UV-Visible (UV-Vis) spectroscopy is an analytical technique used to measure the absorption or transmission of ultraviolet and visible light by a sample. It works in UV region (about 190–400 nm) and visible region (about 400–800 nm). It is a fast and simple method used in both qualitative and quantitative analysis. The absorption is recorded as an absorption spectrum.
In this technique, electromagnetic radiation is passed through the sample and some part of light energy is absorbed by the molecules. The absorbed energy excites the outer valence electrons. The electrons are promoted from ground state to excited state by electronic transition. Compounds having chromophores absorb at specific wavelengths and give a characteristic peak.
The absorption spectrum obtained can be used to identify structural features and chemical composition of an unknown compound. The position of maximum absorption (λmax) and the intensity of absorption are noted for analysis. Different functional groups show different absorption pattern due to difference in electronic structure. This is referred to as UV absorption spectrum.
UV-Vis spectroscopy is also used for quantitative estimation by Beer-Lambert law. The absorbance is directly proportional to concentration of the absorbing molecules and path length of light. By measuring absorbance, concentration of a substance in solution can be calculated. It is commonly used for DNA/RNA and protein quantification, drug quality control, and detection of contaminants in water testing.
Principle of UV Spectroscopy
Principle of UV Spectroscopy is based on the interaction of ultraviolet and visible light with the molecules present in the sample solution. When UV or visible radiation is passed through a compound, a part of the radiation is absorbed. The absorbed energy excites the outer valence electrons. The electrons are promoted from ground state to excited state.
The electronic transition occurs only when the energy of the absorbed light matches the energy gap between the two states. Different bonds and functional groups have different electronic arrangements, so they absorb at specific wavelength range. This gives a characteristic absorption spectrum (absorbance vs wavelength) which is used for identification of compound.
For quantitative estimation, the principle follows Beer-Lambert law. The absorbance is directly proportional to concentration of the absorbing molecules and the path length of light through the solution. The difference between incident light intensity and transmitted light intensity is measured and absorbance is calculated. From this, the concentration of the substance is determined.
Instrumentation (Parts of UV Spectroscopy)
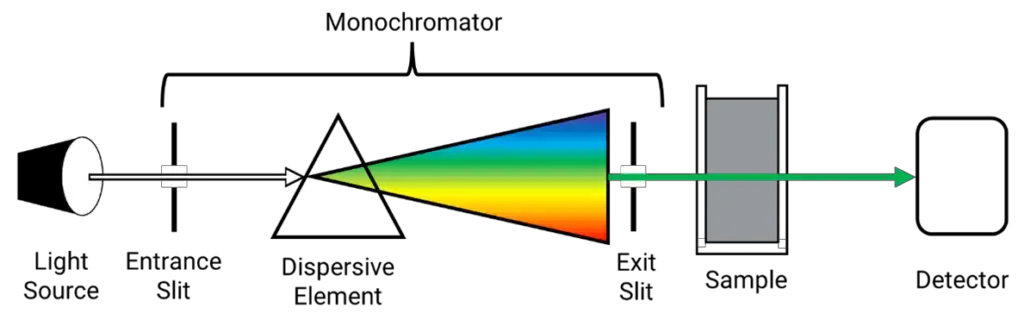
The UV spectrophotometer is made up of different parts which help in producing UV radiation, selecting wavelength and measuring absorbance. The main parts are as follows-
- Light source
It provides ultraviolet radiation for analysis. Tungsten filament lamp is used for emission of red radiations and covers around 375 nm region. Hydrogen-deuterium lamp is used for UV region below 375 nm. Both sources together cover required UV range. - Monochromator
It is used to disperse the light and select a particular wavelength. It contains prism and slits (entrance slit and exit slit). The prism is rotated and different wavelengths are separated. The slit allows only monochromatic light to pass. In double beam instrument, the selected light is split into two beams. - Sample cell and reference cell
These cells hold the sample solution and reference (blank) solution. Usually quartz or silica cells are used because glass absorbs UV light. One beam passes through sample and another beam passes through reference for comparison. - Detector
Detector receives the transmitted beams from sample and reference cells. Generally two photocells are present. The reference beam intensity is higher than sample beam, so alternating electrical currents are produced in the photocells. This electrical signal is proportional to transmitted light. - Amplifier
The alternating current produced in photocells is very weak. It is sent to amplifier to increase the signal strength. Amplifier may be connected with servometer for signal control. The amplified signal is now suitable for recording. - Recording device / readout system
It records the signal and gives the spectrum. Pen recorder or computer system is connected with amplifier. It stores data and shows the spectrum in the form of absorbance or transmittance.
Protocol/Step by step procedure of UV Spectroscopy
- Prepare a blank solution (solvent only) and keep it ready.
- Prepare standard solutions of the given compound with known different concentrations.
- Prepare the unknown sample solution in the same solvent.
- If absorbance is weak, a colour forming reagent or ligand is added to form coloured complex.
- Wash the cuvettes properly and dry it.
- Handle cuvette only from frosted sides, do not touch the clear optical surface.
- Check for scratches, lint or finger print and remove it if present.
- Switch on the UV-Vis spectrophotometer and allow lamp to warm.
- Select the required wavelength or scan and note the λmax of the compound.
- Set the instrument at λmax for maximum sensitivity.
- Fill the cuvette with blank and place it in the sample holder.
- Set zero absorbance (or 100% transmittance) using blank.
- Remove blank cuvette after baseline is set.
- Take first standard solution in a clean cuvette and place it in instrument.
- Record absorbance at λmax.
- Repeat same for all standard solutions and record absorbance of each.
- Plot calibration curve between concentration (x-axis) and absorbance (y-axis).
- A straight line is obtained within Beer-Lambert law range.
- Take unknown sample in cuvette and place in instrument.
- Record absorbance at λmax.
- Compare the absorbance of unknown with calibration curve.
- Find corresponding concentration from the graph.
- Report the concentration of the unknown sample.
Applications of UV Spectroscopy
- Quantitative analysis
Used for estimation of concentration of absorbing compounds by Beer-Lambert law. Used for assay of solutions by measuring absorbance. - Qualitative analysis
Used to identify compounds by absorption spectrum and λmax. Used to detect chromophores and impurities in sample. - Structural elucidation
Used to study electronic structure and functional groups. Used to determine unsaturation and conjugation in organic compounds. It can differentiate conjugated and non-conjugated dienes. Used to identify tautomeric forms and steric hindrance by change in absorption. - Chemical kinetics and equilibrium
Used to study rate of reaction by change in absorbance with time. Used for monitoring enzymatic reactions. Used to determine pKa by spectral change. Used for complex ion formation and stability constant studies. - Biochemistry and molecular biology
Used for quantification and purity checking of DNA and RNA. Used for estimation of proteins. Used to study protein conformational change, denaturation and ligand binding. - Pharmaceutical applications
Used for drug identification and quality control. Used for assay and content uniformity. Used in dissolution test for drug release. Used for stability and degradation studies. - Environmental monitoring
Used for water quality testing and detection of nitrates and nitrites. Used for COD, BOD and suspended solids measurement. Used for monitoring gaseous pollutants and contaminants in soil and sediments. - Food and beverage analysis
Used for estimation of vitamins, caffeine, sugars and preservatives. Used in wine for colour intensity and phenolic content. Used to check oxidation and quality of edible oils. - Clinical diagnosis
Used in routine tests for glucose, cholesterol, uric acid and bilirubin. Used in point-of-care testing like haemoglobin monitoring and pulse oximetry. - Material science and research
Used for characterization of nanoparticles (gold, silver) by size and aggregation state. Used for polymer degradation study. Used to follow oxidation states and intermediates in catalytic research.
Advantages of UV Spectroscopy
- It is a versatile technique and can be used for both organic and inorganic compounds.
- It is used in different fields like molecular biology, pharmaceuticals and environmental analysis.
- It gives good sensitivity and small amount of substance can be detected (about 10⁻⁴ to 10⁻⁵ M).
- It gives good accuracy in quantitative estimation (generally 1% to 3% error range).
- It is a rapid method and results are obtained in short time.
- The procedure is simple and convenient for routine analysis.
- It is useful for kinetics study and continuous monitoring by taking absorbance with time.
- It is a non-destructive method, sample is not destroyed during analysis.
- Small and precious samples can be recovered after measurement.
- It is cost effective compared to many other analytical techniques.
- Single beam instruments are simple, compact and portable.
- Maintenance cost is also low in many instruments.
- It shows moderate to high selectivity for absorbing species and chromophores.
- It can detect specific functional groups by characteristic absorption.
Limitations of UV Spectroscopy
- UV spectrum alone cannot uniquely identify an unknown compound.
- It gives less fine structural detail, so confirmation is needed by other methods (NMR, MS etc.).
- Beer-Lambert law is not followed properly at high concentration, so linearity is lost.
- At higher concentration, molecular interaction and change in refractive index causes deviation.
- It works best with clear solution, cloudy samples gives wrong reading.
- Suspended particles scatter the light and absorbance becomes inaccurate.
- Change in pH can alter the absorption spectrum.
- Association and dissociation of molecules in solution affects the result.
- Complex formation in solution changes peak position and intensity.
- Stray light reaching detector produces error in absorbance.
- Non-monochromatic light (wide bandwidth) reduces accuracy.
- Lamp intensity fluctuation also affects measurement.
- In complex mixtures, absorption bands are broad and overlapping.
- It becomes difficult to analyze many absorbing species together.
- Single beam instrument is affected by lamp drift and frequent re-zero is needed.
- Double beam instrument is costly, complex and bigger in size.
References
- ABB. (n.d.). Using UV/VIS spectrophotometry for real-time BOD, COD, and TOC monitoring.
- Agilent Technologies, Inc. (2025). The basics of UV-Vis spectrophotometry.
- Agiomavriti, A.-A., Bartzanas, T., Chorianopoulos, N., & Gelasakis, A. I. (2025). Spectroscopy-based methods for water quality assessment: A comprehensive review and potential applications in livestock farming. Water, 17(16), 2488.
- Algar, R. (n.d.). UV-Visible spectrophotometers. In Short stories in instrumental analytical chemistry. Pressbooks.
- Analytical principles and technological advancements in ultraviolet-visible spectroscopy. (n.d.).
- Ashenhurst, J. (2025, September 24). What is UV-Vis spectroscopy? And how does it apply to conjugation? Master Organic Chemistry.
- Berhampore Girls’ College. (n.d.). 1. Introduction 2. Principle 3. Beer – Lambert’s law 4. Instrumentation 5. Single beam spectrophotometer 6. Do.
- Canbay, H. S. (2022). Spectrophotometric determination of acid dissociation constants of some arylpropionic acids and arylacetic acids in acetonitrile-water binary mixtures at 25ºC. Brazilian Journal of Pharmaceutical Sciences.
- Delloyd. (n.d.). Ultraviolet-visible spectroscopy instrument. Delloyd’s Lab-Tech Chemistry Resource.
- Drawell. (n.d.). The difference between single and double beam UV-visible spectrophotometers.
- Edinburgh Instruments. (2021, July 8). Beer-Lambert law | Transmittance & absorbance.
- Factors affecting UV visible spectroscopy | PPTX. (n.d.).
- Haffner, H. (2024, January 5). Measurement of enzyme kinetics by UV-visible spectroscopy and advanced calculations. JASCO.
- Hitachi High-Tech Corporation. (n.d.). 8. Structure of a spectrophotometer (3).
- Imran, M. (n.d.). Factors influencing UV-visible spectra. Scribd.
- JASCO Corporation. (n.d.). Application note: Measurement of enzyme activity – ALP activity (UV-0002).
- JASCO Inc. (n.d.). Measurement of enzyme kinetics by UV-visible….
- Khan, S. (n.d.). Spectrophotometry and the Beer–Lambert law [Video]. Khan Academy.
- Labcompare.com. (n.d.). Tech compare: Single vs. double beam spectrophotometers.
- Mettler Toledo. (n.d.). UV/Vis water testing | Your guide to accurate water quality analysis.
- Mohiuddin, K. (n.d.). Factors influencing UV absorption peaks. Scribd.
- NanoQAM. (n.d.). Applications of ultraviolet-visible molecular absorption spectrometry (Chapter 14).
- PatSnap Synapse. (n.d.). What is enzyme kinetics? Understanding Km and Vmax in biochemical reactions.
- PatSnap Synapse. (n.d.). What is UV-Vis spectrophotometry used for in biochemistry?.
- PMC. (n.d.). Rapid determination of ionization constants (pKa) by UV spectroscopy using 96-well microtiter plates.
- PMC. (n.d.). Wastewater quality estimation through spectrophotometry-based statistical models.
- Real Tech Water. (n.d.). Using UV/Vis spectrophotometry for real-time….
- ResearchGate. (n.d.). Application of UV-visible spectroscopy in the quantitative determination and characterization of organic compounds.
- ResearchGate. (n.d.). Solvent, substituents and pH effects towards the spectral shifts of some highly colored indicators.
- Reusch, W. (2023, January 22). Empirical rules for absorption wavelengths of conjugated systems. Chemistry LibreTexts.
- Scribd. (n.d.). UV-Vis spectrophotometer components.
- Slideshare. (n.d.). BEER-LAMBERT’S LAW (2).pptx.
- Slideshare. (n.d.). Factors affecting UV visible spectroscopy | PPTX.
- Sper Scientific. (2025, July 10). What’s the difference between single and dual beam spectrophotometers?.
- Spectroscopy applications: Real-world uses of UV-Vis analysis … (n.d.).
- Thermo Fisher Scientific. (n.d.). Monitoring enzyme kinetics using UV-visible absorption spectroscopy – Michaelis-Menten analysis.
- Torontech Team. (2025, March 12). Spectrophotometer: Single beam vs double beam explained. Torontech.
- U.V. visible spectroscopy. (n.d.).
- Unchained Labs. (n.d.). UV/Vis spectroscopy for DNA & protein analysis.
- University of Toronto Scarborough. (n.d.). UV spectrophotometer.
- Ultraviolet visible spectroscopy – Principle, applications. (2026). Learning Chemistry.
- UV-Vis absorption spectroscopy – Theory. (n.d.).
- UV-Vis lesson 3 – Woodward-Fieser rules. (n.d.). kdsfun.weebly.com.
- Wikipedia. (2023, August 24). Molecular electronic transition.
- Wikipedia. (2025, November 7). Woodward’s rules.
- Wikipedia. (2026, March 17). Beer–Lambert law.
- WOODWARD-FIESER RULES FOR CALCULATION OF – max FOR π-π (TRANSITION) ABSORPTION BAND OF – a, B-UNSATURATED CARBONYL COMPOUNDS*. (n.d.). Googleapis.com.
- Wu, E. (2025, July 30). Single vs double beam spectrophotometers: Key differences. AELAB.
References
- Chemistry LibreTexts. (n.d.). 4.4: UV-Visible Spectroscopy. Retrieved from https://chem.libretexts.org.
- Wikipedia contributors. (n.d.). Ultraviolet–visible spectroscopy. In Wikipedia, The Free Encyclopedia. Retrieved from https:/en.wikipedia.org.
- Technology Networks. (n.d.). UV-Vis Spectroscopy: Principle, Strengths and Limitations and Applications. Retrieved from https://www.technologynetworks.com.
- Chemistry LibreTexts. (n.d.). 4.5: Ultraviolet and Visible Spectroscopy. Retrieved from https://chem.libretexts.org.
- Chemistry LibreTexts. (n.d.). 2.1: Introduction to UV Spectroscopy. Retrieved from https://chem.libretexts.org.
- Chemistry LibreTexts. (n.d.). 3.5: UV-Visible Spectrometer. Retrieved from https://chem.libretexts.org.