Fluorescence microscope is an advanced optical microscope that uses fluorescence for viewing the specimen.
It produces a bright coloured image of the specimen in a dark background. It is mostly used for biological and chemical samples where a specific part of the sample is to be seen clearly.
In this microscope, fluorescent dyes, proteins or labels are used. These fluorescent substances are attached with the particular structure of the specimen.
The specimen is exposed to light of short wavelength. This light is called excitation light.
The fluorescent molecule absorbs this light and becomes excited. After this, it emits light of longer wavelength.
This emitted light is used for the formation of image. The difference between the excitation light and emitted light is referred to as Stokes shift.
The microscope has special filters for controlling the light. The excitation filter allows only the required wavelength of light to pass.
The dichroic mirror reflects the excitation light towards the specimen. It also allows the emitted fluorescence light to pass towards the eyepiece or detector.
The emission filter blocks the remaining excitation light. So only the fluorescence light forms the image.
The image formed is bright and specific. The background remains dark, so the labelled part of the specimen can be seen easily.
Fluorescence microscope is used in cell biology, medical diagnosis, molecular biology and research works.
It is used to study proteins, DNA, living cells and single molecules. Different fluorescent labels can also be used to observe different structures in the same specimen.
Principle of Fluorescence Microscope – How does fluorescence microscopy work?
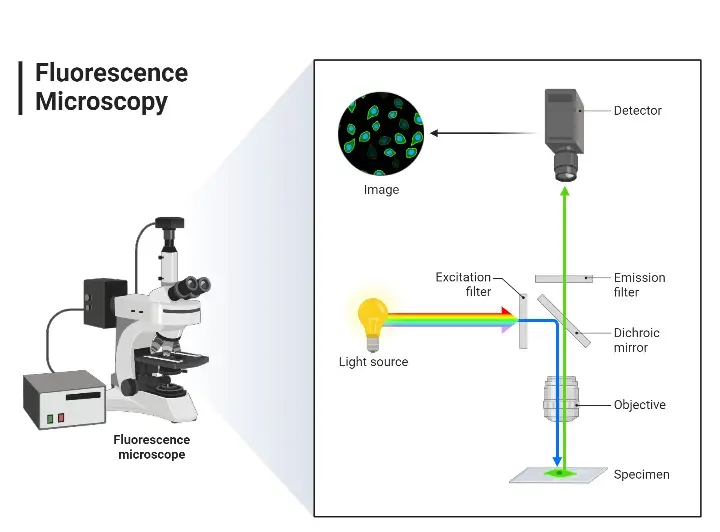
Principle of Fluorescence Microscope is based on fluorescence produced by fluorescent molecules after absorbing light of specific wavelength.
The specimen is first treated with fluorescent dye or fluorophore. These dyes are attached with the required part of the specimen.
The light is obtained from LED, laser or arc lamp. This light is passed through excitation filter.
The excitation filter allows only the required short wavelength light to pass. This light then falls on the dichroic mirror.
The dichroic mirror reflects the short wavelength light towards the specimen. The fluorophore present in the specimen absorbs this light and becomes excited.
After excitation, the fluorophore emits light of longer wavelength. This emitted light has lower energy than the excitation light.
This difference between absorbed light and emitted light is called Stokes shift.
The emitted fluorescent light returns through the objective lens. It passes through the dichroic mirror because it has longer wavelength.
Then the light passes through emission filter or barrier filter. This filter removes the remaining excitation light.
Only the fluorescent light reaches the eyepiece or detector. Thus a bright fluorescent image is formed on a dark background.
Types of Fluorescence Microscopes
Different types of fluorescence microscopes are as follows-
- Widefield fluorescence microscope- It is a type of fluorescence microscope in which the whole specimen is illuminated at a time. It is simple and fast. It is used for quick observation of fluorescent specimen. Out of focus light is also collected, so the image is not very sharp in thick sample.
- Confocal fluorescence microscope- It is a fluorescence microscope that uses pinhole aperture. The pinhole removes the out of focus light. It produces clear and sharp image of the specimen. It is used for optical sectioning and 3D image formation.
- Multiphoton fluorescence microscope- It is based on the absorption of two or more low energy photons by the fluorophore. Near infrared light is used in this microscope. It can pass deep inside the tissue. It produces less damage to the living specimen.
- Total internal reflection fluorescence microscope- It is a special type of fluorescence microscope which uses evanescent wave. It illuminates only the thin region near the cell surface. It is used for observing molecules present near the membrane surface.
- Super resolution microscope- It is used to observe very small structures below the normal resolving power of light microscope. It gives nanoscale image. It is also known as nanoscopy. STED, SIM and SMLM are important types of this microscope.
- Stimulated emission depletion microscope– It uses a depletion laser along with excitation laser. The depletion laser reduces the fluorescent area. So a very small emission spot is formed. It helps to see fine details of the specimen.
- Structured illumination microscope- It uses patterned light for illumination of specimen. The image is then processed by computer. It gives better resolution than ordinary fluorescence microscope.
- Single molecule localization microscope- It is based on detection of blinking of single fluorophore. PALM and STORM are examples of this type. It is used to locate single molecules in the specimen.
- Fluorescence lifetime imaging microscope- It forms image according to the lifetime of fluorescence. It does not depend only on intensity of light. It is used to study the surrounding condition of molecule.
- FRET microscope– It is based on transfer of energy from donor fluorophore to acceptor fluorophore. The two molecules should be very close to each other. It is used to study molecular interaction and change in molecular shape.
- FRAP microscope- In this method, fluorescence of selected area is destroyed by strong light. After this, the recovery of fluorescence is observed. It is used to study movement and diffusion of molecules.
- Fluorescence correlation spectroscopy- It studies the fluctuation of fluorescence intensity in a very small volume. It is used to know molecular movement, concentration and binding reaction.
- Stereo fluorescence microscope- It is used for larger specimen and whole organism. It uses separate path for fluorescence illumination. It is useful for three dimensional observation.
- Single molecule fluorescence microscope- It detects fluorescence from single fluorophore. It is used to study the behaviour of molecule at single molecule level.
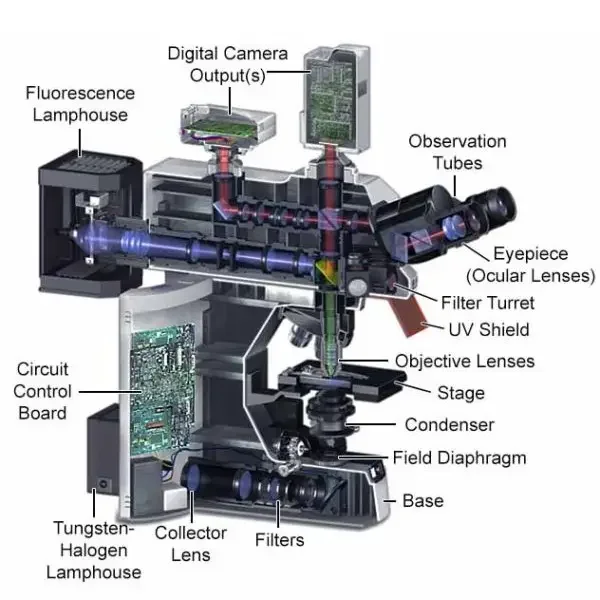
Parts of Fluorescence Microscope
The important parts of fluorescence microscope are as follows-
- Light source- It is used to produce high intensity light for excitation of the specimen. LED, laser, mercury vapour lamp and xenon arc lamp are commonly used as light source.
- Filter cube- It is a special part which holds the optical filters. It separates the excitation light and emitted fluorescence light. It is also called filter turret.
- Excitation filter- It allows only the required short wavelength light to pass. This light is used to excite the fluorophore present in the specimen.
- Dichroic mirror- It is placed at 45 degree angle. It reflects the short wavelength excitation light towards the specimen. It allows the longer wavelength fluorescence light to pass upward.
- Emission filter– It is also called barrier filter. It blocks the remaining excitation light. It allows only the emitted fluorescence light to reach the detector or eyepiece.
- Objective lens- It focuses the excitation light on the specimen. It also collects the fluorescence light coming from the specimen. So it works both as condenser and light collecting lens.
- Aperture diaphragm– It controls the amount of light entering the microscope. It helps to reduce extra light and increase the contrast of image.
- Field diaphragm– It controls the size of illuminated field. It helps to illuminate only the required area of specimen.
- Detector– It receives the weak fluorescent light and forms the image. Camera system, CCD camera, EMCCD, sCMOS camera or photomultiplier tube may be used as detector.
- Eyepiece– It is used to observe the fluorescent image by eye. In modern microscope, camera and monitor are also used for viewing the image.
Sample Preparation for Fluorescence Microscope
The common methods and steps for sample preparation are as follows-
- Direct staining with fluorescent dyes- In this method, the specimen is stained directly with fluorescent dye. Most of the cell molecules do not show fluorescence naturally. So fluorophore or fluorochrome is used for marking the cell part.
- DNA staining- DAPI and Hoechst dyes are used for staining DNA. These dyes bind with the nucleus region and give fluorescence. So the nuclear material can be observed clearly.
- Organelle staining- Specific fluorescent dyes are used for staining cell organelles. Lipophilic dyes are used for endoplasmic reticulum and mitochondria. These dyes help to observe the position and structure of organelles.
- Immunostaining- It is a staining method which uses antibody linked with fluorescent dye. The antibody binds with specific protein present in the specimen. It is used to detect particular protein inside the cell.
- Fixation before immunostaining- In immunostaining, the antibody should enter inside the cell. For this, cells are generally fixed before staining. Fixation makes the cell stable and preserves cellular structure.
- Fluorescent protein tagging- In this method, the cell is genetically changed to produce fluorescent protein. Green Fluorescent Protein (GFP) is commonly used. The fluorescent protein is attached with the protein of interest.
- Use in living cells- Fluorescent protein tagging is useful for living cells and tissues. It is used to observe movement and dynamic process inside the cell.
- Cell fixation- Fixed samples are prepared by using chemicals such as methanol or glutaraldehyde. These chemicals preserve the cell structure. The fixative should be selected carefully because some fixative may damage the target structure or reduce fluorescence.
- Mounting of sample- The stained sample is mounted on good quality glass slide or coverslip. Coverslip of proper thickness is used for better optical resolution.
- Use of mounting medium- Mounting medium is used to keep the sample in proper condition. The refractive index of mounting medium, coverslip and immersion oil should be matched. This reduces scattering of light and gives clear image.
- Minimizing background noise- During preparation, autofluorescence should be avoided. Phenol red, fluorescent plastic and high background antifade medium may produce unwanted fluorescence. This decreases the clarity of the image.
- Final observation- After staining and mounting, the sample is observed under fluorescence microscope. The labelled part emits fluorescence and forms bright image on dark background.
Light Path In Fluorescence Microscopy

The light path in fluorescence microscopy is as follows-
- Light source- The light is first produced from high intensity light source. LED, laser or arc-discharge lamp is used as light source.
- Excitation filter- The light then passes through the excitation filter. This filter allows only the required short wavelength light to pass.
- Dichroic mirror- The filtered excitation light falls on the dichroic mirror. The dichroic mirror is placed at about 45 degree angle.
- Reflection of excitation light- The dichroic mirror reflects the short wavelength light downward. This reflected light is directed towards the specimen.
- Objective lens- The excitation light passes through the objective lens. The objective lens focuses the light on the specimen.
- Excitation of fluorophore- The fluorophores present in the specimen absorb this excitation light. After absorption, the fluorophores become excited.
- Fluorescence emission- The excited fluorophores emit light of longer wavelength. This emitted light is fluorescence light.
- Collection by objective lens- The emitted fluorescence light is collected again by the same objective lens. It travels upward from the specimen.
- Transmission through dichroic mirror- The fluorescence light passes through the dichroic mirror. It is not reflected because it has longer wavelength.
- Emission filter- The emitted light then passes through emission filter or barrier filter. This filter blocks the remaining scattered excitation light.
- Detection- Only the clean fluorescence light reaches the eyepiece or detector. CCD camera or photomultiplier tube may be used as detector.
- Image formation- The final image is formed by the fluorescence signal. The image appears bright on a dark background.
Operating procedure of fluorescence microscopy
The operating procedure of fluorescence microscopy is as follows-
- Preparation of specimen- The specimen is first prepared and stained with fluorescent dye or fluorescent protein. The dye should bind with the required part of the specimen.
- Mounting of specimen- The prepared specimen is placed on the glass slide. Coverslip is placed properly. Then the slide is kept on the microscope stage.
- Selection of filter block- The proper filter block is selected according to the fluorophore used. The filter block contains excitation filter, dichroic mirror and emission filter.
- Position of filter turret- The filter turret is rotated or moved to bring the required filter block in the light path. Wrong filter block does not give proper fluorescence.
- Focusing of objective lens- The objective lens is focused on the specimen. In epi-fluorescence microscope, the same objective lens focuses excitation light and also collects emitted fluorescence light.
- Adjustment of illumination- Aperture diaphragm and field diaphragm are adjusted. This gives uniform illumination on the specimen. It also reduces extra stray light.
- Opening of shutter- The light shutter is opened after focusing and adjustment. The excitation light then reaches the filter cube and falls on the specimen.
- Excitation of fluorophore- The fluorophore absorbs the excitation light. After this, it emits fluorescence light of longer wavelength.
- Observation of image- The fluorescent image is observed through eyepiece. The labelled part appears bright against dark background.
- Capture of image- The image can be captured by camera system. CCD, EMCCD or sCMOS camera may be used for recording the weak fluorescence signal.
- Closing of shutter- The shutter is closed immediately after observation or image capture. This prevents unnecessary exposure of the specimen.
- Prevention of photobleaching- Long exposure of excitation light destroys the fluorophore. This is called photobleaching. So exposure time should be kept less.
- Prevention of phototoxicity- In living cells, strong light may damage the cell. So light intensity and exposure time should be controlled properly.
Application of Fluorescence Microscope
The important applications of fluorescence microscope are as follows-
- Cellular biology- It is used to observe different parts of the cell such as nucleus, cytoskeleton and mitochondria. It is also used to study cell division, cell movement, endocytosis and apoptosis.
- Molecular biology- It is used to study location and expression of proteins inside the cell. Fluorescent tags are attached with molecules and their position can be observed.
- Medical diagnosis- It is used for detecting genetic abnormalities. Fluorescence in situ hybridization (FISH) is used for detecting abnormal genes and chromosomes.
- Pathology- It is used to identify chromosomal translocation in diseases like leukemia. It is also used for detecting bacteria, virus and fungi in clinical samples.
- Cancer research- It is used to identify tumour markers. Immunofluorescence method is used to observe response of cancer cells after treatment.
- Surgical guidance- Fluorescent signal helps to identify tumour margin during surgery. It helps in removing malignant tissue more accurately.
- Dermatology- It is used to study skin abnormality at cellular level. It helps in detection of melanoma and non-melanoma skin cancers.
- Ophthalmology- Fluorescent dyes are injected to observe blood flow in retina. It is useful for detecting vascular problems and macular degeneration.
- Cardiology- It is used to study blood flow and tissue perfusion. It can also help to observe heart function and condition of transplanted tissue.
- Drug discovery- It is used to study drug action, absorption and distribution in cells. It helps to know whether the drug is working or not.
- Toxicology- It is used to observe cell damage caused by toxic substances. The toxic effect can be seen by change in fluorescence signal.
- Material science- It is used to study polymer structure, crystal growth and microscopic defects in materials. It is also used for detecting organic contaminants.
- Semiconductor inspection- It is used to detect small defects and photoresist residue on semiconductor wafer. It helps in checking technical cleanliness.
- Battery manufacturing- It is used for quality checking in high performance battery manufacturing. Small contamination and defect can be detected.
- Molecular interaction study- It is used to study protein-protein and protein-DNA interaction. FRET and FLIM methods are used for studying interaction and structural change at nanometer level.
Advantages of Fluorescence Microscope
The advantages of fluorescence microscope are as follows-
- High sensitivity- It can detect very small amount of specific molecule in the specimen. Fluorescent dyes or proteins are used for this detection.
- High specificity- It can show only the labelled part of the specimen. The fluorophore binds with a particular molecule or structure.
- Multiple labelling- More than one fluorophore can be used in the same sample. Different fluorophores emit different colours. So many targets can be observed separately in one specimen.
- Live cell observation- It can be used to observe living cells and tissues. The cellular processes can be seen without killing the cell in many cases.
- Dynamic imaging- It is used to observe movement and changes inside the cell. Cell division, protein movement and transport of molecules can be followed in real time.
- High resolution- It gives clear image of subcellular structures. Nucleus, mitochondria, cytoskeleton and other small parts can be observed.
- Super resolution- Some fluorescence microscopes can give image below the normal limit of light microscope. It is used to observe nanoscale structure.
- Quantitative analysis- The intensity of emitted fluorescence can be measured. It is used to compare amount of molecule, movement and interaction.
- Wide use- It is used in biology, medical diagnosis, pathology and research work. It is also used in material science and semiconductor inspection.
- Better contrast- The fluorescent part appears bright on dark background. So the target structure can be identified easily.
Limitations of Fluorescence Microscope
The limitations of fluorescence microscope are as follows-
- Photobleaching- In this, the fluorophore loses its fluorescence after continuous exposure to excitation light. The dye becomes fade and does not emit light properly. Due to this, long observation of the specimen is difficult.
- Phototoxicity- The strong light used in fluorescence microscope may damage the living cell. It can form toxic free radicals. So the normal activity of living specimen may be changed.
- Autofluorescence- Some tissues and cell materials show natural fluorescence. This produces background light. Due to this, the actual fluorescent signal becomes less clear.
- Non-specific staining- Sometimes the fluorescent dye binds with unwanted parts of the specimen. This gives false fluorescence. It reduces the specificity of the image.
- Less penetration in thick tissue- In thick specimen, light is scattered and absorbed. So the fluorescence from deep region becomes weak. Clear image is difficult to obtain from deeper part.
- Out of focus fluorescence- In widefield microscope, fluorescence from above and below the focus plane is also collected. This produces hazy background. It is more seen in thick samples.
- Spectral overlap- When many fluorescent dyes are used together, their excitation and emission wavelength may overlap. The signal of one dye may appear in another channel. This is called bleed-through.
- Only labelled structures are seen- Fluorescence microscope shows only those structures which are labelled with fluorophore. The remaining non-fluorescent parts are not observed.
- Artifact formation- Fixation, staining and mounting may produce artificial changes in the cell. These changes are called artifacts. They may give wrong interpretation.
- High cost and handling- Fluorescence microscope is costly instrument. Its light source, filters, camera and dyes are also costly. It also requires trained person for proper use.
Fluorescence Microscopy Images
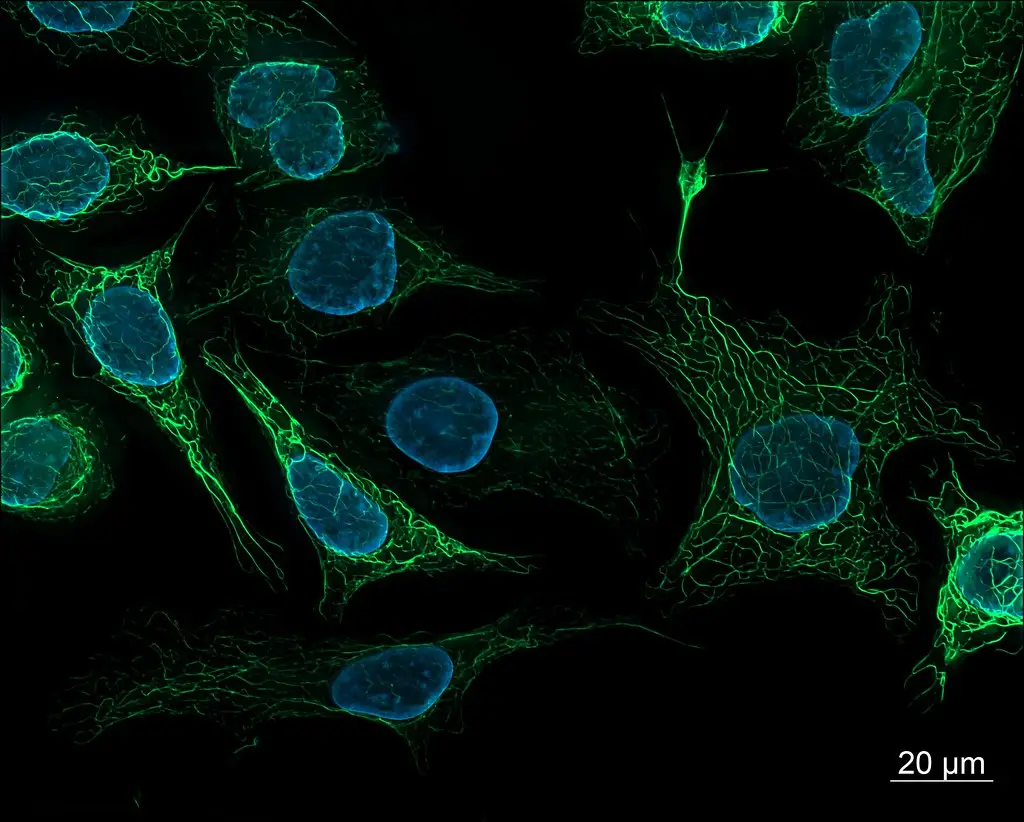

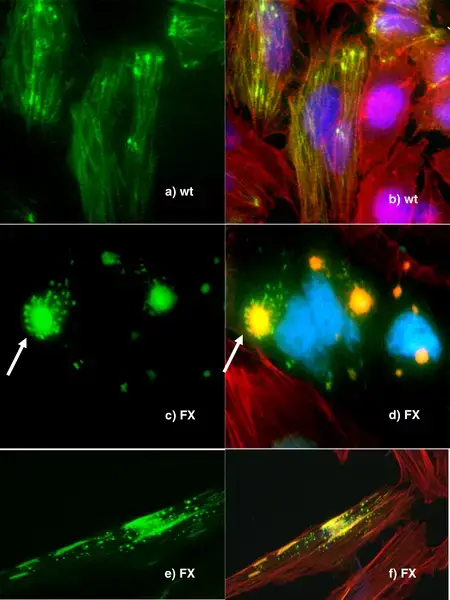
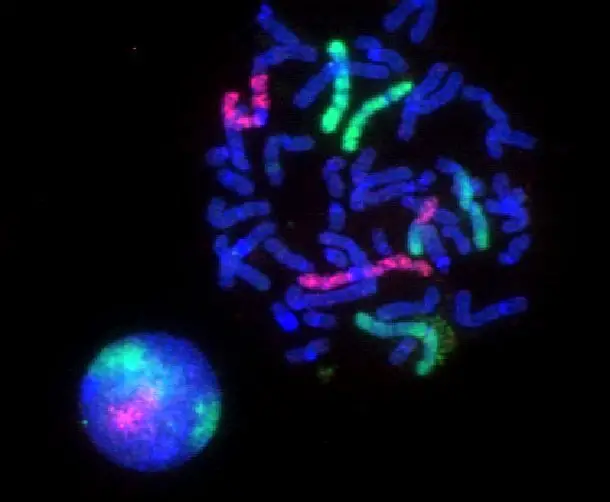
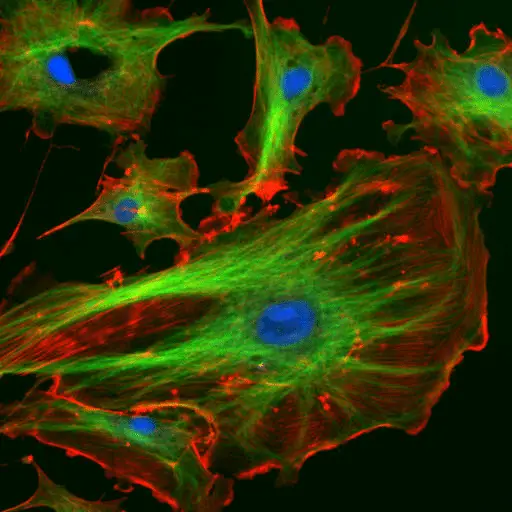
Confocal vs Fluorescence Microscopy
The differences between confocal and fluorescence microscopy are as follows-
- Illumination- In fluorescence microscopy, the whole specimen is illuminated at the same time. LED or arc lamp is generally used. In confocal microscopy, focused laser beam is used and it scans the specimen point by point.
- Light collection- Fluorescence microscopy collects light from the whole illuminated area. So out of focus light is also collected. Confocal microscopy collects light only from the focused plane.
- Pinhole- Fluorescence microscope does not have pinhole for removing out of focus light. Confocal microscope has a pinhole aperture. This pinhole blocks the unwanted light.
- Image clarity- Fluorescence microscopy may produce hazy image in thick specimen. Confocal microscopy gives sharp and clear image. The contrast is also high.
- Optical sectioning- Fluorescence microscopy does not give proper optical sectioning. Confocal microscopy gives thin optical sections of the specimen. These sections are used for 3D reconstruction.
- Imaging speed- Fluorescence microscopy is faster because it captures the whole field at one time. Confocal microscopy is slower because scanning is done point by point.
- Live cell imaging- Fluorescence microscopy is useful for observing fast changes in living cells. Confocal microscopy is also used for living cells but long scanning may affect the cell.
- Photobleaching- Fluorescence microscopy uses less intense light in many cases. So photobleaching is less. Confocal microscopy uses strong focused laser, so photobleaching may be more.
- Phototoxicity- Fluorescence microscopy is comparatively gentle for living specimen. Confocal microscopy may cause more cell damage due to intense laser light.
- Cost- Fluorescence microscope is simpler and less costly. Confocal microscope is costly instrument. It needs laser, pinhole system and sensitive detector.
- Handling- Fluorescence microscopy is easier to use and maintain. Confocal microscopy needs more careful operation and trained person.
- Sample thickness- Fluorescence microscopy is better for thin and flat samples. It is mostly suitable for sample below about 15 µm. Confocal microscopy is better for thick samples about 50-100 µm.
- Best use- Fluorescence microscopy is used for quick observation of bright fluorescent specimen. Confocal microscopy is used when sharp image, less background and 3D study is needed.
The differences between confocal and fluorescence microscopy are as follows-
| Characters | Fluorescence microscopy | Confocal microscopy |
|---|---|---|
| Illumination | The whole specimen is illuminated at the same time. LED or arc lamp is generally used. | Focused laser beam is used. It scans the specimen point by point. |
| Light collection | It collects light from the whole illuminated area. Out of focus light is also collected. | It collects light mainly from the focused plane. |
| Pinhole | Pinhole is absent. So out of focus light is not removed properly. | Pinhole aperture is present. It blocks the out of focus light. |
| Image clarity | Image may become hazy in thick specimen. | Image is sharp and clear. Contrast is also high. |
| Optical sectioning | Proper optical sectioning is not obtained. | Thin optical sections are obtained. These sections are used for 3D reconstruction. |
| Imaging speed | It is faster because whole field is captured at one time. | It is slower because scanning is done point by point. |
| Live cell imaging | It is useful for fast changes in living cells. | It is also used for living cells but long scanning may affect the cell. |
| Photobleaching | Photobleaching is less in many cases because light intensity is low. | Photobleaching may be more because strong focused laser is used. |
| Phototoxicity | It is comparatively gentle for living specimen. | Cell damage may be more due to intense laser light. |
| Cost | It is simpler and less costly. | It is costly instrument. Laser, pinhole and sensitive detector are needed. |
| Handling | It is easier to use and maintain. | It needs careful operation and trained person. |
| Sample thickness | It is better for thin and flat samples, mostly below about 15 µm. | It is better for thick samples, about 50-100 µm. |
| Best use | It is used for quick observation of bright fluorescent specimen. | It is used when sharp image, less background and 3D study is needed. |
References
- Advanced fluorescence microscopy techniques—FRAP, FLIP, FLAP, FRET and FLIM. (n.d.). PubMed Central (PMC).
- Advances in fluorescence microscopy: Theoretical foundations, instrumentation architecture, and multidisciplinary applications. (n.d.).
- Applications of artificial intelligence, deep learning, and machine learning to support the analysis of microscopic images of cells and tissues. (n.d.). PubMed Central (PMC).
- Axiom Optics. (n.d.). Main differences between confocal and widefield microscopy.
- Abramowitz, M., Herman, B., Murphy, D. B., & Davidson, M. W. (n.d.). Anatomy of the fluorescence microscope. Evident Scientific.
- Abramowitz, M., Herman, B., Murphy, D. B., & Davidson, M. W. (n.d.). Comparing confocal and widefield fluorescence microscopy. Evident Scientific.
- Bhattiprolu, S. (2024, November 4). AI in microscopy: Deep learning for image analysis. ZEISS.
- BOC Sciences. (n.d.). Fluorescent probes and fluorescent dyes.
- Bruker. (n.d.). Super-resolution microscopy FAQ.
- BS2010: Bioimaging. (n.d.). 6.2 FRAP, FLIP, FRET, and FLIM. Bookdown.
- Corporate Communications, Leica Microsystems. (2026, March 9). A guide to fluorescence microscopy. Leica Microsystems.
- Field, R. (n.d.). Fluorescence imaging in medical devices – applications and examples. StarFish Medical.
- Fluorescence microscopy. (n.d.). PubMed Central (PMC).
- Fluorescence microscopy: A concise guide to current imaging methods. (n.d.). PubMed Central (PMC).
- Göhler, A. (2015, March). F techniques: FRAP, FLIP, FRET, FLIM, FCS*.
- Grand View Research. (n.d.). U.S. fluorescence microscopy market (2026 – 2033).
- Greb, C., DeRose, J., Kemnitzer, N., & Zilles, A. (2026, March 16). Overview of fluorescent dyes in terms of applications and properties. Leica Microsystems.
- HenriquesLab. (n.d.). CARE.
- Highly fluorescent semiconducting polymer dots for biology and medicine. (n.d.). PubMed Central (PMC).
- Inverted PAINT for material‐specific super‐resolution … (n.d.). PubMed Central (PMC).
- Kappel, C., Kuschel, L., & DeRose, J. (2026, April 21). What is FRET with FLIM or as it is usually known FLIM-FRET? Leica Microsystems.
- Kiderlen, S. (2026, March 24). Super-resolution microscopy. ibidi GmbH.
- Label-free fluorescence microscopy: Revisiting the opportunities with autofluorescent molecules and harmonic generations as biosensors and biomarkers for quantitative biology. (n.d.). PubMed Central (PMC).
- Leica Microsystems. (n.d.). Material science & analysis.
- LICORbio. (2025, April 1). Types of fluorescence imaging.
- Lundsten, K. (2026, March 24). Fluorophores 101: Types, brightness, and how to choose the right dye. FluoroFinder.
- Marine Biological Laboratory. (n.d.). AI@MBL: Machine learning for microscopy image analysis.
- Microscope World. (2024, January 31). Fluorescence microscopy.
- Microscopy Australia. (n.d.). The light path and microscope parts – LFM. MyScope.
- Murphy, D. B., Piston, D. W., Shand, S. H., Watkins, S. C., & Davidson, M. W. (n.d.). Spectral bleed-through artifacts in confocal microscopy. Evident Scientific.
- Nikon’s MicroscopyU. (n.d.). Basics of FRET microscopy.
- Nikon’s MicroscopyU. (n.d.). Introduction to fluorescence microscopy.
- Ockenga, W., & DeRose, J. (2023, June 2). An introduction to fluorescence. Leica Microsystems.
- Omega Optical. (2020, October 12). How microscopy filters work.
- Proteintech Group. (n.d.). IF imaging: Widefield versus confocal microscopy.
- Seeing beyond the limit: A guide to choosing the right super-resolution microscopy technique. (n.d.). PubMed Central (PMC).
- Simultaneous label-free autofluorescence multi-harmonic microscopy. (n.d.). PubMed Central (PMC).
- Simultaneous label-free autofluorescence and multi-harmonic imaging reveals in vivo structural and metabolic changes in murine skin. (n.d.). PubMed Central (PMC).
- Synthetic fluorophores for visualizing biomolecules in living systems. (n.d.). PubMed Central (PMC).
- TOPTICA Photonics. (n.d.). Label free & clinical imaging (Raman, CARS).
- TOPTICA Photonics. (n.d.). Super resolution microscopy (STED, SIM, PALM, STORM).
- University of Warwick. (2026, January 21). Choosing a microscope.
- Vasilev, C. (2022, March 29). Analyzing polymers with microscopy: Principles and applications. AZoOptics.
- VivaScope GmbH. (n.d.). Fluorescence microscope: Unlocking cellular insights.
- Wikipedia contributors. (2025, December 11). Stokes shift. Wikipedia.
- Wikipedia contributors. (2026, April 14). Super-resolution microscopy. Wikipedia.
- ZEISS. (n.d.). Microscopy insights hub. (Interactive Tutorials | Confocal/Widefield Microscopy Comparison).
- ZEISS. (n.d.). Microscopy insights hub. (Introduction to Fluorescent Proteins).