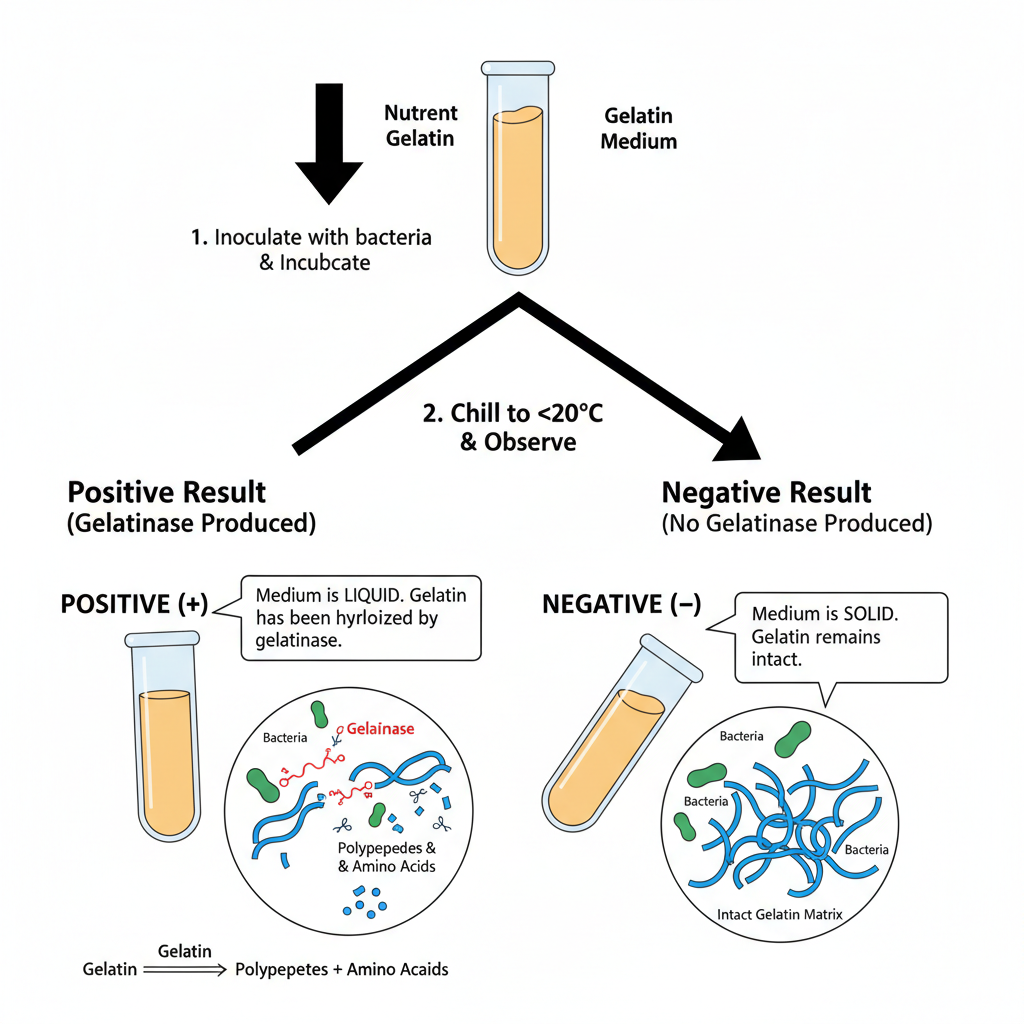
Gelatin hydrolysis test is a biochemical test used to determine whether a microorganism can produce gelatinases. These gelatinases are extracellular proteolytic enzymes, and it helps in breaking down gelatin into smaller polypeptides and then into amino acids which can be absorbed and utilized by the cell for metabolism.
Gelatin is a large protein derived from collagen of vertebrate connective tissue. In normal condition gelatin forms a solid gel at cooler temperatures. This test is evaluated after incubation by chilling the inoculated medium and observing the liquefaction.
If the medium remains liquid even after chilling then the result is positive. This indicates that gelatin has been enzymatically hydrolyzed. If the medium becomes solid again after chilling then the result is negative and gelatin has not been broken down.
This test is used as a diagnostic tool for classifying bacteria. It is used to differentiate gelatinase positive Staphylococcus aureus from gelatinase negative Staphylococcus epidermidis. Other gelatinase producing genera are Serratia, Proteus, Bacillus, Clostridium, and Pseudomonas.
Objectives of gelatin hydrolysis test
- To determine whether an organism can produce gelatinase, which is an extracellular proteolytic enzyme capable of liquefying gelatin.
- To differentiate and group organisms on the basis of their ability to hydrolyze gelatin.
- To distinguish between Staphylococcus species. It is used for differentiating pathogenic gelatinase positive Staphylococcus aureus from non-pathogenic gelatinase negative Staphylococcus epidermidis.
- To aid in the identification of some bacterial genera such as Serratia, Proteus, Bacillus, Clostridium, Pseudomonas and Flavobacterium.
- To separate gelatin positive Serratia and Proteus species from the other members within the Enterobacteriaceae family.
Principle of Gelatin hydrolysis test
Principle of gelatin hydrolysis test is based on the ability of organism to produce gelatinases. These gelatinases are extracellular proteolytic enzymes which hydrolyze gelatin outside the cell. Gelatin is a large protein polymer derived from collagen of vertebrate connective tissue and it forms a solid structure at lower temperatures.
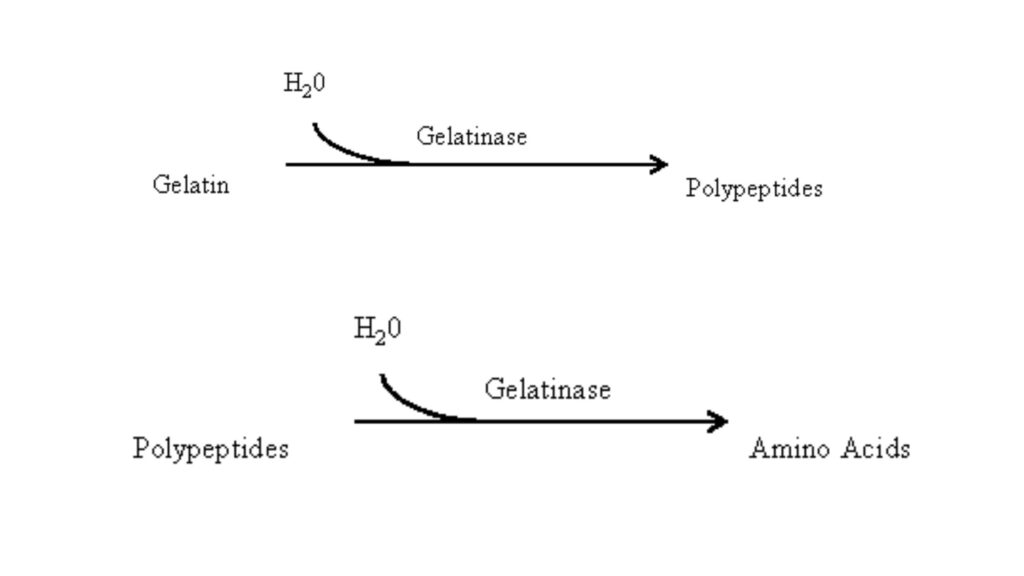
The intact gelatin molecule is too large to be transported across bacterial cell membrane. So some bacteria secrete gelatinase enzyme to break it down externally. This enzymatic degradation occurs in two steps. In first step gelatin is hydrolyzed into smaller polypeptides. In second step these polypeptides are further converted into individual amino acids.
These amino acids are then taken up by bacterial cell and it is used for metabolic purpose. In laboratory this activity is detected by growing the organism in nutrient gelatin medium. If gelatinase is produced then gelatin is digested and the solid medium is permanently liquefied.
Requirements for gelatin hydrolysis test
- Pure culture of the test organism is required. An 18 to 24 hour old culture is generally used. Known gelatinase positive and gelatinase negative control organisms are also taken.
- Nutrient gelatin medium is required for the test. It is usually prepared in test tubes as gelatin deeps. In some methods it can also be prepared in Petri plates.
- Inoculating needle is required for stab inoculation method. Inoculating loop is used if the plate method is performed.
- Bunsen burner and striker is required for sterilization of inoculating instruments and for maintaining aseptic condition.
- Incubator is required for incubation of the inoculated medium. It is generally maintained at suitable temperature about 25°C to 37°C depending upon the organism.
- Refrigerator or ice bath is required in tube method. It is used to cool the tubes after incubation for proper observation of liquefaction.
- Some chemical reagents are required in special methods. Saturated ammonium sulphate, acidic mercuric chloride (Frazier’s reagent) or ninhydrin may be used for precipitation of unhydrolyzed gelatin or detection of amino acids.
- General laboratory supplies such as test tube rack, pipette, marker and labeling tape are also required.
Media preparation composition and preparation steps for gelatin hydrolysis test
Composition of nutrient gelatin medium (per litre)
- Peptone (Peptic digest of animal tissue) – 5.0 g
- Beef extract – 3.0 g
- Gelatin – 120.0 g
- Distilled or deionized water – 1000 mL
- Final pH – 6.8 ± 0.2 at 25°C
Preparation steps
- Suspend 128 g of the dehydrated medium in 1 litre of distilled or deionized water.
- Heat the mixture gently and boil it to dissolve the medium completely.
- Dispense the medium into test tubes. Usually 2 to 3 mL medium is poured in each tube.
- Sterilize the medium by autoclaving at 121°C (15 psi) for 15 minutes.
- Allow the medium to cool in upright position so that gelatin deeps are formed.
- Store the prepared medium in refrigerator at 2°C to 8°C until it is used.
Procedure of gelatin hydrolysis test

1. Nutrient gelatin stab method
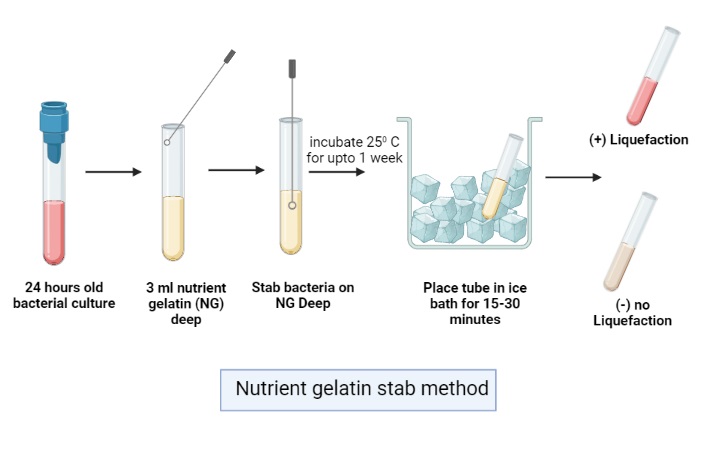
- Take a pure culture of the test organism. An 18 to 24 hour old culture is generally used.
- With the help of a sterile inoculating needle stab the organism into the center of nutrient gelatin deep tube. The stab should be made straight down up to about 0.5 to 1 inch above the bottom.
- In some cases 4 to 5 drops of a 24 hour old broth culture can also be inoculated into the medium.
- At the same time an uninoculated tube of nutrient gelatin is kept as control.
- Incubate the inoculated tube and control tube at suitable temperature. It is generally incubated at 35°C to 37°C or at the optimum temperature of the organism.
- The tubes are incubated for 24 hours up to 14 days because some organisms hydrolyze gelatin slowly.
- After incubation the tubes are removed and cooled before taking the result. This is necessary because gelatin becomes liquid above 28°C.
- Place the tubes in refrigerator at 4°C for about 30 minutes or keep in an ice bath for 15 to 30 minutes.
- When the uninoculated control tube becomes completely solidified gently tilt the inoculated tube and observe the liquefaction.
- Care should be taken not to shake the tube because gelatin hydrolysis may occur only at the surface.
- If the medium remains liquid after cooling, the test is positive.
- If the medium becomes solid, the test is negative. The tube is again incubated and observed up to 14 days for slow gelatinase producing organisms.
2. Gelatin agar plate method
- Prepare a gelatin agar plate and take a pure culture of the test organism.
- With the help of a sterile inoculating loop make a single heavy streak at the center of the gelatin agar plate.
- Incubate the plate in inverted position at 35°C for 24 to 48 hours.
- After incubation flood the plate with a developing reagent such as saturated ammonium sulphate or Frazier’s reagent (acidic mercuric chloride).
- Leave the plate for 5 to 10 minutes and then observe the result.
- The reagent precipitates the unhydrolyzed gelatin and produces a cloudy white appearance.
- If the organism produces gelatinase, a clear zone or halo is formed around the bacterial growth. This indicates positive gelatin hydrolysis test.
Result Interpretation of Gelatin Hydrolysis Test
Nutrient gelatin stab (tube) method
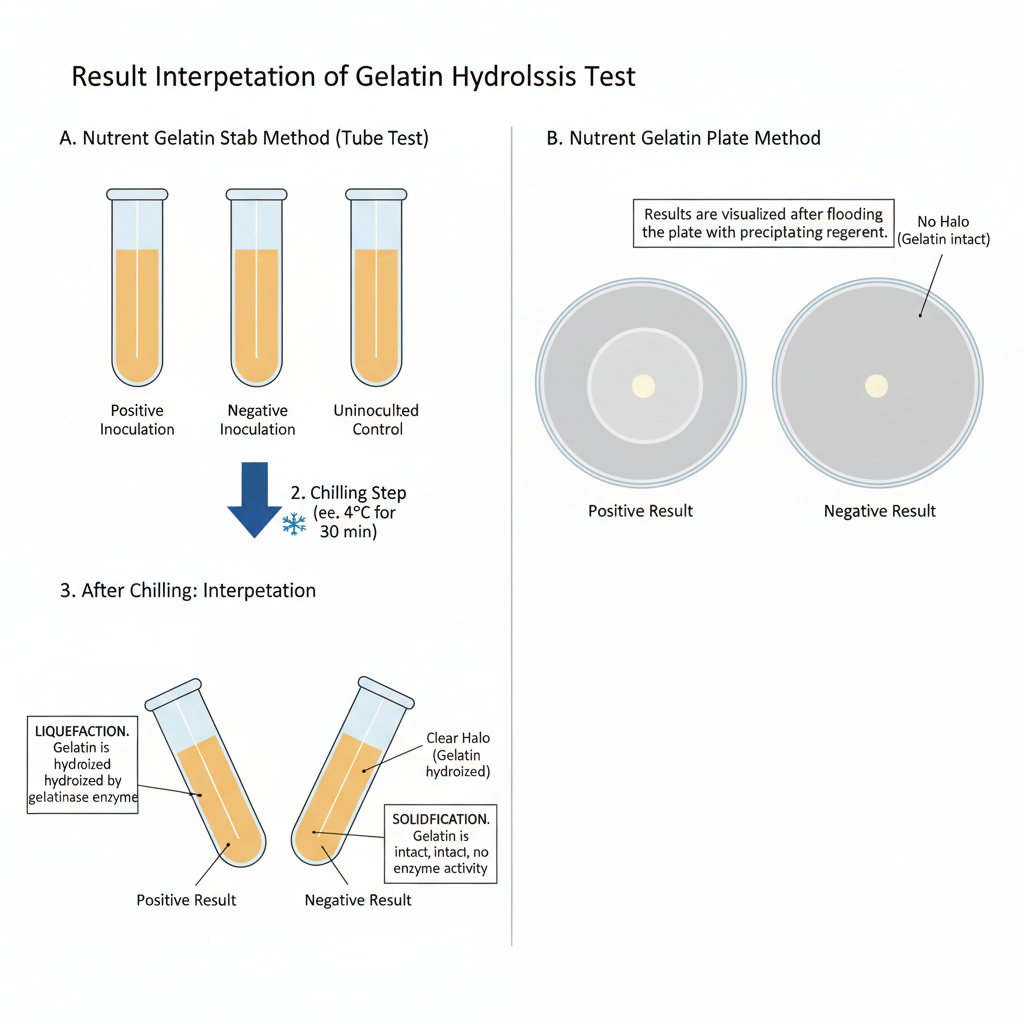
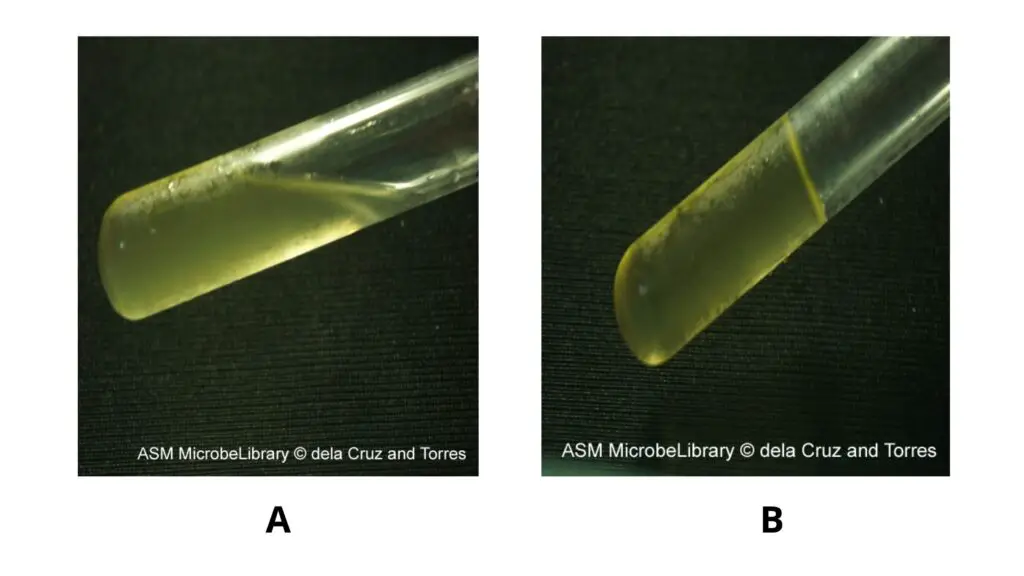
- Positive result – The gelatin medium remains partially or completely liquefied even after cooling in refrigerator or ice bath. This indicates that the organism produces gelatinase and hydrolyzes the gelatin.
- Negative result – The gelatin medium becomes solid again after cooling. It shows that gelatin remains intact and gelatinase enzyme is not produced.
- The result should be read only after the uninoculated control tube becomes completely solidified.
Gelatin agar plate method
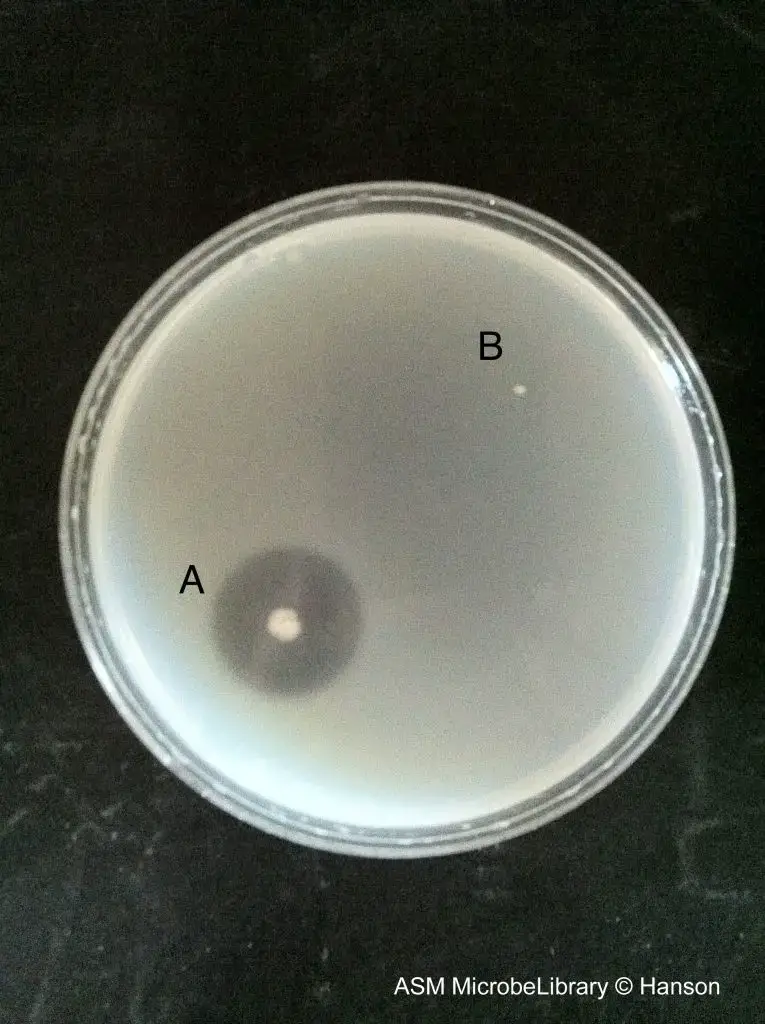
- Positive result – A clear zone or halo is formed around the bacterial growth after flooding the plate with the reagent. This indicates hydrolysis of gelatin by gelatinase enzyme.
- Negative result – No clear zone is seen around the bacterial growth. The unhydrolyzed gelatin forms a cloudy or opaque white precipitate up to the margin of the growth.
Rapid enzymatic methods
- Gelatin strips – Positive result is indicated by the appearance of blue coloured base material of the strip. It is exposed after liquefaction of the outer gelatin coating by the organism.
- Charcoal-gelatin discs – Positive result is observed when charcoal particles are released from the digested gelatin matrix and settle at the bottom of the liquid suspension.
- Ninhydrin detection – Positive result is indicated by the development of deep purple colour (Ruhemann’s purple). It detects the free amino acids released during gelatin hydrolysis.
Quality control organisms of gelatin hydrolysis test
Positive control organisms
- Bacillus subtilis (ATCC 6633) – It produces gelatinase and liquefies gelatin.
- Staphylococcus aureus (ATCC 25923) – It is gelatinase positive and gives positive gelatin hydrolysis test.
- Clostridium perfringens (ATCC 12924 or ATCC 13124) – It hydrolyzes gelatin and is used as positive control organism.
- Proteus vulgaris (ATCC 13315) – It produces gelatinase and liquefies the gelatin medium.
- Pseudomonas aeruginosa – It is used as a positive control because gelatin hydrolysis occurs.
- Serratia marcescens – It is also taken as a positive control organism for gelatin hydrolysis test.
Negative control organisms
- Escherichia coli (ATCC 25922) – It does not produce gelatinase and the gelatin remains solid.
- Staphylococcus epidermidis – It is gelatinase negative and is used as negative control organism.
Uses of Gelatin Hydrolysis Test
- It is used to determine whether a microorganism can produce extracellular gelatinase enzyme.
- It is used to differentiate Staphylococcus species. It helps in distinguishing gelatinase positive pathogenic Staphylococcus aureus from gelatinase negative Staphylococcus epidermidis.
- It is used in the identification of Enterobacteriaceae members. Gelatin positive genera like Serratia and Proteus can be separated from other enteric bacilli such as Escherichia coli, Salmonella and Shigella.
- It is used for classification of Pseudomonas species. It helps in differentiating positive species like Pseudomonas aeruginosa and Pseudomonas fluorescens from negative species like Pseudomonas putida.
- It is used in the identification of spore forming bacteria. Important species of Bacillus and Clostridium can be identified because many of them produce gelatinase.
- It is used for assessing bacterial virulence. Gelatinase producing organisms can degrade extracellular matrix and this indicates tissue invading ability of the organism.
- It is used in environmental and sanitary testing. Nutrient gelatin medium can be used for microbial plate count of proteolytic organisms in water, sewage and other sanitary materials.
Limitations of Gelatin Hydrolysis Test
- Gelatin is temperature sensitive and it liquefies naturally above about 20°C to 28°C. Therefore the tubes must be chilled properly before reading the result. If the tubes are not cooled, false positive result may be obtained.
- The tube should not be shaken or inverted while it is warm. In many cases gelatinase activity occurs only near the surface of the medium and shaking can mix liquefied gelatin with solid part of medium. This may lead to false negative interpretation.
- Some fastidious organisms and obligate anaerobes grow poorly in ordinary nutrient gelatin medium. Due to poor growth the test may become false negative.
- Gelatin is sensitive to excessive heat during sterilization. Over autoclaving may hydrolyze the gelatin and destroy its gelling property permanently. In such case the test becomes invalid.
- The gelling strength of gelatin may vary in different preparations. For this reason an uninoculated control tube is required for proper comparison and correct interpretation.
- In some media or saline suspensions prolonged incubation may sometimes produce false positive liquefaction.
Advantages of Gelatin Hydrolysis Test
- It is a simple and easy test for the detection of gelatinase production by microorganisms.
- The standard nutrient gelatin stab method is safe and does not require addition of hazardous chemical reagent or indicator for interpretation of result.
- It is useful in laboratories with limited resources where expensive molecular methods are not available. It gives a definite phenotypic result for identification of organisms.
- The result is easy to observe and interpret. It is based on the physical change of gelatin from solid to liquid.
- It is useful for assessing virulence of bacteria because gelatinase acts as a virulence factor and helps in tissue invasion.
- It is an important biochemical test for differentiation of closely related species. It helps in distinguishing pathogenic gelatinase positive Staphylococcus aureus from non-pathogenic Staphylococcus epidermidis.
- It is also useful in the identification of some bacterial genera such as Serratia, Proteus, Bacillus, Clostridium and Pseudomonas.
- This test is methodologically adaptable. Besides the standard tube method some rapid enzymatic methods such as gelatin strips and charcoal-gelatin discs are also used and the result can be obtained within short time.
Precautions of gelatin hydrolysis test
- The tubes should always be chilled in refrigerator or ice bath before reading the result. This is necessary because gelatin becomes liquid at incubation temperature above 20°C to 28°C.
- The tubes should not be shaken, tipped or inverted before and during cooling. Gelatinase activity usually occurs at the surface of the medium and shaking may mix the liquefied part with the solid part. This may give false negative result.
- An uninoculated control tube should always be kept with the test tube. It is used to verify the gelling property of the gelatin medium.
- The result should not be interpreted until the control tube becomes completely and firmly solidified.
- Over sterilization of the medium should be avoided. Excess heating or prolonged autoclaving may hydrolyze the gelatin and destroy its ability to form solid gel.
- Negative result should be considered only when sufficient bacterial growth is present in the medium. Some fastidious organisms grow poorly and may give false negative enzymatic result.
- It should be remembered that some organisms are slow liquefiers. Therefore prolonged incubation may be required and the tubes should be observed up to 14 days or more when necessary.
- Gelatin agar plate method is not suitable for fastidious organisms and obligate anaerobes in many cases.
Organisms giving positive and negative result in gelatin hydrolysis test
Organisms giving positive result
- Staphylococcus aureus – It produces gelatinase and liquefies gelatin.
- Bacillus species such as Bacillus subtilis, Bacillus anthracis and Bacillus cereus – These are gelatin hydrolysis positive organisms.
- Pseudomonas species such as Pseudomonas aeruginosa, Pseudomonas fluorescens, Pseudomonas stutzeri and Pseudomonas cepacia – These organisms hydrolyze gelatin.
- Serratia species such as Serratia marcescens, Serratia liquefaciens and Serratia odorifera – These are gelatinase producing organisms.
- Proteus species such as Proteus vulgaris, Proteus mirabilis and Proteus myxofaciens – It gives positive gelatin hydrolysis test.
- Clostridium species such as Clostridium perfringens and Clostridium tetani – These organisms liquefy gelatin.
- Flavobacterium species – It gives positive result in gelatin hydrolysis test.
- Enterococcus faecalis – Some specific clinical and laboratory strains gives positive result.
Organisms giving negative result
- Staphylococcus epidermidis – It does not produce gelatinase and gelatin remains solid.
- Escherichia coli – It gives negative gelatin hydrolysis test.
- Pseudomonas putida – It is gelatinase negative organism.
- Salmonella species – These are generally negative except some rare subspecies such as Salmonella arizonae.
- Shigella species – It does not hydrolyze gelatin.
References
- American Society for Microbiology. (2012, November 1). Protocols gelatin hydrolysis test.
- BYJU’S. (n.d.). Ninhydrin test.
- Cathcart, G. R. A., Quinn, D., Greer, B., Harriott, P., Lynas, J. F., Gilmore, B. F., & Walker, B. (2011). Novel inhibitors of the Pseudomonas aeruginosa virulence factor LasB: A potential therapeutic approach for the attenuation of virulence mechanisms in pseudomonal infection. Antimicrobial Agents and Chemotherapy, 55(6), 2670–2678.
- Comprehensive analysis of microbial gelatin hydrolysis: Principles, diagnostic methodologies, and pathogenic implications. (n.d.).
- dela Cruz, T. E. E., & Torres, J. M. O. (2012, November 1). Gelatin hydrolysis test protocol. American Society for Microbiology.
- Gelatin agar medium for detecting gelatinase production by anaerobic bacteria. (n.d.). PubMed Central (PMC).
- Gelatin as it is: History and modernity. (n.d.). PubMed Central (PMC).
- Gelatin hydrolysis test. (n.d.). Slideshare.
- Gelatinase regulates the egress of intracellular replicating populations during Enterococcus faecalis infection. (n.d.). bioRxiv.
- Hardy Diagnostics. (2020). Nutrient gelatin [Instructions for Use].
- Hartline, R. (2023, February 16). 3.21: Gelatin hydrolysis. Biology LibreTexts.
- HiMedia Laboratories. (2011). Nutrient gelatin M060 [Technical Data].
- Jahn, W. (n.d.). MICROBIOLOGY: GELATIN HYDROLYSIS TEST [Video]. YouTube.
- Kart, D., & Kuştimur, A. S. (2019). Investigation of gelatinase gene expression and growth of Enterococcus faecalis clinical isolates in biofilm models. Turkish Journal of Pharmaceutical Sciences, 16(3), 356–361.
- Key Scientific Products. (n.d.). K250 gelatin strips.
- McDade, J. J., & Weaver, R. H. (1959). Rapid methods for the detection of gelatin hydrolysis. Journal of Bacteriology, 77(1), 60-64.
- Reynolds, J. (2024, February 6). 39: Gelatin hydrolysis. Biology LibreTexts.
- Sapkota, A. (2022, January 27). Gelatin hydrolysis test- Principle, procedure, types, result, uses. Microbe Notes.
- Sigma-Aldrich. (2018). 70151 Nutrient gelatin. Merck KGaA.
- Smith, M., & Selby, S. (2021, March 19). 3.3: Gelatin agar. Biology LibreTexts.
- Taylor & Francis. (n.d.). Gelatinase – Knowledge and references.
- VUMIE. (2022, June 6). Gelatin hydrolysis test. Virtual Microbiology Lab Simulator Software.
- Waters, C. M., Antiporta, M. H., Murray, B. E., & Dunny, G. M. (2003). Role of the Enterococcus faecalis GelE protease in determination of cellular chain length, supernatant pheromone levels, and degradation of fibrin and misfolded surface proteins. Journal of Bacteriology, 185(12), 3613–3623.
- Wikipedia contributors. (2025, July 12). Gelatinase. Wikipedia, The Free Encyclopedia.
- Zeng, J., Teng, F., & Murray, B. E. (2005). Gelatinase is important for translocation of Enterococcus faecalis across polarized human enterocyte-like T84 cells. Infection and Immunity, 73(3), 1606–1612.