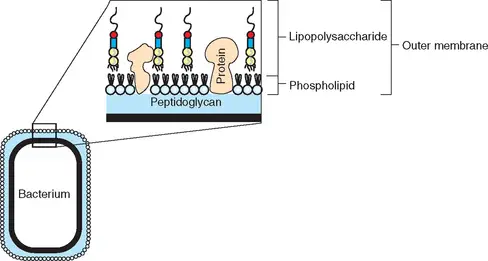
- Endotoxin is a kind of pyrogen. It is an element of the cell wall exterior of Gram-negative bacteria such as E. coli (see image).
- Endotoxin is a type of lipopolysaccharide, LPS. It is a part of the lipid that contains disaccharide disaccharidephosphates, fatty acids as well as core polysaccharides, and the O-antigen (see picture).
- The lipid A component of LPS is responsible for the molecule’s activity as an endotoxin. Although lipid A isn’t able to directly cause harm to any tissue however, those cells that are immune to animals and humans alike consider it to be an indicator of the presence of bacteria.
- This is why these cells produce the body’s response to repel unwanted invaders. This response is completely natural, i.e. there is no prior exposure to the endotoxin has to be done prior to.
- In addition, it might aid in keeping beneficial intestinal bacteria in as well as keeping out the main entrance of pathogenic bacteria from the outside.
- Because of the serious consequences associated with an infection injectable health product like an intravenous or vaccine must be clean or free of live organisms, however, the process used to kill any bacteria may result in the release in the form of LPS as well as endotoxin to the item. Similar to sepsis or a bacterial infection in the event that enough endotoxin enters into our bloodstream or spinal fluid, we could suffer from shock, fever, or organ damage. In extreme cases, it can even result in death.
Endotoxin definition/what is endotoxin?
- The discovery of endotoxins was made in the late in the 19th century. Endotoxins can be toxic complex compounds created by bacteria that are Gram negative. Also called lipopolysaccharides (LPS) endotoxins are among the most commonly used pyrogens (substances that have the ability to cause fever).
- They consist of the hydrophilic polysaccharide along with the hydrophobic part (Lipid A) and form part of the membrane that surrounds Gram-negative bacteria.
- While endotoxins form part that make up the membrane of Gram-negative organisms, these, as numerous other substances are produced within the cell cytosol. They are then transferred to the membrane via the inner membrane, as well as through the space between the periplasmic and outer membranes.
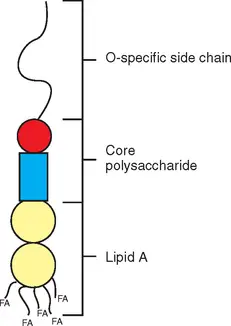
- As previously mentioned, endotoxins are composed of a polysaccharide bonded to the lipid A (a moiety of lipids). In this instance, lipid A is the main culprit behind the harmful effects of the compound.
- The signs and symptoms that are associated with endotoxins are (endotoxin symptoms):
- High fever
- Vasodilation
- Diarrhea
- Shock
- “Endotoxin” is a term that refers to the bacterial “endotoxin” is occasionally used to describe any bacterial toxin associated with cells, in the field of bacteriology, it is reserved for the lipopolysaccharide compound that forms part of the membrane that surrounds Gram-negative pathogens, such as Escherichia Coli, Salmonella, Shigella, Pseudomonas, Neisseria, Haemophilus influenzae, Bordetella pertussis and Vibrio cholerae.
- The bioactivity of endotoxin is linked to its Lipopolysaccharide (LPS).
- Cell wall-wall Antigens (O antigens) of Gram-negative bacteria are part of LPS.
- LPS induces many reactions to inflammation in animals and also activates complement through the second (properdin) route, meaning it could be a component of the pathology that causes Gram-negative bacterial infections.
- The structure of endotoxins differs in different Gram-negative bacteria. Therefore, the degree of toxicity dependent on its structure.
Endotoxin examples
The term”endotoxin” is typically used to describe the pyrogens that Gram-negative bacteria produce. In the field of bacteriology, this complicated compound is also referred to as lipopolysaccharide. It can be located in the membranes on the outside of bacteria such as Escherichia Coli, Salmonella shigella, Vibrio cholerae as well as Haemophilus influenzae. Endotoxins fall into two major categories which comprise:
1. Smooth lipopolysaccharides
Sometimes referred to in the wild type LPS (sLPS) Smooth lipopolysaccharide comprises the O-antigen and complete core Oligosaccharides, and the lipid A. A few of the bacteria that have smooth lipopolysaccharides that are on their surfaces are part of the Brucella genus. Brucella. These include Brucella suis, Brucella, abortus, and Brucella melitensis.
2. Rough lipopolysaccharide
In contrast to smooth lipopolysaccharide lipsaccharide doesn’t have its O antigen. But, they do have lipid A as well as core oligosaccharides. In this instance the oligosaccharides are shown to be gradually shorter. A few of the organisms that have rough lipopolysaccharides (also called R-species) are Brucella canis as well as Brucella Ovis.
Although R species (bacteria species that have tough lipopolysaccharides) also have the lipid A, studies have identified that some of these species (e.g. Brucella) as being less harmful than those with rough lipopolysaccharide.
Characteristics of Endotoxins/Lipopolysaccharide
- Likely, lipopolysaccharides are often found on the exterior membranes of many Gram-negative bacteria.
- Due to differences in structure, some endotoxins aren’t infective and some species aren’t pathogenic.
- While the toxicity of this compound is correlated with its lipid component the ability to induce an immune response (the capability to initiate an immunological response) is because of the polysaccharide region.
- While Gram-negative bacteria are able to release tiny amounts of lipopolysaccharide in small amounts, the endotoxin generally less potent compared to exotoxins.
- Due to their structure they are associated with numerous important roles, such as contributing to an increase in thepermeability membrane.
- Lipopolysaccharides improve the permeability barrier, but only allowing hydrophilic molecules with a small molecular weights to cross.
- They may encourage an adhesion (through their O antigens) of the organism to the host cell, thus helping to spread the infection.
| PROPERTY | ENDOTOXIN |
| CHEMICAL NATURE | Lipopolysaccharide (mw = 10kDa) |
| RELATIONSHIP TO CELL | Part of outer membrane |
| DENATURED BY BOILING | No |
| ANTIGENIC | Yes |
| FORM TOXOID | No |
| POTENCY | Relatively low (>100ug) |
| SPECIFICITY | Low degree |
| ENZYMATIC ACTIVITY | No |
| PYROGENICITY | Yes |
Structure of Endotoxin
The majority of research into the structure and chemical composition of endotoxin was conducted using species of Salmonella as well as E. coli. LPS is extracted from whole cells through treatment with 45 percent phenol at 90 degrees. The mild hydrolysis of LPS produces Lipid A as well as polysaccharides.
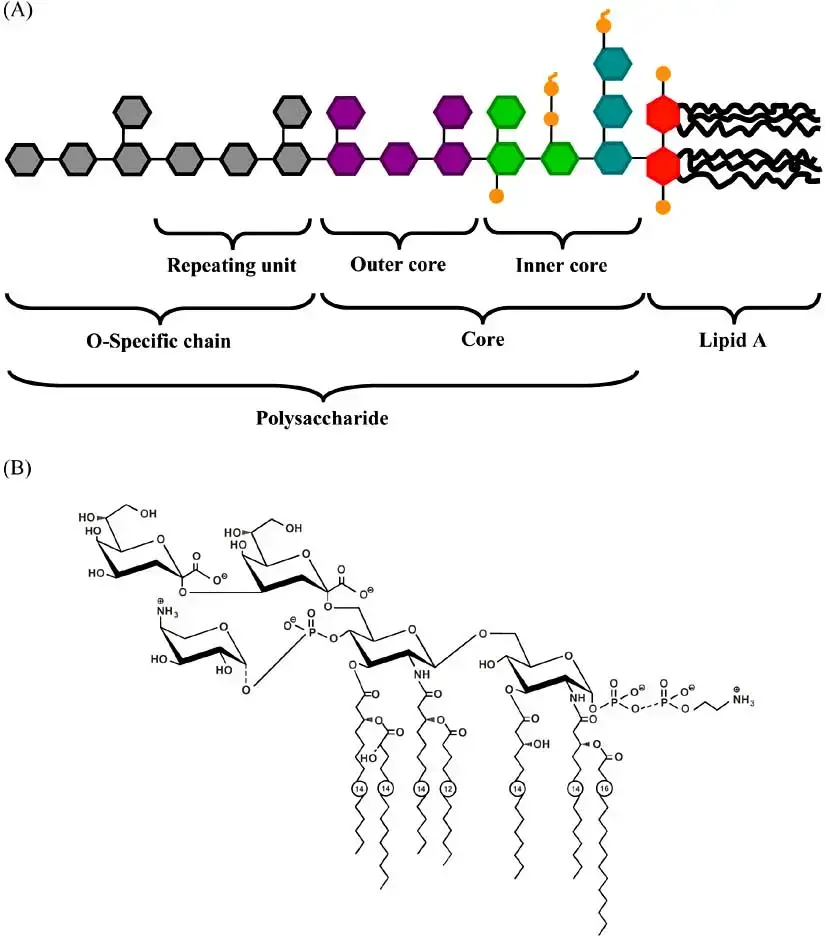
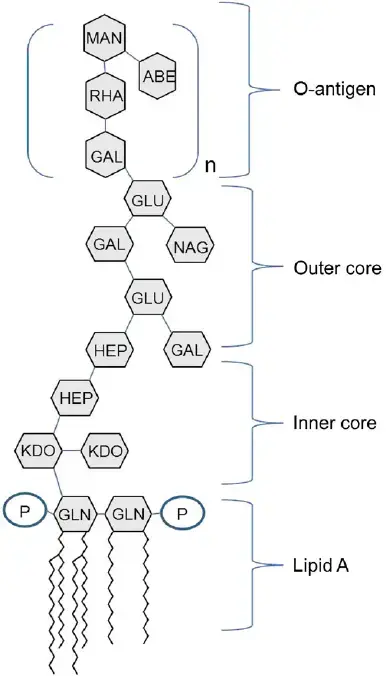
Lipopolysaccharides are amphiphilic complex molecules with MWs of around 10kDa. They differ in chemical composition within and between species of bacteria. LPS is composed of three constituents of regions, namely Lipid A and R polysaccharide as well as the O-polysaccharide.
Lipid A
The physiological actions of LPS are controlled primarily through LPS’s Lipid the major component in LPS. Lipid A is an effective biological response modifier that has the ability to enhance your mammalian’s immune system.
When it comes to infections caused by Gram-negative bacteria endotoxins released by or in part of growing cells can are similar to those that affect animals, and are a significant contributor to the pathology and symptoms of the disease that they encounter.
Because Lipid A exists within the membrane that surrounds the bacterial cells, it may has its toxic effects only in the event that it is released from the cells that are growing in a liquid form or when bacteria are killed by autolysis, complement, and the Membrane Attack Complex (MAC) or the ingestion and killing of phagocytesor killed by specific antibiotics.
There are a variety of signs and symptoms of endotoxin-related infection, including the appearance of fever as well as changes in white blood cells and disseminated intravascularcoagulation shock, hypotension, and even death. The injection of relatively small amounts of endotoxin causes death in the majority of mammals. This sequence follows a consistent order: (1) latent period; (2) physiological distress (diarrhea or prostration, shocked); (3) death. The speed at which death occurs is contingent dependent on the dosage of the endotoxin, the route of administration, as well as the species of animal. Animals have different susceptibility to the endotoxin.
- Lipid A makes up the component of lipids in LPS.
- It has the hydrophobic membrane-anchoring area of LPS.
- Lipid A is made up of an N-acetylglucosamine phosphorylated (NAG) dimer that has either 6 or 7 the fatty acids (FA) connected. In general six fatty acids can be located.
- All fatty acids found in Lipid A contain saturated.
- Certain fatty acids attach directly to the NAG dimer , while other are esterified to 3-hydroxy fatty acids which are typically present.
- Structure of Lipid A is highly conserved in Gram-negative bacteria.
- Within Enterobacteriaceae, Lipid A is almost the only constant.
- The fundamental nature in Lipid A has been elucidated and Lipid A has been chemically synthesized.
- The biological activities of the enzyme appear to be based on a unique configuration that is determined by disaccharide glucosamine and the PO4 groups and the acyl chains as well as the KDO-containing inner core.
Core (R) antigen or R polysaccharide
- Core (R) antigen or R polysaccharide is attached to the 6th position of a NAG.
- The R antigen is a short sugar chain that is consist of KDO – Hep – Hep – Glu – Gal – Glu – GluNAc –
- Two unusual sugars, heptose and 2-keto-3-deoxyoctonoic acid (KDO), are usually present, in the core polysaccharide.
- KDO is unique and always found in LPS which is why it is used to determine the presence of KDO in tests to determine LPS (endotoxin).
- With minor modifications The core polysaccharide is found in all members of the genus Bacteria (e.g. Salmonella) However, it has a distinct structure from other Gram-negative genera. bacteria.
- Salmonella, Shigella and Escherichia share similar, but not exactly the same the cellular structures of these organisms.
Somatic (O) antigen or O polysaccharide
- The somatic (O) Antigen, also known as O Polysaccharide attaches to the polysaccharide’s core.
- It is composed of repeating oligosaccharide oligosaccharide units made by 3 to 5 sugars.
- The individual chains differ in length and can reach 40 units of repeat.
- The O polysaccharide has a longer length than the polysaccharide that is its core and it also houses the hydrophilic portion in the LPS molecule. The major antigenic determinate (antibody-combining website) in the cell wall of Gram negative can be found inside the O polysaccharide.
- There is a wide range of variation with regards to the structure of sugars that make up the O side chain among species, and even between strains from Gram-negative bacteria. More than 20 different sugars have been identified and a large portion of them are distinctively unique dideoxyhexoses that are found only in Gram-negative cell walls.
- Variations in the sugar content in the O polysaccharide can contribute to the variety of antigenic varieties that are found in Salmonella as well as E. coli and presumably other species of Gram-negative.
- Certain sugars in the structure, specifically the ones at the end, provide an immunologically specific O antigen as well as “smoothness” (colony morphology) of the strain.
- The loss of the O specific region through mutation leads to the strain evolving into”rough” or “rough” (colony morphology) or R strain.
- The understanding about the structure LPS (Figure 3.) was heavily dependent on the existence of mutants that were blocked at the same stage of LPS synthesizing.
- The biosynthesis process of LPS is completely sequential. The sugars in the core are added in sequence to Lipid A through successive additions. The O part chain gets added in the last step each preassembled unit at one time.
- The characteristics of mutants that produce incompletion of LPS molecules indicate the biological and cellular functions that are performed by different parts of the LPS molecules.
- When it comes to E. coli and Salmonella In Salmonella and E. coli, the absence of O antigen leads to a some loss of virulence it is believed that this component of LPS is crucial for host-parasite interactions. It is widely known that “rough” mutants are more vulnerable to phagocytosis as well as serum bactericidal reactions.
- The loss of the more proximal areas of the core like in “deep rough” mutants (i.e. in the Rd1, Rd2 and Re mutants shown in Figure 3) make the strains more susceptible to a variety of hydrophobic compounds, such as detergents, antibiotics and mutagens and bile salts. The area is home to a huge number of charged group and is believed to be crucial in maintaining the permeability characteristics of the membrane’s outer.
- Mutants that are part of their assembly Lipid A cannot be isolated other than as lethal conditional mutants and this region is therefore believed to be crucial for the survival of cells. The outermost region of LPS is comprised from Lipid A, as well as the three KDO residues is believed to be vital to the viability of cells, possibly for creating the membrane’s outer.
Virulence of Endotoxin/Virulence mechanism of Gram-negative Bacteria
The Lipid A (the harmful component in LPS), as well as sides chains of polysaccharide (the non-toxic, but immunogenic component of LPS), serve as the indicators of virulence in Gram-negative bacteria.
A. The virulence mechanism of O polysaccharide
Virulence, as well as the characteristic in the form of “smoothness”, is associated with a healthy O polysaccharide. The role of the polysaccharide chains in virulence is evident from the fact that even small modifications to the sugar sequences within those side chains in LPS cause major changes in the virulence. What are the polysaccharide sides chain involved in developing the virulence? There are a variety of possibilities:
1. Adhere to tissues
O-specific antigens may allow organisms to attach to certain tissues, in particular epithelial tissues.
2. Inhibition to phagocytes
The smooth antigens may allow the phagocytes to resist, as rough mutants are more easily consumed and destroyed by phagocytes.
3. Water-solubilizing carriers
The hydrophilic O polysaccharides may be water-solubilizing carriers for the toxic Lipid A. It is well-known that the specific structure of the polysaccharide could profoundly affect the capacity of water to bind at the cell’s surface.
4. Protection
The O antigens may protect from harmful reactions caused by antibody and complement. The Gram-negative bacteria that are rough originated from virulent strains generally not virulent. Smooth strains are made up of polysaccharide “whiskers” which bear O antigens that project out from the cell’s surface.
It is believed that the O antigens are key target for the actions of the complement and antibody of the host However, if the reaction happens in the apex of polysaccharide chains, which is a substantial distance away from the surface of the bacterial cell and complement does not have its normal lytic action.
These bacteria are virulent due to their resistance to the immune forces from the human. When the chains of polysaccharide that are projecting are reduced or removed and antibodies react with antigens that are on the bacteria’s surface or within it. Then, may cause the lysis of the bacteria. This causes the reduction in virulence in “rough” colonial strains.
5. Antigenic variation
The O-polysaccharide, also known as the O antigen is the source of the antigenic variations among the many Gram-negative pathogens that are important, such as E. coli, Salmonella and Vibrio cholerae. The antigenic variation is a guarantee of multiple serotypes for the bacterium, which means that it can have many opportunities to attack its host if it manages to overcome the immune response to an entirely different serotype.
In addition, even though O polysaccharides have antigens that are potent but they rarely trigger immune responses that offer complete protection to the host from an additional challenge from a specific endotoxin.
B. The virulence mechanism of Lipid A
The mechanism is complicated. For humans, LPS binds to a the lipid binding protein (LBP) in serum, which then is transferred onto CD14 at the membrane of cells. This then transfers it to a different non-anchored protein, MD2 that binds to Toll-like receptor 4 (TLR4). This triggers the signaling cascade for macrophage/endothelial cells to secrete pro-inflammatory cytokines and nitric oxide that lead to characteristic “endotoxic shock”. CD14 as well as TLR4 are present on many immune system, including dendritic cells and macrophages. Monocytes and macrophages have three kinds of triggers are activated during their interaction with LPS:
1. Production of cytokines
Production of cytokines, such as IL-1, IL-6 and the IL-8, tumor necrosis factors (TNF) and the platelet-activating factors. They, in turn, trigger the production of prostaglandins and leukotrienes. They are powerful mediators of septic shock and inflammation that is a complication of endotoxin toxemia. LPS triggers macrophages for increased the rate of phagocytosis as well as the cytotoxicity. Macrophages are stimulated as well as release the enzymes lysosomal such as the IL-1 (“endogenous Pyogen”) along with tumor necrosis factor (TNFalpha) and TNFalpha, in addition to other mediators and cytokines.
2. Activation of the complement cascade.
C3a and C5a trigger the release of histamine (leading in vasodilation) and alter the chemotaxis of neutrophils and their accumulation. The result is inflammation.
3. Activation of the coagulation cascade.
The initial induction of Hageman factor (blood-clotting Factor XII) can trigger a variety of humoral systems, resulting in
- Coagulation is a blood clotting process that results in bleeding, thrombosis, and coagulation. disseminated intravascular blood coagulation that reduces platelets as well as other factors that cause clotting, resulting to internal bleeding.
- The activation of the complement alternative pathway (as as previously described, which can lead to inflammation)
- Plasmin activation that results in fibrinolysis and hemorrhaging.
- The activation of kinins releases bradykinins as well as other vasoactive substances that cause hypotension.
The final result is to trigger inflammation as well as hemorrhage and shock. LPS can also act as an B cell mitogen encouraging the polyclonal transformation and multiplication of B-cells as well as the release of immunoglobulins particularly IgG as well as IgM.
References
- Review: Biomaterial-associated thrombosis” roles of coagulation factors, complement, platelets and leukocytes, Maud B. Gorbet, Michael V. Sefton, https://doi.org/10.1016/B978-008045154-1.50025-3
- https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/inspection-technical-guides/bacterial-endotoxinspyrogens
- https://www.pharmamicroresources.com/2017/10/assessment-of-bacterial-endotoxin-by.html
- https://www.horseshoecrab.org/med/endotoxin.html#:~:text=Endotoxin%20is%20a%20type%20of,is%20a%20lipopolysaccharide%20or%20LPS.&text=While%20lipid%20A%20does%20not,for%20the%20presence%20of%20bacteria.
- https://www.merriam-webster.com/dictionary/endotoxin
- https://www.biologicscorp.com/blog/bacterial-endotoxin-definition/#.YgPcXt9BzDc
- http://textbookofbacteriology.net/endotoxin.html
- https://labchem-wako.fujifilm.com/asia/lal/lal_knowledge/about_lal.html
- https://www.biologicscorp.com/blog/bacterial-endotoxin-definition/#.YgPcbt9BzDc
- https://www.microscopemaster.com/endotoxin-vs-exotoxin.html
- https://veteriankey.com/endotoxemia/
- https://www.slideshare.net/dasarimahalaxmi/bacterial-endotoxins-test