Mass Spectrometry (MS) is a analytical method. It is used for measuring mass of atoms and molecules. It is used to know the chemical composition of sample.
In this method, neutral molecules are changed into charged particles. These particles are called ions. Then these ions are separated.
The separation is done on the basis of mass and charge. Electric field and magnetic field are used for this separation.
The ions are measured as mass-to-charge ratio (m/z). This value is important for identification of molecule.
The result is obtained as mass spectrum. It contains different peaks. These peaks are used as molecular fingerprint.
By this method, unknown compound can be identified. Molecular weight can be measured. Structure related information can also be obtained from the spectrum.
Principle of Mass Spectrometry (MS)
Mass Spectrometry (MS) is based on the separation of ionized particles according to their mass and electrical charge. In this method, the compounds present in the sample are first converted into gas phase ions. These ions contain electrical charge, so they can be moved and controlled by electric or magnetic field.
The formed ions are then accelerated with high speed. After this, they are passed through electric field or magnetic field. During this process, the ions are deflected from their path.
The deflection of ions depends on their mass-to-charge ratio (m/z). Ions having lower mass are deflected more easily. Heavy ions are deflected less because they resist the change in their path.
Thus, different ions are separated from each other on the basis of m/z value. The separated ions are then recorded by a detector.
The detector gives a graph which is called mass spectrum. It shows the m/z value and relative abundance of each ion. From this spectrum, molecular weight and structural properties of the sample components are known.
Working of Mass Spectrometry (MS)
The working of Mass Spectrometry (MS) takes place in following steps-
- Sample introduction– The sample is introduced into the mass spectrometer through sample inlet. The instrument is kept under high vacuum condition to prevent collision of ions with air molecules.
- Ionization– In this step, the sample is vaporized and converted into gas phase ions. Usually one electron is removed from the molecule, forming positively charged ion or cation.
- Acceleration– The formed ions are accelerated by electric potential difference. This gives same kinetic energy to the ions before separation.
- Deflection– The ion beam passes through electric field or magnetic field in the mass analyzer. The ions are deflected according to their mass-to-charge ratio (m/z). Light ions are deflected more and heavy ions are deflected less.
- Detection– The separated ions reach the detector such as Faraday cup or electron multiplier. When ions strike the detector surface, electric signal is produced.
- Data processing– The detector signal is sent to a computer. The data is processed and a mass spectrum is formed, which shows relative abundance of ions against their m/z value.
Instrumentation of Mass Spectrometry (MS)
The main components of Mass Spectrometry (MS) are as follows-
- Sample inlet– It is the part through which the sample is introduced into the mass spectrometer. The sample may be solid, liquid or gas. It helps to enter the sample into high vacuum region without changing the internal pressure.
- Ionization source– It is the part where neutral molecules of the sample are vaporized and converted into gas phase ions. This part is considered as important part of mass spectrometer.
- Mass analyzer– It is used to separate the formed ions on the basis of their mass-to-charge ratio (m/z). Electric field or magnetic field is used in this part for separation of ions.
- Ion detector– It is the part where separated ions are detected. When ions strike the detector surface, charge or current is produced. This signal shows the relative abundance of each ion.
- Vacuum system– It maintains high vacuum inside the instrument. The vacuum prevents collision of ions with atmospheric gas molecules. If collision occurs, ions are scattered and signal becomes weak.
- Data processing system– It consists of computer and software. It receives the electrical signal from the detector and processes it. The final result is obtained as mass spectrum graph.
Mass Spectrometry Diagram
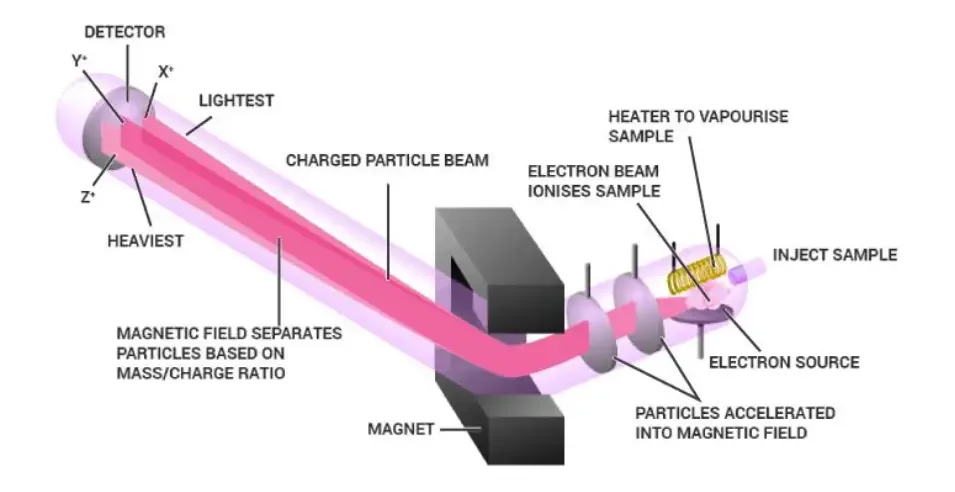
How Does Mass Spectrometry Work?
The working of Mass Spectrometry (MS) takes place in following steps-
- Ionization– The sample is first vaporized into gas. Then the molecules are converted into charged particles called ions. Usually one electron is removed and positive ion is formed.
- Acceleration– The formed ions are accelerated through a potential difference. In this step the ions are focused into a beam. It gives same kinetic energy to the ions.
- Deflection– The ion beam is passed through electric field or magnetic field. The path of ions is bent in this field. Light ions are deflected more and heavy ions are deflected less.
- Detection– The separated ions then reach the detector. When ions strike the detector surface, small charge or current is produced. Faraday cup or electron multiplier is used to detect and count the ions.
- Data processing– The signal is processed by computer software. The result is obtained as mass spectrum. It shows relative abundance of ions against their mass-to-charge ratio (m/z).
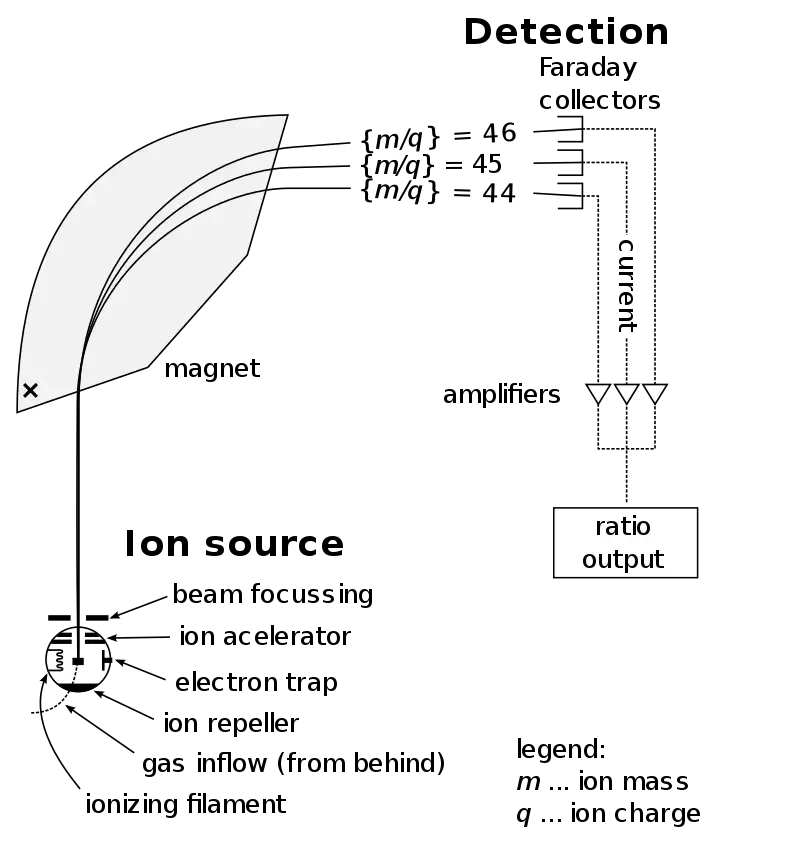
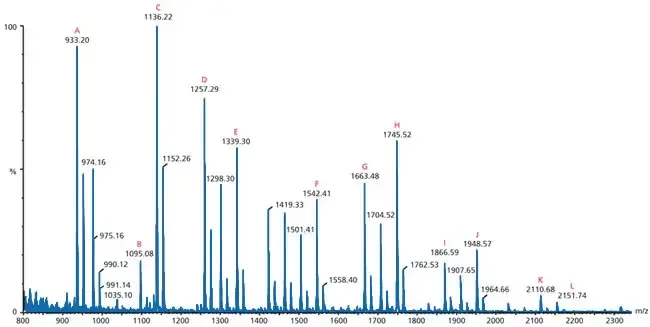
How To Read A Mass Spectrometry Spectrum
The mass spectrum is read by observing the position and height of the peaks. The following are the steps to read the spectrum-
- In mass spectrum, the x-axis shows mass-to-charge ratio (m/z) of the ions and the y-axis shows relative abundance of the ions.
- The peak at the highest m/z value is observed first. It represents the intact molecule after losing one electron and gives the molecular weight of the compound.
- The tallest peak in the spectrum is called base peak. It is taken as 100% relative abundance and represents the most stable and most common ion fragment.
- The difference between two major peaks is calculated. This difference shows the part of molecule which is broken off. For example, loss of water gives 18 amu and loss of methyl group gives 15 amu.
- Small peaks present near the main peak are also observed. M+1 peak may show presence of Carbon-13 and M+2 peak pattern may show presence of Chlorine or Bromine.
- The molecular weight, isotope peaks and fragment peaks are put together. From this, the chemical formula and possible structure of the compound can be known.
- The obtained spectrum is compared with standard reference spectrum. It helps to confirm the identity of the compound. NIST Mass Spectral Library is one of the used database for this purpose.
Applications of Mass Spectrometry (MS)
- It is used in drug discovery to identify lead compounds. It is used to evaluate metabolic profile of drugs. It is used for therapeutic drug monitoring. It is used for newborn metabolic screening. It is used for rapid identification of bacteria and fungi in clinical microbiology.
- It is used to detect trace level pollutants in air, water and soil. It detects pesticides. It detects heavy metals. It detects volatile organic compounds (VOCs). It detects PFAS (forever chemicals). It is also used to detect chemical warfare agents in field.
- It is used for identification of illegal drugs. It is used for new psychoactive substances. It is used for poisons in biological tissues or fluids. It is used to analyze trace crime scene evidence like glass, paint and fibers. It is used in arson investigation by finding traces of ignitable liquids. It is used to identify explosive residues.
- It is used in proteomics. It is used in metabolomics. It is used in lipidomics and glycomics. It is used to weigh and sequence biomolecules. It is used for structural characterization of complex biomolecules like proteins, peptides, carbohydrates and DNA.
- It is used to track food authenticity. It is used against food fraud. It is used to identify pesticide residues in food commodities. It is used to detect trace metal contamination in foods.
- It is used in process control. It is used in semiconductor manufacturing. It is used in vacuum coating. It is used in thin film optical coating. It is used for characterization of battery materials.
- It is used for precision isotope ratio measurement. It is used to evaluate geological materials. It is used to trace geographical source of metal contamination.
- It is used in space exploration missions. It is used in homeland security. It is also used in art conservation.
Advantages of Mass Spectrometry
- It is highly sensitive technique. Very low amount of analyte can be detected. It gives precise measurement in sample analysis.
- It is a rapid method. Data acquisition is quick. Scan speed is high as compared to other techniques.
- It is used for qualitative and quantitative purpose. Unknown compounds can be identified. Structural property can be elucidated. Known materials present in sample can be quantified accurately.
- It gives high reproducibility in results. When it is performed by expert, accuracy remains high. Reproducible data are obtained in repeated analysis.
- It can quantify large number of different molecules at the exact same time. Many analytes can be measured in a single run.
- It is versatile technique and can be integrated with separation methods. It can be combined with GC/MS and HPLC/MS. This gives better separation and very precise quantification.
- It has broad applicability due to precision and adaptability. It is used in pharmaceuticals. It is used in environmental monitoring. It is used in clinical diagnostics. It is also used in forensic science.
Limitations of Mass Spectrometry
- It is high cost technique. The instrument purchase is costly. Maintenance and service also needs high investment.
- It is a destructive technique. During analysis the sample is consumed. So, sample is destroyed and it cannot be recovered back.
- It requires specialized operators. The instrument is complex. Skilled person is needed for operation and interpretation.
- Sample purity is required for best result. In complex mixtures matrix effect occurs. Background components can suppress or enhance ionization and quantification becomes inaccurate.
- It cannot differentiate isomers if same m/z is present. Optical isomers and geometric isomers gives same mass to charge ratio. For separation additional tools are required like chiral column.
- Ionization problem is there for some compounds. Non-polar molecules shows poor ionization. Thermally unstable molecules are also difficult to detect by conventional ionization methods.
- Standardization is lacking in many cases. Sample preparation, instrument settings and data reporting differs. So, result comparison between different studies becomes inconsistent.
- Reference library limitation is there. New unknown or emerging contaminants are difficult to identify. Because spectral libraries are not always comprehensive for comparison.
References
- AdamR1996. (n.d.). How to read a simple mass spectrum : 7 steps. Instructables.
- Advanced architectures and analytical principles of mass spectrometry. (n.d.).
- Ali, O. (2025, April 11). How to read mass spectrometer graph: A beginner’s guide. AZoOptics.
- Analyzers. (n.d.). Chemistry at Emory.
- Bains, B. (2023, February 9). A beginner’s guide to mass spectrometry. ACD/Labs.
- Brucker, G., & Van Antwerp, K. (n.d.). Comparison of ion trap mass spectrometer and quadrupole mass spectrometer. The Society of Vacuum Coaters.
- Clark, J. (2014, February). Fragmentation patterns in the mass spectra of organic compounds. Chemguide.
- Detecting ions in mass spectrometers with the Faraday cup. (n.d.). Spectroscopy Online.
- Detection of large ions in time-of-flight mass spectrometry: Effects of ion mass and acceleration voltage on microchannel plate detector response. (n.d.). PMC.
- Double focussing mass spectrometer – overview – SIMS. (n.d.). MyScope.
- Drug chemistry, fire debris & explosives. (n.d.). St. Louis County Police Department.
- elina. (2020, June 6). Learn more about turbomolecular pumps. VacCoat.
- Factors to consider when choosing vacuum pumps. (n.d.). Leybold USA.
- Forensic applications of DART-MS: A review of recent literature. (n.d.). PMC.
- Forensic mass spectrometry: Scientific and legal precedents. (n.d.). PMC.
- Garg, E., & Zubair, M. (2024, November 22). Mass spectrometer. In StatPearls. StatPearls Publishing.
- Hatton, P., & Lundie, D. (n.d.). Mass spectrometry in real time. Hiden Analytical.
- Interpreting MS. (n.d.). OpenOChem Learn.
- Ionization methods in mass spectrometry: Types & applications. (n.d.). Danaher Corporation.
- Ionization modes. (n.d.). Mass Spectrometry & Proteomics Facility – University of Notre Dame.
- Ionization source technology overview. (n.d.). Thermo Fisher Scientific – US.
- Manning, K. (2023, September 11). Turbomolecular pump advantages & disadvantages. Provac Sales.
- Mass analyzer technology overview. (n.d.). Thermo Fisher Scientific – US.
- Mass analyzers for mass spectrometry. (n.d.). Technology Networks.
- Mass spectrometers. (n.d.). DGMS.
- Mass spectrometry basics. (n.d.). JEOL USA.
- Mass spectrometry for environmental pollutant monitoring. (n.d.). Lab Manager.
- Mass spectrometry ionization methods. (n.d.). Chemistry at Emory.
- Meher, A. K., & Zarouri, A. (2025). Environmental applications of mass spectrometry for emerging contaminants. Molecules, 30(2), 364.
- Microchannel plate detector. (2025, June 12). In Wikipedia.
- Parashar, A. K. (2026). Heavy metals in air, soil and water: Major sources and insights into plant responses, monitoring and management. In N. A. Anjum, A. Masood, N. A. Khan, & S. Umar (Eds.), Heavy metals in air, soil and water. IntechOpen.
- Rawat, B. S., Kumar, N., Ghosal, D., Aflyatunova, D., Rienäcker, B., & Welsch, C. P. (2025). Faraday cups: Principles, designs, and applications across scientific disciplines—A review. Instruments, 9(3), 20.
- Richardson, S. D. (2000). Environmental mass spectrometry. Analytical Chemistry, 72(18), 4477–4496.
- Ristivojević, P., Otašević, B., Todorović, P., & Radosavljević-Stevanović, N. (2025). Forensic narcotics drug analysis: State-of-the-art developments and future trends. Processes, 13(8), 2371.
- The working principle of a mass spectrometer. (n.d.). Hiden Analytical.
- Top 6 ion sources in mass spectrometry: EI, CI, ESI, APCI, APPI, and MALDI. (n.d.). MetwareBio.
- Tucker, K. R. (n.d.). Mass spectrometry. Introduction to Instrumental Analysis and Evaluation of Data.
- Types of MS/MS systems and their key characteristics. (n.d.). Shimadzu Scientific Instruments.
- Wang, D. (2022). Mass spectroscopy: The fundamental implement in forensic science. Journal of Mass Spectrometry & Purification Techniques, 8, 159.
- What is mass spectrometry. (n.d.). Scripps Research.