Inoculating loops and needles are hand held laboratory instruments used in microbiology. These are used to pick and transfer small amount of microorganisms from one culture medium to another. The small amount of microorganism which is transferred is called inoculum.
These instruments are also called smear loops, inoculation wands or microstreakers. They have a handle, insulated shaft and a tip at the end. The tip is the main working part of the instrument.
Inoculating loop has a wire tip which is bent into small circle or oval shape. It can hold liquid by surface tension. Commonly it holds about 1 µL or 10 µL liquid. It is used for transferring liquid culture and for streaking on agar plates.
Inoculating needle has a straight pointed or slightly blunt tip. It has no loop at the end. It is used for picking single colonies and for stab inoculation in semi-solid media. It can enter deep into the medium.
Traditional loops and needles are made up of heat resistant metals like nichrome or platinum. These are sterilized by flame before and after use. Now disposable plastic loops and needles are also used. These are pre-sterilized and reduce chance of cross contamination.
The development of these instruments is related with the golden age of microbiology. Earlier, Robert Koch used sterilized wood splinters for transferring pathogens. Later solid culture media like gelatin and agar were used, so a more delicate tool was needed for spreading culture.
Metal wires were then made into wand like instruments with looped tips. The platinum wire loop is related with Mazyck P. Ravenel. In 1960, O’Sullivan and others standardized loop as volumetric measuring tool by using 3 mm loop for quantitative urine culture. This made the inoculating loop a more accurate instrument for clinical and diagnostic laboratories.
Principle of Inoculating Loops and Needles
Principle of Inoculating Loops and Needles is based on aseptic and precise transfer of microorganisms. It is used to transfer bacteria or yeast from one source to another growth medium. During this process contamination should not occur.
In inoculating loop, the circular loop is dipped into liquid culture. The liquid is held inside the loop due to surface tension. It forms a small droplet and carries fixed amount of sample.
The loop is used for spreading or streaking microorganisms on solid agar medium. It helps to distribute the organisms on the surface of medium. This is used for isolation of colonies.
Inoculating needle works by mechanical piercing. It has straight and pointed tip. It is used to pick single colony and to inoculate deep or semi-solid medium.
During stab inoculation, the needle enters vertically inside the medium. The microorganism is delivered into the inner part of the medium and not only on surface.
Reusable loops and needles are sterilized by heat. The wire is heated in flame until it becomes red hot. This burns and kills all contaminating organisms present on the wire.
After heating, the loop or needle is cooled properly. If it is used while hot, it may kill the required microorganisms. Then it is used for taking the desired microbial sample.
Parts of Inoculating Loops and Needles
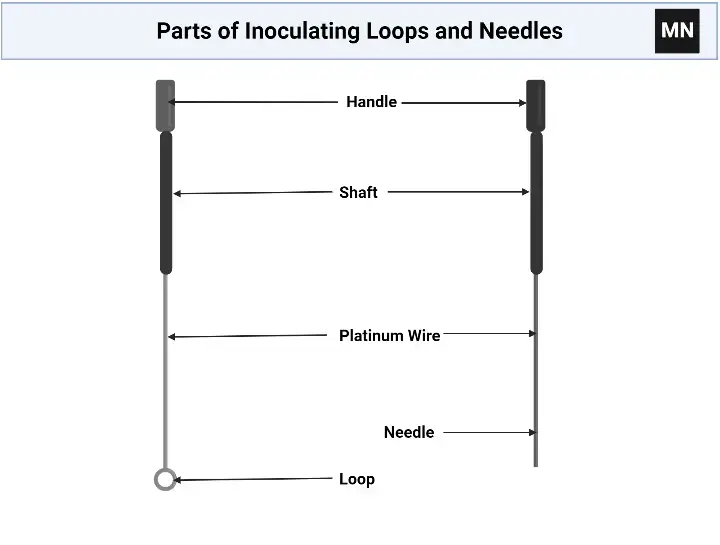
Inoculating loops and needles are made up of few simple parts. These parts help in holding, heating and transferring microorganisms. The main parts are as follows-
- Handle
It is the part which is held by the user. It is generally 6 to 8 inches long. It is made up of heat resistant materials like aluminium, brass or moulded plastic. In reusable metal instruments, the handle may have PVC sleeve for protecting the hand from heat. - Shaft or wire shaft
It connects the handle with the terminal tip. It is made up of nichrome, platinum, nickel plated brass or plastic. It should be rigid but slightly flexible. This helps to prevent shaking and scratching of agar surface. - Turret
It is present in reusable tools. It holds the nichrome or platinum wire tightly in proper place. It helps to fix the wire with the handle. - Terminal end
It is the working end of the instrument. This part directly touches the microorganisms. It is used for picking, transferring or inoculating the culture. - Loop
The loop is a wire bent into small circular or oval shape. Its diameter may be about 1 mm to 10 mm. It holds liquid drop by surface tension. It is used for transferring liquid culture and streaking agar plate. - Needle
The needle is a straight wire with pointed or slightly blunt end. It is used for picking particular colony. It is also used for puncturing deep into semi-solid media during stab inoculation.
Types of Inoculating Loops and Needles
The types of inoculating loops and needles are as follows-
- Calibrated loops
These loops are used to hold fixed amount of liquid culture. Commonly 1 µL and 10 µL loops are used. It is used for quantitative culture, such as urine culture. - Non-calibrated loops
These are general purpose loops. They are available in different internal diameter like 2 mm, 3 mm, 4 mm or 5 mm. It is used for ordinary transfer and streaking work. - Metal reusable loops
These loops are made up of heat resistant metals like nichrome, platinum or tungsten. They can be used again and again after sterilization. They are sterilized by open flame or electric incinerator. - Plastic disposable loops
These are single use loops. They are already sterilized before use. They do not need flame sterilization and reduce the chance of cross contamination and infectious aerosols. - Rigid loops
These are hard plastic loops. They are used for streaking and for picking colonies where little pressure is needed. They are more firm than soft loops. - Flexible loops
These are soft and bendable loops. They are used to prevent cutting or scratching of agar surface. They are useful for gentle streaking. - Metal reusable needles
These are straight pointed wires made up of nichrome or platinum. They are attached with handle. They are sterilized by flame before and after use. - Plastic disposable needles
These are single use needles made from plastic resin. They are pre-sterilized. They are useful where open flame is not safe, such as inside anaerobic chamber. - Regular needles
These are standard size plastic needles. They may be about 20 cm long with 1.45 mm diameter. They are used for stab inoculation and picking colonies. - Mini-needles
These are shorter and thinner needles. They may be about 15 cm long with 1.15 mm diameter. They are more flexible and used for delicate inoculation work.
How to Use Inoculating Loops and Needles
- First the loop or needle is sterilized. If it is metal loop or needle, it is passed through Bunsen burner flame. The wire is heated until it becomes red hot. This kills the contaminating microorganisms present on it.
- If disposable plastic loop or needle is used, flame sterilization is not done. These are already sterilized. They are used once and then discarded.
- After heating, the metal loop or needle is allowed to cool for few seconds. Hot loop should not be touched with culture. If it is too hot, it may kill the microorganisms.
- The loop may be cooled by touching it on sterile uninoculated part of agar. It should be touched gently. Then it is used for taking the sample.
- If sample is taken from liquid broth, the cap of culture tube is removed. The mouth of tube is passed quickly through flame. The loop is dipped into the broth and small drop of culture is taken.
- After taking the broth culture, the mouth of tube is again flamed. Then the cap is replaced. This is done to prevent contamination.
- If sample is taken from agar plate, the lid of petri dish is slightly opened. The loop or needle is touched gently on the bacterial colony. A small amount of colony is picked from the surface.
- For streaking on agar plate, loop is used. The loop is moved gently on the surface of new agar plate. It is moved in zig-zag manner. The agar surface should not be cut or scratched.
- For liquid inoculation, the loop is dipped into sterile broth tube. It is moved back and forth in the liquid. This helps to release the microorganisms into the broth.
- For stab inoculation, needle is used. The straight needle is inserted vertically into semi-solid or deep agar. It is pushed down near about 0.5 inch above the bottom.
- The needle is taken out slowly through the same line. It should not be moved side wise. This prevents tearing of the agar medium.
- After use, metal loop or needle is again heated in flame until red hot. This destroys remaining culture on it. Disposable plastic loop or needle is not heated and is thrown into proper biohazard container.
Applications of Inoculating Loops and Needles
- Inoculating loops and needles are used for aseptic transfer of microorganisms. Bacteria, yeasts and fungal spores are transferred from one source to another growth medium.
- They are used for sample preparation in microbiology. Serial dilution, sterile sampling and spreading of microbial sample are done by these instruments.
- Inoculating loop is used for streak plating. The microbial culture is spread on agar plate surface. This helps to get pure and separate colonies.
- It is used for liquid culture transfer. The loop takes small droplet of culture and transfer it into another medium.
- Calibrated loops are used for quantitative transfer of liquid. Commonly 1 µL and 10 µL loops are used. It is useful in urine culture.
- Inoculating loop is used for slide preparation. A small amount of culture is placed on glass slide. Then smear is made for staining and microscopic examination.
- It is used in biochemical tests. Small amount of colony is transferred to glass slide or reagent paper. Tests like oxidase and catalase can be done by this method.
- Inoculating needle is used for colony picking. It can pick a very small and specific colony. Nearby colonies are not disturbed much.
- It is used for stab inoculation. The needle is pierced straight into semi-solid or deep solid medium. The bacteria are delivered inside the medium.
- It is used for motility testing. The needle is stabbed into semi-solid medium like SIM medium. If growth spreads away from the stab line, the organism is motile.
- It is used for oxygen requirement test. Microorganisms are inoculated deep into media like thioglycollate medium. This helps to study oxygen need and growth of anaerobic organisms.
Advantages of Inoculating Loops and Needles
- Inoculating loops and needles are simple to use. They are small hand held instruments. They are used for transferring, streaking and picking microbial sample.
- They are less costly instruments. They can be used easily in microbiology laboratory. Special complex arrangement is not required.
- Some loops are calibrated. Common sizes are 1 µL and 10 µL. These loops transfer fixed amount of liquid sample.
- They are useful for different microbiological works. Loop is used for surface streaking and liquid transfer. Needle is used for deep stab inoculation and colony picking.
- Metal loops and needles are reusable. They can be used many times after sterilization. So the total cost becomes less for long time use.
- Metal wires are strong and durable. They also give good control during delicate work. It helps in picking and transferring small microbial growth.
- Platinum and nichrome wires heat quickly and cool also fast. This saves time during repeated sterilization and inoculation work.
- Platinum loop is resistant to oxidation and chemical damage. So it can be used for long period without easy damage.
- Disposable plastic loops and needles are safe to use. They are already sterilized. Flame sterilization is not needed, so aerosol formation and burn injury is avoided.
- Plastic disposable tools reduce cross contamination. A new sterile loop or needle is used each time. So one sample is not mixed with another sample.
- They are useful in restricted places. Plastic loops and needles can be used in anaerobic chamber, biosafety cabinet and clean room where open flame is not allowed.
- Disposable tools save time. Heating and cooling step is not needed. So it is useful when many samples are handled together.
- Many plastic loops have colour coding and smooth edges. Colour coding helps to identify size easily. Smooth edge prevents scratching or cutting of agar surface.
Limitations of Inoculating Loops and Needles
- Platinum wire loops are costly. Disposable plastic loops and needles also become costly when used in large number. So the laboratory cost may increase.
- Disposable plastic tools produce biohazard waste. They are used only once and then discarded. So they are not much environment friendly.
- Metal loops and needles are sterilized by flame. They must be cooled before touching the culture. If the wire is hot, it kills the microorganisms.
- Wet loop should not be heated suddenly. The liquid may boil and splatter. This can produce infectious aerosols.
- If the loop vibrates on agar surface, it may flick the culture. This also can spread microbial aerosol. So it may be dangerous during handling of pathogens.
- The use of loop and needle depends on proper hand skill. Aseptic technique should be maintained. If not done properly, contamination and cross contamination may occur.
- Manual inoculation takes more time. When many samples are handled, chance of human error is more.
- Repeated heating and cooling may change the shape of metal loop. Carbonized organic matter may also collect on the wire. This can affect the fixed volume of calibrated loop.
- Calibrated loops need proper technique for correct volume transfer. Thick or viscous sample may not be taken accurately. Inoculating needle has no fixed volume control.
- Nichrome or nickel loops may interfere with some biochemical tests. In oxidase test, it may give false positive reaction.
- The agar surface may be scratched or cut during streaking. This occurs when the tool is too rigid or too much pressure is applied.
- Open flame is needed for reusable tools. But flame is not safe in some places like Biological Safety Cabinet (BSC). It can disturb laminar air flow and may cause fire hazard.
Precautions of Inoculating Loops and Needles
- Reusable metal loops and needles should be sterilized before and after use. The wire is heated in flame until it becomes red hot. This kills the microorganisms present on it.
- Disposable plastic loops and needles should not be reused. They are already sterile. After one use, they are discarded into proper biohazard container.
- During flaming, the wire is heated from base to tip. The wet loop should not be directly placed into flame by tip first. The liquid may boil and splatter.
- The hot loop or needle is allowed to cool for 5 to 10 seconds. If hot wire touches the culture, it kills the microorganisms. It may also make microbial mist.
- The loop should not be waved in air for cooling. It should not be blown by mouth. This may bring airborne contamination.
- The cooled loop can be checked on uninoculated part of agar. If agar melts or hissing sound comes, then the loop is still hot.
- Sterile loop or needle should not be kept on the work bench. The tip should not touch hand, table or any non-sterile surface.
- During streaking, the loop is moved gently on agar surface. Too much pressure should not be applied. It may cut or gouge the agar.
- The loop should not be pulled very hard. It may snap or flick the culture. This can spread infectious droplets.
- Metal loop should be heated red hot only for sterilization. Continuous overheating should be avoided. It may damage the wire slowly.
- After the work is over, the working area is disinfected. Especially the area around the heating device should be cleaned. About 12 inch ring around the base is disinfected to remove settled microorganisms.
References
- Accuracy and precision of quantitative calibrated loops in transfer of bronchoalveolar lavage fluid. (n.d.). PubMed Central (PMC). https://pmc.ncbi.nlm.nih.gov/articles/PMC86741/
- Advanced synthesis of inoculating instruments in microbiology: Principles, methodologies, and technical evolution. (n.d.). [Markdown Document].
- American Society for Microbiology. (n.d.). The streak plate protocol. https://asm.org/asm/media/protocol-images/the-streak-plate-protocol.pdf
- American Type Culture Collection (ATCC). (n.d.). Introduction to microbiology. https://www.atcc.org/resources/culture-guides/introduction-to-microbiology
- Australian Wine Research Institute. (n.d.). Aseptic technique. https://www.awri.com.au/industry_support/winemaking_resources/laboratory_methods/microbiological/aseptic/
- Becton, Dickinson and Company (BD). (n.d.). BD Difco™ loops and needles. https://www.bd.com/en-us/products-and-solutions/products/product-families/bd-difco-loops-and-needles
- Bios, L. (2025, February 19). In lab inoculating loops are sterilized using. Ucallmlabs. https://ucallmlabs.com/in-lab-inoculating-loops-are-sterilized-using/
- Brennan-Krohn, T. (2021, February). Urine good hands: Diagnosing UTIs with urine cultures. American Society for Microbiology. https://asm.org/articles/2021/february/urine-good-hands-diagnosing-utis-with-urine-cultur
- Bunzl Asia Pacific. (n.d.). INOCULATING LOOPS, NEEDLES and SPREADERS. https://www.bunzlasiapacific.com/extension/IP/document_72_5be51cf9f20b0.pdf
- Charles, A. D. (2023). Robert Koch | History | Research starters. EBSCO. https://www.ebsco.com/research-starters/history/robert-koch
- Comparative analysis of different loop types for urine culture collection: Implications for quantitative bacterial growth and culture results. (n.d.). PubMed. https://pubmed.ncbi.nlm.nih.gov/38330685/
- Copan Diagnostics. (n.d.). INOCULATING LOOPS, NEEDLES and SPREADERS. https://www.copanusa.com/wp-content/uploads/2024/01/Loops-Needles-and-Spreaders-Brochure.pdf
- Geisinger Medical Laboratories. (n.d.). Quantitative urine culture guide. https://www.geisingermedicallabs.com/Catalog/Quantitative_Urine_Culture_Guide.pdf
- Greenwood, M. (2019, February 26). Aseptic techniques in microbiology. News-Medical.Net. https://www.news-medical.net/life-sciences/Aseptic-Techniques-in-Microbiology.aspx
- Hardy Diagnostics. (n.d.). Speedstreaks™ – Disposable inoculation loops. https://hardydiagnostics.com/media/assets/product/documents/SpeedStreaks.pdf
- Hawach Scientific. (n.d.). Difference between inoculating needle and inoculating loop. https://m.hawach.com/news/what-is-the-difference-between-inoculating-needle-and-inoculating-loop.html
- Hawach Scientific. (n.d.). What is an inoculating loop. https://www.hawach.com/news/what-is-an-inoculating-loop.html
- Huida Medical. (n.d.). Choosing the right calibrated inoculating loop: 1 μL vs 10 μL – Knowledge. https://www.huidainstrument.com/info/choosing-the-right-calibrated-inoculating-loop-103114558.html
- Huida Medical. (n.d.). Metal vs plastic inoculating loops: Pros, cons, and use cases. https://www.huidainstrument.com/info/metal-vs-plastic-inoculating-loops-pros-cons-103114482.html
- inoculating loops: nichrome/nickel vs. platinum wire? : r/microbiology. (2017). Reddit. https://www.reddit.com/r/microbiology/comments/64av8z/inoculating_loops_nichromenickel_vs_platinum_wire/
- Karki, P. (n.d.). Inoculating loops and needles- Principle, parts, types, uses. Microbe Notes. https://microbenotes.com/inoculating-loops-needles-principle-parts-types-uses/
- Kashi Scientific. (n.d.). Loops and needles – Cell culture. https://kashiscientific.com/cell-culture/loops-and-needles/
- Key Scientific Products. (n.d.). Microloops® loops and needles for perfect inoculation. https://www.keyscientific.com/files/Other%20Manufacturers/Medical%20Wire/Microloops%C2%AE%20for%20perfect%20inoculation.pdf
- LAB-Net, University of Antwerp. (2023, February 8). Sample collection and management manual for the clinics and local laboratory procedures within the ECRAID-Base POS-cUTI. Ecraid. https://www.ecraid.eu/sites/default/files/2024-01/Laboratory%20Manual%20UTI_V3.0.pdf
- Lab Consumables Supplier. (n.d.). Inoculating needle vs loop. https://www.sd-labio.com/inoculating-needle-vs-loop/
- Medical Wire & Equipment. (n.d.). MICROLOOP® platinum range – wire inoculating loop. https://www.mwe.co.uk/product/microloop-platinum-wire-inoculating-loop/
- Membrane Solutions. (n.d.). How to make an inoculating needle or loop. https://www.membrane-solutions.com/News_974.htm
- Microbiology. (n.d.). Quantitative | MI. https://microbiology.mlsascp.com/quantitative.html
- Milne Publishing. (n.d.). Bacteriological culture methods – Microbiology: A laboratory experience. https://milnepublishing.geneseo.edu/suny-microbiology-lab/chapter/bacteriological-culture-methods/
- MiniPlast. (n.d.). Mastering microbial culturing: The essential role of inoculating loops. https://www.mini-plast.com/mastering-microbial-culturing-the-essential-role-of-inoculating-loops/
- National Institute of Standards and Technology. (2019). SOP 14 recommended standard operating procedure for gravimetric calibration of volumetric standards using an electronic balance. https://www.nist.gov/document/sop-14-gravimetric-calibration-20190508pdf
- PakGent Bioscience. (n.d.). Inoculation loop & needle. https://www.pakgentbio.com/product_26.html
- Pharmaceutical Microbiology. (2017, January 1). What is an inoculation loop? Principle, types, sterilization, calibration & GMP requirements. https://www.pharmaceuticalmicrobiology.in/2016/12/inoculation-loop.html
- PV Equip. (n.d.). INOCULATING LOOPS, NEEDLES and SPREADERS. https://www.pvequip.cl/wp-content/uploads/2015/03/Asas-Brochure-Copan.pdf
- Robert Koch: From anthrax to tuberculosis – A journey in medical science. (n.d.). PubMed Central (PMC). https://pmc.ncbi.nlm.nih.gov/articles/PMC11617125/
- Spectrum Chemical. (n.d.). Reusable-nichrome-wire-inoculating-loops-without-handles. https://www.spectrumchemical.com/reusable-nichrome-wire-inoculating-loops-without-handles-336763
- Sterilizing a loop. (n.d.). YouTube. https://www.youtube.com/watch?v=SzmoYgyf2bs
- St. Olaf College. (n.d.). Preventing aerosol production – Laboratory safety. https://wp.stolaf.edu/chemical-hygiene/preventing-aerosol-production/
- The basic concept of microbiology. (n.d.). PubMed Central (PMC). https://pmc.ncbi.nlm.nih.gov/articles/PMC7123386/
- The legacy of Robert Koch: Surmise, search, substantiate. (n.d.). PubMed Central (PMC). https://pmc.ncbi.nlm.nih.gov/articles/PMC3916274/
- Thermo Fisher Scientific. (n.d.). Quantifying and comparing the flexibility of inoculating loops. https://documents.thermofisher.com/TFS-Assets%2FLPD%2FApplication-Notes%2Finoculating-loops-appnote.pdf
- Thomas Scientific. (n.d.). Inoculating loops & needles. https://www.thomassci.com/laboratory-supplies/inoculating
- The University of Tennessee, Knoxville. (2023, April 24). Managing infectious aerosols. https://ehs.utk.edu/index.php/table-of-policies-plans-procedures-guides/managing-infectious-aerosols/
- University of Toronto. (n.d.). Techniques for minimizing aerosols – Environmental health & safety. https://ehs.utoronto.ca/our-services/biosafety/techniques-for-minimizing-aerosols/
- Wikipedia. (2026, March 5). Griffith’s experiment. https://en.wikipedia.org/wiki/Griffith%27s_experiment
- Wikipedia. (2026, February 1). Inoculation loop. https://en.wikipedia.org/wiki/Inoculation_loop
- Wikipedia. (n.d.). Inoculation loop. https://en.wikipedia.org/wiki/Inoculation_loop#:~:text=An%20early%20report%20of%20the,volume%20of%20urine%20for%20analysis.
- Wikipedia. (2025, September 18). Inoculation needle. https://en.wikipedia.org/wiki/Inoculation_needle
- Wikipedia. (2026, May 14). Petri dish. https://en.wikipedia.org/wiki/Petri_dish
- Wikipedia. (2026, May 5). Robert Koch. https://en.wikipedia.org/wiki/Robert_Koch
- Zhejiang SKG Medical Technology Co.,Ltd. (2025, December 31). Inoculation loop cleaning & handling tips for better results. https://www.skgmed.com/Inoculation-loop-Cleaning-Handling-Tips-for-Better-Results-id41111375.html