Chromatography is an analytical technique which is used to separate the components of a mixture.
It is used in laboratory for separation, identification and also estimation of substances present in a complex mixture. The method was first developed by Russian botanist Mikhail Tsvet for separation of plant pigments.
In chromatography, there are two phases. One is mobile phase and other is stationary phase. Mobile phase may be liquid, gas or supercritical fluid. Stationary phase is fixed material which may be solid, gel or liquid supported on a solid surface.
The mixture is dissolved in the mobile phase. Then the mobile phase carries the mixture through the stationary phase. During this process, the components of mixture show different affinity towards the two phases.
The substances which have more attraction towards stationary phase move slowly. They remain for more time in the system. The substances which have more attraction towards mobile phase move faster.
Due to this different movement, the components are separated from each other. They form separate bands. These separated components can be detected, identified and measured.
Thus chromatography is based on differential movement of components between mobile phase and stationary phase. It is an important method for analysis of complex mixtures.
Chromatography Definition
Chromatography is a laboratory technique used to separate, identify, and purify the components of a mixture based on their differential affinities for a stationary phase and a mobile phase.
Principle of Chromatography – How does chromatography work
Principle of Chromatography is based on the differential distribution of components of a mixture between two phases. One is mobile phase and another is stationary phase.
The sample mixture is first dissolved in the mobile phase. Mobile phase may be gas, liquid or supercritical fluid. It carries the mixture through the stationary phase.
Stationary phase is a fixed phase. It may be solid, gel or liquid supported on solid surface. During this movement, the components interact with stationary phase in different degree.
The components which have more affinity towards stationary phase move slowly. They are retained for longer time. The components which have more affinity towards mobile phase move faster.
Due to this difference in rate of movement, the components get separated from each other. They form separate bands or spots. These separated components can be isolated, identified and analysed.
Components/Parts of Chromatography
The following are the components and parts of chromatography-
- Stationary Phase- Stationary phase is the fixed phase used in chromatography. It may be solid, gel or liquid coated on a solid support. It interacts with the components of the sample and slows their movement in different way.
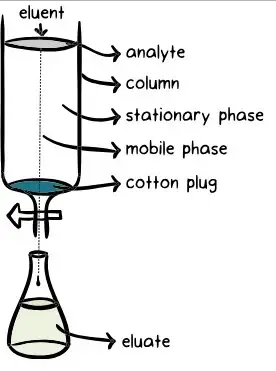
- Mobile Phase- Mobile phase is the moving phase of chromatography. It may be liquid, gas or supercritical fluid. It dissolves the sample and carries it through the stationary phase. It is also called eluent.
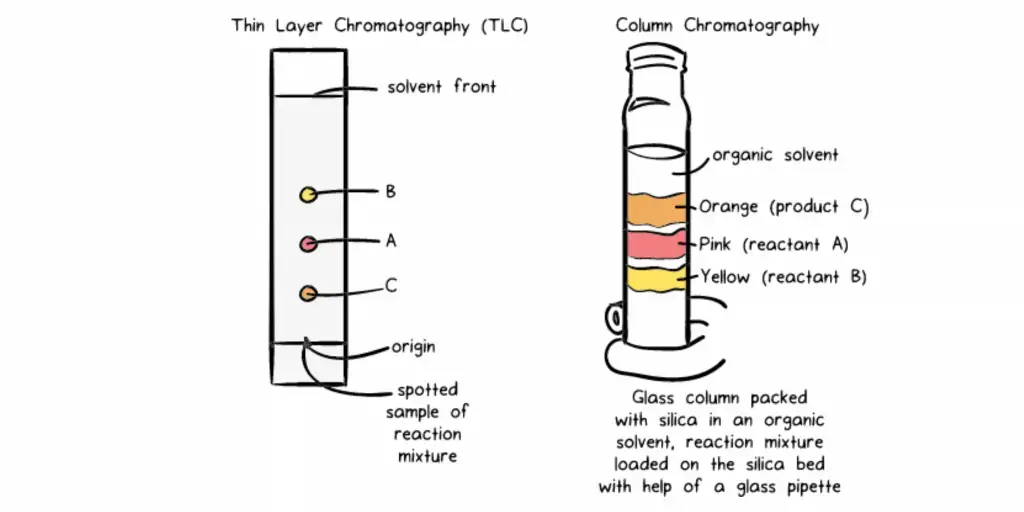
- Column or Planar Support- Column or planar support is the part which holds the stationary phase. In column chromatography, the stationary phase is present inside a tube. The tube may be made up of glass, stainless steel or fused silica. In paper chromatography and TLC, the support is a flat paper or plate.
- Sample or Analyte- Sample is the mixture which is introduced into the chromatography system. It contains the components which are to be separated. The substance to be detected or measured is called analyte.
- Pump- Pump is used to move the mobile phase through the system. It is mainly used in liquid chromatography and HPLC. It gives a constant flow of mobile phase. In HPLC, it also helps to pass mobile phase under high pressure.
- Injector- Injector is the part used to introduce the sample into the mobile phase. A fixed volume of sample is injected. In advanced instruments, autosampler is used for automatic injection of sample.
- Detector- Detector is placed at the end of column. It detects the separated components when they come out from the column. It also measures the amount of components. Common detectors are UV-visible detector, fluorescence detector, mass spectrometer and flame ionization detector.
- Data System- Data system is a computer and software unit attached with detector. It records the signal produced by detector. The signal is converted into chromatogram. The chromatogram shows separated peaks of different components.
- Chromatographic Chamber- Chromatographic chamber is used in paper chromatography and thin layer chromatography. It is a closed container. It contains mobile phase solvent at the bottom. The paper or plate is kept inside it and solvent moves upward by capillary action.
Types of Chromatography
The following are the types of chromatography-
A. On the basis of physical state or bed shape
- Liquid Chromatography (LC)- Liquid chromatography is a type of chromatography where liquid mobile phase is used. The stationary phase is packed in the column. The sample is moved through the column by the liquid phase. HPLC is a modified liquid chromatography, where mobile phase is passed with high pressure.
- Ultra-High-Performance Liquid Chromatography (UHPLC)- Ultra-High-Performance Liquid Chromatography is a developed form of HPLC. It uses higher pressure and very small particles. The separation occurs in less time and gives more clear separation.
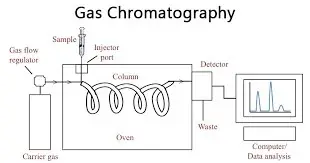
- Gas Chromatography (GC)- Gas chromatography is a type of chromatography where gas is used as mobile phase. The gas is generally inert. It is used for volatile and heat stable substances. The types are Gas-Liquid Chromatography (GLC) and Gas-Solid Chromatography (GSC).
- Supercritical Fluid Chromatography (SFC)- Supercritical Fluid Chromatography is a chromatography where supercritical fluid acts as mobile phase. The fluid remains above its critical temperature and pressure. Carbon dioxide is commonly used in this technique.
- Thin Layer Chromatography (TLC)- Thin Layer Chromatography is a planar chromatography. The stationary phase is spread as a thin layer over a plate. The plate may be made of glass, plastic or metal. Silica gel and alumina are used as adsorbent. The separated substances appear as spots.
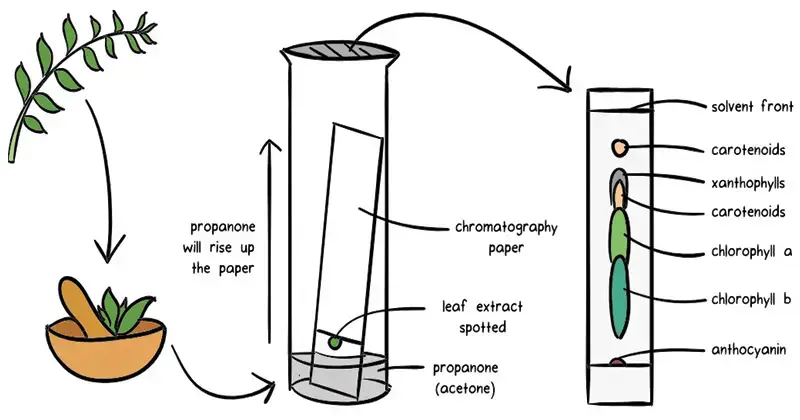
- Paper Chromatography (PC)- Paper chromatography is a simple chromatographic method. In this method cellulose paper is used as stationary phase. The solvent moves through the paper by capillary action. The substances move in different distance and become separated.
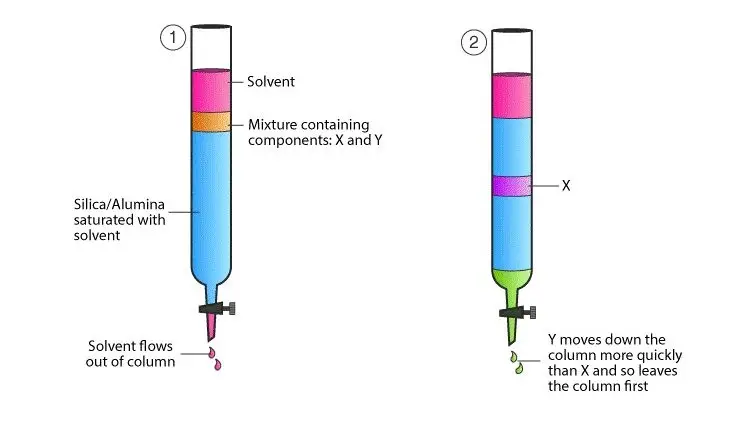
- Column Chromatography- Column chromatography is a type where stationary phase is filled in a column. The sample is placed at the upper part of the column. The mobile phase moves down through the column. The components are separated and collected as fractions.
B. On the basis of separation mechanism
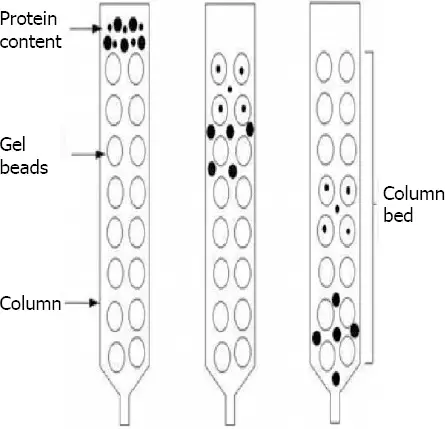
- Size-Exclusion Chromatography (SEC)- Size-exclusion chromatography is a type where molecules are separated according to size. The column contains porous beads. Larger molecules cannot enter the pores and comes out first. Smaller molecules enter the pores and comes out later. It is also called gel filtration or gel permeation chromatography.
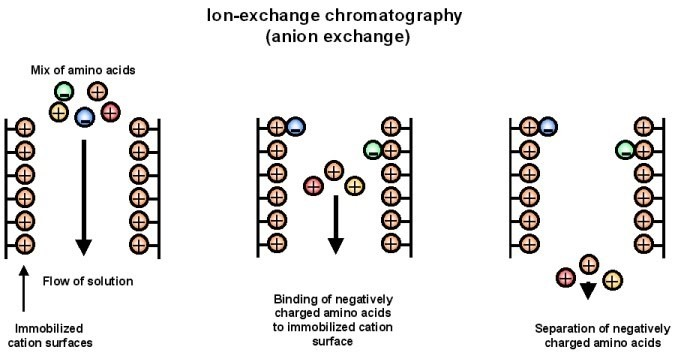
- Ion Exchange Chromatography (IEX)- Ion exchange chromatography is based on charge. The stationary phase contains charged group. Molecules of opposite charge bind with it. The bound molecules are removed by changing pH or salt concentration.
- Affinity Chromatography- Affinity chromatography is based on specific binding. A ligand is attached to the stationary phase. The specific molecule binds with the ligand and other molecules are washed out. It includes protein-ligand, antibody-antigen and enzyme-substrate binding.
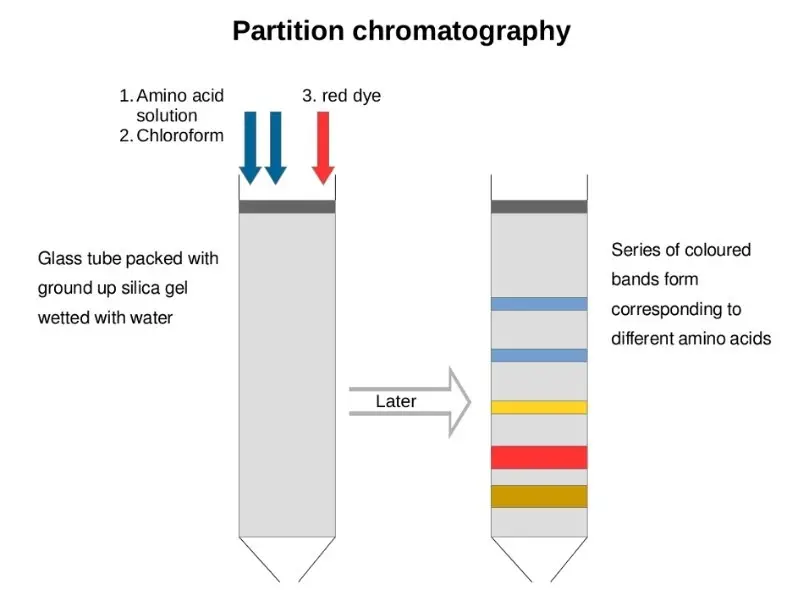
- Partition Chromatography- Partition chromatography is based on distribution of substances between two liquid phases. One liquid is stationary phase and another liquid is mobile phase. The components are separated due to their different solubility in these phases.
- Adsorption Chromatography- Adsorption chromatography is based on adsorption of substances on solid stationary phase. The substance which is adsorbed strongly moves slowly. The substance which is adsorbed weakly moves faster.
- Reversed-Phase Chromatography (RPC)- Reversed-phase chromatography is a type of liquid chromatography. In this method stationary phase is non-polar and mobile phase is polar. Non-polar substances are retained more and polar substances move faster.
- Normal-Phase Chromatography (NPLC)- Normal-phase chromatography is a traditional chromatography. In this method stationary phase is polar and mobile phase is non-polar. Polar substances are retained more strongly. Non-polar substances moves faster.
- Hydrophobic Interaction Chromatography (HIC)- Hydrophobic interaction chromatography is based on hydrophobic interaction. It is mostly used for proteins. High salt concentration helps the hydrophobic part of protein to bind with the matrix. The protein is removed by decreasing the salt concentration.
- Hydrophilic Interaction Chromatography (HILIC)- Hydrophilic interaction chromatography is used for polar substances. It has hydrophilic stationary phase and highly organic mobile phase. Polar analytes are retained due to hydrophilic interaction.
- Chiral Chromatography- Chiral chromatography is used for separation of chiral substances. It separates stereoisomers and enantiomers. A chiral stationary phase is used. Different enantiomers interact in different manner and moves differently.
- Displacement Chromatography- Displacement chromatography is a method where displacer molecule is used. The displacer has high affinity for stationary phase. It binds strongly and displaces other molecules with less affinity.
C. Specialized chromatographic techniques
- Two-Dimensional Chromatography- Two-dimensional chromatography uses two different separation systems. First separation is done in one direction. Then the second separation is carried out in another direction. It is used for complex mixture.
- Fast Protein Liquid Chromatography (FPLC)- Fast Protein Liquid Chromatography is a liquid chromatography used for purification of proteins. A buffer is used as mobile phase. The proteins are separated by size, charge or affinity.
- Pyrolysis Gas Chromatography- Pyrolysis gas chromatography is a special gas chromatography. The sample is first heated strongly. It breaks into smaller fragments. These fragments are separated by gas chromatography.
- Simulated Moving-Bed Chromatography (SMB)- Simulated Moving-Bed Chromatography is a continuous preparative chromatography. Many columns and valve system are used. It gives continuous separation by simulating a moving stationary bed.
- Hydrodynamic Chromatography (HDC)- Hydrodynamic chromatography separates particles according to size. Larger particles move faster in the column. Smaller particles move slowly. It is used for macromolecules, droplets and particles.
- Countercurrent Chromatography (CCC)- Countercurrent chromatography is a liquid-liquid chromatography. Both phases are liquid. The stationary liquid phase is held by centrifugal force. The substances are separated by partition between two liquid phases.
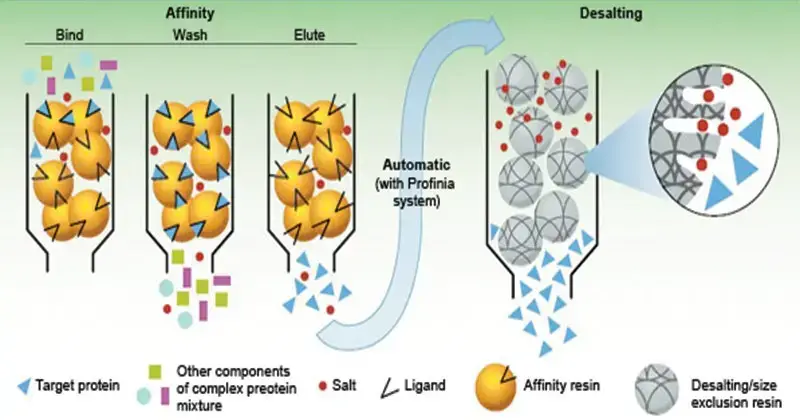
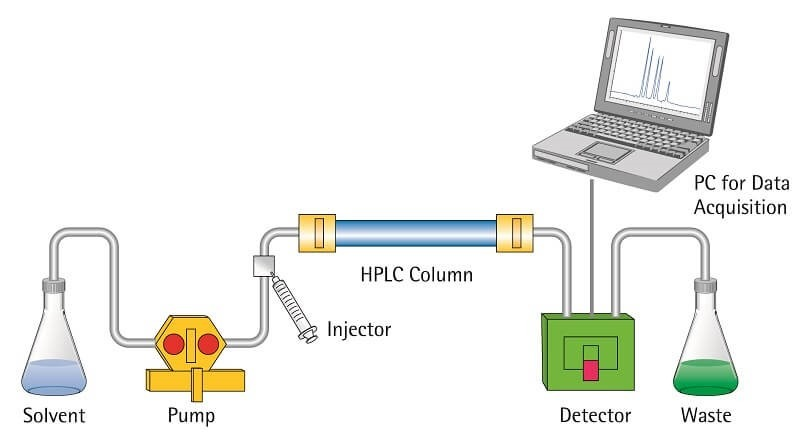
Factors Affecting Chromatography
The following are the factors affecting chromatography-
- Affinity and Phase Composition- The separation in chromatography depends on the affinity of the substance towards mobile phase and stationary phase. The nature, polarity and chemical composition of both phases are important. The substance which has more affinity towards stationary phase moves slowly. The substance having more affinity towards mobile phase moves faster.
- Particle Size- Particle size of stationary phase affects the separation. Small particle size gives more surface area. So the interaction between sample and stationary phase becomes more. It gives better separation and resolution. But very small particles need high pressure for the flow of mobile phase.
- Column Dimensions- The size of the column also affects chromatography. Longer column gives more chance for interaction of components with stationary phase. So separation becomes better. The length and width of the column should be proper for good result.
- Temperature- Temperature affects the movement of components in chromatography. Increase in temperature usually increases the rate of elution. In gas chromatography, it also affects volatility of the compounds. Very high temperature may cause tailing of peak. It may also damage heat sensitive compounds.
- Flow Rate- Flow rate is the speed of mobile phase moving through the system. Slow and uniform flow rate gives better separation. If the flow rate is very high, the components may not separate properly. It may cause band broadening and poor peak shape.
- Solvent Properties- The nature of solvent affects the separation. Solvent should have suitable polarity for the sample. Low viscosity solvent gives better flow and better resolution. In ionizable compounds, small change in pH of solvent can change the retention time.
- Diffusion Coefficient- Diffusion of molecules affects the width of bands. If diffusion is more, the band becomes broad. Longitudinal diffusion and mass transfer occur during movement of sample. These process affects the sharpness of peak and separation efficiency.
- Sample Overloading- Sample overloading occurs when too much sample is added into the system. The column cannot separate the components properly. The peaks become distorted. Fronting and poor resolution may occur due to overloading.
- Contaminants and Buffer Quality- Contamination also affects chromatography. Moisture, air bubbles, precipitated protein and bacterial growth may disturb the separation. Poor buffer quality can change retention time. It can also produce ghost peaks, smearing and irregular result.
Applications of Chromatography
The following are the applications of chromatography-
A. Pharmaceutical industry
- Quality Control (QC)- Chromatography is used in quality control of medicines. It is used to check the identity, purity and strength of active pharmaceutical ingredients (APIs). It is also used for excipients and finished drug products.
- Drug Development- Chromatography is used during development of new drugs. It is used to analyse chemical reactions and monitor the synthetic process. It also helps to study stability and solubility of new drug molecules.
- Impurity Profiling- Chromatography is used to detect impurities in drug formulation. It is used to measure impurities, degradation products and residual solvents. It also helps in removing unwanted substances from the drug.
- Chiral Separation- Chromatography is used for separation of chiral drugs. It separates different stereoisomers or enantiomers. This is important because one form may be active and safe, while another form may be harmful.
- Bioavailability Studies- Chromatography is used to measure drug concentration in biological sample. Blood, plasma or urine samples may be used. It helps to study pharmacokinetics and also support approval of generic drugs.
B. Forensic science and criminology
- Toxicology Screening- Chromatography is used to detect toxic substances in human body samples. It can detect drugs, poison and alcohol from blood, urine, sweat or hair. It is important in forensic toxicology.
- Arson Investigation- Chromatography is used in fire investigation. It helps to identify volatile hydrocarbon accelerants like petrol, gasoline and kerosene. These substances may remain in fire scene samples.
- Explosives Analysis- Chromatography is used for analysis of explosives. It separates and identifies chemical compounds present in bomb materials and post-blast debris. It helps in crime investigation.
- Forgery Detection- Chromatography is used to analyse inks and dyes. It is used in questioned documents and banknotes. The chemical composition of ink can help to detect fraud and forgery.
- Trace Evidence- Chromatography is used for small evidence found at crime scene. Paint, fibres, rubber and other small particles can be analysed. It helps to compare the evidence with suspected source.
C. Food and beverage industry
- Nutritional Profiling- Chromatography is used to measure nutrients in food. It can estimate amino acids, sugars and vitamins. It helps to check the nutritional label of food products.
- Contaminant Testing- Chromatography is used to detect harmful contaminants in food. It can detect pesticide residues, herbicides, heavy metals and food additives. This is important for food safety.
- Mycotoxin Detection- Chromatography is used to detect fungal toxins in food. Aflatoxin and other mycotoxins may be present in grains, nuts and stored food materials. These are harmful for health.
- Allergen Confirmation- Chromatography is used to confirm allergens in food. It helps to detect allergenic proteins like gluten and milk protein. It reduces false result in food testing.
- Flavor and Aroma Analysis- Chromatography is used to analyse flavour and aroma compounds. Volatile compounds, fatty acids and essential oils can be separated. It helps to maintain the quality of food and beverage.
- Shelf-Life Determination- Chromatography is used to study food spoilage. It detects chemical changes during storage. It helps to decide the expiry date and shelf life of the food product.
D. Clinical diagnostics and medicine
- Newborn Screening- Chromatography is used in newborn screening. Dried blood spot is used for detection of metabolic and genetic disorders. Phenylketonuria and other disorders can be detected early.
- Disease Diagnosis- Chromatography is used in diagnosis of different diseases. It is used to measure HbA1c in diabetes. It is also useful in thalassemia, porphyrias and some cancer related tests.
- Therapeutic Drug Monitoring- Chromatography is used to measure drug level in patient sample. It helps to adjust the dose of drug. It also helps to check drug safety and effectiveness.
- Biomolecule Separation- Chromatography is used for separation and purification of biomolecules. Proteins, hormones, viruses and nucleic acids can be isolated. It is used in biomedical research.
E. Environmental monitoring
- Water and Soil Quality- Chromatography is used to detect pollutants in water and soil. It can detect pesticides, non-volatile pollutants and PFAS like chemicals. It is used for drinking water, wastewater and soil testing.
- Air Pollution Testing- Chromatography is used for analysis of air pollutants. Volatile Organic Compounds (VOCs) and smog forming compounds can be measured. It helps in monitoring atmospheric pollution.
F. Chemical and petrochemical industries
- Petrochemical Analysis- Chromatography is used in petroleum industry. It separates complex hydrocarbon mixtures present in crude oil, refinery gas, fuel and lubricants. It helps to analyse the quality of petroleum products.
- Materials Science- Chromatography is used in analysis of polymers and industrial materials. It helps to know molecular weight, structure and degradation products. It is also used for industrial additives.
- Synthesis Monitoring- Chromatography is used to monitor chemical synthesis. It checks purity of raw materials and progress of organic reactions. It also helps to know the yield of product.
Advantages of Chromatography
The following are the advantages of chromatography-
- High Sensitivity- Chromatography is a sensitive technique. It can detect very small amount of substance present in the sample. Trace level compounds can also be separated and analysed by this method.
- High Specificity- Chromatography gives specific separation of components. The substances are separated according to their different affinity, polarity, size, charge or other property. So the required component can be identified from a mixture.
- Broad Versatility- Chromatography can be used for different types of samples. It can be used for gases, liquids and solids. Different chromatographic methods are present for different sample nature.
- Separation of Complex Mixtures- Chromatography is useful for complex mixture. It can separate many components present in one sample. Biological samples, food samples, drug samples and environmental samples can be analysed.
- Detector Compatibility- Chromatography can be joined with different detectors. UV detector, fluorescence detector, mass spectrometer and flame ionization detector can be used. This helps in detection, measurement and identification of separated components.
- Rapid Analysis- Modern chromatographic instruments give fast result. Automated system can run many samples in less time. So it is useful in routine laboratory analysis.
- Reproducible Result- Chromatography gives reproducible result when the condition is maintained properly. Same mobile phase, same flow rate and same column condition gives similar result. This is important in quality control work.
- Separation of Similar Compounds- Chromatography can separate closely related compounds. It is useful for compounds having similar physical and chemical property. Chiral chromatography can separate enantiomers which are optical isomers.
- Qualitative Analysis- Chromatography is used for identification of unknown substances. The retention time or spot position can be compared with standard. Thus the component present in the sample can be known.
- Quantitative Analysis- Chromatography is used to measure the amount of substance present in a mixture. The peak area or peak height is measured. This gives concentration of the component.
- Purity Checking- Chromatography is used to check purity of a sample. Pure sample generally gives single peak or single spot. Presence of extra peak or spot indicates impurity.
- Small Sample Requirement- Chromatography needs small amount of sample. Very little sample can be used for separation and analysis. This is useful when the sample is costly or available in low amount.
Limitations of Chromatography
The following are the limitations of chromatography-
- High Cost- Chromatography is costly technique. The instruments like HPLC and Gas Chromatography need high initial cost. The columns, pure solvents and maintenance also need more cost. Disposal of chemical waste also increases the running cost.
- Need of Skilled Person- Chromatographic instrument cannot be operated properly without trained person. The selection of mobile phase, stationary phase and flow condition needs proper knowledge. Method development and optimization is also difficult process.
- Limited Identification- Chromatography alone gives mainly retention time of the compound. It does not give complete structural information of the unknown substance. For proper identification, it is combined with other detector like mass spectrometer. NMR and IR gives more structural information than chromatography alone.
- Matrix Interference- Complex sample may contain many unwanted substances. These substances may come out with the target analyte at the same time. This is called co-elution. It affects the accurate measurement. So sample preparation and extraction is needed before analysis.
- Sensitivity to Operating Condition- Chromatography is affected by small change in condition. Change in pH, temperature, solvent composition and flow rate may change the retention time. In ionizable sample, small pH change can change the separation. Air bubble, moisture and bacterial contamination in buffer also affects the result.
- Column Contamination- Column may be contaminated by old sample, solvent or precipitated proteins. It gives ghost peaks and smearing in chromatogram. The column efficiency becomes low. So proper washing and storage of column is required.
- Peak Distortion- Too much sample loading causes peak fronting. Unwanted interaction between analyte and stationary phase causes peak tailing. These distortions make the peak shape poor. It also reduces the separation efficiency.
- Sample Limitation- Some chromatographic methods cannot be used for all type of samples. Gas chromatography needs vaporization of sample at high temperature. So it is not suitable for heat sensitive, highly polar and high molecular weight compounds.
- Time Consuming Sample Preparation- Many samples need extraction, filtration, dilution and purification before loading. Complex biological and environmental samples need more preparation. This increases the total analysis time.
- Solvent and Waste Problem- Many chromatographic methods use toxic organic solvents. Methanol, acetonitrile and other solvents may be harmful. They also produce hazardous waste. This creates safety problem and environmental problem.
- Limitation of Green Chromatography- Green chromatography uses eco-friendly solvents. But these solvents may give less selectivity and less sensitivity. They may not detect trace amount properly. Existing methods also need revalidation when green solvent is used.
Troubleshooting and Safety Considerations of Chromatography
The following are the troubleshooting and safety considerations of chromatography-
A. Troubleshooting considerations
- Contaminated Columns- Column may be contaminated by solvent, old sample or precipitated protein. It gives ghost peaks and smearing in the chromatogram. The previous sample may also remain in the column. So the column should be washed properly with suitable solvent which can remove the contaminant.
- Sample Overloading- Sample overloading occurs when excess amount of sample is injected. Due to this the components do not separate properly. It may give broad peak or fronting peak. In gas chromatography, this problem is more common. The column should be allowed to elute the sample completely before the next sample is injected.
- pH Sensitivity- The pH of mobile phase affects the retention time of ionizable compounds. Small change in pH may change the separation. Sometimes mobile phase absorbs carbon dioxide from air and becomes acidic. So the pH of buffer and mobile phase should be checked before use.
- Buffer and Mobile Phase Quality- Poor quality buffer and mobile phase affects chromatography. Air bubbles, moisture, precipitate, colour change and turbidity may disturb the run. Bacterial contamination in buffer also changes the result. The mobile phase should be clear, fresh and properly filtered before use.
- Sample Degradation- Sample may degrade due to wrong extraction, storage and handling. Light, heat and long storage time may also affect the sample. Due to this the composition of analyte changes. So the sample should be prepared freshly or stored under proper condition.
- Peak Tailing- Peak tailing means the peak becomes stretched at the end. It occurs due to unwanted interaction between analyte and stationary phase. It may also occur in complex sample matrix. This problem can be reduced by changing mobile phase composition or using proper stationary phase.
- Poor Resolution- Poor resolution occurs when two components are not separated clearly. It may be due to wrong solvent, high flow rate, poor column condition or excess sample. The mobile phase, flow rate and column should be adjusted for proper separation.
- Irregular Retention Time- Retention time may change due to change in flow rate, pH, temperature or mobile phase composition. Air bubble in the system may also cause this problem. The system should be equilibrated before sample run.
- Baseline Noise- Baseline noise may occur due to dirty detector cell, air bubbles, impure solvent or unstable flow. It makes detection difficult. The solvent should be filtered and degassed. The detector and flow system should be cleaned properly.
B. Safety considerations
- Hazardous Chemicals- Different hazardous chemicals are used in chromatography. Sodium hydroxide, ethanol, methanol, acetonitrile and tetrahydrofuran may be used. Some of them are toxic and some are skin irritant. Gloves, lab coat and safety glass should be used during handling.
- Material Safety Data Sheet (MSDS)- MSDS should be present for all chemicals used in the laboratory. It gives information about risk, exposure symptom, first aid and safe handling. The worker should read the MSDS before using chemical and instrument.
- Gas Cylinder Hazards- Gas cylinders are used in gas chromatography. These cylinders are under high pressure. They should be kept upright and properly fixed. Hydrogen gas needs more care because it is highly combustible and may cause explosion.
- Ventilation- Proper ventilation is needed in chromatography laboratory. Organic solvent vapour and carrier gases may come out during the process. These vapours should not be inhaled. Fume hood or exhaust system should be used where volatile solvents are handled.
- Biological Sample Handling- Blood, urine and other body fluids may be used in clinical chromatography. These samples may contain infectious agents. Aseptic technique should be followed. Gloves should be used and cross contamination should be avoided.
- Waste Disposal- Chromatography produces hazardous waste. Used solvent, sample vial, filter and column may contain chemical residue. These wastes should not be thrown with normal waste. They should be collected in proper waste container and disposed by safe method.
- Fire Safety- Many solvents used in chromatography are flammable. Methanol, ethanol, acetonitrile and other organic solvents can catch fire easily. They should be kept away from flame and heat source. Fire extinguisher should be present near the working area.
- Instrument Safety- The instrument should be operated according to instruction. High pressure system should not be opened during running condition. Leakage of solvent or gas should be checked. Electrical connections should be dry and safe.
References
- Allan Chemical Corporation. (n.d.). HPLC vs GC: Application differences. Allan Chemical Corporation.
- Bio-Rad. (n.d.). Types of chromatography. Bio-Rad.
- Chrom Tech, Inc. (2024). Understanding chromatography diagrams: Principles and types. Chrom Tech, Inc.
- Chromatography Today. (n.d.). HPLC vs GC – A beginner’s guide.
- Davey, R. (2021). Chromatography and forensics. News-Medical.Net.
- Eurofins. (n.d.). Nutrition analysis services – Food testing.
- Fluegemann, J. (n.d.). Solving crimes with chromatography.
- Forensic Science Simplified. (2013). Toxicology: How it’s done.
- Khan Academy. (n.d.). A closer look at types of chromatography (article). Khan Academy.
- Kromasil. (n.d.). Principles of chromatography. Kromasil.
- Lab Manager. (n.d.). Environmental analysis: Water, soil, and air testing.
- Lab Manager. (n.d.). Food safety testing: Analytical methods for contaminants and …
- Lab Manager. (n.d.). Forensic applications of analytical chemistry.
- LCGC International. (n.d.). Advancing green chromatography: Sustainable separation strategies for industrial and agricultural applications. LCGC International.
- Mahadevarao Premnath, S., & Zubair, M. (2024). Chromatography. In StatPearls. StatPearls Publishing.
- MetwareBio. (n.d.). Mastering chromatography: Everything you need to know. MetwareBio.
- Nair, V. S. (2025). Chromatographic techniques in pharmaceutical and chemical analysis: Principles, methods, and applications. Research & Reviews: Journal of Pharmaceutical Analysis, 14(4).
- NJ Labs. (2025). Essential applications of HPLC in the pharmaceutical industry. NJ Labs.
- Panda, S., & Gorantla, S. (2025). Green analytical approaches and eco-friendly solvents: Advancing industrial applications and environmental sustainability: A comprehensive review. Oriental Journal of Chemistry, 41(2).
- PatSnap Eureka. (2025). How to implement green HPLC techniques for reduced waste. PatSnap.
- Pharma Now. (n.d.). Chromatography in pharma: Techniques, case studies & trends. Pharma Now.
- PubMed Central. (n.d.). An overview of nutritional profiling in foods: Bioanalytical techniques and useful protocols.
- PubMed Central. (n.d.). Analytical methods for determining environmental contaminants of …
- PubMed Central. (n.d.). Chromatography – PubMed.
- PubMed Central. (n.d.). Different chromatographic techniques and recent advancements for …
- PubMed Central. (n.d.). Innovative applications and future perspectives of chromatography-mass spectrometry in drug research.
- PubMed Central. (n.d.). Separation techniques: Chromatography.
- Romanello, D. (2025). HPLC vs GC: What sets these methods apart. Phenomenex.
- Sep Science. (n.d.). Green chromatography: Reducing environmental impact in lab practices.
- Shelke, A., Yede, P., & Deokar, R. (n.d.). Chromatography: Types, basic principles and uses. International Journal of Scientific Research and Engineering Development (IJSRED), 257-264.
- Southwest Research Institute. (n.d.). Environmental testing. SwRI.
- Thermo Fisher Scientific. (n.d.). Nutritional & food label testing information. Thermo Fisher Scientific.
- Thomson Higher Education. (2007). Chapter 26: An introduction to chromatographic separations.
- Unknown Author. (n.d.). Comprehensive analysis of chromatographic principles, methodologies, and global industrial applications.
- U.S. Environmental Protection Agency. (2026). Methods for water research. US EPA.
- U.S. Environmental Protection Agency. (2025). The SW-846 compendium. US EPA.
- U.S. Food and Drug Administration. (2025). Chemical contaminants transparency tool.
- U.S. Food and Drug Administration. (n.d.). Elemental analysis manual (EAM) for food and related products.
- Waters Corporation. (n.d.). Amino acids testing | Amino acid profiling in food & beverages. Waters Corporation.
- Watrex. (2025). Green chromatography: How to make HPLC more eco-friendly. Watrex.
- Wikipedia. (2026). Chromatography. Wikipedia.