Gas chromatography is a chromatographic technique which is used for separation and analysis of volatile substances. It is mainly used for the compounds which can be vaporized without decomposition.
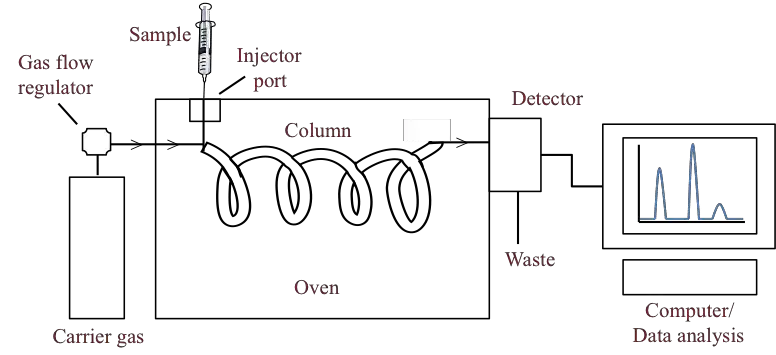
In this technique, the sample is injected into a heated chamber. The sample is converted into vapour and carried by an inert gas through a column. The column contains stationary phase.
The components of the mixture moves through the column at different speed. Some components are retained for longer time and some components pass quickly. Thus the components are separated and detected in the form of chromatogram.

Definition of Gas chromatography
Gas chromatography (GC) is an analytical technique used to separate, identify, and quantify volatile compounds in a mixture by passing them through a column with a stationary phase and a carrier gas.
Principle of Gas chromatography
Principle of Gas Chromatography is based on the distribution of vaporized sample between two phases, mobile phase and stationary phase. The mobile phase is an inert carrier gas and stationary phase is present inside the column.
The sample is first injected into a heated inlet where it is vaporized. This vaporized sample is then carried by the carrier gas through the column. During this movement, the components of the sample interact with the stationary phase in different degree.
The compounds which have more affinity towards stationary phase are retained for longer time and move slowly. The compounds having less affinity moves faster and comes out first from the column. This difference in movement causes separation of the mixture into different components.
The separated components comes out from the column one by one and detected by detector. Thus a chromatogram is formed which is used for identification and quantitative estimation of the compounds.
How does gas chromatography work? (Mechanism of Separation in Gas Chromatography)
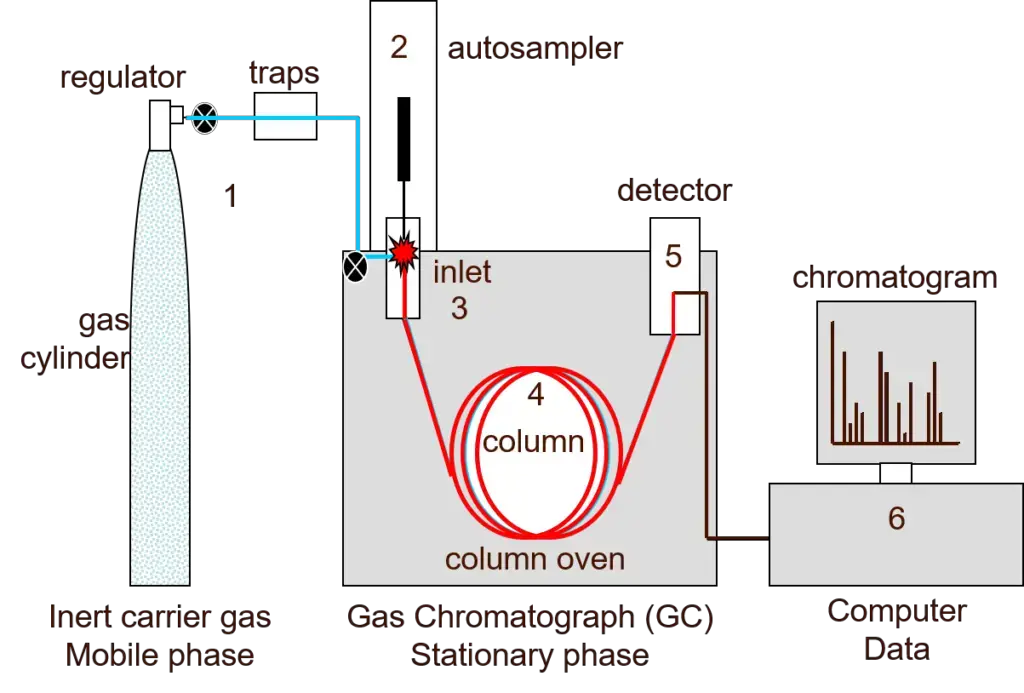
Gas chromatography works by separating the volatile compounds on the basis of their volatility and interaction with stationary phase. The sample may be liquid or gas, which is injected into a heated injection port.
In this step, the sample is immediately converted into vapour. The inert carrier gas such as helium, nitrogen or hydrogen carries the vaporized sample into the column. This carrier gas acts as the mobile phase.
The column contains stationary phase. It may be a thin liquid film or solid coating present on the inner wall or packing material of the column. When the vapour mixture passes through the column, the different components interacts with stationary phase in different degree.
The compounds having low boiling point and weak interaction with stationary phase moves faster. The compounds having high boiling point or more affinity towards stationary phase are retained for longer time and moves slowly.
Due to this differential movement, the components of the mixture are separated into different bands. This is referred to as differential migration. Each component comes out from the column at different time, which is called retention time.
After separation, the compounds passes through the detector one by one. The detector gives electrical signal to the recorder or computer. Thus a chromatogram is formed.
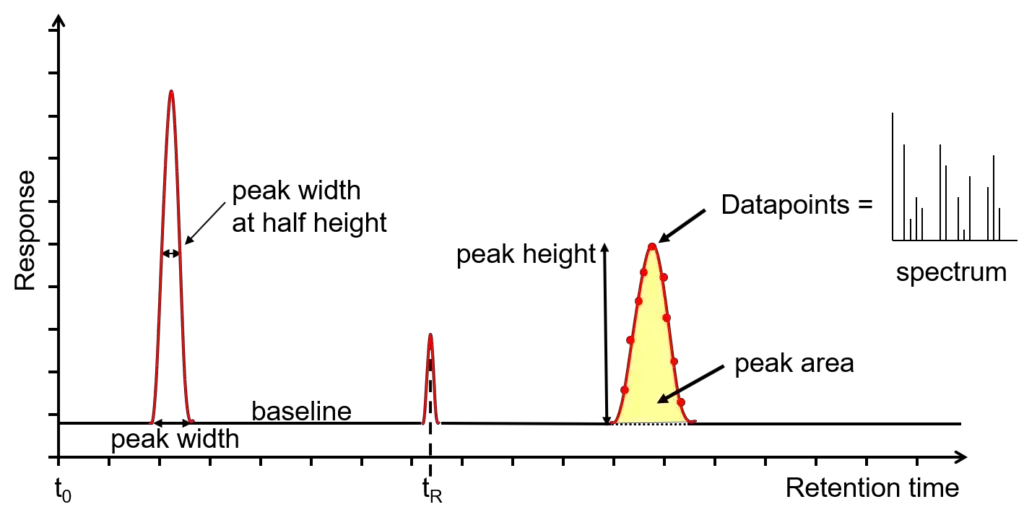
In chromatogram, each compound is shown as a separate peak. The retention time is used to identify the compound and the peak area is used to determine the amount of compound present in the sample.
Instruments/Parts of Gas Chromatography
The following are the important instruments or parts of gas chromatography-
- Carrier gas supply- It supplies inert carrier gas such as helium, nitrogen or hydrogen. It acts as mobile phase and carries the sample through the column.
- High pressure cylinder- It stores the carrier gas under high pressure.
- Pressure regulator and flow meter- It controls the pressure and flow rate of carrier gas.
- Sample injector- It is used to introduce the sample into the carrier gas stream. The sample is vaporized in the heated injection port.
- Heated metal block- It heats and vaporizes the liquid sample before entering into the column.
- Chromatographic column- It is the main part of gas chromatography where separation of components takes place.
- Stationary phase- It is present inside the column. It may be solid adsorbent or liquid coating and helps in separation.
- Liquid phase- It is used as coating material in the column. It is selected according to the nature of sample components.
- Support material- It gives surface for holding the stationary phase.
- Column oven- It maintains the required temperature of the column for proper separation.
- Detector- It detects the separated components as they comes out from the column.
- Temperature control system- It controls the temperature of injector, column and detector.
- Flow rate control system- It maintains constant flow of carrier gas through the system.
- Recorder and data processing system- It records the signal from detector and forms chromatogram.
Stationary Phase in Gas Chromatography System
Stationary phase is the fixed phase present inside the column of gas chromatography. It helps in separation of different components of the sample.
It may be solid material or liquid film coated on a solid support. The sample components interact with stationary phase in different degree and separation takes place.
The following are the types of stationary phase in gas chromatography-
- Gas-Solid Chromatography (GSC)- In this type, the stationary phase is a solid material packed in the column. The sample molecules are adsorbed on the surface of solid phase.
- Adsorption- The compounds are separated due to different adsorption capacity on the solid stationary phase.
- Common solid materials- Porous polymers, silica gel, molecular sieves and activated charcoal are used as solid stationary phase.
- Gas-Liquid Chromatography (GLC)- In this type, the stationary phase is a liquid film coated on a solid support material. The sample molecules distribute between liquid stationary phase and carrier gas.
- Partitioning- The compounds are separated due to their different partition between liquid phase and mobile gas phase.
- Common liquid phases- Polyethylene glycol (PEG), polydimethylsiloxane (PDMS) and cyanopropylphenyl are used as liquid stationary phase.
Selection of stationary phase- It depends on nature of the sample compounds. Polar stationary phase is used for polar compounds and non-polar stationary phase is used for non-polar compounds.
- Volatility- The stationary phase should have high boiling point. It should not evaporate during analysis.
- Molecular size- The stationary phase should be selected according to the size of the analyte molecules.
- Separation efficiency- A good stationary phase gives proper separation and clear peaks in chromatogram.
Procedure or Steps of Gas Chromatography (GC)
The following are the steps of gas chromatography-
- Sample preparation- The sample is first prepared before analysis. It may be extracted, purified, diluted or derivatized to make it suitable for gas chromatography.
- Sample injection- A small amount of prepared sample is injected into the heated injection port by syringe or autosampler.
- Vaporization- In this step, the sample is immediately converted into vapour form due to high temperature.
- Separation- The vaporized sample is carried by inert carrier gas through the column. The components interact with stationary phase in different degree and move at different speed.
- Differential movement- The volatile compounds and weakly retained compounds moves faster. The less volatile or strongly retained compounds moves slowly through the column.
- Detection- The separated components comes out from the column one by one and enters into the detector.
- Signal production- The detector detects each component and produces electrical signal.
- Data analysis- The signal is processed by computer and chromatogram is formed.
- Chromatogram reading- Each component gives separate peak in chromatogram. Retention time is used for identification and peak area is used for quantitative estimation.
Types of Gas Chromatography
The following are the types of gas chromatography-
- Gas-liquid chromatography (GLC)- In this type, the stationary phase is a liquid coated on a solid support or on the inner wall of the column. It is the most common type of gas chromatography. In GLC, separation takes place due to partition of compounds between carrier gas and liquid stationary phase.
- Gas-solid chromatography (GSC)- In this type, the stationary phase is a solid adsorbent material. The separation occurs due to adsorption of compounds on the solid surface. It is mainly used for separation of low molecular weight compounds and trace gases.
- Comprehensive two-dimensional gas chromatography (GC×GC)- It is an advanced type of gas chromatography. In this method, two columns with different stationary phases are used. The two columns are connected by a modulator. It gives better separation of very complex mixtures.
- Fast gas chromatography (Fast GC)- It is a modified type of gas chromatography which is used to reduce the analysis time. In this method, short and narrow columns are used with rapid temperature change. It can complete the analysis in very short time.
Factors Affecting Gas Chromatography (GC)
The following are the factors affecting gas chromatography-
- Sample properties- The boiling point, polarity and molecular size of sample components affect the separation. These properties decide the retention time of the compounds.
- Boiling point- Compounds having low boiling point comes out faster from the column. Compounds having high boiling point are retained for longer time.
- Polarity- Polar compounds interact more with polar stationary phase. Non-polar compounds interact more with non-polar stationary phase.
- Molecular size- Large molecules moves slowly through the column. Small molecules usually moves faster.
- Column temperature- Temperature of column oven affects the vapour pressure and movement of analytes. High temperature decreases retention time.
- Excessive temperature- Very high temperature may reduce separation efficiency. It may also cause poor peak shape.
- Column length- Longer column gives better separation. But it also increases analysis time.
- Column diameter- Narrow column gives better resolution. But it may increase pressure in the system.
- Stationary phase- Nature, polarity and thickness of stationary phase affects separation. It should be selected according to the nature of sample.
- Film thickness- Thick film increases retention of compounds. Thin film gives faster separation.
- Carrier gas- Type of carrier gas affects the speed and efficiency of separation. Helium, hydrogen and nitrogen are commonly used.
- Flow rate- Proper flow rate of carrier gas is necessary for good separation. Very high or very low flow rate may give poor resolution.
- Sample injection volume- The injected sample amount should be controlled. Large amount of sample may overload the column.
- Column overloading- It causes distorted peak and poor separation. Peak fronting may also occur due to overloading.
Applications of Gas Chromatography
The following are the applications of gas chromatography-
- Pharmaceuticals- It is used in drug development and testing of active pharmaceutical ingredients. It is also used to detect impurities, residual solvents and metabolites.
- Environmental analysis- It is used to detect pollutants in air, water and soil. Volatile organic compounds (VOCs), pesticides, PAHs and PCBs can be analysed by this method.
- Food and beverage- It is used for food safety testing. It helps to detect pesticides, contaminants, flavour compounds and aroma compounds in food products.
- Forensic science- It is used in toxicology screening and drug analysis. It is also used for blood alcohol estimation, arson investigation, explosive residue and trace evidence analysis.
- Petrochemical industry- It is used for analysis of crude oil, natural gas and fuel samples. It also helps to detect impurities and quality of fuels.
- Clinical diagnosis- It is used for detection of disease biomarkers in breath, blood or other body fluids. It is also used in newborn screening, drug monitoring and clinical toxicology.
- Material science- It is used to study polymers, plastics, paints and other materials. It helps to analyse chemical composition and stability of these materials.
- Consumer products- It is used for analysis of cosmetics, perfumes, fragrances and household cleaning products.
- Agriculture- It is used to detect pesticide residues in crops. It is also used for analysis of soil and fertilizer composition.
- Cannabis industry- It is used for potency testing, terpene profiling and detection of pesticides, mycotoxins or residual solvents.
- Research- It is used in scientific research, method development and metabolic studies.
Advantages of Gas Chromatography
The following are the advantages of gas chromatography-
- High sensitivity- It can detect compounds present in very low concentration. It can detect trace amount of compounds in ppm or ppb level.
- High resolution- It gives good separation of the components of complex mixtures.
- Fast analysis- The analysis is completed in short time. Many samples can be analysed within few minutes.
- Small sample requirement- It needs very small amount of sample for analysis.
- Versatility- It can be used for different volatile and semi-volatile compounds.
- Accuracy- It gives accurate and reliable result in quantitative analysis.
- Reproducibility- The same sample gives almost same result when the condition is maintained properly.
- Automation- Modern gas chromatography system can be operated automatically. It reduces manual work.
- Cost effective- The instrument is simple and reliable to operate. Some systems are also portable and can be used in field testing.
- Detector compatibility- It can be attached with advanced detectors such as mass spectrometer (GC-MS). This helps in proper identification of compounds.
Limitations of Gas Chromatography
The following are the limitations of gas chromatography-
- Limited to volatile compounds- It can analyse only those compounds which can be converted into vapour form.
- Thermal stability- The compound should not decompose at high temperature. Thermally unstable compounds cannot be analysed directly.
- Molecular weight limitation- It is mainly suitable for low and medium molecular weight compounds. Large molecules like proteins and polymers cannot be analysed directly.
- Sample preparation- Some samples need extraction, purification or derivatization before analysis. This makes the process time taking.
- Derivatization requirement- Non-volatile compounds are converted into volatile form by chemical derivatization.
- Identification problem- Gas chromatography mainly separates the compounds. For exact identification, it is often connected with mass spectrometer or other detector.
- Precise control needed- Temperature, carrier gas flow rate and pressure must be controlled properly. Small change may affect the result.
- Skilled operator- It needs trained person for handling, maintenance and interpretation of chromatogram.
- Safety problem- High pressure gas cylinders are used in this technique. Hydrogen gas may also cause fire or explosion if not handled properly.
- Coelution- In complex sample, two or more compounds may come out together and form single peak.
- Contamination- Carry over of previous sample or contamination of column may affect the chromatogram.
Troubleshooting and Safety Considerations of Gas Chromatography
The following are the common troubleshooting of gas chromatography-
- Peak tailing- In this condition, the peak becomes stretched at the end side. It may occur due to active sites, damaged column end or contaminated inlet.
- Solution- Trim the front end of column, replace inlet liner and use clean deactivated parts.
- Peak fronting- In this condition, the peak becomes sharp at front side like shark fin. It generally occurs due to overloading of sample in the column.
- Solution- Decrease the injection volume, dilute the sample or increase the split ratio.
- Ghost peak or carry over- These are extra peaks which are not from the present sample. It may occur due to contamination from previous run, septum bleed or sample backflash.
- Solution- Run blank injection, clean the injector and replace the septum.
- Baseline drift- In this condition, the baseline slowly rises or changes. It may occur due to column bleed, high temperature or contaminated carrier gas.
- Solution- Check the gas purity, replace gas filter and clean or bake the column.
- Baseline noise or spiking- It is caused by detector contamination, air leak or electronic disturbance.
- Solution- Check the system for leak, clean the detector and check electrical connection.
- Missing peaks- In this condition, no peak or some peaks are not seen in chromatogram. It may occur due to blocked syringe, broken column, detector flame off or absence of carrier gas.
- Solution- Check syringe, gas pressure, column connection and detector condition.
- Retention time shift- In this condition, the peaks come out at different time than normal. It may occur due to gas leak, change in carrier gas flow or column contamination.
- Solution- Check carrier gas flow rate, leak in the system and column oven temperature.
- Condensation test- The GC is kept at 40-50°C for long time and blank run is done. It is used to check contamination in carrier gas or inlet line.
- Jumper tube test- In this test, one part is bypassed to find the source of problem. It helps to know whether problem is from injector, column or detector.
The following are the safety considerations of gas chromatography-
- Hydrogen gas hazard- Hydrogen is used as carrier gas but it is highly flammable. It may cause fire or explosion if leakage occurs.
- Safety measure- Proper ventilation, hydrogen sensor and pressure monitor should be used. On-site hydrogen generator is safer than high pressure cylinder.
- High pressure cylinder- Carrier gases are stored in high pressure cylinders. These cylinders should be fixed properly and handled carefully.
- Toxic chemicals- Some samples, solvents and derivatization reagents are toxic or corrosive.
- Safety measure- Gloves, lab coat, goggles and other PPE should be used. Chemicals should be handled according to MSDS.
- Exhaust and venting- Carrier gas and vapours come out from the GC system. Proper exhaust and ventilation system should be present in laboratory.
- Thermal hazard- Injection port, column oven and detector remain at high temperature.
- Safety measure- The heated parts should not be touched directly. Care should be taken during maintenance and column changing.
References
- My Green Lab. (2026). 2026 Predictions for Sustainable Science. [URL]
- Harvey, D. (2023). 27.3: Gas Chromatographic Columns and Stationary Phases. Chemistry LibreTexts. [URL]
- Buchi. (n.d.). As Easy as ABC: How to Use the Van Deemter Equation to Optimize Your Chromatography. [URL]
- Romanello, D. (2025). Basic Principles of Gas Chromatography. Phenomenex. [URL]
- Basis of Interactions in Gas Chromatography – Part 2: Polar Interactions. (n.d.). [URL]
- Romanello, D. (2025). Capillary column vs. packed column in GC. Phenomenex. [URL]
- uHPLCs. (2023). Chromatographic quantitative internal or external standard method, how to choose_. [URL]
- Larsen, D. (2024). Chromatography. Chemistry LibreTexts. [URL]
- Premnath, S. M., & Zubair, M. (2024). Chromatography. StatPearls – NCBI Bookshelf. [URL]
- SCION Instruments. (n.d.). Chromatography In Forensic Science | Gas & HPLC. [URL]
- Chromatography Industry Trends 2025: AI, Automation, and Workforce Challenges. (n.d.). [URL]
- Thermo Fisher Scientific. (n.d.). Chromatography Troubleshooting Guides-Gas Chromatography. [URL]
- Romanello, D. (2025). Comprehensive 2D (GC×GC) explained. Phenomenex. [URL]
- Comprehensive Analytical Framework of Gas Chromatography: Theory, Instrumentation, and Multi-Industry Applications. (n.d.). [Markdown]
- Shimadzu. (n.d.). Detector. [URL]
- ERROR: The request could not be satisfied. (n.d.). [URL]
- Phenomenex. (n.d.). Essential Components of GC Instruments. [URL]
- Stein, S. E., Babushok, V. I., Brown, R. L., & Linstrom, P. J. (2007). Estimation of Kováts Retention Indices Using Group Contributions. Journal of Chemical Information and Modeling, 47(3), 975-980. ACS Publications. [URL]
- Phenomenex. (n.d.). Exploring the Various Types of Detectors Used in Gas Chromatography. [URL]
- From Detector to Decision, Part III: Fundamentals of Calibration in Gas Chromatography. (n.d.). [URL]
- Usha, Y. (2023). Fundamental Principles and Diverse Applications of Gas Chromatography in Analytical Sciences. Pharmaceutical Analytical Chemistry: Open Access, 8(220). Longdom Publishing. [PDF]
- Poole, C. F. (2005). GAS CHROMATOGRAPHY/Detectors. Elsevier Ltd. [PDF]
- Shimadzu. (n.d.). GC Column Types & Selection Guide. [URL]
- Agilent. (n.d.). GC Troubleshooting Guide Poster. [PDF]
- Gerstel. (n.d.). GCxGC vs. GC-GC: What Good is Separation if You Can’t Smell the Difference? [URL]
- National Institute of Standards and Technology. (n.d.). Gas Chromatographic Retention Data. The NIST WebBook. [URL]
- Biocompare. (n.d.). Gas Chromatography (GC). [URL]
- Gilman, J., & Jimenez, J.-L. (2004). Gas Chromatography. CIRES. [PDF]
- LibreTexts. (2023). Gas Chromatography. Chemistry LibreTexts. [URL]
- Kutztown University. (n.d.). Gas Chromatography. [URL]
- Agilent. (n.d.). Gas Chromatography Detectors. [PDF]
- PerkinElmer. (2022). Gas Chromatography Explained: What It Is and How It Works. PerkinElmer Blog. [URL]
- InsightAce Analytic. (n.d.). Gas Chromatography Market Size and Scope Deep Study 2026. [URL]
- Organomation. (n.d.). Gas Chromatography Sample Preparation. [URL]
- Lovestead, T. M. (n.d.). Gas Chromatography. National Institute of Standards and Technology. [PDF]
- Tamang, S. (2026). Gas Chromatography: Principle, Parts, Steps, Procedure, Uses. Microbe Notes. [URL]
- Tentamus Group. (n.d.). Gas chromatography (GC) explained in simple terms. [URL]
- Wikipedia. (2025). Gas chromatography. [URL]
- Green Chromatography: Must Analysts Sacrifice Performance for Sustainability? (n.d.). [URL]
- Restek Corporation. (2021). Guide to GC Column Selection and Optimizing Separations. Restek Resource Hub. [URL]
- Persee. (2025). How Do Different Detectors Work in a Gas Chromatograph? [URL]
- Wallace, R. F., & Sidisky, L. M. (n.d.). Hydrogen: A Carrier Gas Alternative to Helium. Sigma-Aldrich. [URL]
- Alfa Chemistry. (n.d.). Internal Standard vs. External Standard Methods in Chromatographic Quantification: A Comprehensive Protocol Guide. [URL]
- Romanello, D. (2025). Key Applications of Gas Chromatography. Phenomenex. [URL]
- Ford, K., & James, M. (n.d.). The Theory and Practice of Fast GC. MAC-MOD Analytical. [PDF]
- Wikipedia. (2025). Kovats retention index. [URL]
- LCGC International. (n.d.). LC Troubleshooting Essentials: A Guide to Common Problems and Solutions for Peak Tailing, Ghost Peaks, and Pressure Spikes. [URL]
- Lecture 5: Gas Chromatography Injection. (n.d.). [PDF]
- Lecture 8: Gas Chromatography. (n.d.). [PDF]
- Hetman, I. (2025). Life cycle assessment of hydrogen and helium as carrier gases in gas chromatography analysis. Green Chemistry, 28, 839-851. RSC Publishing. [URL]
- La Salle University. (n.d.). MODULE 4. CHROMATOGRAPHY EXPERIMENT 4A. TEMPERATURE PROGRAMMING AND van DEEMTER CURVES IN GAS CHROMATOGRAPHY. [PDF]
- Welch Materials. (2025). Mastering Gas Chromatography in 7 Comprehensive Steps! [URL]
- Analytical Separation Science. (2025). McReynolds’ constants for characterizing stationary phases. [URL]
- Separation Science. (n.d.). Navigating Split and Splitless Injections with Crowdsourced Insights. [URL]
- Research and Markets. (n.d.). North America Gas Chromatography Market Forecast Report. GlobeNewswire. [URL]
- Ucci, A. (2025). Peak Perfection: A Guide to GC Troubleshooting. Agilent. [PDF]
- Cytiva. (n.d.). Peak tailing and peak fronting in chromatography. [URL]
- Phenomenex. (n.d.). Phenomenex technical tip for Baseline problems – offset. LabRulez GCMS. [URL]
- Choudhary, A. (2025). Principle and Working of Gas Chromatography | GC Principle Explained. Pharmaguideline. [URL]
- Persee. (2025). Real-Life Applications of Gas Chromatography: How It’s Used in Various Industries. [URL]
- Sigma-Aldrich. (2002). Retention index mixture for GC (R8769) – Bulletin. [PDF]
- Separation Science. (n.d.). Rethinking Carrier Gases in GC and GC–MS: Moving Beyond Helium. [URL]
- Restek. (n.d.). Split vs Splitless Injection. Restek Resource Hub. [URL]
- Romanello, D. (2025). Split vs. Splitless Injection in Gas Chromatography (GC). Phenomenex. [URL]
- CLU-IN. (2018). Technologies > Characterization and Monitoring > About Characterization and Monitoring Technologies > Gas Chromatography. [URL]
- SCION Instruments. (n.d.). The Different Types of GC Detectors. [URL]
- CHROMacademy. (n.d.). The Theory of HPLC Chromatographic Parameters. Inacom Instruments. [PDF]
- Darji, P. (2025). Top 10 Industries That Rely on GC-MS Analysis: Pharma, Environmental, Food, Forensics & More. ResolveMass. [URL]
- Sensue, A. (n.d.). Troubleshooting GC Column Baseline Issues. Restek Resource Hub. [URL]
- Romanello, D. (2025). Types of stationary phases in gas chromatography. Phenomenex. [URL]
- Cavagnino, D. (2023). Using Hydrogen as a Carrier Gas With GC and GC-MS: Be Safe and Efficient. Thermo Fisher Scientific. [URL]
- Element Lab Solutions. (n.d.). Van Deemter equation – The Lockdown guide. [URL]
- Wikipedia. (2026). Van Deemter equation. [URL]
- Sepsolve Analytical. (n.d.). What is GCxGC? [URL]
- Teledyne Labs. (n.d.). What is Gas Chromatography? [URL]
- PerkinElmer. (n.d.). What is Peak Fronting? [URL]
- AMETEK MOCON. (n.d.). What is Process Gas Chromatography? [URL]
- Chromatography Today. (n.d.). What is the Van Deemter Equation? [URL]
- Aijiren Technology. (2025). Why Choose Internal Standard Over External Standard in Chromatographic Quantification? [URL]
- Agilent. (2007). simpler, more reliable GC x GC. [PDF]
- Air Products. (n.d.). tell me more. [PDF]
- Boag, M. (2025). van Deemter Equation For Chromatography. Phenomenex. [URL]