Ion Exchange Chromatography (IEC) is a chromatographic separation technique used to separate and purify molecules based on their net surface charge. It works mainly for charged biomolecules like proteins, peptides, amino acids and nucleic acids. In this method, separation depends on electrostatic attraction between the sample and the charged stationary phase.
Ion exchange chromatography is based on reversible ionic interaction between oppositely charged groups. The column is packed with ion exchange resin beads having fixed charged functional groups. When a liquid sample is passed through the column, molecules having opposite charge to the resin are bound strongly. Neutral molecules or same charged molecules do not bind and they are washed out.
The stationary phase can be either positively charged or negatively charged. If the resin has positive groups, it binds negatively charged molecules and it is called anion exchange chromatography. If the resin has negative groups, it binds positively charged molecules and it is called cation exchange chromatography. The binding strength depends on the ionic charge of the molecule and the buffer conditions.
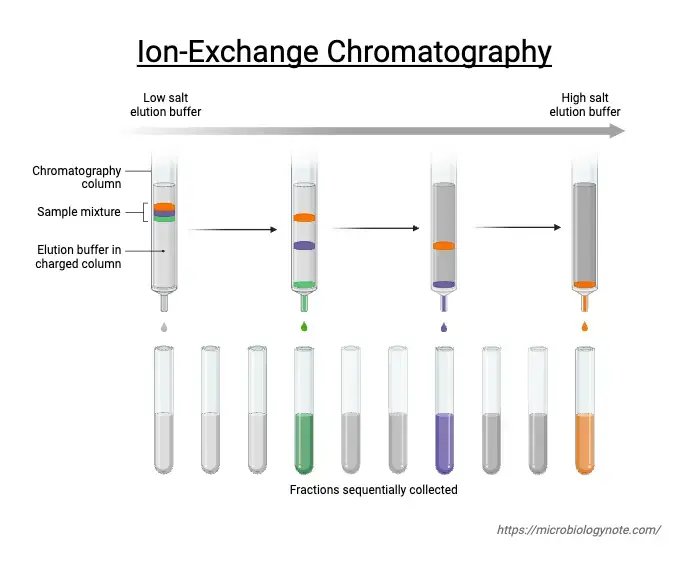
After binding, the target molecules are eluted from the column by changing the buffer condition. In general, elution is done by increasing salt concentration (ionic strength) or by changing pH of the buffer. Salt ions compete with bound molecules and disrupt the electrostatic attraction. pH change alters the charge on the molecules and reduces binding, then different components come out at different condition and fractions are collected.
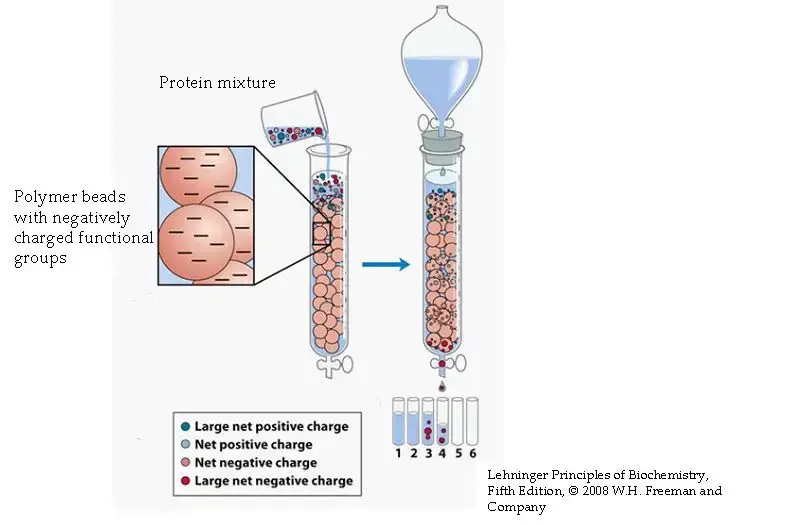
Definition of Ion Exchange Chromatography
Ion exchange chromatography is a separation technique that separates ions and polar molecules based on their affinity to ion exchangers. It involves the reversible exchange of ions between the target ions in the sample solution and ions attached to an ion exchanger.
Working Principle of ion exchange chromatography
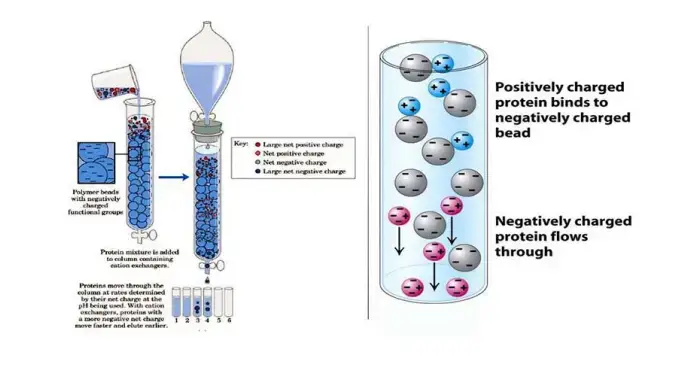
Ion exchange chromatography working principle is based on reversible electrostatic attraction between charged molecules present in the mobile phase and oppositely charged functional groups attached on an ion exchange resin (stationary phase). The resin contains fixed ionic groups, and counter ions are already present around the matrix. When the sample is applied, opposite charged molecules get attracted and are bound on the resin by displacing the counter ions.
In this process, neutral molecules or same charged molecules do not bind with the resin. They pass through the column with the buffer flow and are removed in the wash step. The binding mainly depends on net surface charge of the molecule and the pH and ionic strength of the buffer.
For proteins (amphoteric nature), the charge is decided by the buffer pH in relation to isoelectric point (pI). If pH is below pI, the protein becomes positively charged and it will bind with a negatively charged resin (cation exchanger). If pH is above pI, the protein becomes negatively charged and it will bind with a positively charged resin (anion exchanger).
After binding, the column is washed so that unbound impurities are removed. Then elution is carried out by changing the buffer condition. Increasing salt concentration is used so that salt ions compete and displace the bound molecules. pH can also be changed so that the net charge of the molecule is reduced or changed and affinity is lost, then molecules come out from the column in separated fractions.
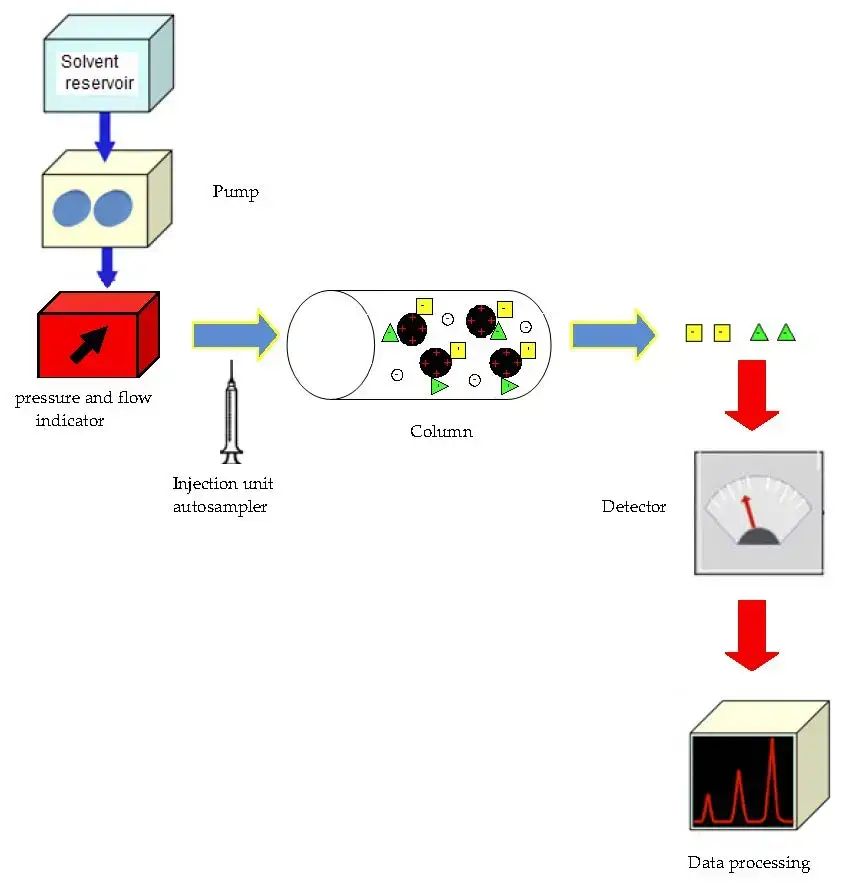
Instrumentation/Parts of Ion Exchange Chromatography
- Solvent reservoirs (Buffer bottles)
It is used to store different mobile phase buffers. Buffers are taken from here for running the column. - Pump
It is used to move the buffer and sample through the system at a constant flow rate. In some systems high pressure pumping is used. - Tubing and connectors
These are the flow lines that carry buffer, sample and eluent between different parts. It connects reservoir, pump, injector, column and detector. - Valves (inlet/selection/outlet valves)
These are used to direct the flow of solvent and sample in proper path. It helps in switching buffers and controlling the system flow. - Mixer (Gradient mixer)
It is used to mix two or more buffers uniformly. It is important during gradient elution to maintain proper composition. - Sample injection device / injection valve
It is used to introduce the sample into the flowing mobile phase. Sample volume is controlled in this step. - Chromatography column (Ion exchange column)
It is the main part that contains ion exchange resin beads (stationary phase). The charged molecules are separated here by electrostatic interaction. - Suppressor (in some ion chromatography systems)
It is used to reduce background conductivity of the eluent. It increases detector sensitivity by lowering salt interference. - Detectors and sensors
Detector is used to identify the eluted molecules. Common detectors are conductivity detector and UV-Visible detector, sometimes refractive index or mass detector is also used. Sensors like pH and temperature sensors may be present. - Fraction collector
It is used to collect the separated components in different tubes/bottles. Fractions are collected sequentially as peaks come out. - Computer interface and software
It is used to set run parameters, monitor pressure/flow and record detector output. Data is analysed and chromatogram is obtained by software.
Protocol/Procedure of Ion Exchange Chromatography
- Column equilibration
In this step, the column is equilibrated with starting buffer. About 5 to 10 column volumes of buffer is passed through the column. This sets the required pH and ionic strength and makes the resin ready with exchangeable counter ions. - Sample preparation
The sample is prepared in the same starting buffer condition. pH and ionic strength is adjusted so that binding will occur properly. - Sample loading
The sample is loaded on the equilibrated column. Oppositely charged molecules bind with the resin by displacing the counter ions. Uncharged or same charged molecules will not bind. - Washing
The column is washed with the starting buffer. Unbound and weakly bound impurities are removed in this step. Washing is continued till no impurity/protein is detected in the flow through. - Elution
The bound molecules are eluted by changing the buffer condition. Salt concentration is increased gradually (ionic strength) so that ions compete and displace the bound molecules. pH can also be changed so that target molecules lose the charge and binding is reduced. Elution can be done by linear gradient or step wise change. - Detection and fraction collection
The eluted sample coming out is monitored by detector (commonly UV absorbance and conductivity). Fractions are collected in separate tubes according to peaks. - Regeneration of column
After run, the column is cleaned by passing high salt buffer (about 1 to 2 M NaCl) or chemical wash like NaOH. This removes tightly bound materials from the resin. - Re-equilibration and storage
The column is again equilibrated with starting buffer (5 to 10 column volumes) till pH and conductivity become stable. Then the column is kept ready for next sample run or stored in suitable buffer.

Sample Preparation Procedure (Step by step) for Ion Exchange Chromatography
- Selection of start buffer
First, the start buffer is selected (pH and ionic strength). The same buffer condition will be used for preparing the sample and equilibrating the column. - Buffer matching (desalting/dialysis)
The sample is brought into the same condition as the starting buffer. Desalting column or dialysis is used to remove excess salts and change the sample into the selected buffer. This step improves binding and separation. - pH adjustment
The pH of the sample is adjusted to control charge on the target molecule. For proteins, pH is usually kept about 0.5 to 1.0 unit away from its isoelectric point (pI) so that proper net charge is present for binding. - Ionic strength adjustment (salt reduction)
The ionic strength is kept low. If salt is high, the sample is diluted with start buffer or desalted further. High salt can prevent binding with the resin, so this step is important. - Clarification (centrifugation)
The sample is centrifuged to remove debris, foam and insoluble particles. Supernatant is taken carefully without disturbing pellet. - Filtration
The sample is filtered to make it completely clear. Syringe-tip filter (like cellulose acetate or PVDF) is used so that particulates do not block the column and no back pressure is created. - Viscosity control
If the sample is highly viscous, it is diluted with starting buffer. This maintains normal flow through the column and avoids irregular flow pattern and pressure problem. - Concentration control
The sample concentration is adjusted before loading. Very concentrated protein samples are avoided, and in general about 50–70 mg/mL range is maintained depending on column capacity. - Removal of interfering substances
Major interfering contaminants are removed before loading. Lipids, ionic detergents, and competing ions are reduced because they can occupy binding sites or disturb separation. - Temperature matching
Finally, sample is brought to same temperature as buffers and column. This reduces bubble formation and gives stable run during loading and binding.
Resin Selection in Ion Exchange Chromatography
- Decide charge of target (pI and pH)
First the net charge of target molecule is considered. It depends on buffer pH in relation to isoelectric point (pI). If pH is above pI, the molecule becomes negative, then anion exchanger resin (positive resin) is selected. If pH is below pI, the molecule becomes positive, then cation exchanger resin (negative resin) is selected. - Check pH stability of sample
The target should be stable at the selected working pH. pH is kept in a range where protein is not denatured and binding is still possible. - Select exchanger type (strong or weak)
Strong ion exchangers remain charged in wide pH range, so binding capacity remains stable. Weak ion exchangers change ionization with pH, so capacity varies, but sometimes gives different selectivity in complex separation. - Select porosity of resin (pore size)
Highly porous resin with large pore volume is selected for large molecules like proteins and antibodies. Low porous or non-porous resin is selected for small molecules like inorganic ions and organic acids. - Select particle size (bead size)
Small bead size gives more surface area, so resolution is high and peaks are sharp, but back pressure is also high and flow rate becomes low. Large bead size gives less resolution but it allows higher flow and it is used for preparative or large scale runs. - Check ionic form (counter ion)
Resins are supplied with counterions like Na+ or Cl−. The counterion should be easily displaced by the target molecule. If counterion binds too strongly, target binding becomes poor. - Consider dynamic binding capacity
Dynamic binding capacity is checked, means how much target can bind in the real flow condition. It depends on flow rate, buffer, and sample property, so resin is chosen with suitable capacity. - Matrix material and stability
The resin matrix (agarose, cellulose, silica, synthetic polymer etc.) should be physically stable. It should tolerate pressure, salt changes and cleaning solutions like NaOH without breaking or losing capacity.
Applications of ion exchange chromatography
- It is used for purification of proteins.
- It is used for purification of enzymes.
- It is used to separate peptides and amino acids.
- It is used in purification of monoclonal antibodies (mAbs).
- It is used for purification of DNA.
- It is used for purification of RNA.
- It is used for plasmid and oligonucleotide separation.
- It is used in purification of viral vectors (AAV etc.) in gene therapy work.
- It is used in mRNA purification for vaccine and related products.
- It is used to separate protein isoforms and charge variants.
- It is used in analysis of proteins having post translational modifications (phosphorylation, acetylation).
- It is used for drug impurity analysis and quality control in pharmaceuticals.
- It is used for biomarker estimation in analytical samples.
- It is used in dairy industry for demineralization of whey (infant formula use).
- It is used in sugar industry for decolorization and demineralization of sugar liquor.
- It is used in glucose and fructose separation or isomerization processes.
- It is used in fruit juice processing for bitterness removal and colour reduction.
- It is used for haze stabilization in apple juice and acidity reduction in syrups.
- It is used in winemaking for tartrate stabilization and total acidity reduction.
- It is used for water softening by removing Ca2+ and Mg2+ ions.
- It is used in ultrapure water production for electronics and power plant use.
- It is used in recovery of useful chemicals (example acetic acid) from industrial effluent.
- It is used for de-acidification and purification steps in industrial oils.
- It is used in environmental monitoring of ionic contaminants in drinking water.
- It is used in metabolomics for small organic acid and metabolite analysis.
- It is used in forensic testing for biomarker detection in urine samples.
Precautions of Ion Exchange Chromatography
- Sample, buffers and column should be kept at same temperature. If temperature is different, air bubbles can form inside the packed bed.
- Buffer should be prepared at the same temperature at which it will be used. pH of some buffers (example Tris) changes with temperature.
- Sudden or extreme change in pH and ionic strength should be avoided. It can denature or inactivate proteins and sometimes precipitation occurs inside the column.
- Samples and buffers should be filtered or centrifuged before use. Particles can clog the column and contamination also occurs. High purity water and reagents are used.
- Column should be handled carefully. Hitting the column or giving sudden pressure change should be avoided. Very high pressure can damage resin bed.
- Buffer should be pumped from the top of the column. Buffer should not be sucked from below, because suction creates bubbles in the column.
- Column should not be overloaded beyond dynamic binding capacity. Overloading reduces resolution and proper separation is not obtained.
- Highly viscous samples should be diluted with starting buffer. Viscosity gives irregular flow, peak distortion and back pressure problem.
- Column should be washed, cleaned and re-equilibrated after every run. It prevents carryover, resin blinding and fouling of the column.
Advantages of ion exchange chromatography
- It gives high resolution separation. Even small charge difference molecules can be separated.
- It is highly precise technique for charged biomolecules, so clean fractions are obtained.
- It has high sample loading capacity. Large volume samples can be processed.
- It is selective and can be customised. Different resins are selected and buffer pH and salt can be adjusted.
- It is scalable method. Small lab scale to large industrial scale can be done with same basic principle.
- It is cost effective. Running cost is low and resin can be regenerated and reused.
- It is compatible with automation. Automated systems and continuous operation can be done.
- It is used in many fields. Biopharmaceutical, food processing, water treatment and environmental testing.
- It produces less waste compared to some other separation methods. Effluent handling is easier.
Limitations of ion exchange chromatography
- It is suitable mainly for charged molecules. Neutral compounds are not separated properly.
- It depends highly on buffer condition. Small change in pH and ionic strength can give poor resolution or incomplete separation.
- Sometimes extreme pH or high salt is needed for elution. These conditions can denature proteins and sensitive biomolecules can get degraded.
- Resin fouling and resin blinding can occur. Non specific adsorption blocks the resin pores and binding capacity reduces.
- Column has limited loading capacity. If sample is overloaded, selectivity and resolution becomes low.
- It can be time consuming method. Gradient elution with salt or pH takes more time for complete run.
- Co-elution problem can occur for closely related species. Charge variants may not separate clearly in some cases.
- High salt in fractions gives difficulty in identification and also needs desalting step for further use.
- Regeneration produces high salt waste. Waste stream contains high salinity and it creates disposal problem.
- It is not directly compatible with mass spectrometry (MS). High electrolyte and large volume mobile phase interferes, so salt removal or special system is needed.
- Kinetic limitation can be seen in complex bioseparation. Binding and elution may become slow in some samples.
- In food and beverage use, desired colour and flavour compounds can also be removed. Product quality can be affected.
References
- Alfa Chemistry. (2026). What are the main components of ion exchange resins?. Alfa Chemistry.
- Ames National Laboratory. (n.d.). Manhattan project roots. Ames Laboratory.
- Atomic Heritage Foundation. (2022). Frank Spedding. Nuclear Museum.
- AXEON Water Technologies. (n.d.). 6 advantages of ion exchange systems for industrial applications. AXEON Water.
- Bio-Rad Laboratories. (2026). Aminex cation exchange resins. Bio-Rad.
- Bio-Rad Laboratories. (2026). Anion exchange chromatography. Bio-Rad.
- Bio-Rad Laboratories. (2026). Introduction to ion exchange chromatography. Bio-Rad.
- Bio-Rad Laboratories. (2026). Ion exchange resins. Bio-Rad.
- Bio-Rad Laboratories. (2026). Saving lives: Innovative therapeutic applications of ion exchange resins. Bio-Radiations.
- Bionex Scientific. (n.d.). Cytiva ÄKTA Pilot 600R chromatography system. Bionex Scientific.
- Boag, M. (2025, June 18). Principles of ion exchange chromatography: A comprehensive guide. Phenomenex.
- Chrom Tech, Inc. (2025, October 20). Evolution and impact of ion-exchange chromatography. Chrom Tech, Inc.
- Chrom Tech, Inc. (2025, October 20). Mastering ion exchange chromatography: Essential guide. Chrom Tech, Inc.
- Conduct Science. (2026, February 16). Ion-exchange chromatography protocol. Conduct Science.
- Currie, S. (n.d.). What is ion exchange chromatography used for? 4 important applications for protein purification. GoldBio.
- Cytiva. (2018, April 25). Ion exchange chromatography handbook. Drug Discovery Online.
- Cytiva. (2020). Tips for successful ion exchange chromatography. Cytiva.
- Cytiva. (2023). Lab-scale protein purification [Brochure]. Gene X-Press.
- Cytiva. (2024, November 12). Fundamentals of ion exchange chromatography. Cytiva.
- Cytiva. (2025). ÄKTApure™. Cytiva.
- Cytiva. (2026). Chromatography systems. Cytiva.
- Cytiva. (n.d.). Ion exchange. Cytiva.
- Department of Energy’s Office of History and Heritage Resources. (n.d.). Manhattan project: Processes > Uranium isotope separation. OSTI.gov.
- Fluence Corporation. (2026). Food and beverage processing solutions. Fluence Corporation.
- Gold Biotechnology. (2026). The 2 common types of ion exchange chromatography: Anion vs. cation. GoldBio.
- Harvard Apparatus. (n.d.). Guide to ion-exchange chromatography. Harvard Apparatus.
- LCGC International. (n.d.). How it works: Ion-exchange SPE. LCGC International.
- López, F. (1999). Ion-exchange processes in the food industry. Environment Protection Engineering, 25(1-2), 104-106.
- Mattick, L. R., & Gogel, E. V. (1980). Acid reduction in wine by ion exchange (U.S. Patent No. 4,205,092A). U.S. Patent and Trademark Office.
- Merck KGaA. (2026). Practical considerations for IEX separation. Sigma-Aldrich.
- Mira, H., Leite, P., Ricardo-da-Silva, J. M., & Curvelo-Garcia, A. S. (2006). Use of ion exchange resins for tartrate wine stabilization. OENO One, 40(4), 223–246.
- Qiu, L. (2023). Principle and applications of ion exchange chromatography. Pharmaceutical Analytical Chemistry, 8, 205.
- Resonac Europe GmbH. (n.d.). Anion and cation exchange chromatography columns. Shodex.
- Sartorius AG. (2026). Ion exchange chromatography. Sartorius.
- Sino Biological. (n.d.). Protein purification by ion exchange chromatography. Sino Biological.
- The Protein Man. (2023, May 22). Ion exchange chromatography resins and methods. G-Biosciences.
- The University of Iowa. (2025). History of Ames Laboratory. Former Worker Medical Screening Program.
- Thermo Fisher Scientific. (2026). Ion exchange chromatography resins. Thermo Fisher Scientific.
- Unknown Author. (n.d.). Ion exchange chromatography: A comprehensive analysis of principles, protocols, and multidisciplinary applications.
- Weiss, J. (2016). Handbook of ion chromatography (4th ed.). Wiley-VCH.
- Wikipedia contributors. (2026, January 22). Frank Spedding. In Wikipedia, The Free Encyclopedia.