Autoclave is a sterilizing instrument that is used to sterilize culture media, glasswares, surgical instruments and other materials by steam under pressure.
It is a strong metallic chamber with a tight lid. The materials are kept inside the chamber. Then steam is passed into the chamber after removing air.
Autoclave works at 121°C temperature and 15 lbs pressure for 15 minutes. In some cases 134°C temperature is used for short time.
The steam under pressure kills microorganisms. It kills bacteria, fungi, viruses and spores. This is done by coagulation of cellular proteins.
It is used in microbiology laboratory, hospital, dental clinic, pharmaceutical industry and research laboratory.
Definition of Autoclave
An autoclave is a machine that uses steam under pressure to sterilize materials by killing bacteria, viruses, and spores.
Autoclave Pressure and Temperature Chart
The following are the important autoclave pressure and temperature chart–
Autoclave Pressure and Temperature Chart
| Cycle / Method | Used for | Temperature | Pressure | Time |
|---|---|---|---|---|
| Gravity displacement cycle | Unwrapped items, liquids, waste | 121°C (250°F) | 15 psi | 15 to 30 minutes |
| Pre-vacuum cycle | Wrapped instruments, hollow tools | 132°C to 134°C (270°F to 273°F) | 27 to 30 psi | 3 to 10 minutes |
| Lightly wrapped items | Light wrapped articles | 132°C (270°F) | 30 psi | 8 minutes |
| Heavily wrapped items | Heavy wrapped articles | 132°C (270°F) | 30 psi | 10 minutes |
| Low temperature cycle | Heat sensitive media, plastics | 115°C to 118°C (239°F to 244°F) | About 15 psi | 20 to 45 minutes |
| Prion cycle | Neurosurgical tools | 134°C or above (273°F or above) | About 30 psi | 18 minutes or above |
| Adjustable lab cycle | Custom laboratory process | 105°C to 135°C (221°F to 275°F) | Variable pressure | 1 to 999 minutes |
| Dry heat wrapped items | Wrapped items | 170°C (340°F) | Not used | 60 minutes |
| Dry heat wrapped items | Wrapped items | 160°C (340°F) | Not used | 120 minutes |
| Dry heat wrapped items | Wrapped items | 150°C (300°F) | Not used | 150 minutes |
| Dry heat wrapped items | Wrapped items | 140°C (285°F) | Not used | 180 minutes |
| Dry heat wrapped items | Wrapped items | 121°C (250°F) | Not used | 12 hours |
| Rapid flow dry heat | Unwrapped items | 190°C (375°F) | Not used | 6 minutes |
| Rapid flow dry heat | Packaged items | 190°C (375°F) | Not used | 12 minutes |
| Chemical vapor sterilization | Articles sterilized by chemical vapor | 132°C (270°F) | 20 to 40 psi | 20 minutes |
| Ethylene oxide sterilization | Heat sensitive articles | Ambient temperature | Not used | 8 to 10 hours |
Principle of an Autoclave – How an Autoclave Works?
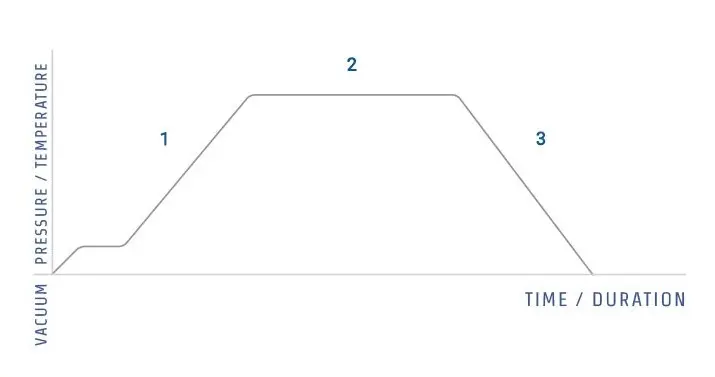
Autoclave is based on moist heat sterilization.
It is based on steam under pressure. In this process water boils at high temperature due to pressure. The steam formed is saturated steam. It is generally 121°C to 134°C.
The steam is entered into the chamber. The articles are exposed to this steam. The steam comes in contact with the articles and condenses on it. In this time latent heat is released.
This heat kills the microorganisms. The proteins of microbial cell are coagulated. The enzymes are denatured. So the cell activity is stopped and the cell is destroyed.
Air is removed from the chamber before sterilization. Because air does not allow proper penetration of steam. After removal of air, steam is maintained for required time. It is usually 15 to 30 minutes.
After this the steam is released. Pressure becomes normal. Thus bacteria, viruses, fungi and bacterial spores are killed by saturated steam under pressure.
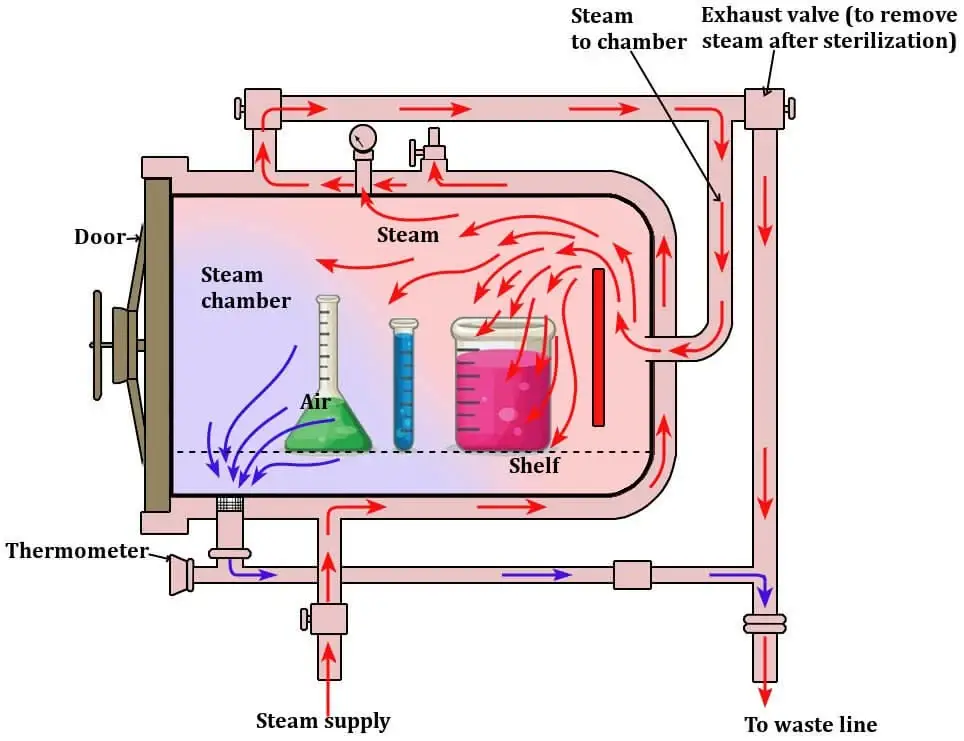
Mode of Action of Autoclave
The following are the mode of action of autoclave–
- In autoclave, saturated steam at high temperature is entered into the pressure chamber. The temperature is generally 121°C to 134°C. The steam surrounds the microorganisms and comes in direct contact with them.
- When the steam touches the cooler microbial cells, it condenses into water. During condensation, large amount of latent heat is released on the surface of microorganisms. This heat produces the killing action.
- The moisture from the steam hydrates the microbial cells. Due to this, the resistance of microorganisms is reduced. Even dry and resistant bacterial spores become more sensitive to heat.
- The high moist heat damages the outer cell membrane of bacteria. The membrane becomes weak and ruptured. So the normal protective covering of the cell is lost.
- The moist heat breaks the bonds present in proteins and enzymes. The essential proteins are denatured and coagulated. Due to this enzyme activity is stopped and the cell metabolism is destroyed.
- The heat also damages the DNA and RNA of microorganisms. So replication and growth is stopped. Finally microorganisms are killed by destruction of membrane, proteins and genetic materials.
Parts of Autoclave
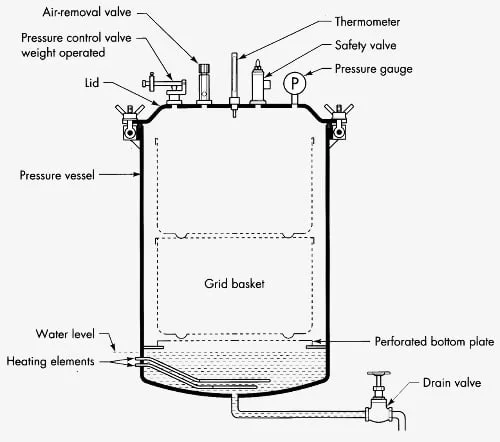
The following are the important parts of autoclave–
- Pressure Chamber or Vessel
It is the main body of the autoclave. It is generally made up of stainless steel. The materials are kept inside this chamber for sterilization. - Steam Jacket
It is the outer covering present around the inner chamber. It is filled with steam. It helps to preheat the wall of chamber and reduces condensation. - Lid or Door
It is used to close the chamber from outside atmosphere. It allows the pressure and temperature to develop inside the autoclave. It has locking system for safe working. - Door Gasket
It is a rubber seal present around the door. It gives air tight closing of the chamber. It prevents leakage of steam during sterilization. - Steam Generator or Electrical Heater
It is used to produce saturated steam under pressure. This steam is used for killing microorganisms inside the chamber. - Vacuum Generator or Pump
It is present in pre-vacuum type autoclave. It removes air from the chamber before entry of steam. It also helps in removing moisture after sterilization. - Control System and Interface
It is the control panel of the autoclave. It is used to set time, temperature and pressure. The cycle is controlled automatically by this system. - Sensors and Gauges
These are used to measure temperature, pressure and humidity inside the chamber. They show the condition of the sterilization cycle. - Safety Valve
It is an important safety part of autoclave. It releases extra steam when the pressure becomes high. Thus it prevents over pressure inside the chamber. - Whistle
It is present in some autoclaves. It releases small amount of extra steam. It helps in controlling pressure like pressure cooker. - Drain and Exhaust System
It is used to remove condensate and steam from the chamber. After sterilization, it helps to reduce the pressure of chamber. - Wastewater Cooling System
It cools the hot waste water, steam and condensate coming out from autoclave. It prevents damage of drainage pipe. - Drying System
It is an optional part of autoclave. It uses vacuum or heat for drying the sterilized materials. It prevents moisture on wrapped load during storage.
Approximate Conditions for Autoclave sterilization
The following are the important approximate conditions for autoclave sterilization–
- Standard or Gravity displacement cycle
Temperature- 121°C (250°F)
Pressure- 15 psi or about 100-103 kPa
Time- 15 to 30 minutes
The time may be increased upto 60 to 90 minutes when the load is large, dense or more in amount. - Pre-vacuum cycle
Temperature- 132°C to 134°C (270°F to 273°F)
Pressure- 27 to 30 psi
Time- 3 to 10 minutes
It is used for wrapped instruments, porous loads and hollow tools. - Low-temperature cycle
Temperature- 115°C to 118°C (239°F to 244°F)
Pressure- about 15 psi
Time- 20 to 45 minutes
It is used for heat-sensitive plastics and laboratory media. - Prion decontamination cycle
Temperature- 134°C (273°F) or above
Pressure- about 30 psi
Time- at least 18 minutes
It is used for very resistant infectious proteins. Sometimes chemical treatment is also used with it. - Steam quality
The steam used in autoclave should be saturated and good quality steam. It should contain about 97% vapour and 3% moisture. This moisture helps in proper transfer of heat and makes sterilization effective.
Operating Procedures of Autoclave – How to Use an Autoclave?
The following are the important operating procedures of autoclave–
1. Preparation
- The instruments are first washed and rinsed properly.
Any dust, blood, chemical or other residues should be removed before sterilization. - The materials are kept in proper autoclave safe containers.
Liquid containers should not be filled completely. It should be filled upto two-third part only. - The caps and lids of bottles are kept slightly loose.
This allows entry of steam and prevents breaking of containers due to pressure. - Autoclave indicator tape is placed on the materials or covering foil.
It is used to show that sterilization temperature has been reached. - The materials are kept in a secondary heat resistant tray.
It may be made up of stainless steel or autoclavable plastic. It helps to collect any leakage or spill.
2. Loading the Autoclave
- The autoclave chamber is checked before loading.
The power is switched on and drain valve is closed. The drain strainer is also checked and cleaned. - In manual-fill autoclave, water is added upto the marked level.
Generally deionized water is used. - The tray or basket containing the materials is placed inside the chamber.
The articles should not be overcrowded. They should not touch the chamber wall or bottom directly. - Solid materials and liquid materials are not mixed in same load.
They are sterilized separately because their heating and cooling time is different. - The door or lid is closed properly.
The handle is locked to make the chamber air tight.
3. Operating the Autoclave
- The required cycle is selected according to the type of load.
Gravity cycle is used for dry goods and glassware. Liquid cycle is used for fluids. Pre-vacuum cycle is used for wrapped and porous materials. - The required temperature, pressure and time are set.
Generally 121°C (250°F) temperature and 15 psi pressure is used. - The sterilization time is usually 15 to 30 minutes.
It may vary according to the type and amount of materials. - The start button is pressed.
Then the sterilization cycle is allowed to complete.
4. Unloading the Autoclave
- The autoclave is opened only after completion of the cycle.
The pressure gauge should show 0 psi before opening the door. - Proper protective materials are used before unloading.
Heat resistant gloves, lab coat, closed shoes and face shield or eye protection are used. - The door is opened slowly and slightly first.
This allows remaining hot steam to escape safely. - The sterilized articles are removed carefully.
Liquid containers should not be shaken because it may boil suddenly. - The glasswares are allowed to cool for about 15 minutes.
Liquids are allowed to cool for about 1 hour before handling without gloves.
Types of Autoclave
The following are the important types of autoclave–
A. On the basis of sterilization function and air removal
- Gravity Displacement Autoclave (Class N)
In this autoclave steam enters into the chamber and pushes the air downward. The air comes out through the drain port. It is used for unwrapped and non-porous solid materials. - Pre-Vacuum or High-Vacuum Autoclave (Class B)
In this autoclave air is removed by vacuum pump before entry of steam. After removal of air, steam enters properly into the load. It is used for wrapped, hollow and porous materials. - Specific Load Autoclave (Class S)
This autoclave is used for some specific products. It uses partial vacuum or steam pulsing. The materials which can be sterilized are given by the manufacturer. - Steam Flush Pressure Pulse Autoclave (SFPP)
In this type, air is removed by repeated steam flushing and pressure pulses. It is used for sensitive and complex instruments. It is also energy saving type. - Liquid Cycle Autoclave
This autoclave is used for sterilization of liquids. It has slow exhaust system. It prevents boiling over of media, broth and other laboratory liquids. - Prion Cycle Autoclave
This type is used for prion contaminated instruments. It uses higher temperature and longer time. It is used for resistant infectious proteins.

B. On the basis of size and orientation
- Tabletop or Benchtop Autoclave
It is small and compact autoclave. It is front loading type. It is used in dental clinic, tattoo centre and small laboratory. - Vertical Autoclave
It is top loading cylindrical autoclave. It occupies less space. It is commonly used in research laboratory. - Horizontal Autoclave
It is large front loading autoclave. It can sterilize large amount of materials at a time. It is used in hospitals, large laboratories and pharmaceutical production. - Industrial Autoclave
It is very large autoclave. It is made for heavy and bulky load. It is used for medical waste, rubber vulcanization and industrial materials. - Mobile or Portable Autoclave
It is small and light weight autoclave. It can be carried from one place to another. It is used in field hospitals and rural clinics.
C. On the basis of chamber and door design
- Single Door Autoclave
It has only one door. The materials are loaded and unloaded from same side. It is the common type of autoclave. - Double Door or Pass-Through Autoclave
It has two doors on opposite sides. One side is used for loading and another side is used for unloading. It separates sterile and non-sterile area and prevents cross contamination. - Hinged Autoclave
It is a single door autoclave with strong hinged door. The door is opened and closed by hinge system. - Tower Autoclave
It has two chambers arranged one above another. It saves space and used for high amount of sterilization work.
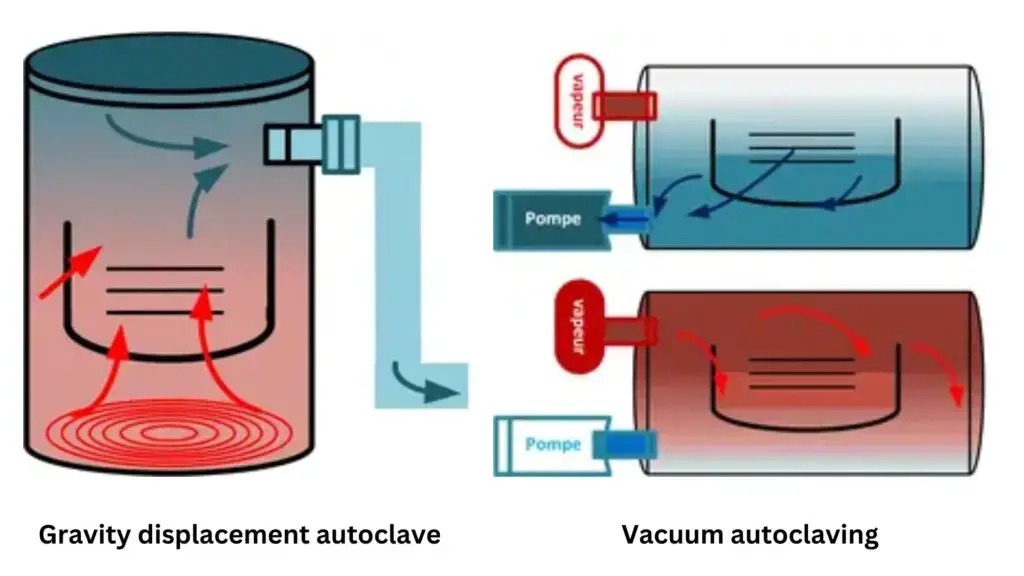
Classes of Autoclaves
The following are the classes of autoclaves–
- Class N Autoclave (Naked) – Class N autoclave uses gravity displacement method where steam removes air from the chamber. It is used only for unwrapped, non-porous solid materials and it is not suitable for wrapped, porous or hollow loads.
- Class S Autoclave (Specified) – Class S autoclave uses partial vacuum or steam pulsing system. It is made for specific products and load types as given by the manufacturer, so it is more versatile than Class N autoclave.
- Class B Autoclave (Big or Universal) – Class B autoclave uses full pre-vacuum and post-vacuum system for deep removal of air. It is used for all types of loads like wrapped, double-wrapped, porous textiles and hollow instruments, so it is considered as the universal class of autoclave.
Compatible/incompatible materials for the autoclave
The following are the compatible and incompatible materials for autoclave–
A. Compatible materials
- Glassware
Heat resistant borosilicate glass can be autoclaved. Example- Pyrex and Kimax. - Metals
Stainless steel materials can be autoclaved. Surgical instruments and biological research instruments are also autoclaved. - Plastics
Heat resistant plastics can be autoclaved. Example- Polypropylene (PP), PTFE (Teflon), Polyetheretherketone (PEEK), PFA, Polyacetal and Polymethylpentene. - Polycarbonate and Polysulfone
Polycarbonate (PC) and Polysulfone can be autoclaved. But they become weak after repeated autoclave cycle. - Elastomers
Silicone and Viton can be autoclaved. - Liquids and solutions
Aqueous solutions, tissue culture media, water and glycerol can be autoclaved. - Fabrics
100% cotton, natural fibres, hospital linens and bedding can be autoclaved. - Laboratory items
Pipette tips made of PP, culture flasks and packed biohazardous waste can be autoclaved.
B. Incompatible materials
- Heat sensitive plastics
Polyethylene (PE), HDPE, LDPE, Polystyrene (PS), Polyurethane, Polyvinyl chloride (PVC), Acrylic, Nylon, Polyester and Phenol cannot be autoclaved. These may melt, bend or release harmful chemicals. - Hazardous chemicals
Flammable, volatile, reactive, toxic and corrosive chemicals cannot be autoclaved. Acids, bases, organic solvents and Trizol are not autoclaved. - Chlorine and bleach
Household bleach, chlorine, chlorides and sulfates cannot be autoclaved. Bleach may release toxic chlorine gas and damage the autoclave. - Dangerous biological and chemical agents
Radioactive materials, carcinogens and mutagens cannot be autoclaved. - Sealed containers
Tightly closed bottles or containers cannot be autoclaved. The vapour expands inside and may cause bursting. - Oils and waxes
Oils and waxes cannot be autoclaved properly. They repel water and steam cannot penetrate into them. - Other incompatible items
Seawater, glucose, non-stainless metals, synthetic fabrics and paper products are not generally autoclaved. Paper products are used only when packed for waste disposal.
Autoclave Cycle Time Frame
The following are the autoclave cycle time frame–
- Total cycle time – The complete autoclave cycle includes heating, sterilization exposure and cooling or drying. It generally takes about 30 to 60 minutes.
- Gravity or standard steam cycle – This cycle is done at 121°C (250°F). The exposure time is generally 15 to 30 minutes for normal load, but dense load or biohazardous waste may require about 60 minutes.
- Pre-vacuum or high-temperature cycle – This cycle is done at 132°C to 135°C (270°F to 275°F). The exposure time is generally 3 to 10 minutes because air is removed actively. Unwrapped items may take 3 minutes, lightly wrapped items take 8 minutes and heavily wrapped items take 10 minutes.
- Liquid cycle – This cycle is done at 121°C (250°F). The exposure time is generally 15 to 45 minutes, depending on the volume of liquid. The slow exhaust cooling time is not included in this time.
- Prion cycle – This cycle is done at 134°C (273°F) or above. It is used for highly resistant infectious proteins and it needs at least 18 minutes exposure time.
- Dry heat sterilization – This method needs longer exposure time than steam sterilization. It takes 60 minutes at 170°C, 120 minutes at 160°C, or 12 hours at 121°C. In rapid flow dry heat cycle, it may take 6 to 12 minutes at 190°C.
- Note – The exact exposure time depends on size, shape, weight, density and material of the load. So time may change according to the materials which are sterilized.
Factors Affecting Sterilisation Effectiveness of autoclave
The following are the important factors affecting sterilisation effectiveness of autoclave–
- Time, Temperature and Pressure – These are the main factors of autoclave sterilisation. The autoclave should reach proper temperature like 121°C or 134°C with required pressure and it should be maintained for sufficient time, otherwise all microorganisms are not destroyed.
- Steam Quality – The steam should be saturated and proper quality steam. It should contain about 97% vapour and 3% moisture, because this helps in good heat transfer. Very wet steam or very dry steam reduces the sterilisation effect.
- Air Removal – Air should be removed from the chamber before sterilisation. Air acts as an insulator and it does not allow steam to reach all parts of the load. If air remains inside, cold spots are formed and sterilisation is not proper.
- Direct Steam Contact – Steam should come in direct contact with all surfaces of the materials. If any surface is covered or blocked, steam cannot touch that part and that part may remain unsterile.
- Load Configuration and Density – The load should not be overcrowded or packed very tightly. If materials are placed too closely or pouches are stacked flat, steam circulation becomes poor and steam cannot enter into the centre of the load.
- Type and Quantity of Material – The nature and amount of material affects sterilisation. Porous textiles, solid metals and instruments with long narrow lumen need different penetration of steam and different time for sterilisation.
- Container Size and Shape – The size and shape of container also affects autoclaving. Some containers may block removal of air and entry of steam. So proper container should be used for good steam penetration.
Sterilization control / Quality Control of Autoclave / Validation of Autoclave
The following are the different methods of sterilization control of autoclave–
- Physical Monitoring – Physical monitoring is done by using built-in display, sensors, pressure gauge and cycle printout of autoclave. It checks the temperature, pressure and exposure time during the cycle. If required temperature, pressure and time are reached, then the cycle condition is considered proper. But it only shows the physical condition of the cycle and does not directly prove sterility.
- Chemical Indicators (CIs) – Chemical indicators are heat sensitive strips or tapes which change colour after exposure to autoclave condition. Type 1 process indicator such as autoclave tape is used outside the pack and it shows that the pack is exposed to heat. Type 5 and Type 6 indicators are placed inside the pack and they change colour only when time, temperature and steam condition are properly reached.
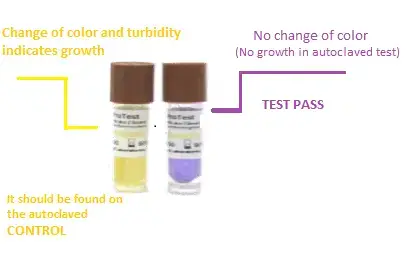
- Biological Indicators (BIs) – Biological indicator is considered as the best method for checking autoclave sterilization. It contains highly resistant bacterial spores, generally Geobacillus stearothermophilus. After autoclaving, the vial or strip is incubated for 24 to 48 hours. If there is no growth of spores, then it indicates that the cycle has killed microorganisms properly.
- Process Challenge Device (PCD) – Process challenge device is used to test the most difficult condition of sterilization. It is made to act like a difficult load where steam penetration is not easy. It may contain chemical indicator or biological indicator inside it. It is placed in the difficult area of the autoclave chamber to check whether sterilization is effective or not.
- Bowie-Dick Test or DART – Bowie-Dick test is used for pre-vacuum autoclave. It is generally done daily before routine work. It checks whether air is removed properly from the chamber and steam is penetrating uniformly into porous load. If air remains inside, the test result becomes improper.
- Helix Test – Helix test is used for hollow instruments. It checks whether steam can enter into long and narrow lumen. It is useful for dental handpiece, tubing and hollow surgical instruments. Proper colour change in the indicator shows that steam penetration into lumen is complete.
- Vacuum Leak Test – Vacuum leak test is done to check the air tightness of the autoclave chamber. In this test, deep vacuum is created inside the chamber. Then it is observed whether air is entering from outside or not. If air leaks inside, steam sterilization may fail because air prevents proper steam contact.
- Installation Qualification (IQ) – Installation qualification is the documented checking of autoclave installation. It shows that the autoclave is installed in correct place and connected properly with water, steam, electricity and drain. Calibration and basic parts are also checked in this method.
- Operational Qualification (OQ) – Operational qualification is done to check the working of autoclave without load or with test condition. It checks whether the autoclave can reach and maintain required temperature, pressure and time repeatedly. Alarm, safety control and empty chamber temperature distribution may also be checked.
- Performance Qualification (PQ) – Performance qualification is the final validation step. In this method, actual load or worst-case load is sterilized repeatedly. It proves that the autoclave can sterilize the real materials used in the laboratory or hospital. If all test cycles pass, then the autoclave is considered suitable for routine use.
Uses of Autoclave
The following are the important uses of autoclave–
- Autoclave is used to sterilize surgical instruments, dental tools, implants, catheters, surgical drapes and hospital linens. It helps to prevent infection in hospital and clinical work.
- It is used to sterilize laboratory glassware, reusable instruments, pipette tips, culture media, agar plates, animal cages and bedding. It is commonly used in microbiology and research laboratory.
- Autoclave is used to decontaminate pathogenic medical and laboratory waste before disposal. Used gloves, culture plates, samples and other infected materials are sterilized before municipal disposal.
- It is used for sterilization of containers, bioreactors, reagents and processing equipment. It is also used in vaccine and biological drug production work.
- Autoclave is used in curing aerospace composites, vulcanizing rubber and making pressure treated wood. It is also used for porous concrete and laminated safety glass production.
- It is used to sterilize packaged foods, canned goods, dairy products and food processing utensils. It kills microorganisms and increases shelf life of food materials.
- Autoclave is used to sterilize needles and reusable tools used in tattooing, body piercing and nail salon work.
- It is used to sterilize instruments used in veterinary clinic and funeral homes.
- Autoclave is used for artificial aging of materials. It helps to test quality, lifespan and physical resistance of materials.
Advantages of Autoclave
The following are the important advantages of autoclave–
- Autoclave is highly effective method of sterilization. It uses steam under pressure, which can penetrate the contaminated articles and kills microorganisms including resistant bacterial spores.
- It is used for sterilizing different types of materials. Surgical instruments, glasswares, liquids and some porous materials can be sterilized by autoclave.
- It is an environment friendly method. It uses only steam and water. So no harmful chemical, reagent or detergent is required in this process.
- It is also used for safe treatment of medical waste before disposal. So it reduces the problem of infected waste and also avoids the use of incineration in many cases.
- The process is reliable and gives repeated result when it is operated properly. Temperature, pressure and time can be controlled during the sterilization cycle.
- It is cost effective in long term use. Many laboratory and medical instruments can be reused after sterilization. The machine also works for many years if maintained properly.
- It is safer for operator than chemical sterilization. Steam is non-toxic for the operator but it is lethal for microorganisms.
Limitations of Autoclave
The following are the important limitations of autoclave–
- Autoclave cannot be used for heat sensitive materials. Some plastics like PET, HDPE, LDPE, PVC and polystyrene may melt or change shape due to high heat and moisture.
- It is not suitable for moisture sensitive materials. Paper products and electronic items may be damaged by steam and water. Paper is autoclaved only when it is packed for waste disposal.
- Flammable, volatile, corrosive, toxic, explosive and radioactive materials cannot be autoclaved. Chlorine bleach should also not be autoclaved because it may release toxic gas and damage the autoclave.
- Oils and waxes are not sterilized properly by autoclave. These materials repel water. So steam cannot penetrate into them and sterilization is not proper.
- Tightly sealed bottles or containers should not be autoclaved. The vapour expands inside the container and it may cause bursting or explosion.
- Autoclave is costly in installation and operation. Large and advanced autoclaves need high cost. They also use electricity and water during operation.
- It needs trained operator for proper use. Wrong loading, wrong cycle selection and poor handling may cause sterilization failure or accident.
- Autoclave needs regular maintenance and calibration. If temperature, pressure and safety parts are not checked, the sterilization process may not be reliable.
- Standard autoclave cycle may not destroy some highly resistant agents like prions and some bacterial toxins. They need special long cycle and chemical pre-treatment like sodium hydroxide.
- Gravity displacement autoclave is not suitable for porous materials, wrapped packs and hollow instruments. Air may remain inside and steam penetration becomes poor.
- Liquid cycle takes longer time than normal cycle. This is because liquids need slow exhaust and cooling to prevent boiling over.
Precautions
The following are the important precautions of autoclave–
- Proper PPE should be used while loading and unloading the autoclave. Heat resistant gloves, eye protection or face shield, closed shoes and lab coat should be used.
- The bottles or containers should not be closed tightly. The caps should be kept slightly loose. It prevents pressure formation inside the container and also prevents bursting.
- The chamber should not be overcrowded with materials. There should be enough space between the articles. This allows proper circulation and penetration of steam.
- Flammable, volatile, toxic, reactive, corrosive and radioactive materials should not be autoclaved. Oils, waxes and household bleach should also be avoided because these are not suitable for steam sterilization.
- Liquid containers should be filled only upto two-third part. It prevents boiling over of liquid. Liquids or leaking materials should be kept in secondary heat resistant tray.
- Only autoclavable bags and containers should be used. Ordinary plastic should not be used. Items should not be wrapped very tightly with aluminium foil because it prevents steam penetration.
- Sharp objects like broken glass should not be placed in autoclave bag. They should be kept in proper sharp container before disposal or treatment.
- The door or lid should be closed and locked properly before starting the cycle. It gives proper sealing of the chamber. The door should never be opened while the autoclave is running.
- Autoclave should be opened only after the cycle is completed. The pressure gauge should show 0 psi before opening the door.
- Hot liquid containers should not be shaken after removal. Their caps should not be opened immediately. Super heated liquid may boil suddenly and cause injury.
- Sterilized materials should be allowed to cool before touching with bare hands. Glasswares should be cooled for at least 15 minutes and liquid loads for about 1 hour.
- The autoclave should be kept on stable surface and the door area should not be blocked. If strange smell, sound or leakage is observed, the power should be turned off and the machine should not be repaired by untrained person.
References
- All Retorts. (n.d.). Autoclave.
- Alpha Scientific. (n.d.). What Are The Primary Parts Of An Autoclave?
- AmeriWater. (n.d.). What You Need to Know About AAMI ST108 [PDF].
- Association for the Advancement of Medical Instrumentation (AAMI). (n.d.). AAMI STANDARD 108 [PDF].
- Baron, A. (n.d.). Autoclave | Definition, Parts & Uses [Video]. Study.com.
- Berlin Packaging. (n.d.). What is Autoclaving?
- Beta Star Life Science Equipment. (n.d.). What Can and Cannot Be Autoclaved.
- Centers for Disease Control and Prevention. (n.d.). Infection Control for CJD.
- Cn-Meditech. (n.d.). Autoclave Cycle Types Explained: Gravity vs Pre-Vacuum vs Liquid Cycle.
- Collins, F. M. (n.d.). Proper loading of autoclaves: the dos and don’ts. Tuttnauer.
- Collins, F. M. (n.d.). Wet Packs in Tabletop Autoclaves: A Common and Preventable Problem. Tuttnauer.
- Complete Smiles Bella Vista. (n.d.). Study: Causes of Autoclave Failures in Dental Clinics.
- Consolidated Sterilizer Systems. (n.d.). 1 How to Validate an Autoclave [PDF].
- Consolidated Sterilizer Systems. (2025). AAMI ST108 & Critical Water for Autoclaves: What ASCs Need to Know.
- Das, P. C. (2012, November 1). Application Of The F0 Value in Steam Sterilizer. Pharmaceutical Resource & Education Services.
- Ellab. (2019, November 21). The Steam Sterilization Process. AZoM.
- Euronda Pro System. (n.d.). Bowie & Dick tests: what is it and why is it important?
- Fisher Scientific. (n.d.). Sterilization Verification Indicators.
- Frankel, E. (2025, January 30). Autoclave Working Principle Explained Simply: What You Need to Know. Surplus Solutions.
- Grifols. (n.d.). Autoclave: Functions, Importance, and Types in Pharma.
- Jacobs School of Medicine and Biomedical Sciences. (n.d.). Operating Procedure for Steam Autoclaves.
- jibimed. (n.d.). The Science Behind Saturated Steam Sterilization and Why It Matters.
- Malhotra, V. (2025, November 5). Autoclave: Definition, Principle, Parts, Uses, Applications. Presto Group.
- Mascherpa, V. (2014). Highlights on “equivalent time” F0. Fedegari [PDF].
- Mason Technology. (n.d.). Autoclaves: Applications and Uses.
- Medical Equipment Services. (n.d.). What Materials Cannot Be Autoclaved?
- MedicalExpo. (n.d.). Choosing the Right Sterilizer – Buying Guides.
- MedSolut AG. (n.d.). Insight into the historical development of autoclaves: importance of.
- My Green Lab. (n.d.). How to Save Water in the Lab: 10 Practical Sustainability Tips for Scientists.
- My Green Lab. (n.d.). Laboratory Water Conservation: Reducing Your Lab’s Hidden Footprint.
- Nevsink. (n.d.). What are Biological Indicators for Autoclaves.
- Office of Energy & Sustainability. (n.d.). BRB Water Conservation – Autoclave Timers. Weill Cornell Medicine.
- PharmaJia. (n.d.). What is F0 value in moist heat sterilization process.
- Pharmaguideline. (n.d.). Principle and Working of Autoclave | Steam Sterilization [Video]. YouTube.
- Pistolesi, D., & Mascherpa, V. (2014). F0 a technical note. Fedegari [PDF].
- Presto Group. (n.d.). What is an Autoclave? Working Principle, Types, and Applications Explained.
- Princeton University. (n.d.). Autoclave Use | Office of Environmental Health and Safety.
- Princeton University. (n.d.). Autoclave Validation | Office of Environmental Health and Safety.
- RAYPA. (n.d.). Bowie & Dick Test: What is it and why is it important?
- RAYPA. (n.d.). Essential Tips for Proper Autoclave Loading.
- Roson, F. S. (n.d.). Autoclave Maintenance: Avoid Mistakes, Ensure Reliability. Roson Dental.
- Sakudo, A., Yamashiro, R., & Onodera, T. (2022). Recent Advances in Prion Inactivation by Plasma Sterilizer. International Journal of Molecular Sciences, 23(18), 10241.
- Schwenke, K. A., Wagenführ, K., Thanheiser, M., & Beekes, M. (2022). Kinetics of the reduction of Creutzfeldt-Jakob disease prion seeding activity by steam sterilization support the use of validated 134°C programmes. Journal of Hospital Infection, 132, 125-132.
- Singh, A. (2023, July 18). Autoclave: Principle, Types, and Precautions. Plant Cell Technology.
- Spire Integrated Solutions. (n.d.). What are the Critical Steps for Loading a Steam Sterilizer.
- STERI-Green. (n.d.). STERI-Green® Water Re-Circulating Systems [Video]. YouTube.
- STERIS. (2022, March 24). Everything About Autoclaves. Knowledge Center.
- STERIS. (n.d.). The Importance of Water Quality in Medical Device Processing.
- Tank Fab. (n.d.). Autoclave Systems: Acceptable Vs. Unacceptable Materials.
- The George Washington University. (n.d.). Autoclave Safety – Office of Research Safety.
- TOMY. (n.d.). Autoclave Guide: What Can & Can’t Be Autoclaved.
- Total Medical Compliance. (n.d.). Sterilization and Disinfection: Common Mistakes to Avoid!
- Trapotsis, A. (n.d.). Autoclave Steam Sterilization Cycles, Part 6: F0 Cycle. Consolidated Sterilizer Systems.
- Trapotsis, A. (n.d.). Bowie-Dick & Vacuum Leak Tests: Autoclave Sterilization Cycles. Consolidated Sterilizer Systems.
- Trapotsis, A. (n.d.). Prion Sterilization: Common Challenges. Consolidated Sterilizer Systems.
- Tuttnauer. (n.d.). Autoclave Sterilization: Cycle Selection, Validation & Tips.
- Tuttnauer. (n.d.). Autoclave Temperature Range for Effective Sterilization.
- Tuttnauer. (n.d.). Autoclave Troubleshooting: Sterilization Failures, Causes, and Fixes.
- Tuttnauer. (n.d.). Bowie-Dick Test and Cycle – Ensuring Autoclave Sterilization Efficacy.
- Tuttnauer. (n.d.). EN 13060 Class B Tabletop Autoclaves.
- Tuttnauer. (n.d.). How Does an Autoclave Work?
- Tuttnauer. (n.d.). Key Components of Autoclaves for Effective Sterilization.
- Tuttnauer. (n.d.). Steam in the Autoclave.
- Tuttnauer. (n.d.). Types of Autoclave Sterilizers – A Comprehensive Guide.
- Tuttnauer. (n.d.). Who Invented the Autoclave and Its Historical Impact.
- UCSD Blink. (2024, March 2). Autoclave Overview.
- UMBC. (n.d.). Autoclave Validation – Environmental Safety and Health.
- University of Pittsburgh. (n.d.). Efficient Autoclaves – Pitt Sustainability.
- Unknown Author. (n.d.). Autoclave Time Temperature Pressure Chart. Sterilizer.
- Unknown Author. (n.d.). Comprehensive Engineering and Applied Science of Saturated Steam Sterilization Systems [Markdown].
- Waheed, M. R., Abdulhussein, D. A., Hassan, M. H., Hssein, H. J., & Salman, M. D. (2025). The autoclave: Engineering and applications in medical sterilization. International Journal of Research in Medical Science, 7(1), 118-124.
- W&H Impex Inc. (n.d.). Types of sterilizers.
- Wikipedia. (n.d.). Autoclave.
- Xylem US. (n.d.). ANSI/AAMI ST108 – Water for the Processing of Medical Devices.
Helpful Note, Thank You