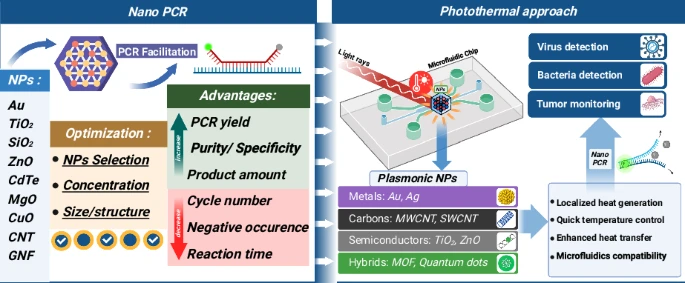
Nanoparticle-Assisted PCR (nanoPCR) is an advanced modification of Polymerase Chain Reaction (PCR) in which small nanoparticles are added into the PCR reaction mixture. These nanoparticles are usually 1-100 nm in size and they help in better amplification of DNA.
It is used to increase the efficiency, sensitivity, specificity and speed of the PCR reaction. Gold nanoparticles, carbon nanotubes and metal oxide nanoparticles are commonly used in this technique. These particles interact with DNA template, primers and DNA polymerase enzyme and make the reaction more suitable for amplification.
The nanoparticles have high thermal conductivity. So, they transfer heat rapidly during denaturation, annealing and extension steps. This helps the reaction to reach the required temperature very fast and reduces the total time of PCR.
Some nanoparticles also act like Single-Stranded DNA Binding (SSB) proteins. They bind temporarily with single stranded DNA and prevent wrong rejoining of DNA strands. Thus, non-specific amplification and background noise are reduced.
Nanoparticles can also bind reversibly with DNA polymerase. This works like a hot-start system and keeps the enzyme less active at low temperature. So, unwanted DNA copying before proper cycling is prevented.
nanoPCR is used in clinical diagnosis, forensic science, food safety and environmental monitoring. It is useful for rapid detection of viral pathogens like SARS-CoV-2, rare genetic mutation, degraded forensic DNA sample and foodborne pathogens.
Working Principle of Nanoparticle-Assisted PCR (nanoPCR)
Nanoparticle-Assisted PCR (nanoPCR) is based on the use of nanoparticles in the normal PCR reaction mixture. These nanoparticles improve the amplification of DNA by changing the thermal and biochemical condition of the reaction.
The principle is based on the high thermal conductivity of nanoparticles. When nanoparticles such as gold nanoparticles, carbon nanotubes or metal oxide nanoparticles are added, the PCR mixture behaves like a heat conducting nanofluid. It transfers heat rapidly and uniformly during each PCR cycle.
During denaturation, the double stranded DNA is separated into single stranded DNA. The nanoparticles adsorb these single stranded DNA molecules for a short time. This prevents early re-annealing of the strands and also reduces formation of secondary structures.
In annealing step, the primers bind to their complementary sequence on the template DNA. Nanoparticles help in proper primer-template binding by reducing mismatched primer attachment. Thus, non-specific amplification is decreased.
The large surface area of nanoparticles also helps to bind DNA polymerase reversibly. At low temperature, the enzyme may remain attached with nanoparticle surface and this acts like a hot-start mechanism. When the temperature increases, the enzyme is released and it starts DNA synthesis.
In extension step, DNA polymerase extends the primers and new DNA strands are formed. Due to better heat transfer and controlled enzyme activity, the amplification becomes faster, more specific and more sensitive.
Thus, in nanoPCR, nanoparticles improve heat distribution, stabilize single stranded DNA, control polymerase activity and increase correct primer binding. As a result, rapid and specific amplification of target DNA is produced.
Steps / Protocols of Nanoparticle-Assisted PCR (nanoPCR)
Steps / Protocols of Nanoparticle-Assisted PCR (nanoPCR)
- Selection of nanoparticles
The suitable nanoparticles are selected for nanoPCR reaction. The commonly used nanoparticles are gold nanoparticles, titanium dioxide nanoparticles, carbon nanotubes and metal oxide nanoparticles. - Optimization of nanoparticles
The size and concentration of nanoparticles are optimized. Very high amount of nanoparticles may inhibit the reaction. Very low amount may not increase the amplification properly. - Order of mixing
The order of mixing is also maintained. Mg²⁺, template DNA, dNTPs, primers, polymerase and nanoparticles are added in proper sequence. This can affect the acceleration of PCR reaction. - Preparation of reaction mixture
The reaction mixture is prepared in a sterile PCR tube. It contains nuclease-free water, PCR buffer or nanobuffer, nanoparticles, dNTP mix, forward primer, reverse primer, template DNA and DNA polymerase. - Addition of nanoparticles
If nanobuffer is not used, nanoparticles are added separately into the master mix. Gold nanoparticles of 20 nm size may be added in the reaction mixture. - Mixing and centrifugation
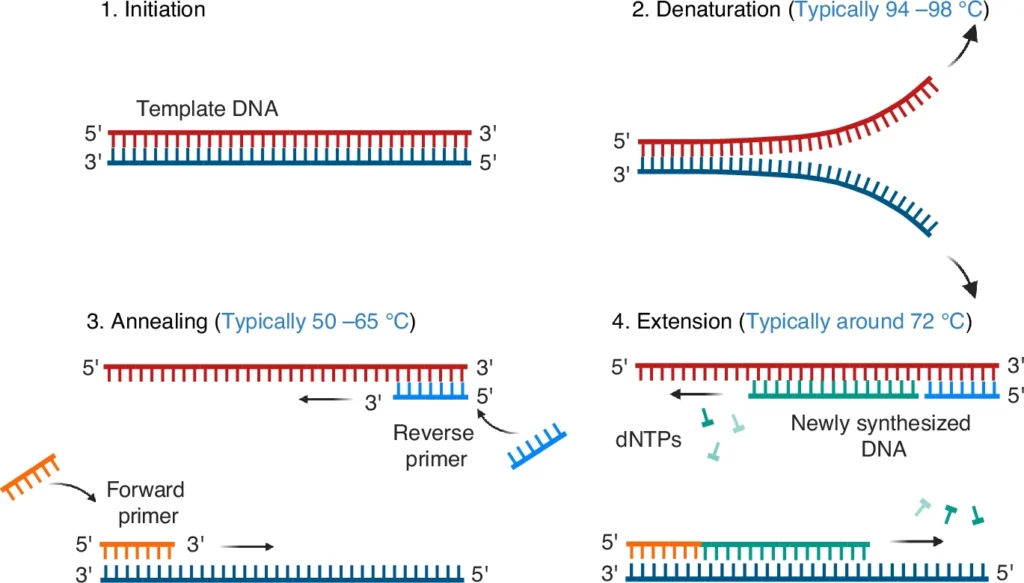
All the reagents are mixed gently. The tube is centrifuged for short time. The components are collected at the bottom of the PCR tube. - Initial denaturation
The reaction mixture is heated at 94°C to 95°C for 3-5 minutes. The double stranded DNA is denatured into single stranded DNA. - Denaturation
The temperature is kept at 94°C to 95°C for 30 seconds. In this step, the DNA strands are separated in each cycle. - Annealing
The temperature is reduced to the optimized annealing temperature. It is usually 50°C to 55°C for 30 seconds. The primers bind with complementary sequence of template DNA. - Extension
The temperature is kept at 72°C for 15-30 seconds. In this step, DNA polymerase adds nucleotides and new DNA strand is formed. - Cycle repetition
The denaturation, annealing and extension steps are repeated for about 30-31 cycles. The target DNA is amplified in repeated cycles. - Final elongation
The reaction is kept at 72°C for 5-10 minutes. The incomplete DNA strands are fully extended in this step. - Product analysis
The amplified DNA product is checked by agarose gel electrophoresis. It confirms the presence, size, specificity and yield of the target amplicon.
Applications of Nanoparticle-Assisted PCR (nanoPCR)
The following are the applications of Nanoparticle-Assisted PCR (nanoPCR)–
- nanoPCR is used for rapid detection of human infectious diseases. It is used to detect viral pathogens such as SARS-CoV-2. It gives more sensitive and specific result than conventional PCR.
- nanoPCR is used to detect single base mutation and cancer related biomarkers. It is used for detection of EGFR T790M mutation in lung cancer, PSA in prostate cancer, CA 125 in ovarian cancer and HER2 in breast cancer.
- nanoPCR is used for monitoring tumor progression, therapeutic response and drug resistance. It helps in cancer diagnosis and treatment related molecular analysis.
- nanoPCR is used for detection of animal pathogens. It can detect feline panleukopenia virus (FPV), mink enteritis virus (MEV), pseudorabies virus (PRV), canine coronavirus (CCoV), porcine parvovirus and bovine respiratory syncytial virus (BRSV).
- nanoPCR gives about 100-1000 fold higher sensitivity than some traditional methods in veterinary diagnosis. So, it is useful for early detection of animal disease.
- nanoPCR is used in forensic samples where normal PCR may fail. It helps to overcome inhibitors such as melanin from hair and indigo dye from denim.
- It is useful for degraded DNA, fragmented DNA, skeletal remains and trace touch DNA samples. It improves primer binding and stabilizes enzyme during amplification.
- nanoPCR is used in wastewater based monitoring. It is used for detection of viral load such as SARS-CoV-2 in municipal wastewater and H5N1 avian influenza in agricultural runoff.
- It is used to detect bacterial aerosols and environmental pathogens. It can also detect organisms such as Acanthamoeba from water and other environmental samples.
- nanoPCR is used for rapid detection of foodborne pathogens. It is used to detect Salmonella, Listeria, Staphylococcus aureus and E. coli O157:H7 from food samples.
- It gives result faster than traditional culture method in food safety testing. So, it is useful for quick screening of contaminated food materials.
- nanoPCR is used in basic molecular biology for gene analysis. It is used in gene typing, haplotyping and detection of specific DNA sequence.
- nanoPCR is used in archaeological study to amplify ancient DNA from preserved samples. It is useful where DNA is degraded and present in very small amount.
Advantages of Nanoparticle-Assisted PCR (nanoPCR)
The following are the advantages of Nanoparticle-Assisted PCR (nanoPCR)–
- nanoPCR can detect target DNA at very low concentration. It has about 100-1000 times higher detection sensitivity than conventional PCR.
- It increases the specificity of DNA amplification. The nanoparticles bind with single stranded DNA and prevent early re-annealing of the strands.
- It reduces false positive result and non-specific amplification. It also decreases primer-dimer formation during the reaction.
- nanoPCR requires less reaction time. Gold nanoparticles, carbon nanotubes and metal oxide nanoparticles transfer heat very fast in the reaction mixture.
- The reaction mixture reaches the required temperature rapidly. So, denaturation, annealing and extension steps are completed in short time.
- It can amplify DNA from degraded and complex samples. It is useful in samples containing inhibitors like melanin, indigo dye and wastewater contaminants.
- It has high tolerance against biochemical inhibitors. So, the reaction can be successful where normal PCR may fail.
- nanoparticles can bind reversibly with DNA polymerase at low temperature. This keeps the enzyme inactive before proper thermal cycling.
- This reversible binding acts like hot-start PCR system. It prevents early and non-specific DNA synthesis.
- nanoPCR is useful for detection of rare genetic mutation. It can detect single base mutation by reducing background noise from mismatched DNA.
- It helps in cancer related mutation detection. It can detect rare mutation where wild type DNA is present in high amount.
- nanoPCR gives sensitive and specific result without costly specialized instrument. It can be used in standard laboratory condition.
- It is cost-effective and convenient technique. It is also suitable for rapid point-of-care testing and routine molecular diagnosis.
Limitations of Nanoparticle-Assisted PCR (nanoPCR)
The following are the limitations of Nanoparticle-Assisted PCR (nanoPCR)–
- nanoPCR needs exact concentration of nanoparticles for proper amplification. If the concentration is very high, the reaction may be inhibited.
- High amount of nanoparticles can bind strongly with DNA polymerase, primers or template DNA. So, the enzyme and template may not be available for amplification.
- Very low amount of nanoparticles may not improve the reaction. In this condition, the sensitivity and specificity may remain almost like conventional PCR.
- Nanoparticles may aggregate or form clumps in the reaction mixture. Due to aggregation, their active surface area is decreased.
- Aggregated nanoparticles may work as reaction inhibitors instead of reaction enhancers. This can reduce amplification efficiency.
- Removal of nanoparticles from final PCR product is difficult. Gold nanoparticles, carbon nanotubes and graphene derivatives may have similar size or density with amplified DNA.
- Normal filtration or centrifugation may not remove the nanoparticles properly. This creates problem in further applications like sequencing, cloning and other high-throughput analysis.
- Some nanomaterials may show toxicity. Cadmium-based quantum dots, some carbon nanotubes and graphene oxide may be cytotoxic or mutagenic.
- Toxic nanoparticles need special handling and proper disposal. Otherwise it may create risk for laboratory workers and environment.
- Some nanoparticles can interfere with fluorescence detection in qPCR. Gold nanoparticles may absorb or quench fluorescent dyes like SYBR Green I.
- Fluorescence interference may change the amplification curve. So, wrong diagnostic reading may be produced.
- The protocol of nanoPCR is not fully standardized. The effect of nanoparticles depends on size, surface charge, concentration and reaction condition.
- Reproducible result is sometimes difficult in different laboratories. Same nanoparticles may not give same effect in all PCR systems.
- The fidelity of nanoPCR is still not fully clear. There is limited study on error rate and mutation formation during amplification.
- Due to unverified mutation rate, nanoPCR may create doubt in highly sensitive genetic analysis. So, careful validation is required before using it for critical diagnosis.
References
- Enhancement in the efficiency of polymerase chain reaction by TiO2 nanoparticles: crucial role of enhanced thermal conductivity. (n.d.). ResearchGate.
- A nanoparticle-assisted PCR assay to improve the sensitivity for rapid detection and differentiation of wild-type pseudorabies virus and gene-deleted vaccine strains. (n.d.). PubMed.
- A review on detection methods used for foodborne pathogens. (n.d.). PMC.
- Ahmedabad University student develops new forensic tool to address forensic challenges. (n.d.).
- Application of nanomaterials to enhance polymerase chain reaction. (n.d.). PMC – NIH.
- Applications of nanotechnology in forensic science: A comprehensive review. (n.d.).
- Jiang, X., Yang, M., & Liu, J. (2022). Capping gold nanoparticles to achieve a protein-like surface for loop-mediated isothermal amplification acceleration and ultrasensitive DNA detection. ACS Applied Materials & Interfaces, 14(24), 27666–27674.
- Classification and applications of nanomaterials in vitro diagnosis… (n.d.).
- Xue, H., Liang, Y., Gao, X., Song, Y., Zhu, K., Yang, M., Hao, J., Ma, H., & Yu, K. (2023). Development and application of nanoPCR method for detection of feline panleukopenia virus. Veterinary Sciences, 10(7), 440.
- Lee, C. Y., Degani, I., Cheong, J., Weissleder, R., Lee, J.-H., Cheon, J., & Lee, H. (2021). Development of integrated systems for on-site infection detection. Accounts of Chemical Research, 54(21), 3991–4000.
- Lee, C. Y., Degani, I., Cheong, J., Weissleder, R., Lee, J.-H., Cheon, J., & Lee, H. (2021). Development of integrated systems for on-site infection detection. Accounts of Chemical Research, 54(21), 3991–4000.
- Wang, J., Cheng, Y., Zhang, M., Zhao, H., Lin, P., Yi, L., Tong, M., & Cheng, S. (2015). Development of a nanoparticle-assisted PCR (nanoPCR) assay for detection of mink enteritis virus (MEV) and genetic characterization of the NS1 gene in four Chinese MEV strains. BMC Veterinary Research, 11, 1.
- Development of integrated systems for on-site infection detection. (n.d.). PMC.
- Development of nanoparticle-assisted PCR assay in the rapid detection of brain-eating amoebae. (n.d.). PubMed.
- Effect of PCR inhibitor removal kit. Environmental water samples were… (n.d.). ResearchGate.
- Emerging biomarkers for early cancer detection and diagnosis: Challenges, innovations, and clinical perspectives. (n.d.). PMC.
- MyBioSource. (n.d.). Environmental applications of PCR: Microbial ecology and pollution monitoring. MyBioSource Learning Center.
- Evaluating the mechanism of indigo-mediated PCR inhibition and validation of nano-based mitigation for forensic STR profiling. (n.d.). ResearchGate.
- Future of DNA fingerprinting: Application of NGS in forensic science. (n.d.). ResearchGate.
- Pan, D., Mi, L., Huang, Q., Hu, J., & Fan, C. (2012). Genetic analysis with nanoPCR. Integrative Biology, 4(10), 1155–1163.
- nanoComposix. (2026). Introduction to gold nanoparticles.
- PHC Holdings Corporation. (n.d.). Is qPCR more accurate than PCR? PHCbi.
- Lou, X., & Zhang, Y. (2013). Mechanism studies on nanoPCR and applications of gold nanoparticles in genetic analysis. ACS Applied Materials & Interfaces, 5(13), 6276–6284.
- Nano-PCR for the early detection of tomato leaf curl virus. (n.d.). PMC – NIH.
- Pirzada, M., & Altintas, Z. (2022). Nanomaterials for virus sensing and tracking. Chemical Society Reviews, 51, 5805–5841.
- Yang, Z., Shen, B., Yue, L., Miao, Y., Hu, Y., & Ouyang, R. (2022). Nanomaterials to enhance polymerase chain reaction. Encyclopedia.pub.
- Nanoparticle PCR: Nanogold‐assisted PCR with enhanced specificity. (n.d.). ResearchGate.
- Nanoparticle probes for the detection of cancer biomarkers, cells, and tissues by fluorescence. (n.d.). PMC.
- Nanoparticle-assisted PCR: A comprehensive analysis of mechanistic principles, procedural architectures, and multi-disciplinary applications. (n.d.).
- Nanoparticle-assisted PCR: Fundamentals, mechanisms, and… (n.d.).
- Nanoparticle-assisted PCR: Fundamentals, mechanisms, and forensic implications. (n.d.). ResearchGate.
- Nanoparticle-based detection of foodborne pathogens: Addressing… (n.d.).
- Nanoplasmonic on-chip PCR for rapid precision molecular diagnostics. (n.d.). ResearchGate.
- Nanotechnology for early cancer detection. (n.d.). PMC – NIH.
- Nanotechnology in tumor biomarker detection: The potential of liganded nanoclusters as nonlinear optical contrast agents for molecular diagnostics of cancer. (n.d.). PMC.
- Nanotechnology-enabled PCR with tunable energy dynamics. (n.d.). PMC.
- Zhao, X., Peng, H., Hu, J., Wang, L., & Zhang, F. (2024). Nanotechnology-enabled PCR with tunable energy dynamics. JACS Au, 4(9), 3370–3382.
- Overview of pathogen testing. (n.d.). Eurofins USA.
- GenScript. (2026). PCR protocol – PCR steps – PCR design tool.
- Khehra, N., Padda, I. S., & Zubair, M. (2025). Polymerase chain reaction (PCR). In StatPearls. StatPearls Publishing.
- Yuce, M., Kurt, H., Mokkapati, V. R. S. S., & Budak, H. (n.d.). Employment of nanomaterials in polymerase chain reaction: Insight into the impacts and putative operating mechanisms of nano-additives in PCR. RSC Advances.
- MilliporeSigma. (n.d.). Standard PCR protocol.
- Unveiling the power of nanomaterials in the area of forensics. (n.d.). ResearchGate.
- Wastewater as sentinel for emerging viral diseases in livestock: A systematic review. (n.d.).
- Ray, U. (2025). What are the different types of nanoparticles? AZoNano.
- Petrovic, M. (2024). What are the different types of nanoparticles? NanoSphere.
- Accelerated Materials. (2023). What are the types of nanoparticles?
- QIAGEN. (n.d.). dPCR vs qPCR.
- QIAGEN. (n.d.). dPCR vs qPCR vs end-point PCR.
- Xue, Z., You, M., Peng, P., Tong, H., He, W., Li, A., Mao, P., Xu, T., Xu, F., & Yao, C. (2021). Taqman-MGB nanoPCR for highly specific detection of single-base mutations. International Journal of Nanomedicine, 16, 3695–3705.