Differential centrifugation is a simple technique used for separation of cell organelles and other sub-cellular particles from a mixture.
It is also called differential pelleting or differential velocity centrifugation. It separates the particles mainly according to their size and density. The larger and heavier particles settle first, while smaller particles remain in the liquid part.
It is based on the principle of sedimentation. When centrifugal force is applied to a homogenized sample, the particles move down at different speed. Larger and dense particles sediment faster than the smaller and lighter particles.
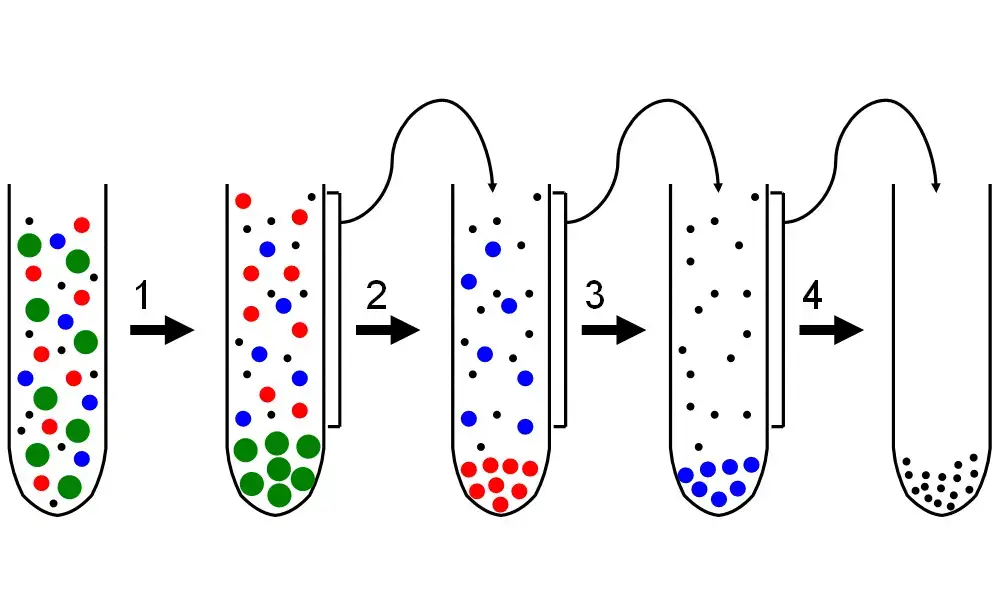
In this method, the sample is first centrifuged at low speed. The largest particles settle at the bottom of the tube and forms a pellet. The liquid part which remains above the pellet is called supernatant.
The supernatant is then carefully taken out and transferred into another tube. It is again centrifuged at a higher speed. In this step, the next larger particles are sedimented and form another pellet.
This process is repeated again and again by increasing the centrifugal force. In each step, a different group of particles are separated as pellet. The previous supernatant is used for the next centrifugation.
Thus, different cell components such as nuclei, mitochondria and ribosomes can be separated step by step. It is mainly used as a primary method for fractionation of cells and for further analysis of separated particles.
Principle of Differential centrifugation
Principle of Differential centrifugation is based on sedimentation of particles under centrifugal force.
When centrifugal force is applied to a cell homogenate, particles start to settle down in the tube. The settling rate is not same for all particles. It depends on size, shape and density of the particles.
Large and dense particles settle down first. Small and less dense particles take more time to settle. So they remain in the supernatant during low speed centrifugation.
In the first step, low speed centrifugation is done. The heaviest particles are sedimented at the bottom and forms pellet. The upper liquid part is called supernatant.
The supernatant is taken into another tube. It is again centrifuged at higher speed. In this step, next group of particles are sedimented.
This same process is repeated with increasing speed and time. In each step, one fraction is obtained as pellet. In this way, cell organelles are separated according to their decreasing size and mass.
Differential centrifugation Protocol
- Sample preparation and homogenization
The tissue sample is minced and suspended in isotonic homogenization medium containing 0.25 to 0.32 M sucrose and buffer.
The sample is homogenized to break the plasma membrane but internal organelles are kept intact.
The homogenate is filtered through cheesecloth or nylon screen to remove unlysed clumps and large debris. - Low speed centrifugation
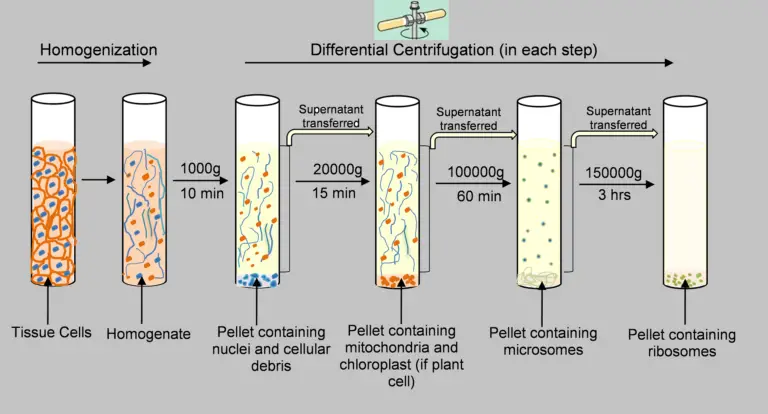
The filtered homogenate is taken in centrifuge tube and centrifuged at 600 to 1,000 × g for 5 to 10 minutes at 4°C.
In this step, intact cells, large debris and nuclei settle down as pellet. - Separation of supernatant
The supernatant is separated carefully from the pellet.
It is transferred into another clean centrifuge tube without disturbing the pellet. - Medium speed centrifugation
The supernatant is centrifuged at 10,000 to 17,000 × g for 15 to 30 minutes.
In this step, mitochondria, lysosomes and peroxisomes form the pellet. - High speed centrifugation
The supernatant is again transferred and centrifuged at 50,000 to 100,000 × g for 60 to 120 minutes.
In this step, microsomal pellet is formed which contain plasma membrane, endoplasmic reticulum vesicles and Golgi fragments. - Ultra-high speed centrifugation
The remaining supernatant is centrifuged at 200,000 × g or more for 120 minutes or more.
In this step, ribosomal subunits and large protein complexes are settled down as pellet.
The final remaining liquid is soluble cytosol fraction. - Washing of pellets
The obtained pellets are resuspended in fresh homogenization buffer and centrifuged again at their respective speeds.
This removes smaller contaminants trapped in the pellet. - Analysis
After separation, the isolated fractions are identified by testing specific marker enzymes or indicators.
These indicators confirms the particular organelles or particles present in the fraction.
Sample preparation for Differential centrifugation
The following are the steps of sample preparation-
- Cooling and preparation- All preparation is done on ice or in cold room at 0–4°C. It prevent enzymatic degradation of sample. In liver tissue, overnight fasting is done to remove glycogen because it can interfere with sedimentation.
- Washing and mincing- The tissue is washed repeatedly to remove blood. It is weighed and then roughly minced into fine pieces by scissors or razor blade.
- Buffer suspension- The minced tissue is suspended in chilled isotonic homogenization medium. Usually 10–20% weight/volume ratio is used. The medium contain 0.25 M to 0.32 M sucrose, mannitol or sorbitol with Tris-HCl, HEPES, PMSF, EDTA and DTT.
- Homogenization- The tissue suspension is homogenized to break the plasma membrane. Internal organelles are kept intact. It is done by Teflon-glass homogenizer or Dounce homogenizer with 5 to 10 gentle strokes.
- Filtration- The homogenate is passed through cheesecloth, gauze or nylon mesh screen. It removes solid particles, large debris, connective tissue and unlysed cell clumps. The filtered homogenate is then used for differential centrifugation.
What is Equilibrium (isopycnic) sedimentation?
Equilibrium sedimentation is a specialized type of density gradient centrifugation in which particles are separated only on the basis of their buoyant density.
It is also called isopycnic sedimentation or buoyant separation. In this method, the size and shape of particles do not decide the final position of particles in the tube.
In this technique, a steep density gradient is used. The gradient is generally made by sucrose or cesium chloride. The highest density of the gradient medium is greater than the density of particles which are to be separated.
During centrifugation, the particles move through the gradient medium. They continue to move until they reach the region where density of medium becomes equal to their own density.
At this position, the particles stop moving and form separate bands. This is called equilibrium state. The particles do not form pellet at the bottom of the tube even if centrifugation is continued for long time.
The size and mass of particles affect only the rate of movement. They do not affect the final position of the particle in gradient.
This method is used for separation of macromolecules which are similar in size but differ in density. It is used for separation of DNA, RNA, proteins and molecules having different isotopes.
Differential centrifugation vs density gradient centrifugation
Differential centrifugation vs Density gradient centrifugation
- Basis of separation- Differential centrifugation separate particles depending on their size. Density gradient centrifugation separate particles depending on their density.
- Medium used- Differential centrifugation is done in homogeneous medium. No sugar solution or density gradient is used. Density gradient centrifugation need density gradient medium like sucrose solution or cesium salts.
- Process- Differential centrifugation is carried out in series of steps with increasing speed. Larger particles settle first and then smaller particles settle in next steps. Density gradient centrifugation is carried out at one speed and particles settle as bands in the gradient.
- Purity- Differential centrifugation gives crude fractions. Smaller particles may be trapped in pellet of larger particles. So contamination is common. Density gradient centrifugation gives purified fractions as separate bands.
- Ease of use- Differential centrifugation is simple, fast and easy method. Density gradient centrifugation is difficult and need careful preparation of gradient and loading of sample.
- Sub-types- Differential centrifugation has no distinct sub-types. Density gradient centrifugation is of two types, rate-zonal centrifugation and isopycnic centrifugation.
| Basis | Differential centrifugation | Density gradient centrifugation |
|---|---|---|
| Basis of separation | It separate particles depending on their size. | It separate particles depending on their density. |
| Medium used | It is done in homogeneous medium. No sugar solution or density gradient is used. | It need density gradient medium like sucrose solution or cesium salts. |
| Process | It is carried out in series of steps with increasing speed. Larger particles settle first and smaller particles settle in next steps. | It is carried out at one speed. Particles settle as bands in the gradient. |
| Purity | It gives crude fractions. Smaller particles may be trapped in pellet of larger particles. So contamination is common. | It gives purified fractions as separate bands. |
| Ease of use | It is simple, fast and easy method. | It is difficult and need careful preparation of gradient and loading of sample. |
| Sub-types | It has no distinct sub-types. | It is of two types, rate-zonal centrifugation and isopycnic centrifugation. |
What is the difference between centrifugation and differential centrifugation?
The following are the applications of differential centrifugation-
- Separation of cellular organelles- Differential centrifugation is used to isolate cell organelles from tissue homogenate. It is used for mitochondria, lysosomes, microsomes and cell membrane fractions.
- Harvesting of intact cells- It is used to separate and collect whole biological cells from suspension. The cells settle as pellet at low speed.
- Nuclear separation- It is used as preliminary method for separation of crude nuclear fraction. The nucleus settle down with large cell debris during low speed centrifugation.
- Removal of large impurities- It is used to remove large debris, unbroken cells and larger contaminants from cell extract. It gives cleared extract for further centrifugation.
- Purification of non-living particles- It is used for crude purification of suspended particles. These include microparticles, nanoparticles, colloidal particles and viruses.
- Isolation of macromolecules- It is used in microbiology and cell biology for separation of macromolecules. It is a basic technique for partial purification of cellular components.
| Application | Description |
|---|---|
| Separation of cellular organelles | Differential centrifugation is used to isolate cell organelles from tissue homogenate. It is used for mitochondria, lysosomes, microsomes and cell membrane fractions. |
| Harvesting of intact cells | It is used to separate and collect whole biological cells from suspension. The cells settle as pellet at low speed. |
| Nuclear separation | It is used as preliminary method for separation of crude nuclear fraction. The nucleus settle down with large cell debris during low speed centrifugation. |
| Removal of large impurities | It is used to remove large debris, unbroken cells and larger contaminants from cell extract. It gives cleared extract for further centrifugation. |
| Purification of non-living particles | It is used for crude purification of suspended particles. These include microparticles, nanoparticles, colloidal particles and viruses. |
| Isolation of macromolecules | It is used in microbiology and cell biology for separation of macromolecules. It is a basic technique for partial purification of cellular components. |
Application of Differential centrifugation
The following are the applications of differential centrifugation-
- Separation of cellular organelles- It is used to isolate cellular organelles from tissue homogenate. Mitochondria, lysosomes, microsomes and membrane fractions are separated by this method.
- Harvesting of intact cells- It is used to separate and collect intact cells from suspension. The cells are settled as pellet at low speed.
- Nuclear separation- It is used for crude separation of nuclear fraction. Nucleus is separated with large debris in low speed centrifugation.
- Removal of large impurities- It is used to remove large debris, unbroken cells and large sized contaminants from extract. This gives cleared extract for further separation.
- Purification of non-living particles- It is used for crude purification of non-living suspended particles. Microparticles, nanoparticles, colloidal particles and viruses can be separated.
- Isolation of macromolecules- It is used in microbiology for separation of macromolecules. It is a basic method for partial purification of cellular components.
Advantages of Differential centrifugation
The following are the advantages of differential centrifugation-
- Simple method- Differential centrifugation is the simplest form of centrifugation. It is easier than density gradient centrifugation.
- Fast process- The process is fast and direct. It does not need long preparation.
- Bulk isolation- It is used for bulk isolation of cells and organelles. Large amount of sample can be processed by this method.
- No complex medium- It does not need complex sugar solution or density gradient medium. The separation is done in simple homogenization medium.
- Preliminary purification- It is useful for crude separation of particles. It removes larger impurities before high resolution purification.
- No added contaminant- It does not use sucrose or cesium chloride gradient. So contamination from added gradient medium is not present.
Limitations of Differential centrifugation
The following are the limitations of differential centrifugation-
- Co-sedimentation and contamination- In differential centrifugation, smaller unwanted particles may settle with larger target particles. So the pellet is not pure and crude fraction is obtained.
- Low recovery- Some larger target particles may remain in the supernatant if they start from upper part of the tube. They do not reach the bottom in proper time and are lost from the desired fraction.
- Overlapping sedimentation- Biological particles differ in size and density. Mitochondria, lysosomes and plasma membrane fragments may have overlapping sedimentation rate and they are mixed in same pellet.
- Damage to organelles- High centrifugal force may damage delicate organelles. The pellet may become very firm and difficult to resuspend, causing further damage during resuspension.
- Need further purification- Differential centrifugation is mostly used as preliminary separation method. For better purity, repeated washing and further method like density gradient centrifugation is needed.
- Experimental inconsistency- The result may vary in different laboratory. Small change in temperature, sucrose concentration and use of RPM instead of RCF can change the sedimentation result.
References
- Eppendorf Denmark. (2018). A short history of centrifugation.
- A comparison of the influence of 2.7% sorbitol-0.54% mannitol and 5% glucose irrigating fluids on plasma serum physiology during hysteroscopic procedures. (n.d.). PMC.
- Addgene. (n.d.). AAV purification by iodixanol gradient ultracentrifugation.
- Danaher Life Sciences. (n.d.). AAV purification – Methods, process and challenges.
- Fournier, L. (n.d.). Advancements in centrifugal microfluidics. Elveflow.
- de Carvalho, J. C., Molina-Aulestia, D. T., Martinez-Burgos, W. J., Karp, S. G., Manzoki, M. C., Medeiros, A. B. P., Rodrigues, C., Scapini, T., Vandenberghe, L. P. d. S., Vieira, S., Woiciechowski, A. L., Soccol, V. T., & Soccol, C. R. (2022). Agro-industrial wastewaters for algal biomass production, bio-based products, and biofuels in a circular bioeconomy. Fermentation, 8(12), 728.
- University of Liège. (n.d.). Albert Claude.
- Chakravarty, P. R. (n.d.). All about the composition of protein purification buffers and why it matters. GoldBio.
- STEMCELL Technologies. (n.d.). Cell centrifugation speed, time, and temperature.
- The Rockefeller University. (n.d.). Cell fractionation, biochemistry, and electron microscopy: The birth of modern cell biology. Hospital Centennial.
- Baruah, M. P. (n.d.). Centrifugation. Mangaldai College.
- Frei, M. (2011). Centrifugation basics. BioFiles, 6(5). Sigma-Aldrich.
- Frei, M. (2011). Centrifugation separations. BioFiles, 6(5), 6-7. Sigma-Aldrich.
- Centrifugation-based purification protocol optimization enhances structural preservation of nucleopolyhedrovirus budded virion envelopes. (n.d.). PMC.
- Corning. (n.d.). Centrifugation: Lab techniques, examples and types.
- Comparative studies of sucrose and mannitol utilization in celery (Apium graveolens). (n.d.). ResearchGate.
- Comparison of sucrose and sorbitol as main carbon energy sources in micropropagation of peach rootstock GF-677. (n.d.). ResearchGate.
- pluriSelect. (n.d.). Density gradient centrifugation explained simply.
- Difference between density gradient centrifugation and differential centrifugation. (n.d.). Youtube.
- Differential centrifugation. (2025). In Wikipedia.
- Drawbacks of dialysis procedures for removal of EDTA. (n.d.). PMC – NIH.
- Emerging microfluidic plasma separation technologies for point-of-care diagnostics: Moving beyond conventional centrifugation. (n.d.). PMC.
- Alberts, B., Johnson, A., Lewis, J., Raff, M., Roberts, K., & Walter, P. (2002). Fractionation of cells. In Molecular biology of the cell (4th ed.). Garland Science.
- Beckman Coulter. (n.d.). Fundamentals of ultracentrifugal virus purification.
- Guidelines for an optimized differential centrifugation of cells. (n.d.). PMC.
- Sankar, M. G. (2025). History today in medicine – Dr. Albert Claude. CME INDIA.
- Investigating centrifugation science: Methods and uses in various fields. (n.d.).
- Isolation of exosomes by differential centrifugation: Theoretical … (n.d.).
- Isolation of mitochondria from cells and tissues. (n.d.). PMC – NIH.
- Rama University. (n.d.). LT 25. Centrifugation.
- Mahlangu, D., Mphahlele, K., De Paola, F., & Mthombeni, N. H. (2024). Microalgae-mediated biosorption for effective heavy metals removal from wastewater: A review. Water, 16(5), 718.
- Eden Tech. (2025). Microfluidics in 2025: Applications, trends & how it work.
- Graham, J. M. (1992). Principles and practices of centrifugation. Beckman Instruments, Inc.
- Abcam. (n.d.). Nuclear extraction and fractionation protocol.
- Overview and challenges of large-scale cultivation of photosynthetic microalgae and cyanobacteria. (n.d.). PMC.
- Beckman Coulter. (n.d.). Principles of continuous flow centrifugation.
- Producing oleaginous microorganisms using wastewater: Methods and guidelines for lab- and industrial-scale production. (n.d.). PubMed.
- Sigma-Aldrich. (n.d.). Protease inhibition using protease inhibitor mix.
- Creative Proteomics. (n.d.). Protocol for differential velocity centrifugation.
- Creative BioMart. (n.d.). Protocol of preparation of the subcellular organelle protein samples.
- Gutka, H. (2018). Rational selection of sugars for biotherapeutic stabilization: A practitioner’s perspective. BioProcess International.
- Lian, S., Li, X., & Lv, X. (2025). Recent developments in SERS microfluidic chips: From fundamentals to biosensing applications. ACS Applied Materials & Interfaces, 17(7), 10193-10230.
- Yuan, H., Miao, Z., Wan, C., Wang, J., Liu, J., Li, Y., Xiao, Y., Chen, P., & Liu, B. F. (2025). Recent advances in centrifugal microfluidics for point-of-care testing. Lab on a Chip, 25(5), 1015-1046.
- The Protein Man. (2018). Role of additives in cell lysis. G-Biosciences.
- Sedimentation coefficient. (2025). In Wikipedia.
- Steiert, C., Busto, J. V., Melchionda, L., & Wiedemann, N. (2024). Subcellular fractionation by differential centrifugation for mitochondrial studies. Methods in Enzymology, 706, 61-73.
- Svedberg, T. (1927). The ultracentrifuge [Nobel Lecture].
- Nobel Prize Outreach. (1974). The Nobel Prize in Physiology or Medicine 1974 – Press release.
- The Rockefeller University. (n.d.). Cell fractionation, biochemistry, and electron microscopy: The birth of modern cell biology. Hospital Centennial.
- The role of density gradient centrifugation in virus purification. (n.d.).
- Theoretical foundations and practical methodologies of differential centrifugation in subcellular fractionation and industrial bioprocessing. (n.d.).
- Merck KGaA. (2021). Viral vaccine bioprocessing handbook. Sigma-Aldrich.
- Bangs Laboratories. (1999). Washing microspheres (TechNote 203).
- AAT Bioquest. (n.d.). What are the different types of centrifugation techniques?