Reverse CAMP test is a biochemical test that is used for presumptive identification of some pathogenic bacteria mainly Clostridium perfringens and also Arcanobacterium haemolyticum and Corynebacterium pseudotuberculosis.
It is based on the interaction of extracellular products of two different bacteria when they are grown on sheep blood agar plate. This interaction can be synergistic or inhibitory and it gives a typical hemolysis pattern.
In this test, a known CAMP-factor producing strain of Streptococcus agalactiae (Group B Streptococcus) is streaked down the center of the blood agar plate. The suspected organism (like C. perfringens) is streaked perpendicularly to it leaving a small gap of about 1 to 2 mm. The plate is incubated under anaerobic condition for about 24 to 48 hours at 37°C.
Positive reverse CAMP test for C. perfringens is seen as a distinct bow-tie or reversed arrowhead zone of enhanced hemolysis pointing towards the S. agalactiae streak. This enhanced hemolysis is produced due to synergistic action between alpha toxin (phospholipase C) of C. perfringens and CAMP factor of S. agalactiae.
In some organisms like Arcanobacterium haemolyticum or Corynebacterium pseudotuberculosis the reverse CAMP test is also performed by streaking the organism perpendicular to Staphylococcus aureus. In this case, positive reaction is seen as an arrow of no hemolysis because the phospholipase produced by test organism inhibits the normal hemolysis produced by S. aureus beta hemolysin.
Objective of Reverse CAMP test
- To identify Clostridium perfringens presumptively and differentiate it from other Clostridium species.
- To detect the production of alpha-toxin (Phospholipase C) by C. perfringens which gives a characteristic bow-tie or reversed arrowhead zone of enhanced hemolysis with Streptococcus agalactiae.
- To identify Arcanobacterium haemolyticum and differentiate it from Corynebacterium species normal flora and beta hemolytic streptococci like Streptococcus pyogenes.
- To demonstrate phospholipase D production in Arcanobacterium haemolyticum which can inhibit staphylococcal beta-lysin (arrow of no hemolysis) or can show synergistic hemolysis with Streptococcus agalactiae.
- To identify Corynebacterium pseudotuberculosis (veterinary and clinical use) by showing inhibition of beta hemolysin produced by Staphylococcus aureus.
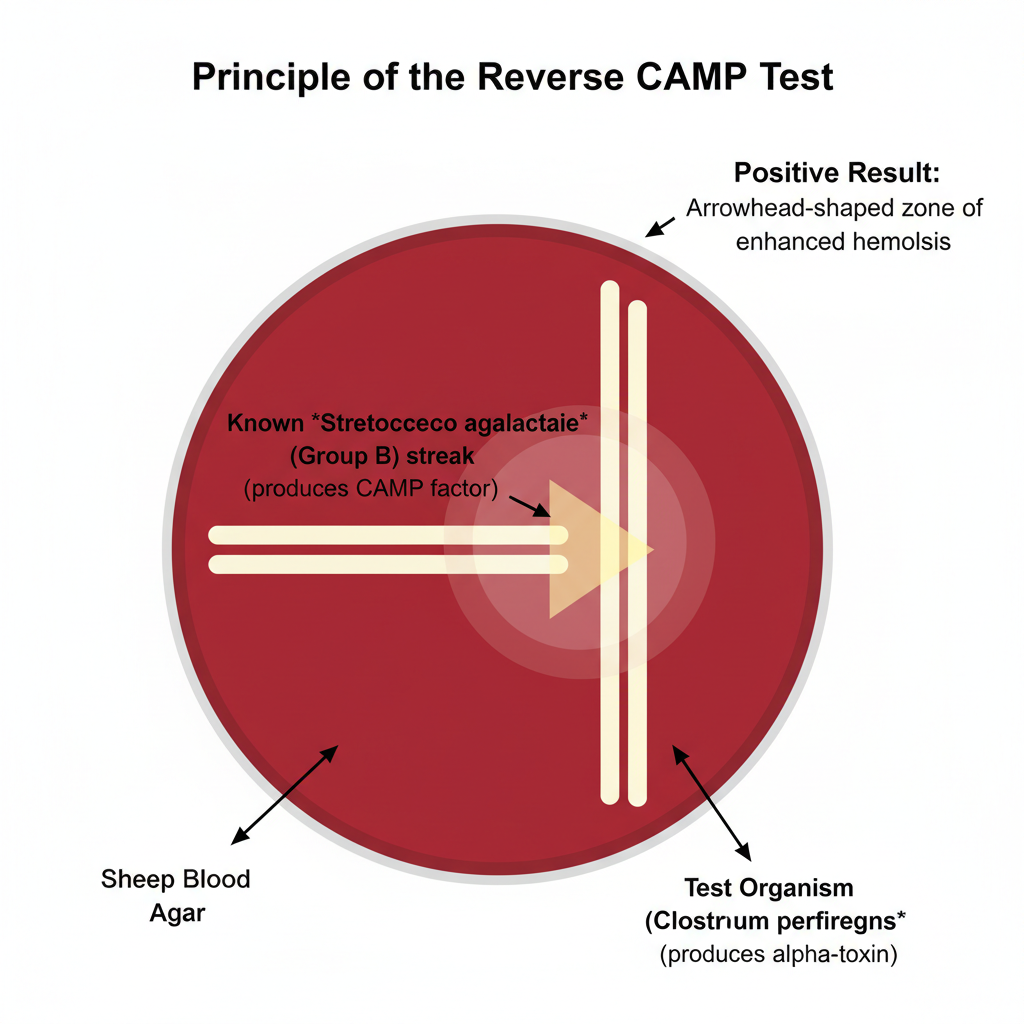
Principle of Reverse CAMP test
Reverse CAMP test works on the principle of interaction between extracellular hemolytic factors produced by two bacteria on sheep blood agar. It is mainly used for presumptive identification of Clostridium perfringens and Arcanobacterium haemolyticum.
In this principle, alpha toxin (phospholipase C) produced by test organism is allowed to diffuse in the agar and act on red blood cell membrane components. The alpha toxin hydrolyzes sphingomyelin of RBC membrane into ceramide, and the RBC becomes sensitized (metastable state).
After this, CAMP factor protein secreted by Streptococcus agalactiae binds non-enzymatically to the exposed ceramide and the lipid bilayer integrity is destroyed. This results in complete lysis of red blood cells and an enhanced hemolysis zone is produced.
The positive reaction is seen as a bow-tie or reversed arrowhead shaped hemolysis area which points towards the S. agalactiae streak. In case of Arcanobacterium haemolyticum, similar synergistic hemolysis can be seen with S. agalactiae or an inhibitory reaction can be seen against Staphylococcus aureus beta hemolysin showing an arrow of no hemolysis.
Material Required for Reverse CAMP test
- 5% Sheep blood agar plates (preferred medium due to high sphingomyelin content in sheep RBC).
- Known CAMP-positive Streptococcus agalactiae strain (Group B Streptococcus) to supply CAMP factor (Protein B).
- Suspected test organism (pure culture) most commonly Clostridium perfringens or Arcanobacterium haemolyticum.
- Anaerobic jar or anaerobic chamber to provide oxygen-free condition (required for C. perfringens growth and toxin production).
- Incubator set at 35°C to 37°C for incubation of plates for 24 to 48 hours.
- Inoculating loop/ needle/ sterile applicator sticks for streaking the organisms in straight perpendicular lines (kept about 1 to 4 mm gap and not touching).
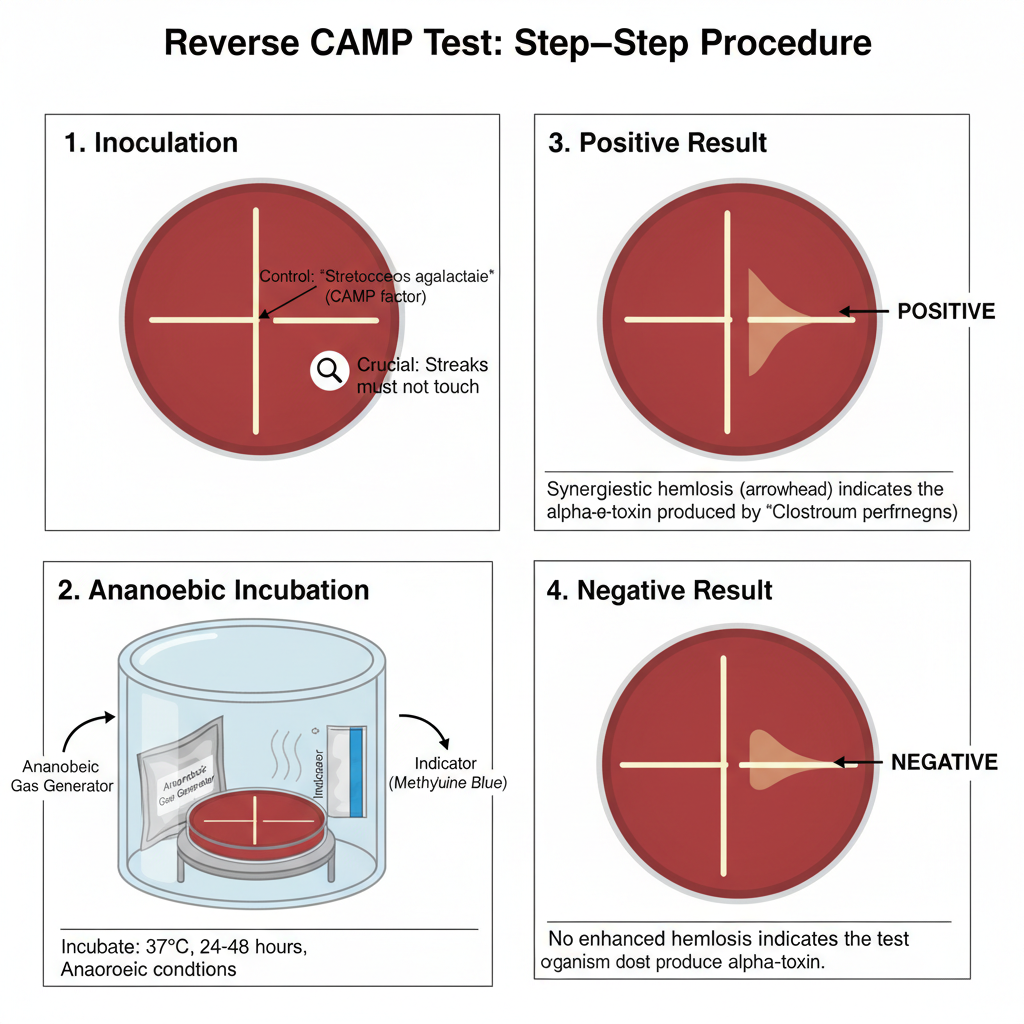
Procedure of Reverse CAMP Test
Procedure of Reverse CAMP test
- Fresh 5% sheep blood agar plate is taken and it is allowed to reach room temperature.
- A known CAMP-positive Streptococcus agalactiae (Group B Streptococcus) strain is streaked in a single straight line across the center/diameter of the plate.
- The suspected test organism (Clostridium perfringens or Arcanobacterium haemolyticum) is streaked in a straight line perpendicular (right angle) to the S. agalactiae streak.
- The streaks should not touch and a small gap is maintained between them (about 1 to 2 mm) to allow proper diffusion zone. (In some protocols, test organism is streaked at center and S. agalactiae is streaked at right angle).
- The plate is placed in anaerobic jar/chamber and incubated anaerobically at 37°C for 24 to 48 hours.
- After incubation, the area near the intersection is observed and a positive reverse CAMP test is indicated by arrowhead/bow-tie shaped zone of enhanced hemolysis pointing towards the center streak.
Results of Reverse CAMP Test

- Positive result for Clostridium perfringens– A distinct bow-tie/arrowhead/reversed arrow shaped zone of enhanced hemolysis is formed pointing towards the Streptococcus agalactiae streak. This indicates the test isolate produces sufficient alpha-toxin which acts synergistically with CAMP factor.
- Negative result for Clostridium perfringens– Growth of organism is seen but no enhanced hemolysis zone is visible at the junction area of the two streaks. It is used to differentiate the isolate from other clostridia like Clostridium septicum.
- Inhibitory result for Arcanobacterium haemolyticum (with Staphylococcus aureus)– The test organism inhibits the staphylococcal beta-lysin and an arrow of no hemolysis is seen or an indentation is formed in the linear hemolytic clearing.
- Synergistic result for Arcanobacterium haemolyticum (with Streptococcus agalactiae)– An arrowhead or crescent shaped zone of enhanced clearing is produced because phospholipase D acts as sensitizing factor and interacts with CAMP factor.

Uses of Reverse CAMP test
- It is used for presumptive identification of Clostridium perfringens in clinical laboratory and it helps in wound workup cases like suspected gas gangrene.
- It is used for confirmation of Arcanobacterium haemolyticum during pharyngeal screening in adolescent pharyngitis and it helps in differentiating it from commensal diphtheroids and beta hemolytic streptococci.
- It is used in veterinary diagnosis to identify Corynebacterium pseudotuberculosis (caseous lymphadenitis in sheep and goats) by showing inhibition of staphylococcal beta hemolysin.
- It is used as a screening method in bovine mastitis where reverse CAMP principle can be applied to detect synergistic pathogens in milk flora.
- It is used in food safety laboratory to confirm C. perfringens isolates from suspected food samples or patient fecal matter in food poisoning outbreaks.
- It is used in environmental and epidemiological studies to differentiate toxigenic C. perfringens from other non-toxigenic clostridia found in environment for epidemiological tracking.
Advantages of Reverse CAMP test
- High sensitivity– It is highly sensitive and it can identify most of the Clostridium perfringens isolates (up to about 97%) by giving clear positive reaction.
- Cost effective and easy availability– It uses standard 5% sheep blood agar which is commonly available and it is not needed to use expensive special media like egg yolk agar (Nagler reaction) which have shorter shelf life.
- Direct proof of toxin production– It provides phenotypic confirmation that the organism is actively producing toxin like alpha-toxin (phospholipase C) or phospholipase D unlike only molecular tests or MALDI-TOF.
- Clear visual interpretation– The reaction gives distinct bow-tie or reversed arrowhead zone which is easily recognized on the agar plate.
- Effective differentiation of pathogens– It helps in differentiating target organisms from similar species like C. perfringens from Clostridium septicum (negative) and Arcanobacterium haemolyticum from Streptococcus pyogenes and other commensal bacteria.
Limitations of Reverse CAMP test
- Strict media requirement– The test depends on sheep or bovine blood agar because high sphingomyelin content is needed for proper reaction. Blood from other species (human horse rabbit) may not give proper hemolysis and results becomes weak or absent.
- Risk of false negative– The reaction depends on amount of toxin secreted by organism. Some Clostridium perfringens strains may produce alpha-toxin in low level and visible synergistic hemolysis is not produced.
- Reagent strain variability– All Streptococcus agalactiae strains do not produce equal CAMP factor. A verified high CAMP producing strain is required otherwise reaction may not be clear.
- Strict incubation atmosphere– For Clostridium perfringens, incubation must be in strict anaerobic condition. If anaerobic condition is not maintained toxin production is reduced and interpretation can be incorrect (controls is needed).
- Slower turnaround time– The test needs 24 to 48 hours incubation so it is slower than rapid methods like MALDI-TOF. In Arcanobacterium haemolyticum, hemolysis is subtle and it may take up to 72 hours to develop.
- Agar quality dependency– If blood agar is poured too thin or already partially hemolyzed then synergistic reaction becomes feeble and it is difficult to interpret.
References
- American Society for Microbiology. (2009). Reverse CAMP test for the identification of Clostridium perfringens. ASM Image Gallery.
- Awad, M. M., Ellemor, D. M., Boyd, R. L., Emmins, J. J., & Rood, J. I. (2001). Synergistic effects of alpha-toxin and perfringolysin O in Clostridium perfringens-mediated gas gangrene. Infection and Immunity, 69(12), 7904–7910.
- Bae, S. Y., Choi, S., Kang, S. J., & Shin, J. H. (2010). A case of Arcanobacterium haemolyticum bacteremia and osteomyelitis diagnosed by 16s rRNA sequencing. Infection and Chemotherapy, 42(4), 241-243.
- Bernheimer, A. W., & Avigad, L. S. (1982). Mechanism of hemolysis by Renalin, a CAMP-like protein from Corynebacterium renale. Infection and Immunity, 36(3), 1253–1256. https://doi.org/10.1128/iai.36.3.1253-1256.1982
- Bernheimer, A. W., Linder, R., & Avigad, L. S. (1979). Nature and mechanism of action of the CAMP protein of group B streptococci. Infection and Immunity, 23(3), 838–844. https://doi.org/10.1128/iai.23.3.838-844.1979
- Camargo, A., Ramírez, J. D., Kiu, R., Hall, L. J., & Muñoz, M. (2024). Unveiling the pathogenic mechanisms of Clostridium perfringens toxins and virulence factors. Emerging Microbes & Infections, 13(1), 2341968. https://doi.org/10.1080/22221751.2024.2341968
- CAMP test. (2025, November 17). In Wikipedia. https://en.wikipedia.org/wiki/CAMP_test
- Centers for Disease Control and Prevention. (1971). Details – Public Health Image Library (PHIL).
- Consonni, A., Briozzo, E., Giubbi, C., Tonolo, S., Luzzaro, F., & Mauri, C. (2026). Insights into Arcanobacterium haemolyticum: A narrative review of an emerging pathogen revisited. Pathogens, 15(3), 335. https://doi.org/10.3390/pathogens15030335
- Gase, K., Ferretti, J. J., Primeaux, C., & McShan, W. M. (1999). Identification, cloning, and expression of the CAMP factor gene (cfa) of Group A Streptococci. Infection and Immunity, 67(9), 4725–4731. https://doi.org/10.1128/iai.67.9.4725-4731.1999
- Hansen, M. V., & Elliott, L. P. (1980). New presumptive identification test for Clostridium perfringens: Reverse CAMP test. Journal of Clinical Microbiology, 12(4), 617-619. https://doi.org/10.1128/jcm.12.4.617-619.1980
- Hanson, A. (2006). CAMP test protocols. American Society for Microbiology.
- Ilhan, Z. (2013). Detection of Corynebacterium pseudotuberculosis from sheep lymph nodes by PCR. Revue Méd Vét, 164(2), 60-66.
- Kang, H., Park, G., Kim, H., & Chang, K. (2016). Haemolytic differential identification of Arcanobacterium haemolyticum isolated from a patient with diabetic foot ulcers. JMM Case Reports, 3(1), e005016. https://doi.org/10.1099/jmmcr.0.005016
- Kiu, R., & Hall, L. J. (2018). An update on the human and animal enteric pathogen Clostridium perfringens. Emerging Microbes & Infections, 7(1), 141. https://doi.org/10.1038/s41426-018-0144-8
- Microbiology pictures. (2015). The CAMP test and the reverse CAMP test. www.microbiologyinpictures.com
- Microbiology pictures. (2015). The reverse CAMP test with Corynebacterium pseudotuberculosis and Staphylococcus aureus on blood agar. www.microbiologyinpictures.com
- Mubarak, A., & Moussa, I. M. (2022). Sero-diagnosis of Corynebacterium pseudotuberculosis affecting camels using recombinant phospholipase D (rPLD) and anti-camels IgY-conjugate. Journal of King Saud University – Science, 34(8), 102344. https://doi.org/10.1016/j.jksus.2022.102344
- Niyomdecha, N., Phakamas, W., & Nana, A. (2016). Modified human blood agar as substitute for sheep blood agar in laboratories of developing countries. Journal of Pure and Applied Microbiology, 10(3), 1773-1779. https://doi.org/10.22207/JPAM.10.3.11
- Pokhrel, P. (2015). Reverse CAMP test for the identification of Clostridium perfringens. Microbiology Notes.
- Rao, S. P. N. (2009). CAMP TEST. Microrao.
- Raza, A., Mahmood, S., Khan, A., Goshi, S., Khalid, R., Ijaz, M., Shaheen, M., Noor, A., Bano, S., Asghar, S., Afzal, T., & Akbar, Z. (2024). Mechanisms of action of toxins released by Clostridium perfringens. BioScientific Review, 6(1), 16-33. https://doi.org/10.32350/bsr.61.i
- Sakurai, J., & Oda, M. (2004). Clostridium perfringens alpha-toxin: Characterization and mode of action. Journal of Biochemistry, 136(5), 569-574. https://doi.org/10.1093/jb/mvh161
- Sapkota, A. (2022). CAMP test- principle, procedure, types, results, uses, limitations. Microbe Notes.
- Syame, S. M., Abuelhag, H. A., Kandil, M. M., Gaber, E. S., & Ebessy, E. A. (2021). Bacteriological, molecular and histopathological evaluation of four vaccines’ protective role against caseous lymphadenitis in sheep. International Journal of Health Sciences, 6(S6), 9941–9954. https://doi.org/10.53730/ijhs.v6nS6.12561
- The reverse CAMP test: Analytical mechanisms, biochemical principles, and clinical utility in diagnostic microbiology. (n.d.).
- Vu, M. L. D., & Rajnik, M. (2024). Arcanobacterium haemolyticum infections. In StatPearls. StatPearls Publishing.
- What is the difference between CAMP and reverse CAMP test. (n.d.). Difference Between.
- Yeh, E., Pinsky, B. A., Banaei, N., & Baron, E. J. (2009). Hair sheep blood, citrated or defibrinated, fulfills all requirements of blood agar for diagnostic microbiology laboratory tests. PLoS ONE, 4(7), e6141. https://doi.org/10.1371/journal.pone.0006141
- Zwart, M. (n.d.). Reverse CAMP test Clostridium perfringens – Microbiology [Video]. YouTube.