- MHC is a group of genes that encode proteins that allow the host to differentiate between self and non-self.
- Histocompatibility is derived from the Greek word for tissue (histo) and compatibility (compatibility).
- The human MHC resides on chromosome 6 and is known as the human leukocyte antigen (HLA) complex.
- HLA proteins fall into three distinct classes: Class I molecules are located on all nucleated cell types, but class II molecules are restricted to cells that can process antigens and transmit them to T lymphocytes.
- As we will see, macrophages, dendritic cells (DCs), and B cells are capable of antigen presentation in this manner.
- Finally, class III molecules include numerous immune-functioning proteins that are secreted. Class III molecules are not necessary for self-versus-non-self discrimination.
What is Class I MHC (Major Histocompatibility Complex) molecules?
- Class I MHC (Major Histocompatibility Complex) molecules are an essential component of the immune system responsible for recognizing and presenting antigens to T-cells. In humans, the Class I MHC molecules are known as Human Leukocyte Antigen (HLA) proteins and consist of HLA types A, B, and C. These molecules are present on the surface of nearly all nucleated cells in the body, except for certain cells in the retina, brain, and non-nucleated red blood cells.
- Class I MHC molecules are composed of two protein chains: an alpha chain, weighing approximately 45,000 Da, and a smaller protein called 2-microglobulin, with a weight of about 12,000 Da. The alpha chain spans the plasma membrane, while the 2-microglobulin is associated with the alpha chain internally. Together, these chains create a pocket-like structure that protrudes from the cell surface.
- This pocket is responsible for binding either self antigens, which helps identify the cell as a host cell, or peptides derived from intracellular pathogens, indicating that the cell is infected. By presenting antigens on their surfaces, Class I MHC molecules enable the immune system to recognize and mount a response against infected or abnormal cells.
- HLA proteins exhibit significant variation among individuals, and this variation is an outcome of the polymorphic nature of HLA genes. Each individual possesses multiple alleles of each HLA gene, inherited from both parents. As a result, an individual can produce six distinct Class I MHC proteins. The greater the genetic relatedness between two individuals, the more similar their HLA molecules will be.
- The presence of Class I MHC proteins on the surface of cells makes them capable of stimulating an immune response when cells from one individual with different Class I molecules are transplanted into another individual. This forms the basis for histocompatibility matching when preparing patients for organ or bone marrow transplantation.
- The Major Histocompatibility Complex (MHC) is a region of the genome found in all vertebrates and is responsible for coding MHC proteins. In humans, the MHC genes are clustered on chromosome 6 and encode the HLA proteins. MHC proteins are crucial for the recognition of foreign molecules by the acquired immune system, determining histocompatibility, and facilitating immune responses. Within the human MHC, genes encoding Class I, Class II, and Class III MHC proteins are considered particularly important.

Cellular Distribution of Major Histocompatibility Complex I (MHC I)
- The majority of nucleated cells express the classical class I MHC molecules, however the level of expression differs amongst cell types.
- Class I molecules are most abundant in lymphocytes, where they make up around 1% of all plasma membrane proteins, or 5×105 molecules per cell.
- Comparatively, fibroblasts, muscle cells, liver hepatocytes, and brain cells express low numbers of class I MHC molecules.
- By limiting the possibility of graft detection by the recipient’s Tc, the low quantity on liver cells may contribute to the high success rate of liver transplants.
- A few cell types (such as neurons and sperm cells at particular periods of development) appear to be completely devoid of class I MHC molecules.
- As previously mentioned, a single MHC molecule can bind several peptides. Due to the codominant expression of the MHC alleles, a heterozygous individual expresses the gene products at each MHC locus that are encoded by both alleles.
- On each nucleated cell of an F1 mouse, the K, D, and L from each parent are expressed (six distinct class I MHC molecules).
- A heterozygous human expresses the A, B, and C alleles from each parent (six different class I MHC molecules) on the membrane of each nucleated cell.
- Each cell can present a large number of peptides in the peptide-binding clefts of its MHC molecules as a result of the creation of so many class I MHC molecules.
- Class I molecules will exhibit self-peptides in normal, healthy cells due to the natural turnover of self proteins. Both viral peptides and selfpeptides will be expressed in infected cells.
- On the membrane of a single virus-infected cell, many class I molecules displaying a variety of viral peptides are present.
- Due to allelic variations in the peptide-binding clefts of class I MHC molecules, diverse sets of viral peptides might be bound by different people within a species.
Major Histocompatibility Complex I (MHC I) Structure
The majority of nucleated cells have an assortment of MHC class I proteins. The peptides bound by these MHC class I molecules are typically endogenous, or derived from the breakdown of intracellularly produced proteins. The vast majority of these peptides will be “self” because the majority of proteins normally synthesised within a cell are of host origin at any given moment (as opposed to proteins of non-self origin, such as those generated during a viral infection). The MHC class I molecule does not distinguish between “self” and “non-self” peptides; CD8+ T cell TCRs provide this function. Due to the formation of central tolerance, self peptide–MHC complexes do not elicit an immunological response because T cells with the relevant specificity are often absent from the T cell repertoire. In contrast, non-self peptides complexed with MHC class I are identified and activate CD8+ T cells.
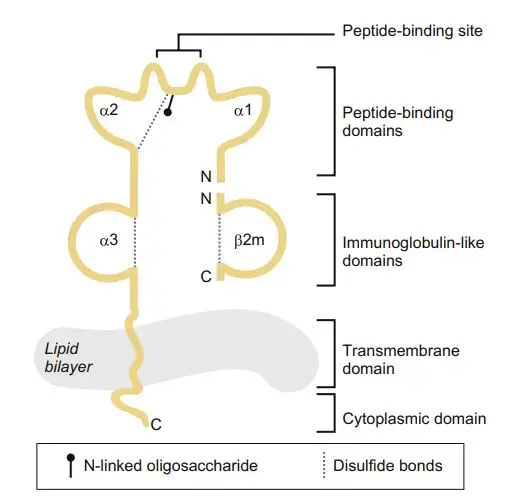
a. MHC Class I Component Polypeptides
- MHC class I α chains in both mice and humans are approximately 44 kDa glycoproteins with three extracellular globular domains.
- N-terminal domains α1 and α2 pair non-covalently to create the peptide-binding site, whereas the Ig-like α3 domain connects non-covalently with the β2m polypeptide.
- The α chain is also responsible for the transmembrane and cytoplasmic domains. The α1 domain maintains its shape without an internal disulfide bond, whereas the α2 an α3 domains include disulfide bonds.
- The β2m protein, the other partner of the MHC class I molecule, is a non-transmembrane polypeptide of around 12 kDa.
- β2m resembles a single Ig-like domain and, through its interaction with the MHC class I α3 domain, aids in the maintenance of the MHC class I molecule’s overall conformation.
- Indeed, the transport of the full heterodimer to the cell surface is dependent on the binding of β2m to the MHC class I α chain shortly after protein synthesis in the ER.
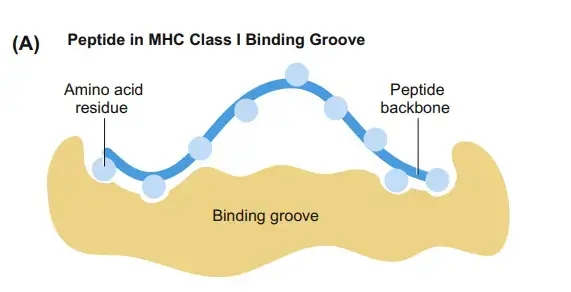
b. MHC Class I Peptide-Binding Site
- The MHC class I peptide-binding groove is relatively tiny. As a result, MHC molecules cannot detect big native antigens.
- Before antigens can be given to T cells, they must be digested into tiny peptides that can fit into the MHC groove.
- It is anticipated that each MHC class I molecule can attach to several hundred distinct peptides with reasonably high affinity, but can only capture one peptide at a time.
- The groove for binding class I MHC peptides is produced by the juxtaposition and contact of the α1 and α2 domains of the chain.
- The β2m chain helps by interacting with the amino acids that form the groove’s floor in α1 and α2.
- When the groove is occupied by a peptide consisting of 8–10 amino acids, these contacts are reinforced and the MHC class I structure is stabilised.
- Interactions between certain amino acids of the α1 and α2 domains and conserved “anchor residues” at the N- and C-termini of the peptide maintain its position in the groove.
- The peptide anchor residues point “down” into the groove, whereas the central residues point “up” toward the TCR.
- There is a considerable amount of conformational flexibility for peptides with widely diverse amino acid sequences to occupy the groove in the region between the anchor residues.
- Because the ends of the MHC class I groove are closed, peptides larger than 8–10 amino acids can only fit if their central residues bulge out of the groove.
How are self and non-self peptides deposited in the Major Histocompatibility Complex I (MHC I) binding pocket?
- Recall that proteasomes breakdown intracellular proteins as part of the natural process by which a cell recycles its protein content.
- Specific self and antigenic non-self peptides are transferred from the cytoplasm to the endoplasmic reticulum during this process (ER).
- The alpha chain of class I MHC and β2-microglobulin interact within the ER lumen. Attachment is restricted to peptides that fit properly into the binding cleft of one of the six distinct MHC I molecules.
- This process, known as endogenous antigen processing, allows the host cell to present peptide antigen to CD8+, or cytotoxic T lymphocytes, a subset of T cells (CTLs).
- Antigen-presenting CD8+ T lymphocytes express a receptor specific for class I MHC molecules. T lymphocytes “ignore” self-antigen presented by the host cell via MHC I molecules.
- T cells bind and destroy the host cell if the MHC class I molecule presents a non-self antigen (e.g., from an intracellular pathogen or a novel peptide found only in malignant cells).
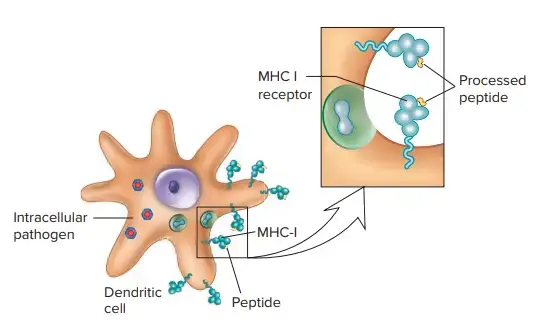
Mechanism of Major Histocompatibility Complex I
- The major histocompatibility complex class I (MHC class I) plays a crucial role in the immune system by presenting antigens derived from intracellular proteins to CD8+ T cells. This mechanism enables the immune system to detect and eliminate infected or transformed cells, such as those infected by viruses or cancer cells.
- The process of antigen presentation by MHC class I molecules begins with the degradation of intracellular proteins, including viral or tumor antigens, through the action of the proteasome. The proteasome breaks down these proteins into smaller peptide fragments. These endogenous peptides then translocate from the cytoplasm to the endoplasmic reticulum (ER).
- In the ER, the peptides are loaded onto MHC class I molecules through the involvement of a complex called the peptide-loading complex. The peptide-loading complex consists of several cellular components, including the ER transporter associated with antigen processing (TAP1/2), tapasin, the oxidoreductase ERp57, and the chaperone protein calreticulin. These components work together to ensure the proper loading of peptides onto MHC class I molecules.
- The cellular components involved in the presentation of endogenous antigens, from the proteasome subunits to the peptide-loading complex, are collectively referred to as the antigen-processing machinery (APM). The APM plays a vital role in generating the peptide repertoire that is presented by MHC class I molecules.
- CD8+ T lymphocytes express CD8 receptors in addition to their T-cell receptors (TCRs). When a cytotoxic T cell CD8 receptor binds to an MHC class I molecule, and the TCR recognizes and fits the specific antigenic peptide displayed within the MHC class I molecule, it triggers a signaling cascade within the T cell. This signaling leads to the activation of the CD8+ T lymphocyte, which can then initiate various immune responses, including the induction of programmed cell death (apoptosis) in the target cell.
- By triggering apoptosis in infected or transformed cells, the CD8+ T lymphocytes help mediate cellular immunity, which is the primary defense mechanism against intracellular pathogens such as viruses and certain bacteria. This immune response aims to eliminate the source of infection or abnormal cellular growth, thus preserving the overall health and integrity of the organism.
Functions of Major Histocompatibility Complex I
Antigen Processing and Presentation
- Peptides are typically seen in nucleated cells, primarily self-peptides produced by protein turnover and faulty ribosomal products.
- During viral infection, infection by intracellular microorganisms, or transformation into cancer, these proteasome-degraded proteins are also loaded onto MHC class I molecules and displayed on the cell surface.
Transplant Rejection
- MHC molecules operate as antigens after transplantation of an organ or stem cells and can provoke an immunological response in the recipient, resulting in transplant rejection.
- Due to the considerable MHC variation in the human population and the fact that no two individuals, with the exception of identical twins, express identical MHC molecules, these molecules can facilitate transplant rejection.
FAQ
What is Major Histocompatibility Complex I (MHC I)?
Major Histocompatibility Complex I (MHC I) is a group of cell surface proteins found on almost all nucleated cells in the body, except for certain cell types. They play a vital role in the immune system by presenting antigens derived from intracellular proteins to CD8+ T cells.
What is the function of MHC I molecules?
The primary function of MHC I molecules is to bind to peptide antigens derived from intracellular proteins and display them on the cell surface. This allows CD8+ T cells to recognize and respond to infected or abnormal cells.
How are antigens loaded onto MHC I molecules?
Antigens are loaded onto MHC I molecules through a process involving the peptide-loading complex. This complex includes components such as the ER transporter associated with antigen processing (TAP1/2), tapasin, ERp57, and calreticulin. These components work together to ensure the proper loading of antigens onto MHC I molecules in the endoplasmic reticulum.
What is the role of the proteasome in MHC I antigen presentation?
The proteasome is responsible for degrading intracellular proteins, including viral or tumor antigens. The degraded protein fragments are then translocated to the endoplasmic reticulum, where they are loaded onto MHC I molecules for presentation to CD8+ T cells.
Are MHC I molecules specific to individuals?
Yes, MHC I molecules are highly diverse and vary between individuals. The genes encoding MHC I proteins are polymorphic, meaning they exist in several forms. This variation allows for recognition of a wide range of antigens, and the more closely related two individuals are genetically, the more similar their MHC I molecules will be.
What happens when a CD8+ T cell recognizes an MHC I-antigen complex?
When a CD8+ T cell receptor (TCR) recognizes a specific antigen presented by an MHC I molecule on an infected or abnormal cell, it triggers a signaling cascade within the T cell. This leads to the activation of the CD8+ T cell and the initiation of immune responses, such as the induction of apoptosis in the target cell.
What is the significance of MHC I in cellular immunity?
MHC I molecules play a critical role in cellular immunity, which is the primary defense mechanism against intracellular pathogens, including viruses and certain bacteria. By presenting antigens from infected or abnormal cells, MHC I molecules activate CD8+ T cells to eliminate the source of infection or abnormal cellular growth.
Do MHC I molecules have any role in organ transplantation?
Yes, MHC I molecules are important in organ transplantation. The presence of different MHC I molecules in individuals can lead to immune rejection if incompatible organs or tissues are transplanted. Histocompatibility matching, which involves matching the MHC I molecules between the donor and recipient, is crucial to minimize the risk of rejection and ensure successful transplantation.
Are red blood cells (RBCs) equipped with MHC I molecules?
No, red blood cells (RBCs) lack MHC I molecules on their surface. Unlike other nucleated cells, RBCs do not possess MHC I molecules. This is one of the reasons why RBCs can be transfused between individuals of different MHC types without causing immune rejection.
Are MHC I molecules involved in the recognition of self-antigens?
Yes, MHC I molecules also play a role in recognizing self-antigens. They continuously sample peptides generated within the cell and present them on the cell surface. This helps maintain immune tolerance by allowing the immune system to recognize and distinguish self-cells from foreign or abnormal cells.
References
- Kuby Immunology Seventh edition.
- Roitt’s Essential Immunology Thirteenth edition
- Natarajan K, Li H, Mariuzza RA, Margulies DH. MHC class I molecules, structure and function. Rev Immunogenet. 1999;1(1):32-46. PMID: 11256571.
- Li XC, Raghavan M. Structure and function of major histocompatibility complex class I antigens. Curr Opin Organ Transplant. 2010 Aug;15(4):499-504. doi: 10.1097/MOT.0b013e32833bfb33. PMID: 20613521; PMCID: PMC3711407.
- Natarajan, K & Li, Hongmin & Mariuzza, RA & Margulies, David. (1999). MHC class I molecules, structure and function. Reviews in immunogenetics. 1. 32-46.
- Wieczorek, M., Abualrous, E. T., Sticht, J., Álvaro-Benito, M., Stolzenberg, S., Noé, F., & Freund, C. (2017). Major Histocompatibility Complex (MHC) Class I and MHC Class II Proteins: Conformational Plasticity in Antigen Presentation. Frontiers in Immunology, 8. doi:10.3389/fimmu.2017.00292
- Janeway CA Jr, Travers P, Walport M, et al. Immunobiology: The Immune System in Health and Disease. 5th edition. New York: Garland Science; 2001. The major histocompatibility complex and its functions. Available from: https://www.ncbi.nlm.nih.gov/books/NBK27156/
- Hohl, T. M. (2015). Cell-Mediated Defense against Infection. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases, 50–69.e6. doi:10.1016/b978-1-4557-4801-3.00006-0
- Mak, T. W., & Saunders, M. E. (2006). MHC: The Major Histocompatibility Complex. The Immune Response, 247–277. doi:10.1016/b978-012088451-3.50012-0
- The Major Histocompatibility Complex. (2014). Primer to the Immune Response, 143–159. doi:10.1016/b978-0-12-385245-8.00006-6
- Rammensee, H.G. (1993). Structure and Function of MHC Class I Molecules. In: Eibl, M.M., Huber, C., Peter, H.H., Wahn, U. (eds) Symposium in Immunology I and II. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-78087-5_9
- Janeway CA Jr, Travers P, Walport M, et al. Immunobiology: The Immune System in Health and Disease. 5th edition. New York: Garland Science; 2001. The major histocompatibility complex and its functions. Available from: https://www.ncbi.nlm.nih.gov/books/NBK27156/
- https://www.ebi.ac.uk/interpro/entry/InterPro/IPR001039/
- https://www2.nau.edu/~fpm/immunology/documents/Ch-08.pdf
- https://www.ndvsu.org/images/StudyMaterials/Micro/MHC-Antigens.pdf
- https://pdb101.rcsb.org/motm/62
- https://www.jrc.ac.in/working_folder/DOWNLOAD-D-11-53-5F0F117D553FB.pdf
- https://www.microbiologybook.org/bowers/mhc.htm
- https://www.azolifesciences.com/article/What-is-the-Function-of-MHC-Molecules.aspx
- https://www.onlinebiologynotes.com/major-histocompatibility-complex-mhc-structure-types-and-functions/
- https://immunobites.com/2018/07/23/what-is-mhc-and-why-does-it-matter/
- https://microbenotes.com/major-histocompatibility-complex-i-structure-mechanism-and-functions/