Wave Bioreactor is a single-use cultivation system that is used for growing cells in a sterile flexible plastic bag.
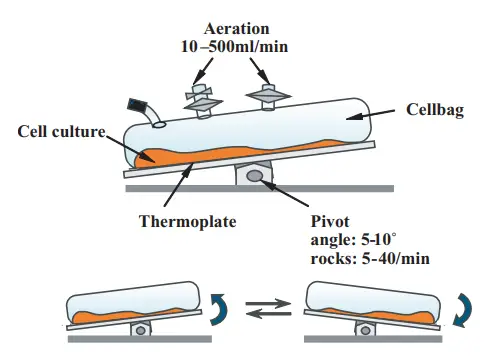
It is also known as rocking bioreactor. In this bioreactor, the culture medium and cells are placed inside a partially filled disposable bag which is kept on a heated motorized platform. The platform moves back and forth and forms a continuous wave inside the liquid.
This wave helps in mixing of nutrients and oxygen transfer without using stirrer or impeller. Aeration takes place at the liquid and gas surface, so bubble formation and high shear stress is avoided.
Wave bioreactor is mainly used for the cultivation of shear sensitive cells such as mammalian cells, insect cells, plant cells and stem cells. It also reduces cleaning and sterilization steps because disposable bags are used. It is used in vaccine production, cell therapy, gene therapy and seed culture preparation before large scale production.

Working Principle of Wave Bioreactor
Wave bioreactor is based on the principle of wave-induced motion for mixing and aeration of cell culture.
In this system, the cells and culture medium are kept in a partially filled single-use flexible plastic bag. The bag is placed on a motorized and temperature controlled rocking platform. When the platform moves back and forth, it produces a continuous traveling wave inside the liquid.
This wave keeps the cells in uniform suspension and distributes the nutrients equally in the medium. It also helps in surface aeration without formation of bubbles. Oxygen from the head space of the bag diffuses across the liquid surface and carbon dioxide is removed from the medium.
There is no internal stirrer or sparger in this bioreactor. So the kinetic energy is distributed in the whole liquid instead of being concentrated at impeller tip. Due to this, very low shear stress is produced and delicate cells are protected from mechanical damage.
Types of Wave Bioreactor
Wave bioreactors are classified into different types depending upon the movement of platform, structure of vessel, operational use and target application. The following are the important types of wave bioreactor-
On the basis of degree of motion
a. One Degree of Freedom (1-DOF) Bioreactors- These are the most common type of commercial wave bioreactors. In this type, the rocking platform moves only along one axis, generally Y-axis. This movement produces wave motion inside the bag and helps in mixing and aeration of culture medium.
Examples include Cytiva WAVE 25, Sartorius Biostat RM and Eppendorf BioBLU Rocking bioreactor.
b. Two Degrees of Freedom (2-DOF) Bioreactors- These bioreactors have standard rocking movement with one additional movement component. This type of motion increases mixing efficiency and oxygen transfer rate (OTR). It is useful for shear sensitive cells because the cells are not damaged by harsh agitation.
CELL-tainer is the main example of 2-DOF wave bioreactor.
On the basis of vessel structure and format
a. Flexible 2D Pillow Bag Bioreactors– This is the standard form of wave bioreactor. It uses a flexible disposable plastic bag which is placed flat on a heated rocking platform. The rocking movement forms waves in the liquid medium.
Examples include Cytiva Cellbag and Sartorius Flexsafe RM bags.
b. Rigid-Walled Rocking Bioreactors– In this type, the flexible bag is replaced by a rigid-walled single-use vessel. The vessel is fixed on the rocking platform. It gives more fixed geometry like traditional glass bioreactor but also has the benefit of single-use system.
Example includes Eppendorf BioBLU rocking bioreactor.
On the basis of operational configuration
a. Standard Batch or Fed-Batch Bioreactors– These are simple wave bioreactors used for normal cell expansion. The bags are provided with basic ports and sensors such as optical pH and dissolved oxygen (DO) sensors. It is mostly used in seed train and basic cell culture process.
b. Perfusion-Capable Bioreactors– These bioreactors are used for continuous processing. They have cell-retention devices which remove spent medium but keep the cells inside the bag. This helps in maintaining high cell density for longer period.
Cell retention is done by floating lily pad filters, internal membranes or dip tube filters.
On the basis of target application
a. Mammalian Cell Culture Wave Bioreactors- These bioreactors are designed for mammalian cells which are shear sensitive. They work at lower agitation range, generally up to 25 rpm. It gives gentle mixing and proper oxygen transfer without damaging the cells.
b. Microbial and Fungal Wave Bioreactors- These bioreactors are designed for bacteria, yeast and fungal cultures. They require higher rocking rate, generally up to 45 rpm. This is because microbial cells need higher oxygen transfer and higher specific power input than mammalian cells.
Parts of Wave Bioreactor
The wave bioreactor is made up of various parts, including-
- Rocking platform or base unit – It is the mechanical foundation of the wave bioreactor. It has motorized drive mechanism that tilts the bag and creates continuous wave motion. It also has bag holders and cam-locks to hold the culture bag tightly on the platform.
- Single-use culture bag – It is a sterile and flexible cultivation chamber. It is pillow-like in shape and made up of multi-layer plastic films. It contains the cells, culture medium and gases for cell growth.
- Control tower and automation system – It is the central unit of the wave bioreactor. It contains electronics, software and controllers. It is used to monitor and regulate the whole bioprocess.
- Gas supply and manifold – It is used to supply gases into the headspace of the bag. The gases include air, oxygen, carbon dioxide and nitrogen. It helps in oxygenation and pH control of the culture medium.
- Heating and temperature control – It consists of heater mats present on the rocking platform. It warms the bottom of the culture bag. Temperature sensors are inserted into protective thermowells to monitor and maintain the required temperature.
- Ports, connectors and tubing – These are pre-installed aseptic connectors, lines and needleless sampling ports. They are used for safe addition of media, inoculum and other solutions. They also allow sampling without breaking the sterile barrier.
- Air filters – These are hydrophobic vent filters present on the gas inlet and exhaust lines. They allow sterile gas exchange and prevent the entry of contaminating microorganisms into the culture bag.
- Sensors and measurement tools – These include load cells and optical sensor spots. Load cells are present in the rocker and used to monitor culture weight and volume. Optical sensor spots are present inside the bag and used to measure pH and dissolved oxygen continuously.
Operating Procedure of Wave Bioreactor
The operating procedure of wave bioreactor is carried out in different steps, including-
- Preparation and equilibration
- The single-use bioreactor bag such as Cellbag is first inflated with air and 10% CO2 until it becomes fully rigid.
- The culture medium is added into the bioreactor bag and the inlet and outlet air filters are closed with clamps.
- The rocking platform is started at low speed such as 15 rocks per minute and with gentle rocking angle such as 6° to 7°.
- The system is allowed to run for 1 to 2 hours. During this time, temperature and pH of the medium become equilibrated.
- A sample is taken before inoculation to check the pH. If required, the pH is adjusted before adding the cells.
- Inoculation
- The cells are added into the bioreactor bag to obtain the required starting cell density.
- In many processes, the culture is started at about 50% of the final culture volume. This helps to increase the surface area to volume ratio during initial growth phase.
- The inlet and outlet filters are kept closed during the early stage of culture. This helps in maintaining steady pH inside the bag.
- Routine operation and monitoring
- The bioreactor bag is kept rigidly inflated during the whole operation. This is necessary for proper gas exchange and also reduces foam formation.
- Cell density, cell viability and metabolic condition are monitored regularly.
- Glucose concentration is checked and pre-warmed fresh medium is added when it is required.
- When active cell growth occurs, the pH and glucose level start decreasing. This generally occurs within 24 to 60 hours.
- At this stage, the filters are opened and continuous sweep gas flow is started through the headspace.
- The sweep gas may be 5% CO2 mixed with air. It helps in maintaining gas exchange and pH control.
- Adjustment of parameters for high cell density
- Dissolved oxygen (DO) level is continuously monitored by integrated optical sensors, controller system or by offline blood gas analyzer.
- As the cell density increases, oxygen demand also increases. So rocking speed and rocking angle are increased slowly in stepwise manner.
- This increases oxygen transfer and helps in proper mixing of the culture medium.
- If too much foam is formed, the rocking angle is increased and rocking speed is decreased. This helps in breaking the foam.
- If oxygen becomes limiting at high cell density, the headspace gas is enriched with pure oxygen (O2).
- pH is controlled by changing the CO2 concentration in the headspace gas.
- If pH falls below the required level due to lactic acid formation, liquid base can be added automatically by the system.
Applications of Wave Bioreactor
- Wave bioreactor is used for seed-train expansion and inoculum preparation. It acts as a bridge between small laboratory flasks and large production bioreactor. It helps to increase healthy cell mass before transferring the culture into main production vessel.
- It is used in cell and gene therapy processes. It provides closed, sterile and low shear condition for growth of sensitive primary cells. Human T cells, CAR-T cells and stem cells can be expanded in this system without cross contamination between patient batches.
- Wave bioreactor is used for production of recombinant proteins and monoclonal antibodies. It is used for cultivation of different mammalian and insect cell lines. CHO, HEK293, NS0 and hybridoma cells are commonly grown in this system.
- It is used for production of vaccines and viral vectors. Viral particles such as lentivirus and adeno-associated virus can be produced in wave bioreactor. It is also used for growth of adherent cells like Vero and MDCK cells for vaccine production.
- Wave bioreactor is used in process development and scale-down studies. It has low capital cost and is easy to operate. So it is useful for rapid prototyping, early clone screening and media optimization.
- It is used for culture of anchorage dependent cells on microcarriers. The gentle rocking motion prevents collision of carriers and reduces cell damage. Hillex, Cultispher, Cytodex beads and Fibra-Cel disks are used in such cultures.
- Wave bioreactor is used for plant cell and tissue culture. It is useful for study of plant physiology and production of recombinant compounds. As there is no internal impeller, hairy root cultures do not get tangled or clog the system.
- It is used for perfusion and continuous bioprocessing. Cell retention devices such as ATF or TFF filters are connected with the system. These filters help to remove spent medium and add fresh nutrient medium, while cells remain inside the bag.
- Wave bioreactor can also be used for microbial and yeast cultivation. By changing rocking speed, rocking angle and oxygen enriched headspace, high oxygen demanding cultures can be grown. E. coli and Pichia pastoris are important examples.
- It is used in early stage of cultivated meat research. It helps in expansion of animal cells such as bovine satellite cells on microcarriers for in vitro meat production.
Advantages of Wave Bioreactor
The following are the advantages of wave bioreactor-
- Wave bioreactor provides ultra-low shear stress and gentle agitation. The wave motion mixes the culture without using mechanical impellers. So shear sensitive mammalian cells, plant cells and stem cells are protected from physical damage.
- It gives efficient bubble-free surface aeration. The continuous rocking motion breaks and renews the liquid surface again and again. This provides good oxygen transfer and gas exchange without using submerged spargers.
- It eliminates complex cleaning procedures. The system uses pre-sterilized disposable bags, so clean-in-place (CIP) and steam-in-place (SIP) processes are not required.
- It reduces the risk of cross-contamination. The culture bag is closed and single-use type. This decreases the contamination entry points and improves the process safety.
- It gives faster turnaround time. The setup is simple and there is no cleaning delay after each batch. So different production batches or products can be changed quickly.
- It requires lower cost and smaller facility space. Wave bioreactor needs less initial capital investment. It also reduces the requirement of large facility utilities like pharmaceutical water and steam system.
- It supports high cell density culture. Low hydrodynamic stress and proper distribution of nutrients and gases allow better cell growth. So high cell density and faster growth rate can be obtained.
- It reduces foaming in the culture system. Oxygenation occurs through the headspace and not by internal gas bubbling. So foam formation is very less and chemical antifoaming agents are generally not required.
Limitations of Wave Bioreactor
The following are the limitations of wave bioreactor-
- Wave bioreactor has limited scale and capacity. It cannot be scaled up like stirred tank bioreactor. The working volume is generally limited between 200 to 1000 liters, so it may restrict large scale commercial production.
- It has lower oxygen transfer rate. This is because it depends mainly on surface aeration and not on spargers. So it is not much suitable for bacteria, yeast and fungi which need high oxygen supply.
- It is sensitive to operating volume. Gas transfer depends on the filling volume of the bag. If the bag is filled more than the optimum level, generally above 50%, headspace and surface area to volume ratio decreases and oxygen transfer is also decreased.
- Carbon dioxide accumulation may occur at high cell density. Removal of CO2 becomes difficult and it affects the pH stability of the culture medium.
- Foaming may occur during high rocking speed and high rocking angle. Foam can block the exhaust filters. This may cause pressure buildup and sometimes bag rupture.
- It is not suitable for highly viscous culture. The rocking motion cannot mix thick culture properly, such as fast growing plant cells producing polysaccharides or very high density perfusion cultures.
- It has fewer sensors and control points. The disposable bag has fixed geometry and limited ports. So inline sensors and controllable parameters are less as compared to stainless steel stirred tank bioreactor.
- The single-use flexible bag may get punctured or leaked. This is a material related limitation of the wave bioreactor.
- Leachables and extractables may come out from the multi-layer plastic films into the culture medium. This may happen more at higher temperature and can inhibit cell growth or affect the final product quality.
- It has continuous cost and waste problem. Single-use bags are expensive and they are needed again and again. This also increases solid waste disposal cost, especially in long term continuous culture.
Precautions of Wave Bioreactor
The following are the important precautions of wave bioreactor–
- Single-use cultivation bag should be handled carefully.
It is made up of plastic material and may be punctured easily. If puncture occurs then leakage of culture medium and contamination may takes place. - The bag should be properly inflated before starting the process.
It should remain rigidly inflated during the whole operation. If the bag has low internal pressure, then proper wave formation and mixing will not occur. - The rocking speed and rocking angle should be controlled carefully.
Very high rocking speed and angle may produce more shear stress. It may also remove cells from the microcarriers. Very slow rocking is also not suitable because it causes poor mixing, poor growth and low oxygen supply. - Excess foam formation should be avoided.
Foam may reach the exhaust filter and block it. This causes pressure build-up inside the bag and may lead to bag rupture. Foam can be controlled by reducing rocking angle, adding antifoam agent or using foam trap. - The exhaust filter should be protected from condensation.
Condensation may wet and block the sterile vent filter. For this, proper exhaust gas flow and heating element should be used. - The temperature probe should touch the culture fluid properly.
If the probe does not remain in proper contact with the fluid, the system may show wrong temperature. This may cause overheating of the cells, especially in low culture volume. - Oxygen limitation should be checked during high cell density culture.
In wave bioreactor, oxygenation mainly occurs from the headspace by surface aeration. So at high cell concentration, gas transfer may become limiting. In such condition, pure oxygen may be supplied to the headspace. - High heat producing process should be operated with proper cooling.
Wave bioreactor cannot remove very large amount of heat. In processes like methanol induction in yeast culture, the bioreactor should be kept in refrigerated condition to prevent overheating. - In perfusion culture, bubble accumulation should be prevented.
During alternating tangential flow (ATF) perfusion, gas bubbles may collect in dip tubes. It can reduce filtration efficiency and may cause shear damage to the cells. So rocking motion should be adjusted properly. - All connections and bag fittings should be checked before operation.
Loose connection may cause leakage, pressure loss and contamination. So inlet, outlet, gas line, sampling line and filter should be fixed properly before starting the culture.
References
- Modeling and validating of oxygen transport in wave bioreactors: optimized experimental mass transfer method and novel Lattice-Boltzmann CFD approach. (n.d.). ResearchGate. [URL]
- 50L, 200L, 500L, 1000L or 2000L Stainless Steel Disposable Bioreactor. (n.d.). lab1st-eshop. [URL]
- Rojewska, O., & Tęczar, M. (n.d.). Advanced Bioreactor Technologies in Bioprocess Operations. Mabion. [URL]
- Advanced Films for Single-Use Bioprocessing Bags. (n.d.). Dow. [URL]
- Advantages of Single-layer Film vs. Multilayer Film for use in Bio-processing Bags. (2018). Entegris. [PDF]
- BioBLU f Single-Use Bioreactors for microbial cultures. (n.d.). Making.com. [PDF]
- BioBLUc Single-Use Bioreactors Product Brochure. (n.d.). Eppendorf. [PDF]
- BioBLU® c Single-Use Bioreactors. (n.d.). Eppendorf. [URL]
- Amanda. (2024, May 24). Bioreactor Manufacturers. The Lab World Group. [URL]
- Biostat STR® Generation 3 Single-Use Bioreactor. (n.d.). Sartorius. [URL]
- Biostat® RM – Wave-Mixed Bioreactor. (n.d.). Sartorius. [URL]
- Seidel, S., Maschke, R. W., Kraume, M., Eibl, R., & Eibl, D. (2022). CFD modelling of a wave-mixed bioreactor with complex geometry and two degrees of freedom motion. Frontiers in Chemical Engineering, 4, 1021416. [URL]
- Cellbag™ bioreactors. (2020). Cytiva. [PDF]
- China WAVE Single-use Bioreactor For Cell Culture Manufacturer and Supplier. (n.d.). LABOAO. [URL]
- Design and Use of the Wave Bioreactor for Plant Cell Culture. (n.d.). [URL]
- Disposable Tank Bioreactor Market Outlook 2026-2034. (2026, April 29). Intel Market Research. [URL]
- Filter concepts for perfusion. (n.d.). CerCell. [URL]
- How Do Optical Dissolved Oxygen Sensors Work? (2022). Scientific Bioprocessing. [URL]
- Leak Resistant Film for Single-Use Bioprocessing Bags. (2026). Sigma-Aldrich. [URL]
- Measurement Principle of Chemical Optical Sensors. (n.d.). PreSens Precision Sensing GmbH. [URL]
- Bell, D. (2025, September 2). Mixing in Bioreactors: Scale-Up Best Practices. Cultivated Meat Shop. [URL]
- Piontek, S., Fitschen, J., Weiland, C., Habicher, T., Schlüter, M., & Wucherpfennig, T. (2026). Modeling and validating of oxygen transport in wave bioreactors: optimized experimental mass transfer method and novel Lattice-Boltzmann CFD approach. Frontiers in Bioengineering and Biotechnology, 13, 1688774. [URL]
- Modeling and validating of oxygen transport in wave bioreactors: optimized experimental mass transfer method and novel Lattice-Boltzmann CFD approach. (n.d.). PMC. [URL]
- Multilayer Film Structure. (n.d.). Saint-Gobain Performance Plastics. [PDF]
- Optical Dissolved Oxygen Sensors & Principles of Operation. (n.d.). Knowledge Center. [URL]
- Optical Fiber pH and Dissolved Oxygen Sensors for Bioreactor Monitoring: A Review. (n.d.). PMC. [URL]
- Shaughnessey, E., Opitz, A., & Prior, J. (2018, June 23). Oxygen Mass Transfer Correlation for a Rocking-Motion Bioreactor System. BioProcess International. [URL]
- Perfusion culture using TFF or ATF as cell retention method. (2020). Cytiva. [PDF]
- Perfusion: hardware needs. (n.d.). Thermo Fisher Scientific. [PDF]
- Principles of quenched fluorescence (QF). (2025, December 9). Endress+Hauser. [URL]
- Shakers, Rockers, and Stirred Tank Bioreactors – Advantages of Each. (n.d.). Alicat Scientific. [URL]
- Single-Use Bioreactor Selection Guide: Wave, Stirred-Tank, and Fixed-Bed Compared. (2026, April). BioProcess Tools. [URL]
- Single-Use Bioreactors. (2026). Eppendorf. [URL]
- Single-Use Bioreactors. (n.d.). American Pharmaceutical Review. [URL]
- Technical Evolution and Bioengineering Characterization of Wave-Induced Rocking Bioreactors. (n.d.). [Markdown]
- Top 20 Companies in Global Bioreactors Market Size Report. (2026, April). Spherical Insights. [URL]
- Top Bioreactor Companies – Key Players & More. (n.d.). Mordor Intelligence. [URL]
- Cytiva. (n.d.). Understanding the Wave Bioreactor™: Cell culture technology [Video]. YouTube. [Youtube]
- Jurkiewicz, E., Husemann, U., Greller, G., Barbaroux, M., & Fenge, C. (2014). Verification of a new biocompatible single-use film formulation with optimized additive content for multiple bioprocess applications. Biotechnology Progress, 30(5), 1171–1176. [URL]
- Clincke, M.-F., Mölleryd, C., Zhang, Y., Lindskog, E., Walsh, K., & Chotteau, V. (2013). Very high density of CHO cells in perfusion by ATF or TFF in WAVE Bioreactor™. Part I. Effect of the cell density on the process. Biotechnology Progress, 29(3), 754–767. [URL]
- Very high density of CHO cells in perfusion by ATF or TFF in WAVE bioreactor™. Part I. Effect of the cell density on the process. (n.d.). PubMed. [URL]
- WAVE Bioreactor Systems Cell culture procedures. (2020). Cytiva. [PDF]
- WAVE Bioreactor 200 system. (2020). Cytiva. [PDF]
- Wave Bioreactor: A Modern Approach to Scalable Cell Culture. (n.d.). BaiLun Biotechnology Co., Ltd. [URL]
- Wave Bioreactor: What It Is, How It Works, and When It Makes Sense. (n.d.). ZETA BIOSYSTEM. [URL]
- Collignon, M.-L. (2025, August 27). What are the different types of bioreactors? Cytiva. [URL]