novobiocin susceptibility test.
- It is mainly reliable for presumptive identification of Staphylococcus saprophyticus from urine isolates. If it is done from other body sites, result may be misleading.
- It gives only presumptive identification. Further confirmatory tests are needed for final identification.
- It is culture based test, so time is required for growth and incubation. Media, chemicals and proper incubation condition is needed.
- False positive or false negative result can occur if procedure is not followed properly. Wrong inoculum density, old disc, improper agar depth and excess moisture can affect the zone size.
- Other coagulase negative staphylococci like S. cohnii and S. xylosus can also show novobiocin resistance, so confusion can occur.
- It should be performed only on pure isolated colonies and organism should be confirmed as aerobic, catalase positive and coagulase negative Gram positive cocci before doing the test.
Quality Control
The quality control strains used to validate the novobiocin susceptibility test include:
- Staphylococcus aureus ATCC 25923: Expected to show a sensitive (susceptible) result with a large zone of inhibition.
- Staphylococcus saprophyticus ATCC 15305: Expected to show a resistant result.
- Staphylococcus saprophyticus ATCC 13518: Expected to show a resistant result.
- Staphylococcus epidermidis ATCC 12228: Expected to show a sensitive (susceptible) result.
- Staphylococcus epidermidis ATCC 14990: Expected to show a sensitive (susceptible) result.
Precautions of Novobiocin Susceptibility Test
The following are the precautions.
- Aseptic technique should be followed. Media should be sterilized properly. PPE should be used.
- Pure isolated culture should be used. Mixed culture gives wrong result.
- Blood agar preparation. Blood should not be added above 45°C. Media should be poured before it falls below 40°C. Air bubbles should be avoided. Agar depth should be about 4 mm.
- Agar plate should be solidified at room temperature (about 6 hours). It should not be kept in refrigerator for solidification.
- Plate surface should be dried properly. Lid can be kept slightly open to avoid water droplets.
- Inoculum density should match McFarland standard. Too heavy or too light inoculum changes zone size.
- After inoculation, plate should be dried before disc placement (not more than 15 minutes). Excess moisture causes uneven diffusion.
- Novobiocin discs should be stored at -20°C to +8°C and protected from light. Disc should not be used if desiccant turns blue to pink.
- Disc should be placed with sterile forceps and pressed gently for full contact. Disc should be kept away from edge (about 25 mm).
- Incubation should be done aerobically. CO2 incubation should be avoided.
References
- Amer Al-Jawabreh. (n.d.). Novobiocin test- Coagulase Negative Staphylococci (S. epidermidis and S. saprophyticus) [Video]. YouTube.
- Clinical and molecular analysis of the novobiocin susceptibility test: A definitive guide for diagnostic microbiology. (n.d.).
- Coagulase-negative, novobiocin-resistant staphylococci … – SciSpace. (n.d.).
- Coagulase-negative, novobiocin-resistant staphylococci on the skin of animals and man, on meat and in milk – PMC. (n.d.).
- Condalab. (n.d.). Novobiocin 30 µg Cat. 7305. BioTrading.
- Dahal, P. (2023, March 6). Novobiocin susceptibility test- Principle, procedure, results. Microbe Notes.
- Dalynn Biologicals. (2014, October). Novobiocin disks.,
- Five-hour novobiocin test for differentiation of coagulase-negative staphylococci – PMC. (n.d.).
- Hardy Diagnostics. (2020). Novobiocin differentiation disks – Instructions for use.,
- InformationBoxTicket Lifestyles. (n.d.). Test for novobiocin susceptibility: Principles, methods & results | Biochemical tests – Microbiology [Video]. YouTube.
- Intrinsic novobiocin resistance in Staphylococcus saprophyticus – PMC – NIH. (n.d.).
- MBBS NAIJA. (n.d.). Novobiocin susceptibility test laboratory method used to identify coagulase negative staphylococci [Video]. YouTube.
- Namavar, F., De Graaff, J., De With, C., & Maclaren, D. M. (1978). Novobiocin resistance and virulence of strains of Staphylococcus saprophyticus isolated from urine and skin. Journal of Medical Microbiology, 11(3), 243. https://doi.org/10.1099/00222615-11-3-243,
- Nobrega, D. B., Naushad, S., Naqvi, S. A., Condas, L. A. Z., Saini, V., Kastelic, J. P., Luby, C., De Buck, J., & Barkema, H. W. (2018). Prevalence and genetic basis of antimicrobial resistance in non-aureus staphylococci isolated from Canadian dairy herds. Frontiers in Microbiology, 9, 256. https://doi.org/10.3389/fmicb.2018.00256,
- Prevalence and genetic basis of antimicrobial resistance in non … (n.d.).
- Rapid determination of novobiocin susceptibility for the identification of Staphylococcus saprophyticus – PubMed. (n.d.).
- Remel. (n.d.). Novobiocin disk instructions for use. Thermo Fisher Scientific.,
- Use of Mueller-Hinton agar to determine novobiocin susceptibility of coagulase-negative staphylococci – PMC. (n.d.).