Native PAGE is a gel electrophoresis technique used for separation of proteins and nucleic acids. It is called native because the molecule remains in its natural form. The protein is not unfolded during the process.
In this method, SDS, urea and reducing agents are not used. So the protein keeps its original charge, shape and three dimensional structure. Its biological activity may also remain present.
The separation occurs in polyacrylamide gel under electric field. The molecules move through the gel according to their net charge, size and shape. So the movement is not only based on molecular weight.
Native PAGE is useful for studying protein-protein interaction. It is also used for subunit composition, multimeric protein complex and enzyme activity study. Active enzymes can sometimes be detected directly inside the gel.
The method was developed from early protein separation work. Ornstein and Davis developed native discontinuous gel system in 1964. It was used for serum protein separation without destroying their native structure.
In the same year, Kenneth A. Ferguson gave the Ferguson plot. It was used to estimate true size and mobility of macromolecules by using different gel concentration.
Later Blue Native PAGE (BN-PAGE) was introduced by Hermann Schägger and von Jagow in 1991. In this method, Coomassie blue dye is used. It helped to study membrane protein complexes like mitochondrial respiratory chain complexes in intact form.
Principle of Native Polyacrylamide Gel Electrophoresis (PAGE)
Principle of Native Polyacrylamide Gel Electrophoresis (PAGE) is based on the movement of proteins or macromolecules through polyacrylamide gel under electric field. The molecules move in the gel without losing their native structure.
In Native PAGE, SDS, urea and reducing agents are not used. So the protein is not denatured. It remains folded and keeps its own charge and shape.
The movement of protein is not only depend on molecular weight. It depends on net surface charge of the protein, its size and its three dimensional shape. The pH of running buffer also affect the charge of protein.
When electric current is applied, the charged proteins move through the gel pores. Smaller and more charged molecules may move faster. Large or less charged molecules move slowly.
Thus the proteins are separated in their natural form. Their biological activity may remain present. So Native PAGE is useful for studying enzyme activity, protein-protein interaction and multimeric protein complexes.
Types of Native Polyacrylamide Gel Electrophoresis
- Standard Native PAGE – It is the common type of native electrophoresis. It separates proteins by their own charge, size and shape. The protein remains in folded form.
- Discontinuous Native PAGE – In this type, discontinuous buffer system is used. It first concentrates the proteins into thin band. Then the proteins enter into resolving gel and separate.
- Anodic Native PAGE – It is used for acidic proteins. These proteins are negatively charged in the running buffer. They move towards the anode during electrophoresis.
- Cathodic Native PAGE – It is used for basic proteins. These proteins have pI more than 7.0. The polarity is reversed and positively charged proteins move towards the cathode.
- Blue Native PAGE (BN-PAGE) – It is used for membrane proteins and protein complexes. Coomassie Brilliant Blue G-250 is added in this method. It gives negative charge to the protein surface.
- Migration in BN-PAGE – In this method, the proteins are not unfolded. The dye helps the proteins to move towards the anode. It also prevents aggregation of hydrophobic proteins.
- Uses of BN-PAGE – It is useful for studying large protein assemblies. Mitochondrial respiratory chain complexes and other multiprotein complexes can be separated in intact form.
- Clear Native PAGE (CN-PAGE) – It is similar to BN-PAGE, but dye is not used. The separation depends on natural charge and size of the protein.
- Features of CN-PAGE – It gives mild condition for proteins. It is useful for fragile protein complexes. But the resolution may be lower than BN-PAGE.
- Uses of CN-PAGE – It is used when Coomassie dye may disturb the next analysis. It is useful for FRET study and in-gel enzyme activity assay.
- High-resolution Clear Native PAGE (hrCNE) – It is improved form of clear native electrophoresis. It uses mixed micelles of sodium deoxycholate and mild detergent like DDM in cathode buffer.
- Principle of hrCNE – These mixed micelles give slight negative charge to the proteins. It prevents aggregation and gives better band separation. Dye is not used in this method.
- Quantitative Preparative Native Continuous PAGE (QPNC-PAGE) – It is a special native electrophoresis method. It is used for isolation and quantification of active metalloproteins.
- Process of QPNC-PAGE – It uses basic buffer of about pH 10.0 and large pore gel. The gel is prepared for long time to make gentle separation. The separation occurs mainly by isoelectric point.
- Use of QPNC-PAGE – In this method, the proteins remain folded with their metal cofactors. So metalloproteins can be collected and studied later by mass spectrometry or NMR spectroscopy.

Requirements for Polyacrylamide Gel Electrophoresis (PAGE)
- Acrylamide – It is the main chemical used for making the gel. It forms the gel matrix after polymerization. The molecules move through this porous gel.
- Bis-acrylamide – It acts as cross-linking agent. It joins the acrylamide chains. Due to this, proper pore size is formed in the gel.
- Ammonium persulfate (APS) – It is used as initiator. It produces free radicals. These free radicals start the polymerization of acrylamide.
- TEMED – It is used as catalyst. It helps APS to form free radicals quickly. So the gel sets in proper time.
- Running buffer – It is poured in electrophoresis tank. It conducts electric current during the run. Tris-Glycine or Bis-Tris buffer may be used.
- Sample loading buffer – It is mixed with sample before loading. It contains glycerol which makes the sample heavy. So the sample settles inside the well.
- Tracking dye – It is present in loading buffer. Bromophenol blue or phenol red may be used. It helps to see how far the electrophoresis has run.
- Electrophoresis chamber – It holds the gel and buffer. The chamber may be vertical or horizontal. In PAGE, vertical chamber is generally used.
- Power supply – It gives electric current to the chamber. The molecules move in the gel due to this electric field. Voltage or current is adjusted by this unit.
- Glass plates and spacers – Glass plates are used for casting the gel. Spacers keep fixed distance between two plates. This gives proper gel thickness.
- Comb – Comb is placed during gel casting. After gel setting, comb is removed and wells are formed. The samples are loaded into these wells.
- SDS – In SDS-PAGE, sodium dodecyl sulfate is used. It denatures the proteins and gives uniform negative charge. It is not used in normal Native PAGE.
- Reducing agents – DTT or beta-mercaptoethanol may be used in denaturing PAGE. They break disulfide bonds of proteins. This helps in complete unfolding.
- Mild detergents – In Native PAGE, mild detergents like digitonin or DDM may be used. These help to solubilize membrane proteins without unfolding them.
- Coomassie Blue G-250 – It is used in Blue Native PAGE. It gives charge shift to proteins but does not denature them. It helps in separation of protein complexes.
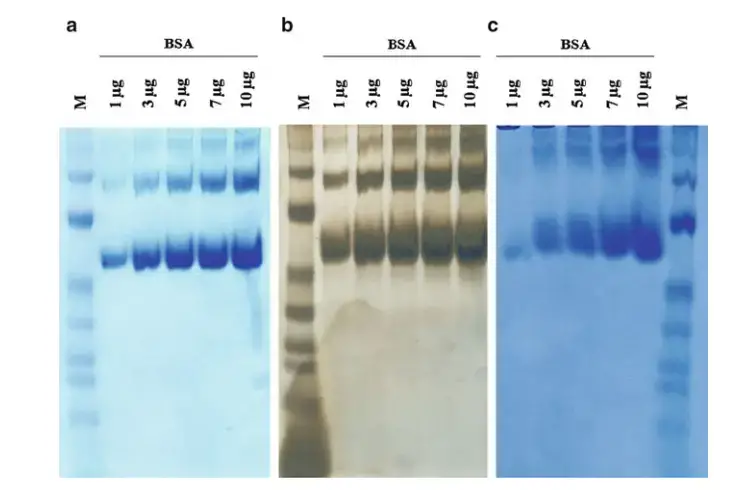
- Staining reagents – After electrophoresis, staining is done to see the bands. Coomassie blue, silver stain or fluorescent stain may be used. The separated proteins become visible after staining.
Sample Preparation for Polyacrylamide Gel Electrophoresis (PAGE)
- The sample is first lysed properly. Cell culture or tissue is broken by sonication or homogenization. This helps to release the proteins from the cells.
- During lysis, protease inhibitor is added. It prevents protein degradation. Phosphatase inhibitor may also be added when phosphorylation study is needed.
- The lysate is then centrifuged at high speed. It may be around 20,000 × g. This removes cell debris, aggregates and insoluble particles.
- Clear supernatant is taken after centrifugation. If debris remain in the sample, the protein may not enter the gel properly. It may also cause vertical smearing.
- Protein concentration is measured before loading. This is done to load equal and proper amount of protein. Usually about 10 µg protein may be loaded in one well.
- Too much sample should not be loaded. Overloading gives smeared, distorted or U-shaped bands. Very less sample gives faint or invisible bands.
- The sample is mixed with loading buffer. The loading buffer contains glycerol. Glycerol makes the sample heavy so that it settles at the bottom of the well.
- Tracking dye is also present in loading buffer. Bromophenol blue or phenol red may be used. It helps to follow the movement of electrophoresis.
For SDS-PAGE
- In SDS-PAGE, the sample is mixed with sample buffer containing 1-2% SDS or LDS. It unfolds the proteins and gives uniform negative charge.
- Reducing agent is added in the sample buffer. DTT, beta-mercaptoethanol (BME) or TCEP may be used. These break the disulfide bonds.
- The sample is heated before loading. It is usually heated at 95°C for 5-10 minutes. In some buffer system, 70-85°C for few minutes is also used.
- Heating helps in complete denaturation of protein. After this, the proteins become linear and can be separated mainly according to molecular weight.
For Native PAGE
- In Native PAGE, SDS, urea and reducing agents are not used. These chemicals are avoided because the protein should remain in native form.
- The sample is not boiled or heated. Heating can denature the protein. It may destroy the biological activity and subunit interaction.
- The sample is mixed with native loading buffer and loaded directly into the gel. The protein keeps its natural charge, shape and three dimensional structure.
- Salt concentration of the sample should be low. High salt may cause band distortion, streaking and heating during electrophoresis. The ionic strength is preferably kept below 0.1 M.
- For membrane proteins in Blue Native PAGE, mild detergent is used. DDM or digitonin helps to solubilize membrane protein without breaking the complex.
- In BN-PAGE, Coomassie Blue G-250 is added after solubilization. It gives negative charge shift to the proteins and helps their movement in the gel.
Buffer Preparation for Polyacrylamide Gel Electrophoresis (PAGE)
A. SDS-PAGE Buffers
- SDS sample buffer – This buffer is used for preparing denatured protein sample. It contains 63 mM Tris-HCl (pH 6.8), 10% glycerol, 2% SDS, 0.0025% bromophenol blue and 50 mM DTT. SDS unfolds the protein and DTT breaks disulfide bonds.
- SDS running buffer – This buffer is used in the electrophoresis tank. It contains 25 mM Tris base, 192 mM glycine and 0.1% SDS. The pH is kept about 8.3. It helps the current to pass through the gel.
- Transfer buffer – This buffer is used after SDS-PAGE for Western blot. It contains 25 mM Tris, 192 mM glycine, 10% methanol and 0.1% SDS. It helps protein to move from gel to membrane.
B. Native PAGE Buffers
- Native sample buffer – This buffer is used for native protein sample. It contains 100 mM Tris-HCl (pH 8.6), 10% glycerol and 0.00025% bromophenol blue. SDS and reducing agent are not added. The sample is not heated.
- Native running buffer – This buffer is used for running native gel. It contains 25 mM Tris base and 192 mM glycine. The pH is about 8.3. It allows protein to move according to its own charge and shape.
C. Blue Native PAGE Buffers
- Cathode buffer – It contains 50 mM Tricine, 15 mM Bis-Tris and 0.02% Coomassie blue G-250. The pH is about 7.0. Coomassie blue gives negative charge to protein complex.
- Anode buffer – It contains 50 mM Bis-Tris. The pH is about 7.0. It is used in anode chamber during BN-PAGE.
- Sample buffer – For water soluble protein, it contains 75 mM Bis-Tris, 750 mM 6-Aminocaproic acid and 10% glycerol. The pH is kept about 7.0. It keeps the protein complex in native condition.
- Membrane protein buffer – For membrane protein, mild detergent is added. Triton X-100 or extra Coomassie blue may be used. It helps to solubilize the membrane protein without breaking the complex.
- 3X gel buffer – It contains 150 mM Bis-Tris and 1.5 M 6-Aminocaproic acid. The pH is adjusted to 7.0. It is used during gel preparation.
D. Cathodal Native PAGE Buffers
- Cathode buffer – This buffer is used for basic proteins having pI > 7.0. It contains 30 mmol/L acetic acid, 0.01% sodium azide and 0.001% Basic blue. The pH is about 3.6.
- Anode buffer – It contains 113 mmol/L ε-aminocaproic acid, 5 mmol/L acetic acid and 0.01% sodium azide. The pH is about 5.8.
- Gel buffer – It contains 0.6 mol/L HEPES, 10 mmol/L arginine, 1 mmol/L acetic acid, 0.01% sodium azide and 0.001% Pyronin. The pH is about 5.5. It is used for preparing gel in cathodal native system.
Procedure of Native Polyacrylamide Gel Electrophoresis
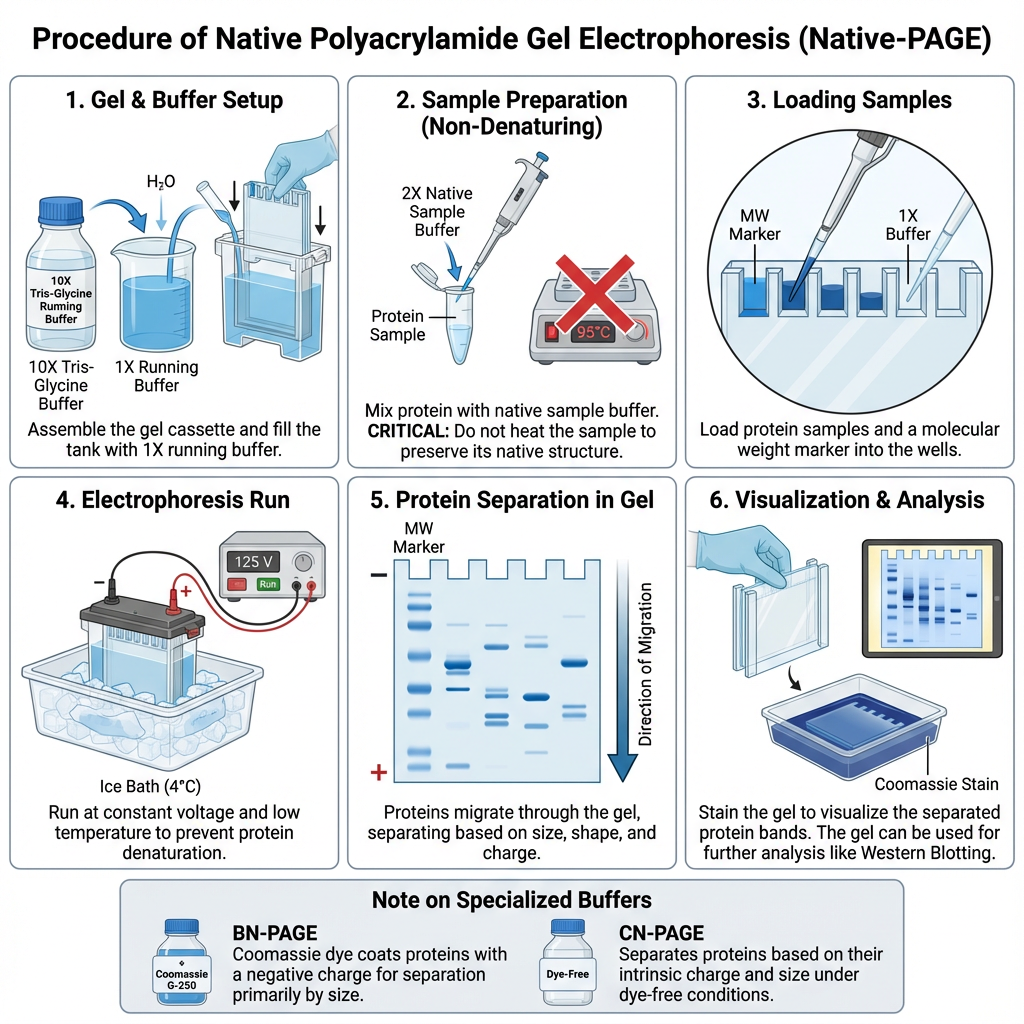
- The glass plates are assembled first. The plates should be clean and fixed properly. Then the polyacrylamide gel is prepared.
- The resolving gel is poured first between the glass plates. It is allowed to polymerize. After this, stacking gel is poured above it.
- The comb is inserted in the stacking gel. It makes wells for loading the sample. After polymerization, the comb is removed slowly.
- The protein sample is mixed with native loading buffer. The buffer contains glycerol and tracking dye. Glycerol helps the sample to settle inside the well.
- SDS and reducing agents are not added in the sample. DTT or beta-mercaptoethanol is also avoided. These can destroy the native condition of protein.
- The sample is not heated or boiled. Heating denature the protein and changes its natural shape. So the sample is loaded in unheated condition.
- The prepared sample is centrifuged for short time. Insoluble particles and protein aggregates are removed. This prevents streaking in the gel.
- The gel cassette is placed in electrophoresis chamber. Upper and lower reservoirs are filled with native running buffer. In some method, gel is pre-run for 30 to 60 minutes.
- The sample is loaded carefully into the wells by micropipette. The salt concentration of sample should be low. High salt may produce distorted bands.
- The chamber is connected with power supply. The gel is run at low voltage, generally 100-150 V. High voltage is avoided because heat may denature the proteins.
- The run is preferably done at cold condition. Pre-cooled buffer or cold room at 4°C may be used. This reduces heating and keeps the protein in native form.
- Electrophoresis is continued until the tracking dye reaches near the bottom of the gel. Then the current is stopped.
- The gel is removed carefully from the cassette. It may be used for Coomassie staining, silver staining, Western blotting or in-gel enzyme activity assay.
- In enzyme activity assay, the protein should remain active in the gel. So harsh staining or denaturing condition is avoided according to the experiment.
SDS PAGE vs. Native PAGE
| SDS-PAGE | Native PAGE |
|---|---|
| It separates proteins mainly by molecular weight. | It separates proteins by size, charge and shape. |
| SDS is used in this method. | SDS is not used in this method. |
| Protein is unfolded into linear chain. | Protein remains in folded native form. |
| Sample is usually heated before loading. | Sample is not heated or boiled. |
| Reducing agents like DTT or beta-mercaptoethanol may be used. | Reducing agents are avoided. |
| Disulfide bonds are broken in reduced SDS-PAGE. | Disulfide bonds and subunit interaction are mostly preserved. |
| SDS gives uniform negative charge to proteins. | Protein keeps its own natural charge. |
| Natural charge of protein is masked by SDS. | Charge depends on buffer pH and protein pI. |
| Protein structure and activity are lost. | Protein structure and activity remain present. |
| Protein complexes are usually dissociated. | Protein complexes can remain intact. |
| It is used to find molecular weight of protein. | It is used to study protein complex and interaction. |
| It is useful for checking purity and expression level. | It is useful for enzyme activity and active protein forms. |
| It is commonly used before Western blotting. | It can be used for in-gel activity assay like zymography. |
| Functional protein recovery is difficult. | Functional protein may be recovered from gel in some cases. |
Applications of Native Polyacrylamide Gel Electrophoresis (PAGE)
- Native PAGE is used to study protein structure in native condition. The protein remains folded. So its natural shape and active form can be observed.
- It is used to study protein complexes. Multimeric proteins and subunit arrangement can be checked. The complex does not break easily because SDS is not used.
- It is used for protein-protein interaction study. If two proteins bind with each other, their movement in gel may change. This gives idea about interaction.
- It is used for protein-ligand interaction also. Ligand binding can change the mobility of protein. So bound and unbound forms can be compared.
- It is used in protein-nucleic acid interaction study. EMSA is one common method. It shows the binding of protein with DNA or RNA.
- It is used for in-gel enzyme activity assay. The enzyme remains active in the gel. Protease, lipase and matrix metalloproteinases can be detected by this method.
- Blue Native PAGE (BN-PAGE) is used for membrane protein study. Hydrophobic membrane protein complexes can be separated without breaking their subunits.
- It is useful for mitochondrial protein complexes. OXPHOS complexes can be studied in intact form. Their arrangement and activity can be checked.
- It is used in clinical diagnosis. Isoenzymes like Lactate Dehydrogenase (LDH) can be separated. These may indicate liver or heart damage.
- It is used for biomarker profiling. Disease related protein forms can be analyzed. Misfolded metalloproteins in disorders like Alzheimer’s disease may also be studied.
- It is used for purification of active proteins. The protein can be recovered from gel in functional form. QPNC-PAGE is used for this type of preparative work.
- The purified native proteins can be used for further analysis. NMR spectroscopy, ICP-MS, FRET and mass analysis may be done after separation.
Advantages of Native Polyacrylamide Gel Electrophoresis (PAGE)
- Native PAGE keeps the protein in natural form. The protein remains folded. Its own charge, shape and biological activity are maintained.
- It is useful for studying protein complexes. The subunits remain attached in many cases. So multimeric protein structure can be observed.
- It is used for protein-protein interaction study. Since SDS and reducing agents are not used, weak non-covalent interactions can remain present.
- It is used for protein and nucleic acid binding study. Protein with DNA or RNA can be separated. The shifted band shows the binding condition.
- Enzyme activity can be detected directly in the gel. The protein remains active after electrophoresis. Zymography is one example of this use.
- Cofactors and metal ions may remain attached with the protein. So catalytic activity of enzyme or metalloprotein can be studied in native condition.
- It can separate proteins having same molecular weight. The separation also depends on charge and shape. So proteins of same size but different conformation can be resolved.
- Active protein can be recovered from the gel. The band is cut and protein may be eluted. It can be used for functional assay, crystallization or mass analysis.
- It helps to know subunit composition of large protein complex. Oligomeric state and homogeneity of the complex can be checked.
- It is useful in clinical and diagnostic work. Changes in native protein complexes can act as biomarker. It helps in study of neurodegenerative and metabolic disorders.
Limitations of Native Polyacrylamide Gel Electrophoresis (PAGE)
- Native PAGE does not give exact molecular weight easily. The movement of protein depends on size, charge and shape. So one band position cannot show only molecular weight.
- Different proteins may migrate to same place in gel. One protein may be small but less charged. Another protein may be larger but more charged. Due to this, interpretation becomes difficult.
- Native protein state is difficult to maintain. During electrophoresis, heat may be produced. This is called Joule heating.
- Heat can denature sensitive proteins. It may also make curved or smiling bands in the gel. So the gel is often run at low voltage and cold condition.
- The sample cannot be heated. SDS, urea and reducing agents are not added. Because of this, some proteins may aggregate before or during loading.
- High salt concentration in sample is not suitable. More than about 0.1 M salt may cause streaking and band distortion. It may also increase heating during run.
- Buffer pH should be selected properly. If the pH is close to the protein pI, the protein becomes nearly neutral. Then it may not move properly in the gel.
- It needs knowledge of protein pI. Without knowing the approximate pI, proper running condition is difficult to select.
- Standard Native PAGE and Clear Native PAGE (CN-PAGE) may give lower resolution. The bands may not be as sharp as SDS-PAGE or Blue Native PAGE.
- In Blue Native PAGE, Coomassie dye may disturb later analysis. It can interfere with mass spectrometry. It also reduces fluorescence signal strongly.
- Native gel preparation is more technical. Continuous gel or gradient gel may be needed. So it is more difficult than ordinary SDS-PAGE in many cases.
- Protein activity may still be lost if condition is not correct. Wrong buffer, high voltage, high temperature or long run can affect the native structure.
Agarose vs polyacrylamide gel electrophoresis
| Agarose Gel Electrophoresis | Polyacrylamide Gel Electrophoresis |
|---|---|
| Agarose gel is made from agarose powder. | Polyacrylamide gel is made from acrylamide and bis-acrylamide. |
| It is prepared by heating agarose in buffer. | It is prepared by chemical polymerization reaction. |
| The melted agarose is cooled at room temperature. Then gel becomes solid. | APS and TEMED are added. These start polymerization and gel is formed. |
| Pore size depends on the percentage of agarose used. | Pore size depends on percentage of acrylamide and cross-linker. |
| It is mostly used for DNA and RNA separation. | It is mostly used for protein separation. |
| It is used for general nucleic acid electrophoresis. | It is used in SDS-PAGE and Native PAGE. |
| It is suitable for larger nucleic acid fragments. | It is suitable for small fragments, usually below 1000 base pairs. |
| The gel preparation is simple and easy. | Gel preparation is more technical and needs careful handling. |
| Agarose gel has larger pores. | Polyacrylamide gel has smaller and more controlled pores. |
| It gives lower resolution for very small fragments. | It gives high resolution for small proteins or small nucleic acid fragments. |
| It can be used for some non-denaturing protein work. Example, LDH isoenzyme study. | It is standard gel matrix for protein analysis and native protein study. |
| During boiling, water loss should be adjusted. Otherwise gel concentration may become high. | During preparation, acrylamide should be handled carefully because it is toxic before polymerization. |
Precautions of Polyacrylamide Gel Electrophoresis
- Gloves, lab coat and protective glass should be used during PAGE. The chemicals and buffer should not touch the skin.
- Acrylamide and bis-acrylamide are toxic before polymerization. They are neurotoxic chemicals. Direct contact and inhalation of powder should be avoided.
- If acrylamide solution spills, it should be cleaned immediately. The waste should be discarded in proper chemical waste container.
- Some buffers may contain sodium azide. It is toxic preservative. So native buffers and ready-made reagents should be handled carefully.
- Gel solution should be degassed before pouring. Oxygen can stop free radical formation. Due to this, polymerization may become poor.
- Resolving gel should be overlaid with water or n-butanol after pouring. It gives flat upper surface. It also helps the gel to polymerize properly.
- Gel should be allowed to polymerize completely. If gel is not set properly, the pores become uneven. Then bands do not separate clearly.
- Comb should be removed slowly from stacking gel. If it is pulled roughly, the wells may tear. Damaged wells give poor sample loading.
- Samples should be centrifuged before loading. Cell debris, aggregates and insoluble particles are removed. Otherwise they may block the well and cause vertical streaking.
- Salt concentration of sample should be low. High salt causes band distortion and heating. It may also make the migration uneven.
- Protease inhibitor and phosphatase inhibitor should be added in lysate when needed. It prevents degradation of proteins. Repeated freezing and thawing should be avoided.
- Wells should be washed with running buffer before loading. This removes unpolymerized acrylamide or other residues. Then sample can enter the well properly.
- Sample should be loaded carefully by micropipette. Air bubbles should not be formed inside the well. The pipette tip should not puncture the gel.
- Too much protein should not be loaded in one well. Overloading gives fuzzy, smeared or U-shaped bands. Around 10 µg protein per well is generally used.
- Empty wells should not be left completely empty. Sample buffer or blank sample may be added. This helps to reduce edge effect in nearby lanes.
- The gel should not be overheated during run. Joule heating can make bands diffuse or smiling type. Low voltage, cold buffer or 4°C condition may be used.
- Fresh running buffer should be used. The pH and ionic strength should be correct. Old buffer may change the movement of proteins.
- Electrode connection should be checked before starting. The polarity should be correct. If it is reversed, proteins may move upward and run out of the gel.
Common Troubleshoot of Polyacrylamide Gel Electrophoresis
- No bands visible – This may occur when protein runs out of the gel. Run time may be too long or voltage may be too high. The electrode connection may also be reversed, so sample moves in wrong direction.
- Less protein loading – If very little protein is loaded, the band may not be seen. Proper amount of sample should be loaded. The sample should not be allowed to diffuse from well before current is started.
- Fuzzy or smeared bands – This is commonly due to overloading of protein. Too much sample in one well gives smear instead of sharp band. Protein amount should be reduced.
- Incomplete denaturation – In SDS-PAGE, smear may come if protein is not denatured properly. SDS, reducing agent or heating may be insufficient. Proper sample buffer and heating should be used.
- Protein degradation – Extra bands or smear may occur due to degraded protein. Protease inhibitor should be added during lysis. Repeated freezing and thawing should be avoided.
- Smiling bands – Bands become curved upward at the edges due to heating. This is called smiling. The gel should be run at low voltage or with pre-cooled buffer.
- Uneven heat distribution – If heat is not uniform, band shape becomes distorted. The run may be done in cold room or on ice. This reduces Joule heating.
- Sample leaking from wells – This happens when wells are damaged. Comb may be removed roughly or pipette tip may puncture the gel. The comb should be removed slowly and loading should be careful.
- Overfilled wells – If well is filled too much, sample may come out. The well should not be filled more than about three fourth part. Air bubbles should also be avoided.
- Sample floating – Sample may float instead of settling in well. This occurs when loading buffer has less glycerol. Proper glycerol should be present to make sample heavy.
- Too many bands – Extra bands may come due to degradation or aggregation of protein. In SDS-PAGE, proper DTT or BME and heating should be used. For native work, aggregation should be avoided by proper buffer condition.
- Poor resolution – Bands may remain too close together. Run time may be short or gel percentage may be wrong. Correct acrylamide concentration should be selected according to protein size.
- Need of gradient gel – If proteins have wide size range, normal gel may not separate well. Gradient gel can be used. It helps to separate both small and large proteins.
- Distorted outer lanes – Empty wells near sample lanes may cause edge effect. The outer bands become distorted. Empty wells should be filled with blank sample buffer.
- Gel running too slow – This may occur when running buffer is too concentrated or voltage is low. Buffer should be prepared correctly. Voltage should be set properly.
- Gel running too fast – This may occur when buffer is too dilute or voltage is too high. The buffer concentration and voltage should be checked before running.
References
- Advantages and limitations of clear-native PAGE. (n.d.). PubMed.
- Analysis of RNA folding by native polyacrylamide gel. (n.d.). PMC – NIH.
- Blue native PAGE: Applications to study peroxisome biogenesis. (n.d.). PubMed.
- Liobikas, J., & Trumbeckaitė, S. (2008). Blue native polyacrylamide gel electrophoresis (BN-PAGE). bioch.eu.
- Liobikas, J., & Trumbeckaitė, S. (2008). Blue native polyacrylamide gel electrophoresis (BN-PAGE). bioch.eu.
- Blue native electrophoresis protocol. (n.d.). Abcam.
- Can anyone suggest why I cannot see the protein band when looking at my native gel? (n.d.). ResearchGate.
- Cathodal electrophoresis with native buffer kit pH 5.5. (1997). Amersham Pharmacia Biotech.
- Clear native gel electrophoresis for the purification of fluorescently… (n.d.). bioRxiv.
- Common artifacts and mistakes made in electrophoresis. (n.d.). PMC – NIH.
- Comprehensive analysis of native polyacrylamide gel electrophoresis: Principles, methodologies, and applications in macromolecular biochemistry. (n.d.).
- Controlling the separation of native proteins with temperature in thermal gel transient isotachophoresis. (n.d.). PMC.
- Differences between SDS PAGE and native PAGE. (2025, July 23). GeeksforGeeks.
- Niepmann, M., & Zheng, J. (2006). Discontinuous native protein gel electrophoresis. Electrophoresis.
- Diverse native-PAGE. (n.d.). Assay-Protocol.
- Heidersbach, R. (2013, December). Cathodic and anodic corrosion protection. Electrochemistry Encyclopedia.
- Ferguson analysis of protein electromigration during single-cell electrophoresis in an open microfluidic device. (n.d.). PMC.
- Gelatin zymography protocol. (n.d.). Abcam.
- How is native PAGE different from SDS-PAGE? (2025, May 9). Patsnap Synapse.
- How do native PAGE markers account for charge? (n.d.). ResearchGate.
- Interpretation & troubleshooting on SDS/native PAGE. (2012, December 15). Protocol Online.
- Lactate dehydrogenase (LDH) isoenzymes test. (2023, October 30). MedlinePlus.
- Lactate dehydrogenase (LDH) staining kit. (2000). Biomedical Research Service Center, University at Buffalo.
- Native PAGE separation technique file no. 120. (1998, August). Amersham Biosciences.
- Native PAGE vs SDS-PAGE: A practical guide to choosing the right electrophoresis technique. (2026, January 26). Biofargo.
- Native protein electrophoresis. (n.d.). National Diagnostics.
- NativePAGE™ sample prep kit. (2011, December 2). Life Technologies.
- Nucleic acid gel electrophoresis troubleshooting. (n.d.). Thermo Fisher Scientific.
- Protein electrophoresis buffers and reagents. (n.d.). Thermo Fisher Scientific.
- Protein electrophoresis methods. (n.d.). Bio-Rad.
- Proteomics/protein separations- electrophoresis/QPNC-PAGE. (2026, January 22). Wikibooks.
- QPNC-PAGE. (n.d.). Bionity.com.
- Kastenholz, B. (n.d.). QPNC-PAGE standardized protocol. Research Centre Juelich.
- Kastenholz, B. (n.d.). QPNC-PAGE: Standard protocol. Forschungszentrum Jülich GmbH.
- SDS-PAGE troubleshooting: Why are my protein bands fuzzy? (2025, April 29). Patsnap Synapse.
- Sample prep & gel electrophoresis troubleshooting. (n.d.). Sigma-Aldrich.
- The determination of lactate dehydrogenase isoenzymes in normal human muscle and other tissues. (n.d.). PMC.
- The principle and method of polyacrylamide gel electrophoresis (SDS-PAGE). (n.d.). MBL Life Sience.
- Tietz, D. (n.d.). The rationales of Ferguson plot analysis. HIS.com.
- The use of blue native PAGE in the evaluation of membrane protein aggregation states for crystallization. (n.d.). PMC.
- Tips for native PAGE. (n.d.). Assay-Protocol.
- Roy Chakravarty, P. (n.d.). Troubleshooting SDS-PAGE sample preparation issues. GoldBio.
- Aref, J., Lee, S., Sriphoosanaphan, S., Falabella, M., Yang, S.-Y., & Taanman, J.-W. (2025, September 18). Validation of blue- and clear-native polyacrylamide gel electrophoresis protocols to characterize mitochondrial oxidative phosphorylation complexes. PLOS One, 20(9), e0332065.
- Western blotting electrophoresis techniques. (n.d.). Bio-Rad.
- Why does my Blue native gel doesn’t run completely? (n.d.). ResearchGate.
- Zymogram gels. (n.d.).
- What is the major difference between native PAGE and SDS-PAGE? (n.d.). ResearchGate.