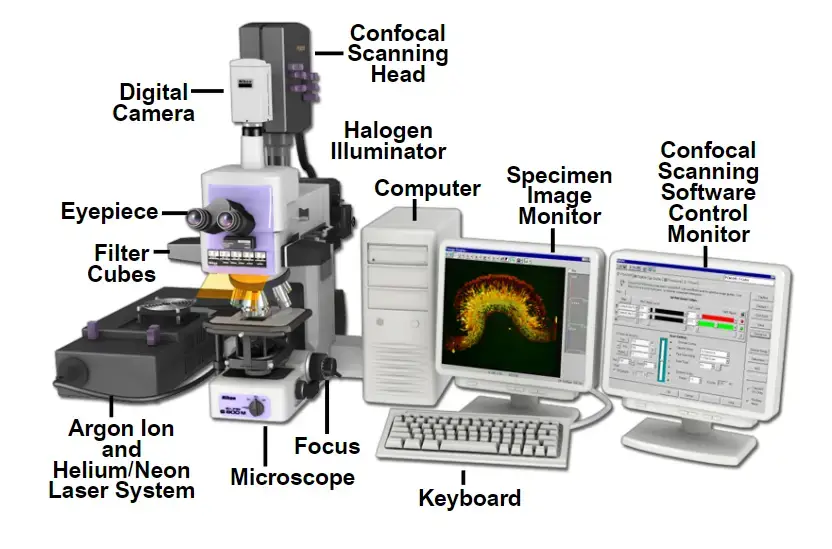
Confocal Microscope is an advanced optical microscope that is used to produce clear and sharp image by removing the out-of-focus light. It uses laser light to focus on a single point of the specimen. It gives better resolution and contrast than the ordinary light microscope.
In confocal microscope, the light is not passed through the whole specimen at once. The laser beam is focused only on a very small point of the sample. A pinhole aperture is present in front of the detector which blocks the scattered light coming from above or below the focal plane.
Only the focused light from the selected depth of the specimen reaches the detector. Due to this, the image formed is very clear and has high contrast. This removal of unwanted light is the main feature of confocal microscope.
The focused laser beam scans the specimen point by point. After scanning, the signals are collected and converted into image by computer. By changing the focus at different depth, many thin optical sections are produced from thick specimen.
These optical sections are combined by computer to form a three-dimensional image of the specimen. So, confocal microscope is very useful for studying cells, tissues and other biological structures in detail. It is used in cell biology, medicine, neuroscience and material science.
Principle of Confocal Microscope
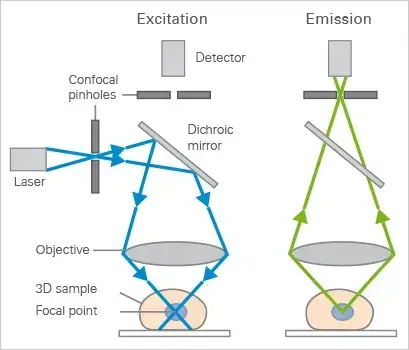
Confocal microscope is based on point illumination and filtering of light. A laser light is focused on one point of the specimen at a time.
The whole specimen is not illuminated at once like ordinary microscope. The focused light scans the specimen point by point. The light coming from the specimen is passed back through the same optical path.
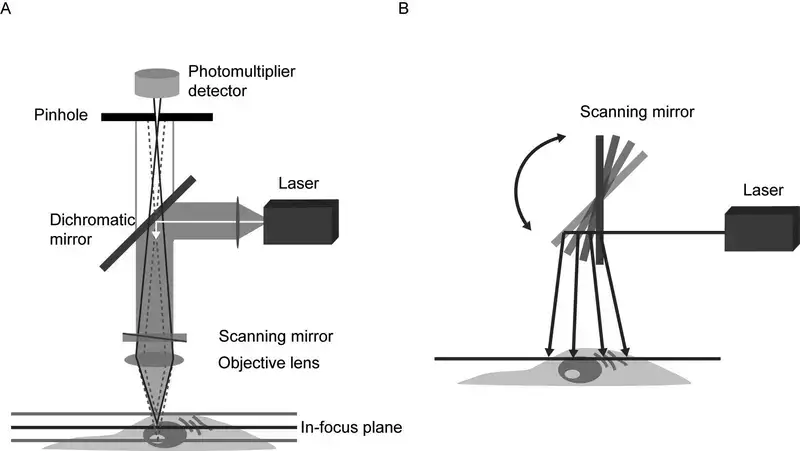
A pinhole aperture is present before the detector. It blocks the light which comes from above and below the focal plane. Only the light coming from the exact focus passes through the pinhole.
So, out of focus light is removed and a clear image is formed. The image has high contrast and better resolution. The part which is not in focus appears dark.
The focal plane is moved up and down through the specimen. In this way, many thin optical sections are obtained. These sections are joined by computer and a three dimensional image is formed.
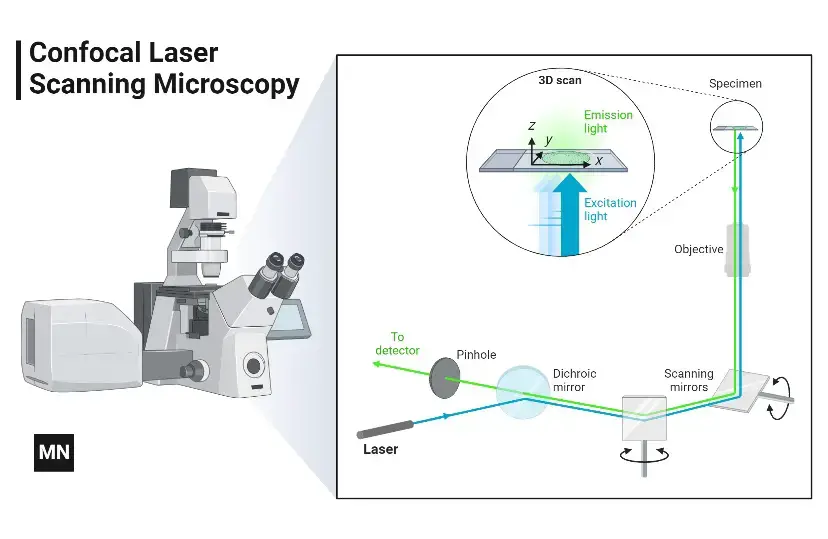
Mechanism of Confocal Microscope – How does a confocal microscope work?
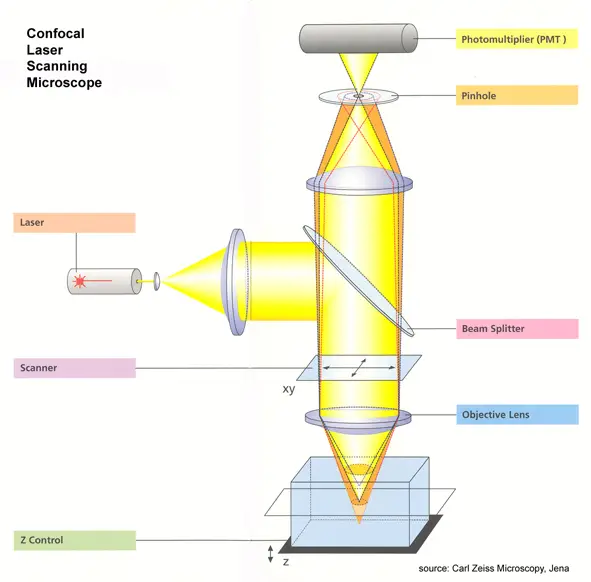
The following are the steps of working of confocal microscope-
- Laser light- Laser light is used in this microscope. It produces a narrow beam of light for illumination of the specimen.
- Dichroic mirror- The laser light strikes on the dichroic mirror. The mirror reflects the light towards the objective lens.
- Focusing- The objective lens focuses the light on a small point of the specimen. This point is present in the focal plane.
- Fluorescence- The focused light excites the fluorescent dye present in the specimen. Then light is emitted from the excited region.
- Return path- The emitted light comes back through the objective lens. It again comes to the dichroic mirror and then passes towards the pinhole.
- Pinhole- The pinhole is present in front of the detector. It passes only the light coming from the focal plane.
- Removal of blurred light- Light coming from upper and lower parts of the focal plane cannot pass through the pinhole. So, out of focus light is removed.
- Detector- The light passed through the pinhole falls on the detector. The detector changes the light into electrical signal.
- Scanning- One point is seen at one time. So, the laser beam is moved point by point and line by line over the specimen.
- Image formation- The signals are collected by computer. A clear optical section is formed.
- 3D image- The focal plane is changed in different depths. Many optical sections are formed and these are joined to make three dimensional image.
Parts of Confocal Microscope
The following are the main parts of confocal microscope-
- Laser- Laser is the light source of confocal microscope. It gives a strong narrow beam of light. This light is used to excite the fluorescent material present in the specimen.
- Dichroic mirror- It is also called beam splitter. It reflects the laser light towards the specimen. It also allows the emitted fluorescent light to pass towards the detector.
- Scanning mirrors- These are fast moving mirrors. They move the laser beam over the specimen. The specimen is scanned point by point and line by line.
- Objective lens- Objective lens focuses the laser beam on a small point of the specimen. It also collects the fluorescent light coming back from the specimen.
- Confocal pinhole- Pinhole is a small aperture present before the detector. It allows only the focused light to pass. The light coming from above and below the focal plane is blocked.
- Detector– Detector receives the light which passes through the pinhole. It changes the light signal into electrical signal. Photomultiplier tube (PMT), GaAsP detector and hybrid detector are used as detectors.
- Computer system- Computer receives the electrical signals from the detector. It arranges the signals and forms the image. It is used to produce two dimensional and three dimensional image of the specimen.
Types of Confocal Microscope
The following are the important types of confocal microscope-
1. Laser Scanning Confocal Microscope (LSCM)- This is the most common type of confocal microscope. It uses laser beam for scanning the specimen. The specimen is scanned point by point and line by line to form high resolution image.
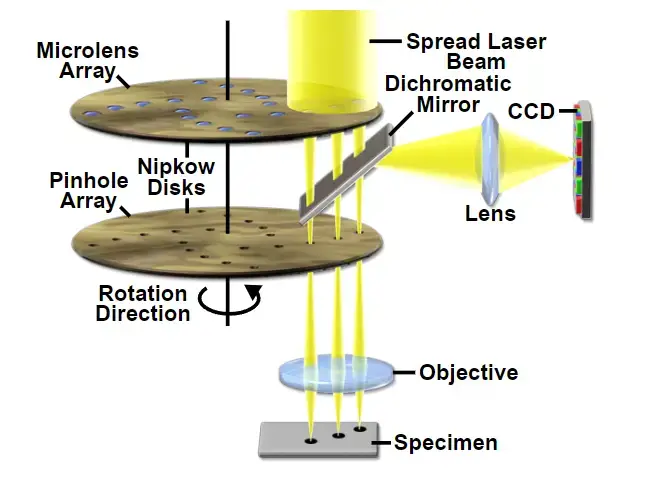
2. Spinning Disk Confocal Microscope- This microscope uses a rotating disk with many pinholes. These pinholes allow scanning of many points at same time. So, image formation is faster. It is useful for study of living cells because less light damage occurs.
3. Dual Spinning Disk Confocal Microscope- This is modified form of spinning disk confocal microscope. It has another disk with microlenses. These microlenses focus the light into the pinholes and increase the sensitivity of the microscope.
4. Programmable Array Microscope (PAM)- This microscope uses electronically controlled light modulator. It forms many moving pinholes by using pixel array. The pinholes can be changed according to the need.
5. Hybrid Scanning Confocal Microscope- This microscope combines laser scanning and spinning disk system. It can be used for fast imaging and also for detailed imaging. So, it is selected according to the type of specimen and imaging work.
Application of Confocal Microscope
The following are the important applications of confocal microscope-
- Biology- It is used to study cell structure and tissue structure. It is also used to study protein interaction and cell organelles.
- Medicine- It is used in medical diagnosis. It is used to study eye diseases, corneal infection and other tissue changes.
- Neuroscience- It is used to study nerve cells and brain tissue. It helps in mapping of neural connections.
- Pharmaceutical research- It is used to study drug delivery system. It is also used to check thin film drugs and their uniformity.
- Material science- It is used to study surface structure of materials. It helps in measuring roughness, coating and thin films.
- Industrial inspection- It is used for checking semiconductor wafer, microelectronics and battery electrodes. It is also used in aerospace parts and solar cell surfaces.
- Microbiology- It is used to study microorganisms in biofilm. It shows their structure, activity and arrangement.
- Live cell imaging- It is used to observe living cells with less background light. Cellular movement and changes can be studied.
- Three dimensional imaging- It is used to form 3D image of thick specimen. Many optical sections are joined by computer.
- Audio preservation- It is used for non-contact scanning of old audio records. Damaged wax phonograph cylinders can be studied.
- Data retrieval- It is used in 3D optical data storage system. It helps to recover stored information from different optical layers.
Advantages of Confocal Microscopy
The following are the advantages of confocal microscope-
- Out of focus light is removed- The pinhole blocks the scattered light coming from above and below the focal plane. So, the image becomes clear and contrast is increased.
- High resolution image- It gives better resolution than ordinary fluorescence microscope. The edges and details of the specimen are seen clearly.
- Three dimensional image- Many thin optical sections are collected from the specimen. These sections are joined by computer and three dimensional image is formed.
- Optical sectioning- Thin sections are obtained by light only. The specimen is not cut physically, so cutting artefacts are not formed.
- Live cell study- It can be used for living cells and fixed cells. The movement and changes inside living cells can be studied.
- Less background light- Background fluorescence is reduced by the pinhole. It helps to see the selected part of the specimen.
- Electronic zoom- Magnification can be changed by changing the scanned area of laser. Objective lens is not needed to change every time.
- Multi colour imaging- More than one fluorescent dye can be used. Different structures of the cell can be seen at the same time.
- Different viewing plane- The image can be reconstructed in x-y, x-z and y-z planes. So, internal arrangement of thick specimen can be studied.
- Depth control- The depth of field can be controlled during imaging. A particular depth of the specimen can be selected for observation.
Disadvantages of Confocal Microscopy
The following are the limitations of confocal microscope-
- High cost- Confocal microscope is costly instrument. Its purchasing, maintenance and operation cost is high.
- Limited wavelength- The laser light has limited excitation wavelength. So, all types of fluorophore cannot be excited easily.
- Phototoxicity- High intensity laser light may damage the living cells. It is more important during long time observation of live cells.
- Photobleaching- Fluorescent dye may lose its fluorescence after continuous laser exposure. So, the image signal becomes weak.
- Low signal intensity- The pinhole blocks most of the emitted light. So, less light reaches the detector and sensitive detector is needed.
- Limited depth imaging- It is difficult to image deep part of thick specimen. In thick specimen, light scattering and refraction reduce the image quality.
- Slow image formation- The specimen is scanned point by point and line by line. So, image formation takes more time than widefield microscope.
- Complex operation- The adjustment of laser, pinhole, detector and scanning system is complex. Skilled person is needed for proper operation.
- Resolution limit- It gives better image than ordinary fluorescence microscope. But its resolution is lower than transmission electron microscope.
- Heat and light damage- Continuous laser illumination may produce heat and light damage in delicate specimen. So, exposure time should be controlled.
Confocal microscope image
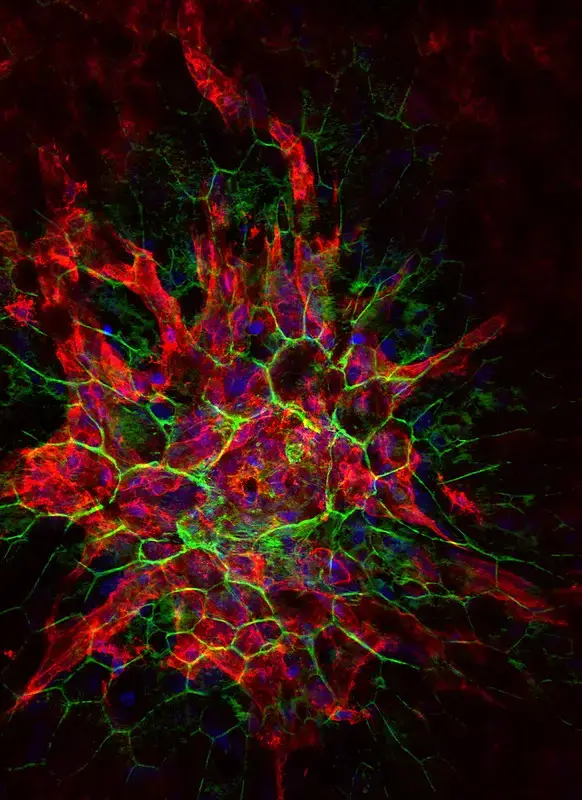
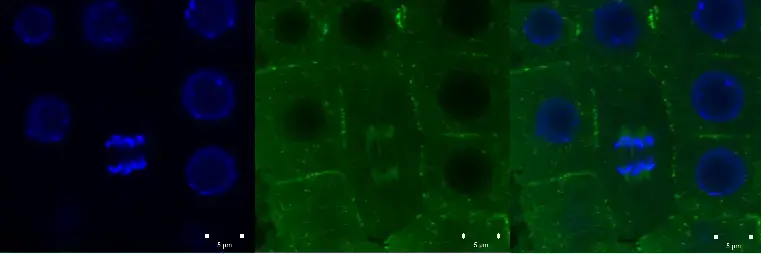
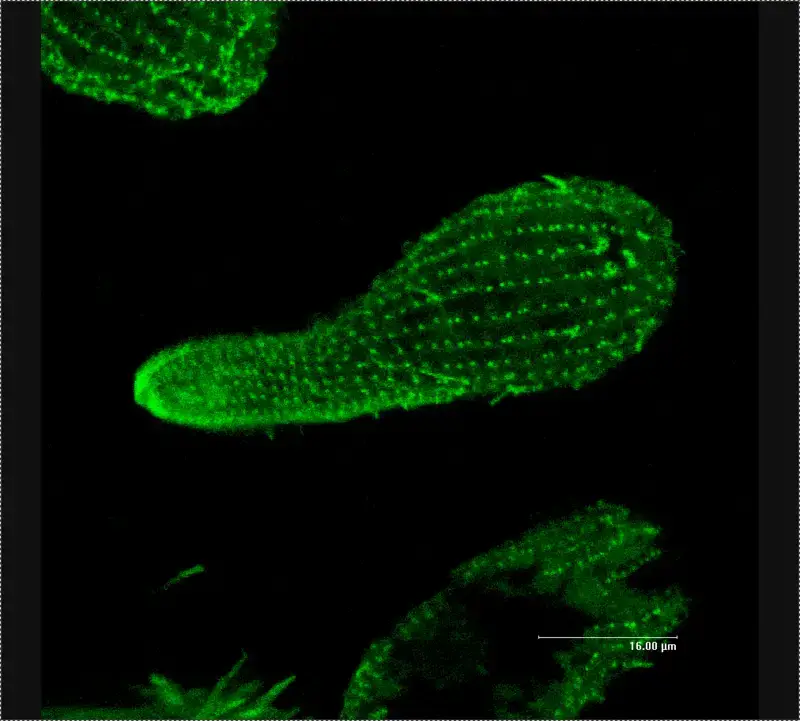
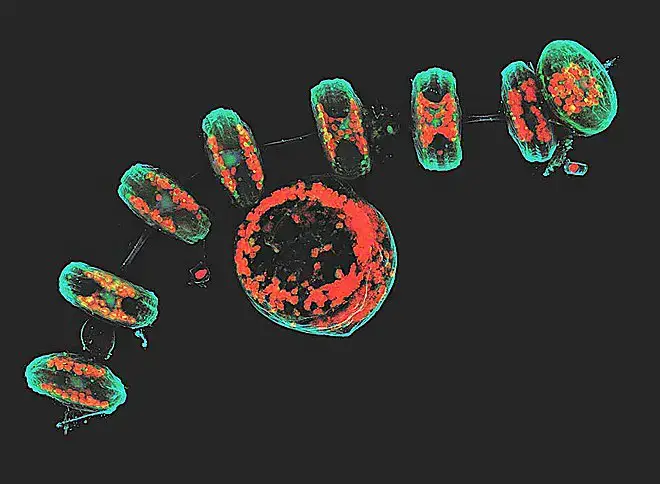
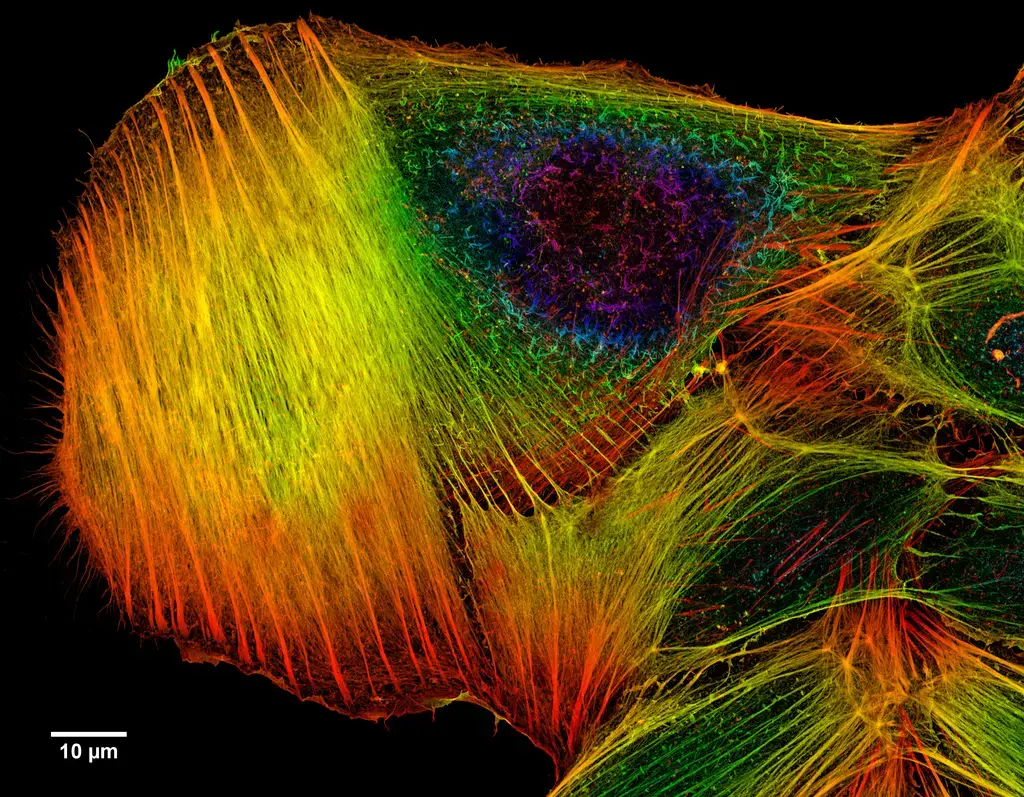

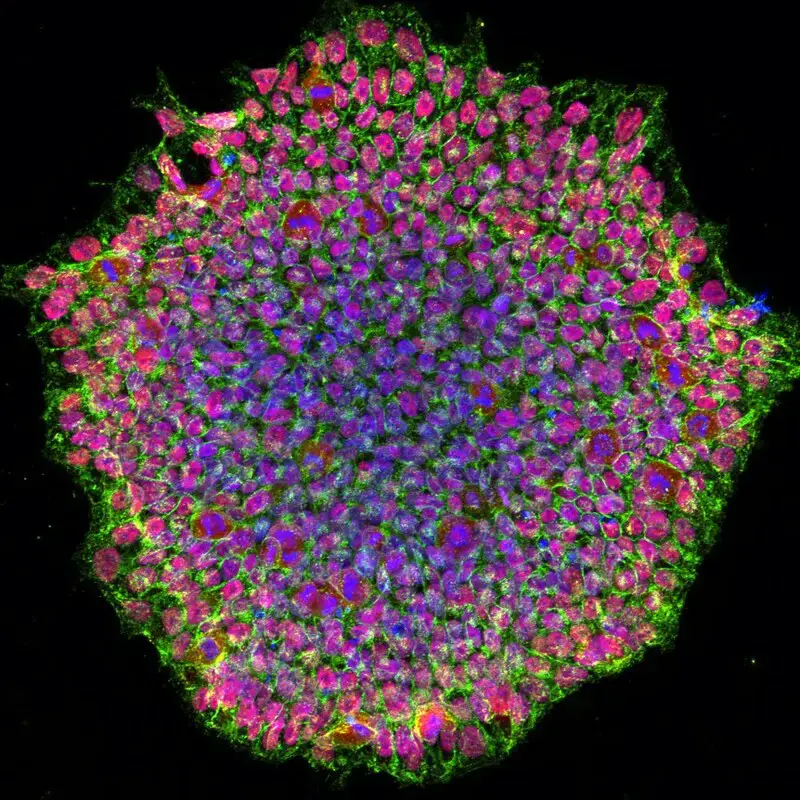
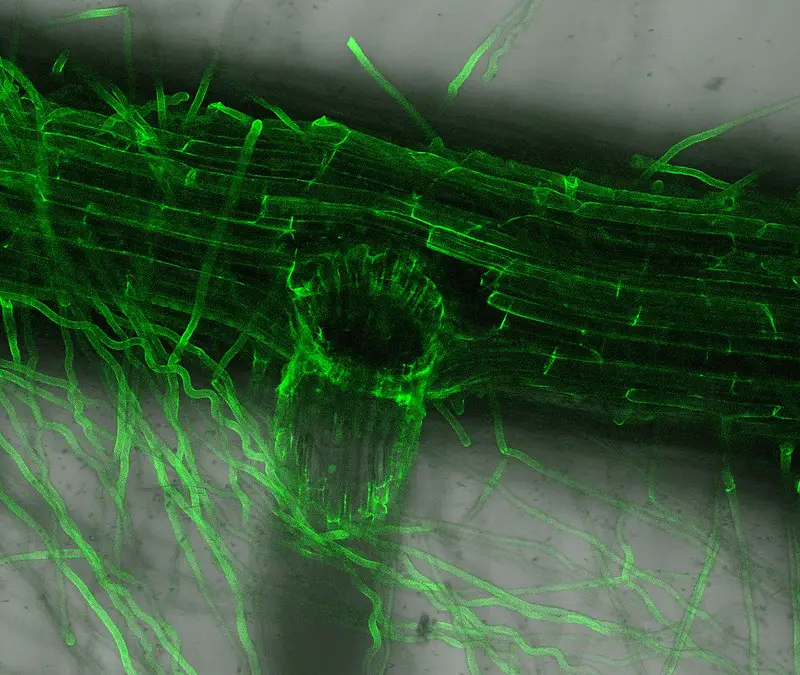
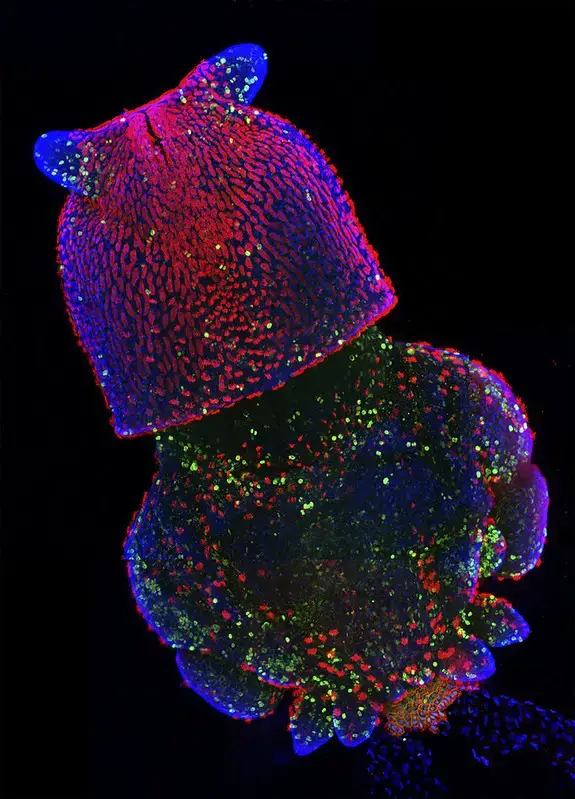
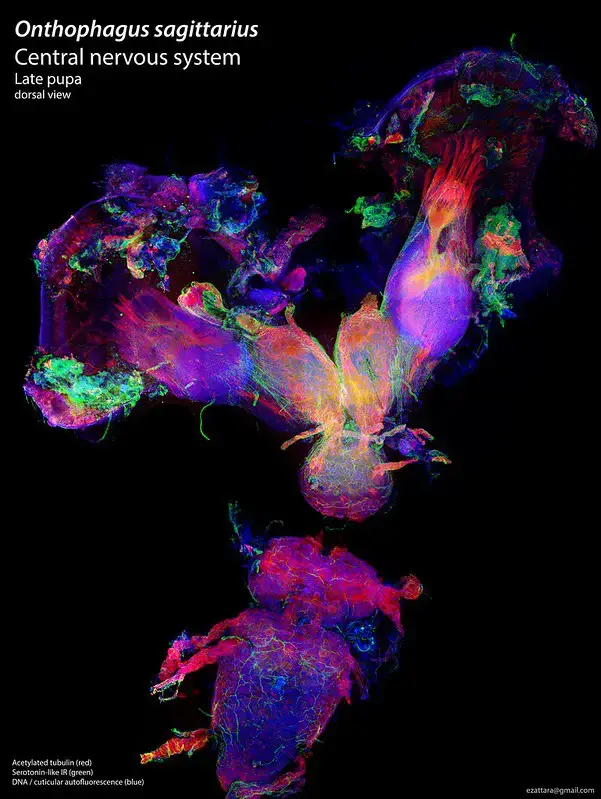
Check more Confocal microscope image – https://www.flickr.com/search/?text=Confocal%20microscope%20image
References
- Abberior. (2026). Diving into the deep: confocal vs multi-photon microscopy.
- Accurate Optics. (2024, March 4). Difference: Dielectric beam splitter vs. dichroic mirror.
- Axiom Optics. (2026). Main differences between confocal and widefield microscopy.
- Baylor College of Medicine. (n.d.). Multi-photon microscopy.
- Bergter, A., & Huff, J. (2016, April). ZEISS LSM 880 with Airyscan: Introducing the fast acquisition mode (Technology Note). Carl Zeiss Microscopy GmbH.
- Biocompare. (n.d.). Focal wars: Widefield vs. confocal.
- Borlinghaus, R. T. (2017, April 26). Pinhole effect in confocal microscopes. Leica Microsystems.
- Borlinghaus, R. T. (2017, May 16). Primary beam splitting devices for confocal microscopes. Leica Microsystems.
- Caston, J. (n.d.). IF imaging: Widefield versus confocal microscopy. Proteintech Group.
- Covalent Corp. (2026). Laser scanning confocal microscopy (LSCM).
- Evident Scientific. (2026). Comparing confocal and widefield fluorescence microscopy.
- Evident Scientific. (2026). Confocal laser scanning microscopes.
- Evident Scientific. (2026). Perform accurate and efficient microscopy image analysis using TruAI based on deep learning.
- Fellers, T. J., & Davidson, M. W. (2026). Confocal microscopy: Principles, optical sectioning & 3D imaging. Evident Scientific.
- FluoroFinder. (2025, January 22). Multiplexing with spectral confocal fluorescence microscopy.
- Gonzaga University. (n.d.). 3D surface profiler.
- Hamamatsu Photonics. (n.d.). The principle of confocal microscopy | MEMS confocal unit.
- Keyence Corporation. (2026). VK-X vs. interferometer.
- Leica Microsystems. (2005, August). AOBS Leica TCS SP5: The broadband confocal.
- Martin-Luther-Universität Halle-Wittenberg. (2017, October 23). General description [LSM 880 Airyscan].
- Microscope World. (2025, January 16). A comprehensive guide to confocal microscopes.
- Minsky, M. (1961, December 19). Microscopy apparatus (U.S. Patent No. 3,013,467A). U.S. Patent and Trademark Office.
- Minsky, M. (1988). Memoir on inventing the confocal scanning microscope. Scanning, 10, 128-138.
- Mokobi, F. (2024, April 16). Confocal microscope: Principle, parts, types, diagram, uses. Microbe Notes.
- National Center for Biotechnology Information. (n.d.). Confocal microscopy: Principles and modern practices – PMC.
- National Center for Biotechnology Information. (n.d.). The Airyscan detector from ZEISS: confocal imaging with improved signal-to-noise ratio and super-resolution.
- National Center for Biotechnology Information. (n.d.). ZEISS Airyscan: Optimizing usage for fast, gentle, super-resolution imaging – PMC.
- Nikon Instruments Inc. (2019, December 14). Nikon announces artificial intelligence (AI) for predictive imaging, image segmentation and processing.
- Nikon Instruments Inc. (2020, January). Nikon NIS-Elements Denoise.ai software: utilizing deep learning to denoise confocal data.
- Nikon Instruments Inc. (2025). Introductory confocal concepts | Nikon’s MicroscopyU.
- Nikon Instruments Inc. (2025). Spectral imaging and linear unmixing | Nikon’s MicroscopyU.
- Nikon Instruments Inc. (2026). Confocal and multiphoton microscopes.
- Nikon Instruments Inc. (n.d.). Denoise.ai | A1 HD25 / A1R HD25 | Confocal and multiphoton microscopes.
- Optolong Optics Co. (2024, March 27). Dichroic mirrors vs beamsplitters: Understanding the differences?.
- ResearchGate. (n.d.). (PDF) Confocal microscopy: Applications in materials science.
- ResearchGate. (n.d.). AOBS, working principle. The AOBS crystal is tuned to the desired….
- Scientific Volume Imaging. (2026). Confocal pinhole and spatial resolution.
- Spring, K. R., Fellers, T. J., & Davidson, M. W. (2026). Confocal microscopy resolution: Lateral, axial & pinhole effects. Evident Scientific.
- The systematic architecture of confocal microscopy: Principles, instrumentation, and analytical evolution in life and materials sciences. (n.d.).
- Wikipedia contributors. (2026, February 24). Confocal microscopy. In Wikipedia, The Free Encyclopedia.
- Wilson, M., DeRose, J., & Greb, C. (2017, June 29). Introduction to widefield microscopy. Leica Microsystems.
- Wilson, M., DeRose, J., & Greb, C. (2023, January 19). Microscope resolution: Concepts, factors and calculation. Leica Microsystems.
- ZEISS. (n.d.). ZEISS Airyscan | Super-resolution imaging and molecular measurements.
- Zimmermann, T. (2005). Spectral imaging and linear unmixing in light microscopy. Advances in Biochemical Engineering/Biotechnology, 95, 245–265.