Spectrophotometer is an analytical instrument which is used to measure the light absorbed, transmitted or reflected by a substance at particular wavelength.
It has a light source, sample holder and detector. The liquid sample is kept in a cuvette. Then the beam of light is passed through the sample.
The detector detects the light which comes out from the sample. The amount of light absorbed is calculated by comparing the incident light and transmitted light.
Different molecules absorb different wavelength of light. So, it is used for finding the concentration and purity of a sample. It works according to Beer-Lambert Law.
Spectrophotometer is used in chemistry, clinical diagnosis, environmental monitoring and quality control.
Principle of Spectrophotometer
Principle of Spectrophotometer is based on the absorption and transmission of light by the sample solution.
When a beam of light is passed through the sample, some light is absorbed by the molecules present in it. Some light passes through the sample.
The absorbed light depends on the concentration of the substance and the length of the path through which light passes.
This follows Beer-Lambert Law. According to this law, absorbance is directly proportional to concentration and path length.
If the concentration is more, absorption of light is also more. So, less light comes out from the sample.
The difference between the incident light and transmitted light is measured. From this, the concentration and chemical composition of the sample is determined.
Types of Spectrophotometer
The following are the types of Spectrophotometer–
- Single-beam spectrophotometer
This spectrophotometer has only one light beam. The beam of light is passed through the sample and then it reaches the detector. The blank is taken separately. - Double-beam spectrophotometer
This spectrophotometer has two light beams. One beam is passed through the sample and another beam is passed through the blank. The reading is compared directly. - Split-beam spectrophotometer
This type uses a beam splitter. A part of light is passed to the reference detector and other part is passed through the sample. It gives stable baseline. - UV-Visible spectrophotometer
This spectrophotometer measures absorbance in UV and visible region. The range is generally 200 nm to 700 or 800 nm. - Ultraviolet spectrophotometer
This type measures absorbance in ultraviolet region. The range is 200-400 nm. - Visible spectrophotometer
This type measures absorbance in visible region. The range is 400-700 nm. - Infrared spectrophotometer
This spectrophotometer measures absorption or transmission of infrared light. It is used for molecular vibration and structure. - Fluorescence spectrophotometer
This spectrophotometer measures fluorescence. The sample gives light after excitation by light. - Atomic absorption spectrophotometer
This spectrophotometer measures absorption of light by free atoms. It is used for detecting and measuring metal ions. - Diode array spectrophotometer
This type has photodiode array detector. It measures many wavelengths at the same time. - Digital spectrophotometer
This is modern type of spectrophotometer. It has digital display, automatic calibration and data storage system.

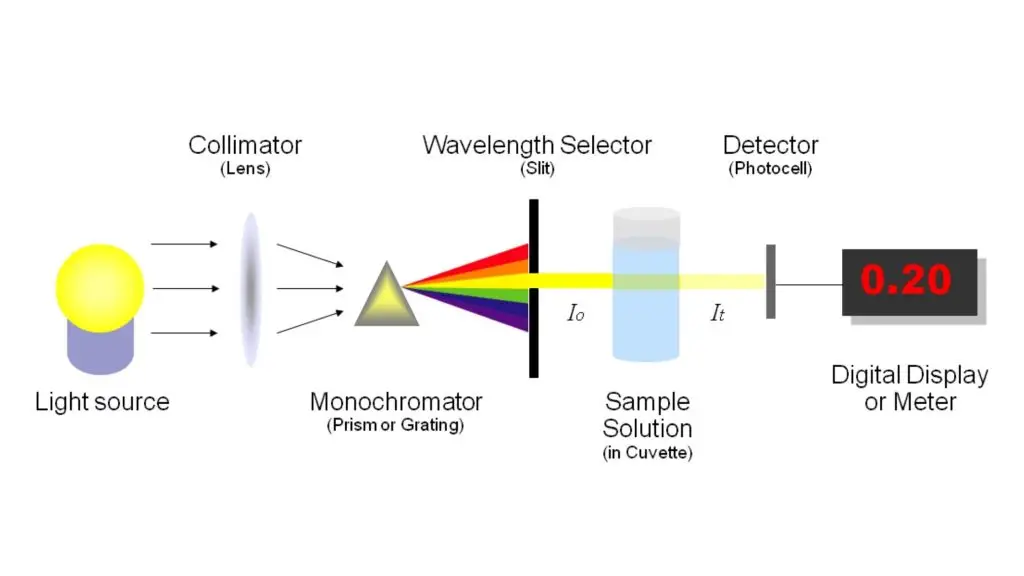
Instrumentation of Spectrophotometer/Parts of Spectrophotometer
The following are the main parts of Spectrophotometer–
- Light source
It gives light for the measurement. The light should be stable and strong. Tungsten-halogen lamp is used for visible light and deuterium lamp is used for ultraviolet light. - Monochromator
It is used to select a particular wavelength of light. It separates the light into different wavelength. It has entrance slit, prism or diffraction grating and exit slit. - Sample holder or Cuvette
It is a small transparent tube like container. The sample solution is kept in it. It is placed in the path of light. Quartz cuvette is used for UV light. - Beam splitter and mirrors
These are present in double beam spectrophotometer. Beam splitter divides the light into two paths. One path is for sample and another path is for blank. Mirrors are used to direct the light beam. - Detector
It detects the light which comes out from the sample. Then it changes the light energy into electrical signal. Photomultiplier tube, photodiode and photovoltaic cell are used as detector. - Signal processor
It receives the electrical signal from detector. It amplifies the signal and changes it into digital signal. - Readout device or Display
It shows the final result. The result is shown as absorbance or transmittance on digital screen or computer.
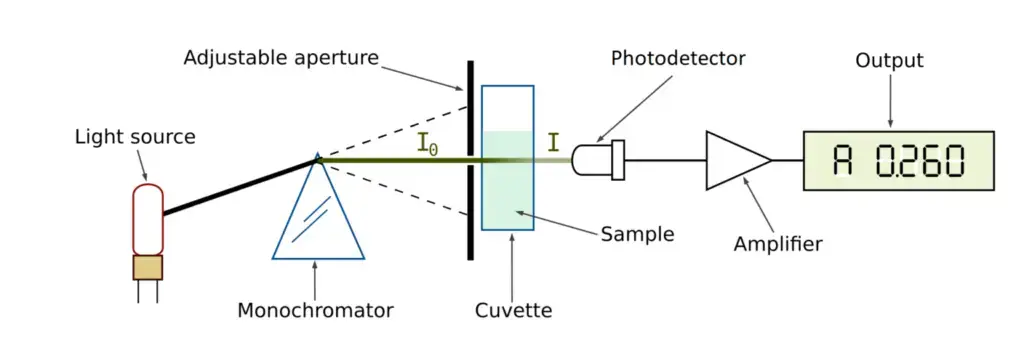
How does a spectrophotometer work? – Working Mechanism of Spectrophotometer
The working mechanism of Spectrophotometer is as follows-
- First the instrument is calibrated by using blank solution. Blank solution contains solvent but it does not contain the test substance. It is used to set the zero reference.
- Then light is produced from the light source. Tungsten-halogen lamp or deuterium lamp is generally used as light source.
- The light is passed through the monochromator. It separates the light and selects a particular wavelength which is required for the test.
- The selected wavelength of light is passed through the sample solution. The sample is kept in a transparent container called cuvette.
- During this process some amount of light is absorbed by the molecules present in the sample. The remaining light passes through the sample and this is called transmitted light.
- The transmitted light is detected by the photodetector. It changes the light energy into electrical signal.
- The instrument compares the original light and transmitted light. Then absorbance is calculated according to Beer-Lambert Law. The result is shown on the display as absorbance or transmittance.
Operating Procedure of spectrophotometer
The operating procedure of Spectrophotometer is as follows-
- First the spectrophotometer is switched on and allowed to warm up for about 45 minutes. It is done for stabilizing the instrument.
- The required wavelength is selected according to the experiment.
- The blank solution is taken in a clean cuvette. The blank contains only solvent or buffer without sample.
- The blank cuvette is placed in the sample holder and the lid is closed. The reading is adjusted to zero.
- The sample solution is taken in another clean cuvette. The outside of the cuvette is wiped with clean tissue to remove dust, liquid and fingerprints.
- The sample cuvette is placed in the holder. The clear side of the cuvette is kept towards the light path and the lid is closed.
- The Read or Measure button is pressed. The reading is allowed to become stable.
- The absorbance value is recorded. Then the cuvette is removed and same process is repeated for other samples.

Advantages of Spectrophotometer
The following are the advantages of Spectrophotometer–
- It gives result very fast. Same sample can be measured again and again and gives repeatable reading.
- It gives accurate and sensitive result. Very small change in absorbance can also be detected.
- It does not destroy the sample. After measurement the sample remains same.
- It requires less sample preparation. The process is simple and can be done easily.
- It can be used for different types of sample. Liquids, solutions and transparent solids can be tested.
- It is a simple and cost effective method. It is used in routine laboratory and industrial work.
- Modern spectrophotometer has digital display and data output system. Some instruments also have software connection and multi wavelength scanning.
- It is used in pharmaceuticals, environmental science, food and beverage, clinical diagnosis and textile industry.
- It is useful in quality control. It helps to reduce waste, maintain product quality and keep batch to batch consistency.
Limitations of Spectrophotometer
The following are the limitations of Spectrophotometer–
- It needs clear liquid sample. Turbid, opaque and highly scattering sample cannot give proper result.
- It works properly only in low concentration. At high concentration Beer-Lambert Law is not followed correctly.
- If molecules associate, dissociate or react in the solution, the reading becomes incorrect. The change of pH also affects the result.
- Stray light can enter into the detector. So, error may be produced in the reading.
- If monochromatic light is not proper, the absorbance value may not be accurate.
- Dirty or scratched cuvette gives wrong result. Fingerprint, dust and liquid drop on cuvette also affects the reading.
- Temperature change, vibration and voltage change can affect the measurement.
- In single-beam spectrophotometer, the blank is measured separately. It is slower and signal drift may occur.
- Double-beam spectrophotometer is more stable but it is costly. It is also large in size and needs more maintenance.
Applications of Spectrophotometer
The following are the applications of Spectrophotometer–
- It is used in clinical diagnosis for testing blood and urine sample. It helps to measure protein, glucose and hemoglobin.
- It is used in diagnosis of diseases like anemia and diabetes.
- It is used in pharmaceutical industry for drug analysis. It helps to measure the concentration of active drug substance.
- It is used to test drug purity, dissolution and stability of drug.
- It is used in biochemistry and molecular biology for measuring DNA, RNA and protein concentration.
- It is used to check purity of DNA, RNA and protein sample.
- It is used in enzyme study and ELISA test.
- It is used in environmental monitoring for testing water, soil and air quality.
- It is used to detect pollutants, heavy metals, phosphate, nitrate and toxic gases.
- It is used in food and beverage industry for checking colour, freshness, sugar and alcohol content.
- It is used to detect harmful contaminants and pesticides in food sample.
- It is used in textile and paint industry for colour matching and dye formulation.
- It is used to maintain same colour quality in different production batch.
- It is used in chemical and material science for studying reaction rate and unknown compounds.
- It is used to study thin films and optical property of semiconductors.
- It is used in petroleum industry for checking fuel quality and impurities.
- It is used in agriculture and soil science for testing soil nutrient and pH.
- It is also used to detect chlorophyll deficiency in plants.
Precautions of Spectrophotometer
The following are the precautions of Spectrophotometer–
- Safety goggles and gloves should be used during handling of chemicals and samples.
- Food and drink should not be kept near the instrument.
- The spectrophotometer should be kept in clean and dry place.
- Direct sunlight, excess moisture and sudden temperature change should be avoided.
- The instrument should be warmed up before taking reading. Generally 15 to 45 minutes is needed for stabilization.
- Blank solution should be used before sample reading. The blank should contain same solvent which is used in the sample.
- Cuvette should be held only from frosted side or non optical side.
- The clear side of cuvette should not be touched by fingers. Fingerprint can scatter light and gives high absorbance.
- Dirty or scratched cuvette should not be used. It gives wrong result.
- The outside of cuvette should be wiped with lint free tissue before placing in the instrument.
- Correct cuvette should be used according to light. Quartz cuvette is used for UV measurement and glass or plastic cuvette is used for visible light.
- Air bubbles and suspended particles should not be present in sample solution.
- Cuvette should not be overfilled or underfilled.
- The sample chamber lid should be closed properly during reading.
- The optical parts inside the instrument should not be touched by bare hand.
Difference between Spectrometer and Spectrophotometer
The following are the difference between Spectrometer and Spectrophotometer–
- Spectrometer is a general instrument which is used to separate and measure different spectral components. Spectrophotometer is a specific instrument which is used to measure absorption, transmission or fluorescence of light by a sample.
- Spectrometer may be used for light, mass or other physical phenomenon. Spectrophotometer is mainly used for light and sample interaction.
- In spectrometer, light is not always passed through the sample. It may be used only to measure the spectrum of a light source.
- In spectrophotometer, light is passed through the sample kept in a cuvette. Then absorption or transmission of light is measured.
- Spectrometer gives spectrum of light at different wavelengths. Spectrophotometer gives absorbance or transmittance value.
- Spectrometer is mainly used for optical analysis, wavelength separation and study of light source.
- Spectrophotometer is mainly used in chemistry, biology and life science for finding concentration of chemical, protein and nucleic acid.
- Modern diode-array spectrometer can record full spectrum very fast. Some traditional spectrophotometer measures one wavelength at a time, so it is slower.
- Spectrophotometer contains spectrometer part inside it. So, spectrometer is a part of spectrophotometer system.
| Basis | Spectrometer | Spectrophotometer |
|---|---|---|
| Definition | Spectrometer is a general instrument which is used to separate and measure spectral components. | Spectrophotometer is a specific instrument which is used to measure absorption, transmission or fluorescence of light by sample. |
| Scope | It may be used for light, mass or other physical phenomenon. | It is mainly used for light and sample interaction. |
| Sample use | In this instrument, light is not always passed through the sample. | In this instrument, light is passed through the sample kept in cuvette. |
| Main work | It measures spectrum of light or energy distribution at different wavelength. | It measures how much light is absorbed or transmitted by the sample. |
| Output | It gives spectrum at different wavelengths. | It gives absorbance or transmittance value. |
| Application | It is used for optical analysis, wavelength separation and study of light source. | It is used in chemistry, biology and life science for finding concentration of chemical, protein and nucleic acid. |
| Speed | Modern diode-array spectrometer records full spectrum very fast. | Traditional spectrophotometer measures one wavelength at a time, so it is slower. |
| Relation | Spectrometer may work as separate instrument. | Spectrophotometer contains spectrometer part inside it. |
Differences between Spectrometer and Spectrophotometer
| Basis | Spectrometer | Spectrophotometer |
|---|---|---|
| Definition | Spectrometer is a general instrument which is used to separate and measure spectral components of a physical phenomenon. | Spectrophotometer is a specific instrument which is used to measure the amount of light absorbed or transmitted by sample. |
| Scope | It has broad scope. It may be used for light, mass or nuclear magnetic resonance. | It has limited scope. It is mainly used for measuring light interaction with sample. |
| Instrument system | It may work as a separate instrument. | It contains spectrometer as a main measuring part inside it. |
| Sample interaction | In this instrument, light is not always passed through the sample. It can measure intensity and wavelength of light source. | In this instrument, light is passed through or reflected from the sample. The sample may be solution kept in cuvette. |
| Main work | It measures the spectrum and separates different wavelengths. | It measures how much light is absorbed or transmitted by the sample. |
| Output | It gives spectrum of light at different wavelengths. | It gives absorbance or transmittance value along with spectrum. |
| Use of result | It is used to know wavelength distribution and optical energy. | It is used to know concentration and purity of substance. |
| Applications | It is used in physical science, optical analysis, wavelength separation and study of light source. | It is used in chemistry, biology, life science and drug analysis. |
| Example work | It can be used for studying emission of different light sources. | It can be used for measuring protein, DNA, RNA or active drug concentration in solution. |
Difference between UV Spectroscopy and Visible Spectrophotometry
| Basis | UV Spectroscopy | Visible Spectrophotometry |
|---|---|---|
| Full form | UV spectroscopy means Ultraviolet spectroscopy. | Visible spectrophotometry means measurement of visible light absorbed by sample. |
| Region | It works in ultraviolet region of electromagnetic spectrum. | It works in visible region of electromagnetic spectrum. |
| Wavelength range | Its wavelength range is about 185 nm to 400 nm. | Its wavelength range is about 400 nm to 700 nm. |
| Light source | Deuterium lamp or hydrogen lamp is used as light source. | Tungsten lamp or tungsten-halogen lamp is used as light source. |
| Cuvette | Quartz cuvette or fused silica cuvette is used. | Glass or plastic cuvette is used. |
| Reason for cuvette use | Glass and plastic absorb UV light, so they are not used. | Glass and plastic allow visible light to pass, so they can be used. |
| Sample type | It is used for compounds which absorb UV light. The sample may not be coloured. | It is used for coloured solution which absorb visible light. |
| Main substances | It is used for DNA, RNA, protein and some pharmaceutical compounds. | It is used for dyes, food colour, pigments and coloured chemicals. |
| Application | It is mainly used in molecular biology, biochemistry and drug analysis. | It is mainly used in food industry, textile dye analysis and colour measurement. |
| Sensitivity | It can detect many colourless compounds which absorb in UV region. | It detects substances which show colour in visible region. |
Difference between Double-beam Spectrophotometer and Split-beam Spectrophotometer
| Basis | Double-beam Spectrophotometer | Split-beam Spectrophotometer |
|---|---|---|
| Beam path | In this spectrophotometer, light is divided into two separate beams. | In this spectrophotometer, light is divided into two parts by beam splitter. |
| Sample and blank | One beam passes through the sample cuvette and another beam passes through the blank cuvette. | One part of light passes through the sample and other part goes to reference detector. |
| Reference cuvette | Reference cuvette is used in this instrument. | Reference cuvette is not used in this instrument. |
| Reference beam | Reference beam passes through the blank solution. | Reference beam only checks the light source intensity. |
| Comparison | Sample and blank are compared continuously. | Sample reading is corrected by monitoring lamp output. |
| Beam splitting system | It may use mechanical chopper for alternating the light between sample and reference path. | It generally uses static beam splitter like half-silvered mirror. |
| Mechanical part | It has more moving mechanical parts. | It has less mechanical moving parts. |
| Stability | It gives very high stability and accurate reading. | It gives better stability than single-beam spectrophotometer. |
| Cost | It is more costly. | It is less costly than double-beam spectrophotometer. |
| Size | It is larger in size and more complex. | It is smaller and simple than double-beam type. |
| Use | It is used for high precision work, kinetic study and very low concentration sample. | It is used for routine work where stable baseline is needed with less cost. |
Difference between Colorimeter and Spectrophotometer
| Basis | Colorimeter | Spectrophotometer |
|---|---|---|
| Definition | Colorimeter is an instrument which is used to measure absorbance or transmittance of coloured solution. | Spectrophotometer is an instrument which is used to measure absorbance, transmittance or reflection of light by sample at selected wavelength. |
| Wavelength selection | It uses optical filters for selecting wavelength. | It uses monochromator for selecting wavelength. |
| Light selection | The filter allows only fixed range of wavelength to pass. | The monochromator gives narrow and specific wavelength of light. |
| Accuracy | It is less accurate than spectrophotometer. | It gives more accurate and precise result. |
| Bandwidth | It has broad bandwidth because filter is used. | It has narrow bandwidth, generally about 1-5 nm. |
| Wavelength change | Wavelength cannot be changed continuously. Only fixed filter wavelengths are used. | Wavelength can be changed continuously according to need. |
| Complexity | It is simple instrument. | It is more complex instrument. |
| Cost | It is cheaper and cost effective. | It is more costly than colorimeter. |
| Use | It is mainly used for coloured sample and simple routine test. | It is used for coloured and many non-coloured sample depending on region of light. |
| Main similarity | It measures light absorbed or transmitted by sample for finding concentration. | It also measures light absorbed or transmitted by sample for finding concentration. |
Single Beam vs Double Beam Spectrophotometers
| Basis | Single Beam Spectrophotometer | Double Beam Spectrophotometer |
|---|---|---|
| Light path | It has only one light path. | It has two light path. |
| Beam | One beam of light passes from light source to sample and detector. | Light beam is divided into two beams. |
| Blank | Blank is measured first and then sample is measured. | Blank and sample are measured at same time. |
| Sample path | The same path is used for blank and sample. | One beam passes through sample and another beam passes through blank. |
| Beam splitting | Beam splitter is not present. | Beam splitter or mechanical chopper is present. |
| Light intensity | More light reaches the detector because beam is not divided. | Less light reaches each path because beam is divided. |
| Stability | It is less stable. Lamp fluctuation and detector drift may affect the reading. | It is more stable. It corrects lamp fluctuation and electronic drift. |
| Speed | It is slower because blank and sample are changed manually. | It is faster because comparison is done continuously. |
| Cost | It is less costly. | It is more costly. |
| Size | It is compact and simple. | It is larger and more complex. |
| Use | It is used in teaching lab, simple clinical test and routine concentration checking. | It is used in research work, pharmaceutical testing, kinetic study and high precision analysis. |
| Main advantage | It is simple, durable and gives strong signal. | It gives stable baseline and accurate reading for long time. |
| Main limitation | It cannot correct signal drift automatically. | It is costly and needs more maintenance. |
References
- ResearchGate. (n.d.). (PDF) Forensic analysis applications in textile and chemistry.
- Haas, K. (2025). 2.2: UV/Vis Instrumentation. Chemistry LibreTexts.
- Band Optics. (2025). A Beginner’s Guide to Using a Spectrophotometer.
- New England Biolabs. (n.d.). A Practical Guide to Analyzing Nucleic Acid Concentration and Purity with Microvolume Spectrophotometers.
- HunterLab. (2022). Application of Double Beam Spectrophotometer.
- Pandya, R. (2025). Applications of Spectrophotometers in Different Industries. Coherent Market Insights.
- O’Kane, M. (n.d.). Beer Lambert Law. Ossila.
- Edinburgh Instruments. (2021). The Beer-Lambert Law.
- Felipe, F., Hellmann-Kolbe, A., & Staab, T. (2022). Comparing Microvolume Spectrophotometers: Colibri+ VS NanoDrop™ One, DS-11 and Nabi. Berthold Technologies.
- ColorWorkDesk. (2025). Colour Analysis in the Textile Sector: Technologies and Industrial Applications.
- Konica Minolta Sensing Europe. (n.d.). Colour Measurement Solutions for the Textile Industry.
- Efstathiou, C. E. (n.d.). Deviations from Beer’s Law: Effect of Polychromatic Radiation.
- Agilent Technologies. (n.d.). Double Beam Spectrophotometers in UV-Vis.
- Hach Company. (n.d.). Split Beam vs Single Beam Optics.
- Shimadzu Scientific Instruments. (n.d.). How is a double beam spectrophotometer different from a single beam instrument?
- Persee. (2025). How the Light Source Works in UV-Vis Spectrophotometers: Single, Split, and Double Beam Compared.
- PatSnap. (2025). Monochromator vs polychromator: Which is better for your spectroscopy setup?
- Whyte, B. (2024). What is the difference between a spectrometer and a monochromator for absorbance measurements? BMG LABTECH.
- National Institutes of Health. (n.d.). NanoDrop Microvolume Quantitation of Nucleic Acids. PMC.
- LabX. (n.d.). Nanodrop Spectrophotometry: A Modern Guide to Nucleic Acid and Protein Quantification.
- HunterLab. (2026). Single Beam vs. Double Beam Spectrophotometer.
- Wu, E. (2025). Single vs Double Beam Spectrophotometers: Key Differences. AELAB.
- Aakash Institute. (n.d.). Spectrophotometer Principle – Beer-Lambert’s Law, Instrumentation, Working, Types and Applications.
- Qualiper. (2024). Spectrophotometer for Color Measurement in Textile Industry.
- Kiron, M. I. (2022). Spectrophotometer in Textile: Components, Principle and Uses. Textile Learner.
- Presto Group. (2025). Spectrophotometer: A Comprehensive Guide to Principles, Types, and Applications.
- Torontech. (2025). Spectrophotometer: Single Beam vs Double Beam Explained.
- Dhruv. (2025). Spectrophotometers: Working Principle, Types, and Applications. Labix Industries.
- Hussein, A. A., & Issa, A. H. (2022). Spectrophotometry.
- Barron, J., & Geary, L. (n.d.). Spectrophotometry Standards. Reagecon Diagnostics Ltd.
- Khan Academy. (n.d.). Spectrophotometry and the Beer–Lambert Law.
- Kapoor, R. V. (2025). Spectrophotometry in Pharmaceutical and Chemical Analysis: Principles, Techniques, and Applications. Research & Reviews: Journal of Pharmaceutical Analysis, 14(019).
- O’Kane, M. (n.d.). Spectrophotometry: How To Use A Spectrophotometer. Ossila.
- Danaher Life Sciences. (n.d.). Spectrophotometry: Uses, Advantages & Applications.
- Danaher Life Sciences. (n.d.). Spectrophotometry: Uses, Advantages & Applications.
- Cole-Parmer. (n.d.). Spectroscopy Applications Selection Guide.
- Filo. (2025). State the beer’s law and provide it limitations?
- Labcompare. (n.d.). Tech Compare: Single vs. Double Beam Spectrophotometers.
- MRC. (n.d.). The Complete Guide to Spectrophotometers.
- Author unknown. (n.d.). The Systematic Evolution and Technical Architecture of Spectrophotometric Instrumentation: A Comprehensive Guide to Analytical Principles, Mechanisms, and Industry Applications.
- Malhotra, G. (2025). The Ultimate Guide to Spectrophotometers: Principles, Uses, and Applications. Presto Group.
- Thermo Fisher Scientific. (2015). Thermo Scientific SPECTRONIC Certified Standards.
- Hellma Analytics. (n.d.). UV/Vis calibration standards.
- Persee. (2026). Understanding Double Beam Spectroscopy Calculation Methods Explained.
- Lab Manager. (n.d.). Validating Photometric Accuracy and Stray Light in UV-Vis Spectrophotometers.
- Upstone, S. (n.d.). Validating UV/Visible Spectrophotometers. PerkinElmer, Inc.
- MRC. (n.d.). What Industries Use Spectrophotometers, and For What Applications?
- Copenhagen Nanosystems A/S. (2023). What is a monochromator in a UV-Vis spectrophotometer? SpectroKnowledge™.
- Malhotra, G. (2025). What is the Spectrophotometer Principle? Working, Uses, and Basics. Presto Group.
- Optosky. (2020). What is the difference between monochromator and spectrometer?
- Sper Scientific Direct. (2025). What’s the Difference Between Single and Dual Beam Spectrophotometers?
- Metash. (2024). Working Principle Of Dual Beam Spectrophotometer.
- Harris, D. C. (2010). Quantitative Chemical Analysis (8th ed.). W. H. Freeman and Company.
- Berhampore Girls’ College. (n.d.). UV-Visible Spectroscopy.