GasPak Anaerobic System is a self-contained airtight apparatus that is used in microbiology laboratory for cultivation of strict anaerobic bacteria. These bacteria cannot grow in presence of oxygen and need oxygen free condition for their survival and growth.
The system contains a sealed jar or container in which culture plates are kept with a gas generating envelope. The envelope generally contains chemicals like sodium borohydride, sodium bicarbonate and citric acid. When water is added, hydrogen and carbon dioxide gases are produced inside the jar.
The hydrogen gas then combines with oxygen present inside the container in presence of palladium catalyst. This reaction forms water, which may be seen as condensation on the inner wall of the jar. Thus the oxygen is removed and anaerobic condition is produced.
Newer GasPak EZ system is a simple waterless and catalyst free system. It uses sachets containing ascorbic acid and activated carbon. These sachets remove oxygen and produce carbon dioxide automatically after exposure to air.
An anaerobic indicator strip is also placed inside the jar. Methylene blue or resazurin indicator is commonly used. The indicator becomes colourless when oxygen is completely removed. This indicates that proper anaerobic condition has been formed inside the GasPak system.
Principle of GasPak Anaerobic System
GasPak Anaerobic System is based on the principle of chemical gas generation and catalytic removal of oxygen from a sealed jar. In this system, the oxygen present inside the jar is removed and anaerobic condition is produced for the growth of anaerobic bacteria.
In the classic GasPak system, a disposable gas generating envelope is placed inside the jar. This envelope contains sodium borohydride, sodium bicarbonate and citric acid. When water is added to the envelope, sodium borohydride produces hydrogen gas, while sodium bicarbonate and citric acid produce carbon dioxide.
The hydrogen gas reacts with the residual oxygen present inside the jar in presence of palladium catalyst. This reaction forms water and the oxygen becomes completely depleted from the jar. The reaction is exothermic and produces heat. Due to this, dew or condensation is seen on the inner wall of the jar.
Carbon dioxide produced inside the jar helps in stimulation of bacterial growth. Thus, the combined action of hydrogen, carbon dioxide and palladium catalyst creates an oxygen free environment in the GasPak jar.
In newer GasPak EZ system, the principle is based on waterless and catalyst free oxygen scavenging method. The sachet contains ascorbic acid and activated carbon. These chemicals directly remove free oxygen from the air and produce carbon dioxide.
An indicator strip is placed inside the sealed jar to confirm anaerobic condition. Methylene blue indicator is blue in presence of oxygen. It becomes colourless when the environment becomes completely oxygen free.
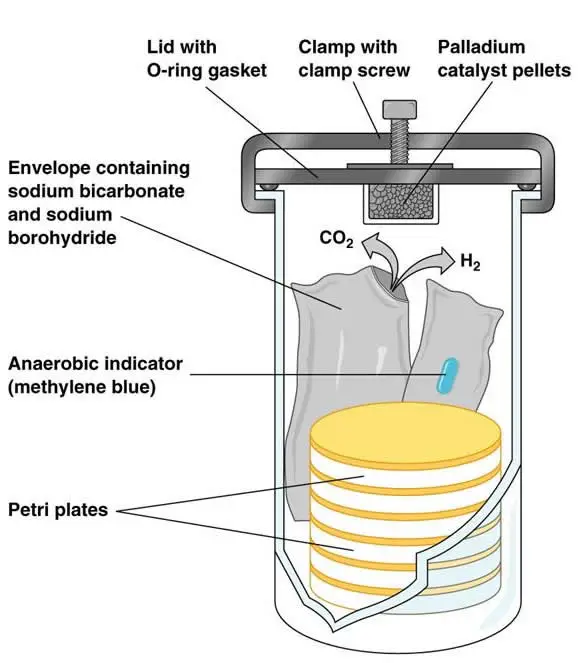
The GasPak Anaerobic System is made up of various parts, including
- Polycarbonate jar or plastic container – It is the main airtight vessel of the GasPak system. It is used to hold the culture plates or tubes during incubation. It provides closed condition for removal of oxygen.
- Lid assembly – It is the cover of the jar or container. It may have clamp, clamp screw, thumbscrew or easy closing latches. It helps to close the jar tightly and prevent entry of outside air.
- O-ring gasket – It is a rubber or silicone ring present in the lid. It provides a tight seal between the lid and the jar. It helps in maintaining airtight condition inside the GasPak system.
- Wire rack or Petri plate carrier – It is a removable holder present inside the jar. It is used to keep Petri dishes or culture tubes in proper position. It also prevents movement of culture plates inside the container.
- Gas-generating envelope or sachet – It is a disposable packet placed inside the jar. In traditional system, it produces hydrogen and carbon dioxide after addition of water. In newer GasPak EZ system, it removes oxygen by ascorbic acid and activated carbon without adding water.
- Palladium catalyst – It is used in the traditional GasPak system. It is usually present as palladium coated alumina pellets. It helps hydrogen to react with oxygen and form water.
- Anaerobic indicator strip or tablet – It is a chemical indicator placed inside the jar. Methylene blue or resazurin is commonly used. It becomes colourless when oxygen is removed and indicates that anaerobic condition has been formed.
How does a gaspak anaerobic system work?
The working procedure of GasPak anaerobic system can be divided into following steps:
1. Setup
The inoculated Petri plates or culture tubes are placed inside the airtight polycarbonate jar. The plates are arranged in a wire rack or plate carrier. The lid is then closed properly to make a sealed condition.
2. Addition of gas generating envelope
In the classic GasPak system, a gas generating envelope is placed inside the jar. Water is added to the envelope for activation. The chemicals present inside the envelope start reacting after addition of water.
3. Production of gases
The envelope contains sodium borohydride, sodium bicarbonate and citric acid. Sodium borohydride produces hydrogen gas. Sodium bicarbonate and citric acid produce carbon dioxide gas. These gases are released inside the sealed jar.
4. Removal of oxygen
The hydrogen gas reacts with the trapped oxygen inside the jar in presence of palladium catalyst. This reaction forms water. In this way, oxygen is removed from the jar and anaerobic condition is formed. Water formation may be seen as dew or condensation on the inner wall of the jar.
5. Modern GasPak EZ system
In newer GasPak EZ system, dry sachet is used. The sachet contains ascorbic acid, activated carbon and inorganic carbonate. When it is removed from foil packet and exposed to air, it starts removing oxygen directly. It does not need water and palladium catalyst.
6. Carbon dioxide generation
In both systems, carbon dioxide is produced inside the container. Carbon dioxide helps in the growth of anaerobic bacteria. It also maintains suitable atmosphere for culture of anaerobes.
7. Verification of anaerobic condition
An anaerobic indicator strip is placed inside the sealed container. Methylene blue is blue in presence of oxygen and becomes colourless in anaerobic condition. Resazurin is pink in presence of oxygen and becomes colourless when oxygen is removed. This confirms that strict anaerobic state has been reached.
Indicator of anaerobiosis
The following are the important indicators used for checking anaerobic condition in GasPak system:
- Methylene blue – It is the standard and commonly used redox indicator strip in GasPak anaerobic system. It is placed inside the jar before sealing. In presence of oxygen, the indicator remains blue in colour. In absence of oxygen, it becomes colourless or white due to reduced form.
- Resazurin – It is another redox indicator used for detecting oxygen inside the anaerobic jar. It may be used as strip, tablet or in liquid culture media like thioglycollate broth. In presence of oxygen, it appears blue, purple or pink depending on the oxygen level and product type. In absence of oxygen, it becomes completely colourless.
These indicators help to confirm that oxygen has been removed from the sealed jar. Colourless indicator shows that proper anaerobic condition has been formed for the growth of anaerobic bacteria.
Applications of GasPak Anaerobic System
The following are the applications of GasPak anaerobic system-
- Clinical diagnostics- GasPak anaerobic system is used for isolation of anaerobic pathogens from clinical samples. It is used in diagnosis of botulism, tetanus, gas gangrene and bacterial vaginosis.
- Food and beverage production- It is used in food processing and brewing industry. It helps to detect spoilage organisms in vacuum packed foods and other stored products.
- Pharmaceutical research- It is used in drug research and development. It is useful for testing antibiotics against anaerobic pathogens.
- Environmental microbiology- It is used to study anaerobic microorganisms present in soil, sediments and wastewater treatment plants. These organisms grow in absence of oxygen.
- Industrial fermentation- It is used to provide oxygen free condition for microbial fermentation. It helps in industrial microbial processes where anaerobic condition is needed.
- Bacterial characterization- It is used to test oxygen tolerance of unknown bacteria. It helps in identification, characterization and basic study of bacteria.
Advantages of GasPak Anaerobic System
The following are the advantages of GasPak anaerobic system-
- Cost effective and compact- GasPak anaerobic system is cheaper than anaerobic chamber. It is small in size and can be kept easily on laboratory bench or in incubator.
- Easy to use- It is simple to operate. It does not need vacuum pump, gas cylinder or pressure manometer.
- Safe method- Modern GasPak system is safer to use. In GasPak EZ system, waterless and catalyst free oxygen removal is done, so hydrogen gas is not produced.
- Self contained system- It uses ready-made disposable sachet or envelope for anaerobic atmosphere. The indicator strip shows whether anaerobic condition is formed or not.
- Rapid growth condition- It forms anaerobic condition within short time. Carbon dioxide is also produced which helps in growth of many fastidious anaerobic bacteria.
- Flexible capacity- The jar has removable racks for holding many Petri plates or test tubes. It is useful for comparative study and routine laboratory work.
Limitations of GasPak Anaerobic System
The following are the limitations of GasPak anaerobic system-
- Catalyst poisoning- In traditional GasPak system, palladium catalyst may be inactivated by hydrogen sulfide gas. Excess moisture, organic acids and chemical fumes also poison the catalyst.
- Maintenance requirement- The palladium catalyst need regular rejuvenation after use. It is heated in dry oven at 160°C to 170°C for about two hours to remove moisture and volatile contaminants.
- Explosion hazard- Classic GasPak system produce hydrogen gas. Hydrogen is flammable, so explosion hazard may occur if spark or ignition source is present.
- Leakage problem- The jar parts may be damaged after repeated use. O-ring may dry, split or not fit properly, so oxygen can enter inside the jar.
- Damage of lid and clamp- Plastic lid and clamp may develop cracks due to long use or over-tightening. This can disturb anaerobic condition inside the jar.
- Inconsistent oxygen removal- Oxygen removal is based on chemical reaction. So oxygen is not removed at constant and controlled rate.
- Less useful for fastidious organisms- GasPak system is useful for common anaerobes like Bacteroides and Clostridium. But it may be less effective for very fastidious microaerophiles like some Campylobacter species.
- Manual activation- Traditional GasPak envelope need addition of water for starting gas generation. This adds extra step in the procedure.
References
- BD Diagnostics. (n.d.). BD GasPak EZ gas generating systems. Grosseron.
- BD Diagnostics. (2010). BD GasPak EZ gas generating system [Brochure]. Becton, Dickinson and Company.
- Becton, Dickinson and Company. (2004). Package insert, GasPak anaerobic system envelopes. Scribd.
- Becton, Dickinson and Company. (2008). Package insert, BBL GasPak anaerobic systems. Labchem Catalog.
- Becton, Dickinson and Company. (2022). GasPak EZ gas generating pouch systems [Package insert]. Nelson-Jameson.
- Becton, Dickinson and Company. (2024). GasPak EZ gas generating container systems [Package insert]. Labchem Catalog.
- Becton, Dickinson and Company. (2026). BD GasPak EZ container systems.
- BenchChem Technical Support Team. (2026, March). The role of methylene blue as a redox indicator: A technical guide. BenchChem.
- Biokar Diagnostics. (2016). Thioglycollate broth with resazurin [Technical data sheet]. FUJIFILM Wako Chemicals.
- Chemical stability of ascorbic acid integrated into commercial products: A review on bioactivity and delivery technology. (n.d.). PubMed Central.
- Collee, J. G., & Watt, B. (n.d.). An evaluation of the Gaspak system in the culture of anaerobic bacteria. Semantic Scholar.
- Comparative study regarding the properties of methylene blue and proflavine and their optimal concentrations for in vitro and in vivo applications. (n.d.). PubMed Central.
- Cox, M. E. (1996). Palladium catalyst: Testing, validation, reactivating and cleaning. 46PP.
- Cox, M. E. (1996). Palladium catalyst: Testing, validation, reactivating and cleaning. 46PP – Anaerobe Society of the Americas Congress on Anaerobic Bacteria and Anaerobic Infections, Chicago, IL.
- Creating and protecting anaerobic environments with anaerobic systems and jars. (n.d.). Thomas Scientific.
- Evaluation of the AnaeroPack system for growth of anaerobic bacteria. (n.d.). PubMed Central.
- Gutsanu, V., & Baerle, N. (2022). Interaction of L-ascorbic acid with activated carbon: Kinetic studies and the effect of pH. Scilit.
- Gutsanu, V., & Baerle, N. (2022). Interaction of L-ascorbic acid with activated carbon: Kinetic studies and the effect of pH. Colloid Journal, 84(3), 353–363.
- Haque, A. (n.d.). Special culture techniques.
- Laboratory maintenance and cultivation of Bacteroides species. (n.d.). PubMed Central.
- mbavictor376. (n.d.). McIntosh anaerobic jar overview. Scribd.
- MRC Lab. (n.d.). Anaerobic jars.
- Proficiencies of three anaerobic culture systems for recovering periodontal pathogenic bacteria. (n.d.). PubMed Central.
- Reynolds, J. (2024). 6: Oxygen requirements and anaerobes. Biology LibreTexts.
- Roth, J. N. (2020, April 21). Use of redox compounds in microbiological media for the differentiation of microbial growth from non-microbial debris in test samples. Micrology Labs.
- Seip, W. F. (2003). Exothermic chemistry and method for generating an anaerobic environment (U.S. Patent No. 6,642,047 B2). U.S. Patent and Trademark Office.
- Seip, W. F., & Evans, G. L. (1980). Atmospheric analysis and redox potentials of culture media in the GasPak System. Journal of Clinical Microbiology, 11(3), 226-233.
- Sikkenga, D. L., Zaenger, I. C., & Williams, G. S. (1991). Palladium catalyst reactivation (U.S. Patent No. 4,999,326 A). U.S. Patent and Trademark Office.
- Slideshare. (n.d.). Anaerobic jar [PowerPoint slides].
- Spears, B., Stern, E., & Flentie, K. (2019). Metabolic assay for bacterial growth and gram typing (U.S. Patent Application Publication No. US 2019/0300927 A1). U.S. Patent and Trademark Office.
- Sudibya, A., & Widyaningsih, I. (2023). Unleashing the power of palladium catalyst: Unveiling the role in dew formation and anaerobiosis within anaerobic jars. Anatomica Medical Journal, 6(3), 149-162.
- The effect of palladium catalyst on dew formation and anaerobiosis in anaerobic jars. (n.d.). AWS.
- Theoretical and practical framework of the GasPak anaerobic system in clinical and industrial microbiology. (n.d.).
- TM Media. (2025, November 13). Anaerobic jar systems shaping modern microbiology. Titan Biotech Ltd.
- University of Nebraska–Lincoln. (2026). Anaerobic chamber. Biomedical and Obesity Research Core.
- Van Gray, J. (2025). 5.5: Bacterial oxygen requirements. Biology LibreTexts.
- Weber Scientific. (2026). GasPak EZ gas generating container system (BD diagnostics).
- Wehrspann, P. (1976). [Comparison of diverse methods for the cultivation of anaerobians, with special reference to the GASPAK system]. Zentralblatt für Bakteriologie, Parasitenkunde, Infektionskrankheiten und Hygiene, 234(1), 121-128.
- Wikipedia contributors. (2023, May 28). Gas-pak. In Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (2026, April 6). Chemistry of ascorbic acid. In Wikipedia, The Free Encyclopedia.