Simmons Citrate Agar is a specialized solid microbiological culture medium which is used mainly to differentiate Gram-negative bacteria especially the members of Enterobacteriaceae family. It is used to test the ability of an organism to survive by using citrate as the sole carbon and energy source and ammonium salts as the sole nitrogen source.
It contains a pH indicator called bromothymol blue which is forest green in neutral pH. If the bacterial species have the required enzyme such as citrate permease then citrate and ammonium salts are utilized for growth and alkaline byproducts are produced.
During this process ammonia and sodium carbonate is formed which increases the alkalinity of medium. This change in alkalinity causes the bromothymol blue indicator to change the colour of agar from green to deep blue and this is referred to as positive citrate test.
If the bacteria do not have the ability to metabolize citrate then growth is not seen and the agar remains green which indicates negative test result.
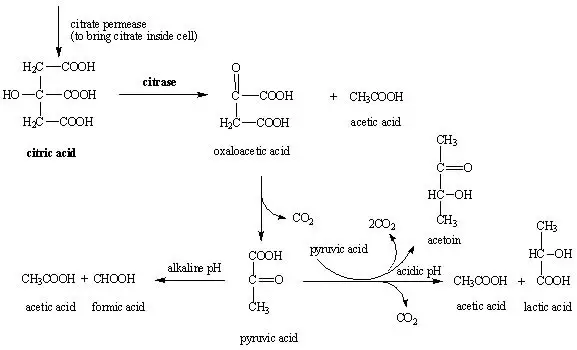
Principle of Simmons Citrate Agar

Simmons citrate agar works on the principle of determining the ability of an organism to use citrate as the sole carbon and energy source and inorganic ammonium salts as the sole nitrogen source. This is a strictly defined medium and growth is possible only when citrate and ammonium salt is utilized.
For citrate utilization the organism must produce transport enzyme citrate permease which brings citrate inside the cell. In this step citrate is metabolized by citrate lyase (citritase) and it is converted into pyruvate and carbon dioxide.
As citrate and ammonium salts are consumed alkaline byproducts are produced such as ammonia and sodium carbonate. These products increases the pH of agar and the medium becomes alkaline (pH above 7.6).
The pH indicator bromothymol blue reacts with this alkaline condition and the colour changes from green to deep royal blue. Growth with green to blue colour change indicates positive citrate utilization test.

Requirements for Simmons Citrate Agar test
- Simmons citrate agar medium– It is used as prepared slants or it is prepared from dehydrated powder using distilled/deionized water.
- Pure bacterial culture– 18 to 24 hour pure culture of test organism is required and it should be taken from solid medium not from broth to avoid nutrient carryover.
- Sterile inoculating needle– Needle is preferred over loop and a very light inoculum is transferred to avoid false positive due to extra nutrients or dead cellular material.
- Incubator– Aerobic incubation is required at 35–37°C for most organisms and some organisms may require 30°C (Yersinia enterocolitica).
- Test tubes with caps– Tubes are used for slant medium and caps should be kept loosened during incubation for oxygen exchange in citrate metabolism.
- Sterilization equipment– Incinerator or Bunsen burner is used to flame the inoculating needle and to maintain aseptic conditions.
Composition of Simmons Citrate Agar (For 1 litre)
- Sodium chloride (NaCl)– 5.0 g
- Sodium citrate– 2.0 g
- Ammonium dihydrogen phosphate (NH₄H₂PO₄)– 1.0 g
- Dipotassium phosphate (K₂HPO₄)– 1.0 g
- Magnesium sulfate (MgSO₄)– 0.2 g
- Bromothymol blue– 0.08 g
- Agar– 15.0 g
- Distilled/deionized water– 1000 mL
Preparation of Simmons Citrate Agar

- Weigh the dehydrated Simmons citrate agar powder (about 24.2 to 24.3 g) and suspend it in 1 litre distilled/deionized water. If it is prepared from individual chemicals then salts are dissolved first and pH is adjusted to about 6.8 to 6.9 then agar and bromothymol blue is added.
- The medium is heated with frequent shaking and it is brought to boil for 1 to 2 minutes so that agar is dissolved completely.
- The liquid medium is dispensed into test tubes (about 4.0 to 5.0 mL per 16 mm tube).
- Sterilization is done by autoclaving at 121°C (15 psi) for 15 minutes.
- After autoclaving tubes are kept in slanting position and allowed to cool and solidify to form slants. Long slant is formed (about 4 to 5 cm) and a small butt is also formed at the bottom (about 1.5 to 2.5 cm).
- The prepared uninoculated citrate agar slants are stored in refrigerator and it can be used for about 6 to 8 weeks.
Procedure of Simmons Citrate Agar

- A pure 18 to 24 hour culture of the organism is taken from a solid medium. Broth culture is avoided because external nutrients can be carried over.
- A sterile inoculating needle is used and a very light inoculum is picked from a single well isolated colony. Needle is preferred over loop to limit the amount of cellular material.
- The Simmons citrate agar slant is inoculated by lightly streaking the surface in a zigzag/back and forth manner from bottom to top. Stabbing of butt is generally avoided because the reaction needs aerobic condition.
- The caps of the tubes are kept loosened to allow oxygen exchange which is required for oxidative citrate metabolism.
- The tubes are incubated aerobically at 35–37°C for 24 to 48 hours. For some organisms incubation can be done at 30°C (Yersinia enterocolitica). Slow growing organisms may require extended incubation up to 4 to 7 days.
- The slants are observed daily for growth and colour change. Growth with green to intense blue colour change indicates positive citrate test result.
Result of Simmons citrate test

Positive result (Growth with colour change)
Visible growth is seen on the citrate agar slant and the medium changes from green to intense royal/Prussian blue. This indicates citrate is utilized and alkaline byproducts is produced.
Positive result (Growth without colour change)
Growth is seen on the slant but the medium remains green. It is still considered positive because growth indicates citrate is being used and blue colour may develop after additional 24 hours incubation.
Negative result
No visible growth is seen (or only trace growth) and the medium remains dark green. This indicates the organism do not have the required enzymes for citrate utilization.
Equivocal/Invalid result
Yellow or tan discoloration is seen on the slant. This occurs when heavy inoculum is used and acid is produced from breakdown of dead cellular material not true citrate utilization so it should not be taken as positive result.

List of bacteria in Simmons citrate test
Bacteria which gives positive result (Blue colour)
- Enterobacter aerogenes
- Klebsiella pneumoniae
- Salmonella enteritidis
- Salmonella typhimurium
- Serratia proteamaculans
- Proteus rettgeri
- Yokenella regensburgei
- Acidovorax delafieldii
- Frateuria aurantia
- Most of Enterobacter, Citrobacter, Klebsiella, Proteus, Providencia and Serratia species gives positive result.
Bacteria which gives negative result (Green colour)
- Escherichia coli
- Shigella dysenteriae
- Other Shigella species
- Salmonella typhi
- Salmonella paratyphi A
- Salmonella pullorum
- Salmonella gallinarum
- Yersinia enterocolitica
- Yersinia pseudotuberculosis
- Edwardsiella species
- Morganella morganii
- Klebsiella rhinoscleromatis
- Hafnia alvei
- Acidovorax facilis
- Acidovorax temperans
Uses of Simmons Citrate Agar
- It is used to differentiate Enterobacteriaceae and other Gram-negative bacilli as a part of IMViC test panel (Indole, Methyl red, Voges-Proskauer and Citrate).
- It is used in food and water quality testing to detect fecal contamination by differentiating fecal coliforms such as Escherichia coli (citrate negative) from environmental coliforms such as Enterobacter aerogenes (citrate positive).
- It is used to subtype Salmonella species by separating citrate positive strains (S. enteritidis, S. typhimurium) from citrate negative human pathogens (S. typhi, S. paratyphi A, S. pullorum).
- It is used as secondary identification test to confirm Yersinia enterocolitica (citrate negative) and differentiate it from other Yersinia species which are citrate positive or variable.
- It is used to distinguish closely related species such as Proteus rettgeri (+) from Morganella morganii (–), Yokenella regensburgei (+) from Hafnia alvei (–), and Serratia proteamaculans (+) from Yersinia pseudotuberculosis (–).
- It is also used as a secondary tool for isolation and identification of some fungi and yeasts which can utilize citrate due to restricted nutrient profile of medium.
Advantages of Simmons Citrate Agar
- Bromothymol blue is used as pH indicator and it gives a distinct colour change from forest green to deep blue which makes the result easy to read.
- It is a solid agar medium (slant) so true growth and colony formation can be seen clearly and turbidity judgement is not required like liquid broth.
- False positive results are reduced because heavy transfer of dead cells or carryover matter in broth can mimic growth but in Simmons agar growth with colour change is observed more clearly.
- Slant surface provides ideal oxygen exposure and it gives maximum aerobic surface area required for oxidative citrate metabolism.
- The medium is highly selective because only sodium citrate is present as carbon source and ammonium salt is present as nitrogen source so organisms lacking citrate permease cannot grow.
Limitations of Simmons Citrate Agar
- This test cannot identify organism upto species level and it must be used with other biochemical tests for complete identification.
- False positive can occur due to nutrient carryover (glucose or other nutrients) from previous isolation media so inoculation from broth culture is discouraged.
- Inoculum size can affect result– heavy inoculum can give false positive or can produce yellow/tan discoloration due to breakdown of dead cellular material not true citrate utilization.
- Very light inoculum can sometimes give false negative result because growth may not be sufficient to be observed.
- Citrate metabolism is an oxidative process and it requires aerobic condition so stabbing the agar or tightening the caps restricts oxygen exchange and leads to false negative.
- Some organisms shows growth without colour change because pH may not rise enough to turn blue. It can be confusing and may require additional 24 hours incubation.
- Some organisms reacts slowly and extended incubation (4 to 7 days) may be required for clear positive reaction and equivocal results should be repeated.
- Medium is pH sensitive during preparation and if pH is not maintained (about 6.5 to 7.0) initial colour can be incorrect. If medium becomes dry or old it can change colour even before inoculation.
- Due to nutritional variations some strains may grow poorly or fail to grow even if they belong to citrate utilizing species.
Precautions of Simmons citrate test
- A very light inoculum should be used. Heavy inoculum can give false positive or yellow/tan discoloration because bacteria may survive by breaking down dead cellular material instead of citrate utilization.
- Inoculating needle should be used instead of loop because it transfers less biomass and less carryover of medium.
- Sample should be taken from pure 18 to 24 hour culture grown on solid medium. Broth culture should not be used because unwanted nutrients can be carried over.
- Nutrient carryover should be prevented. Simmons citrate agar should be inoculated first when multiple biochemical tests are done or needle should be reflamed before streaking. Diluting inoculum in sterile saline or water can also be done to reduce carryover.
- Agar should not be stabbed. Only surface of slant should be streaked because citrate metabolism needs aerobic condition.
- Tube caps should be kept loosened during incubation for proper oxygen exchange. Tight caps restrict oxygen and can affect growth and pH change.
- During preparation water pH should be maintained between 6.5 to 7.0 so that initial green colour of medium is not altered.
- Citrate test alone is not sufficient for species level identification so it should be confirmed with other biochemical or molecular tests.
References
- Biolife Italiana. (2023, April). Simmons citrate agar: Dehydrated culture medium [Instructions for use].
- Bio-Rad. (2011, August 26). Simmons’ citrate/agar [Technical sheet].
- Condalab. (n.d.). Simmons citrate agar ISO Cat. 1014. BioVendor.
- Dalynn Biologicals. (2014, October). Simmons citrate agar [Technical sheet].
- Difco. (n.d.). Simmons citrate agar. In Difco manual (pp. 514-515). Interlab.
- DrChika. (2022, December 28). Citrate test. Microbiology Class.
- Food and Drug Administration. (2017, October 17). BAM media M138: Simmons citrate agar. U.S. Department of Health and Human Services.
- Hardy Diagnostics. (2020). Simmons citrate agar [Instructions for use].
- Hartline, R. (2023, February 18). 1.20: Citrate test. In Microbiology laboratory manual. Biology LibreTexts.
- MacKenzie, E. (2025, August 12). 31.12: Simmon’s citrate agar test. In IVC microbiology lab manual. Biology LibreTexts.
- MacWilliams, M. P. (2009, December 8). Citrate test protocol. American Society for Microbiology.
- Mast Group. (2024, August). Simmons citrate agar – DM211 [Instructions for use].
- Merck KGaA. (2008, October 17). SIMMONS citrate agar. Merck Millipore.
- Merck KGaA. (2018). 85463 Simmons citrate agar NutriSelect basic [Datasheet]. Sigma-Aldrich.
- MilliporeSigma. (2026). Simmons citrate agar for microbiology citrate utilization test. Sigma-Aldrich.
- Neogen. (n.d.). Technical specification sheet: Simmons citrate agar (NCM0168).
- Remel. (2011, February 9). Citrate agar (Simmons) [Instructions for use]. Thermo Fisher Scientific.
- Remel. (2011, March 1). Simmons citrate agar [Instructions for use]. Thermo Fisher Scientific.
- Sapkota, A. (2022, January 10). Citrate utilization test- Principle, procedure, results, uses. Microbe Notes.
- Sigma-Aldrich. (2004, March). Simmons citrate agar (S3681) [Product information].
- Smith, M., & Selby, S. (2021, March 19). 3.9: Simmons citrate agar. In Microbiology for allied health students: Lab manual. Biology LibreTexts.
- Comprehensive analytical report on Simmons citrate agar: Biochemical principles, formulation dynamics, and diagnostic applications in clinical and environmental microbiology. (n.d.).
- Virtual Microbiology Lab Simulator Software. (2022, November 1). Citrate utilization test. VUMIE.
- Wikipedia. (2025, November 4). Citrate test.
- Wikipedia. (2025, October 10). Simmons’ citrate agar.
Thank you