Beta (β) Lactamase Test is a rapid diagnostic test that is used to detect whether a particular bacterial strain produces beta-lactamase enzyme or not. It is used to identify resistance against beta-lactam antibiotics like penicillins and cephalosporins. This enzyme gives protection to bacteria by breaking open the beta-lactam ring of antibiotic and the drug is inactivated.
The principle of Beta (β) Lactamase Test is based on hydrolysis of beta-lactam ring by beta-lactamase. When the antibiotic ring is broken the breakdown products are formed and these products is detected by rapid biochemical indicators. This is referred to as beta-lactamase activity and it indicates that the organism can resist the action of beta-lactam antibiotics.
The test is performed by rapid methods. In chromogenic method (nitrocefin test) a chromogenic cephalosporin is used and presence of enzyme produces a distinct colour change from yellow to red. In acidimetric method a pH indicator is used and when antibiotic is degraded into penicilloic acid acidity is produced which changes the indicator colour. In iodometric method a starch–iodine complex is used and clearing of the dark blue complex is seen when enzyme is present.
This test gives results much faster than conventional susceptibility testing like disk diffusion. It provides early information for selecting targeted antimicrobial therapy and helps in choosing effective antibiotics for treatment.
Objectives of Beta (β) Lactamase Test
Objectives of Beta (β)-Lactamase Test are as follows-
- To detect the beta-lactamase enzyme produced by a bacterial organism using rapid methods like chromogenic acidimetric or iodometric test.
- To identify beta-lactamase mediated resistance in bacteria against beta-lactam antibiotics such as penicillins and cephalosporins.
- To provide rapid diagnostic results in comparison to traditional susceptibility tests like MIC or disk diffusion method.
- To guide appropriate clinical therapy by predicting resistance profile so that broad spectrum empirical antibiotics can be shifted to targeted and effective treatment.
Principle of Beta (β)-Lactamase Test
Principle of Beta (β)-Lactamase Test is based on detection of beta-lactamase enzyme produced by some bacteria. This enzyme gives resistance to beta-lactam antibiotics by cleaving the beta-lactam ring hence the antibiotic becomes inactive. The test works by observing this enzymatic hydrolysis by rapid biochemical methods which are chromogenic method acidimetric method and iodometric method.
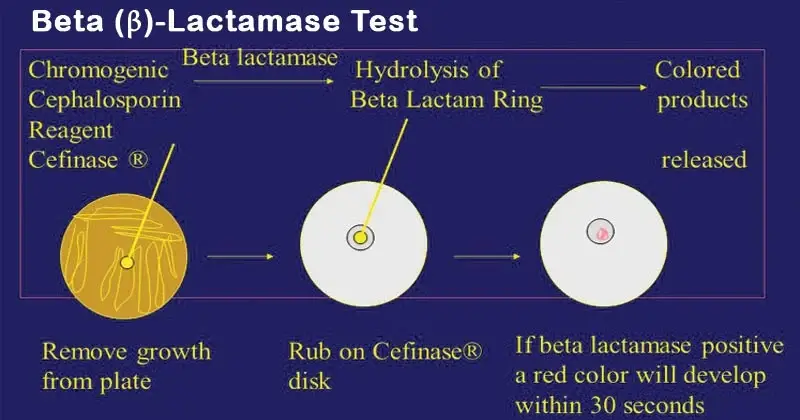
In chromogenic method (Nitrocefin test) a test disc is impregnated with nitrocefin which is a chromogenic cephalosporin substrate. If beta-lactamase is present it hydrolyzes the amide bond in beta-lactam ring of nitrocefin and a visible color change is produced from light yellow to red or deep pink. If the yellow colour remains unchanged it is considered as negative test and it indicates absence of the enzyme.
In acidimetric method penicillin-phenol red substrate is used which initially has alkaline pH with purple-pink or violet colour. When bacteria produces beta-lactamase penicillin is hydrolyzed and penicilloic acid is formed. This formation lowers the pH of the solution due to extra carboxyl group and phenol red indicator changes colour from violet or pink to yellow showing positive test.
In iodometric method starch–iodine complex is used. Iodine combines with starch to form dark blue or purple complex. When beta-lactamase hydrolyzes penicillin G penicilloic acid is released and it acts as a reducing agent. This acid reduces iodine and it prevents the formation of starch iodine complex and the dark blue colour fades to colorless or white indicating positive result.
Requirement for Beta (β) Lactamase Test
Requirement for Beta (β) Lactamase Test are as follows-
Chromogenic (Nitrocefin) test
- Nitrocefin disks (2-8°C).
- Sterile distilled water.
- Glass slide or empty petri dish.
- Sterile Pasteur pipettes.
- Sterile wooden sticks and inoculating loops.
- Test organism colonies grown overnight (18-24 hours) on non-selective media.
Acidimetric test (disk/strip method)
- Acidimetric disks or strips (2-8°C).
- Sterile distilled water.
- Glass slide or empty petri dish.
- Sterile Pasteur pipettes.
- Sterile wooden sticks and inoculating loops.
- Test organism colonies grown overnight (18-24 hours) on non-selective media.
Acidimetric test (tube method)
- 0.5% phenol red solution.
- Crystalline potassium penicillin G (20 million U).
- 1 N NaOH (Sodium Hydroxide) solution.
- Sterile 1-ml and 10-ml pipettes with a pipette bulb.
- Sterile polystyrene capped tubes (12×75 mm).
- Sterile wooden applicator sticks or inoculating loops.
Iodometric test
- Penicillin solution (6,000 µg/ml in pH 6.0 phosphate buffer).
- Starch reagent (1 g soluble starch in 100 ml heated distilled water).
- Iodine reagent (2.03 g iodine + 53.2 g potassium iodide in 100 ml distilled water).
- Empty sterile microdilution tray or small test tubes.
- Sterile 1-ml pipettes and a pipette bulb.
- Sterile wooden applicator sticks or inoculating loops.
- Test organism to create an opaque milky suspension.
Procedure of Beta (β)-Lactamase Test
Detail step by step Procedure of Beta (β)-Lactamase Test are as follows-
A. Chromogenic method (Nitrocefin disk test)
- Using sterile forceps required number of nitrocefin disks is placed on a clean microscope slide or in an empty petri dish.
- The disks are allowed to reach room temperature before inoculation.
- Each test disk is moistened with one drop of sterile distilled water.
- Several well isolated colonies of test organism is picked with sterile inoculating loop or wooden applicator stick and smeared directly on the moistened disk surface.
- Positive and negative control strains can also be tested at same time.
- The disk is observed for colour change from yellow to red or pink and positive result is usually seen within 15 seconds to 5 minutes.
- In some Staphylococcus species positive reaction may take up to 1 hour.
B. Acidimetric method (Tube test)
- Prepared penicillin-phenol red reagent tubes are taken from freezer and allowed to thaw at room temperature.
- Four or five colonies of bacteria is picked with sterile loop or applicator stick and mixed into the test solution to make an opaque milky suspension.
- The tube is observed for colour change and positive reaction is indicated when solution changes from violet/red to yellow.
- The positive reaction is usually seen in less than 15 minutes and colour change after 15 minutes is generally not considered positive.
- Some Staphylococcus strains may take up to 1 hour to show reaction.
C. Iodometric method
- 0.1 ml of prepared penicillin solution is dispensed into small test tube or a well of microdilution tray.
- Test organism is added to the solution and an opaque milky suspension is prepared.
- Two drops of starch solution is added and mixed and it is kept at room temperature (about 25°C) for 30 to 60 minutes.
- One drop of iodine reagent is added and the mixture is shaken or stirred for 1 minute.
- The mixture is observed and if dark blue colour rapidly fades to colorless (white) within 10 minutes it indicates positive reaction.
Result Interpretation of Beta Lactamase Test
Result Interpretation of Beta (β)-Lactamase Test are as follows-
Chromogenic method (Nitrocefin test)
- Positive result- Test disk or solution changes colour from yellow to red deep pink or orange-red. It indicates the organism produces beta-lactamase enzyme.
- Negative result- Test disk remains yellow and no colour change is seen.
Acidimetric method
- Positive result- Test disk or solution changes colour from violet/red to yellow. It indicates drop in pH due to formation of penicilloic acid.
- Negative result- Colour remains violet or red and it indicates no beta-lactamase activity.
Iodometric method
- Positive result- Dark blue or purple starch-iodine mixture fades to colorless or white. It occurs due to hydrolysis products reducing iodine.
- Negative result- Solution retains dark blue or purple colour.
Clinical interpretation notes
- Positive test predicts resistance to penicillinase-labile penicillins such as ampicillin amoxicillin and penicillin.
- Negative test does not completely rule out resistance because other mechanisms may be present like altered penicillin-binding proteins (example BLNAR strains in Haemophilus influenzae).
Organisms giving positive and negative result in Beta (β) Lactamase Test are as follows-
Positive result (QC strains)
- Staphylococcus aureus ATCC 29213.
- Staphylococcus aureus ATCC 43300.
- Haemophilus influenzae ATCC 33533.
Negative result (QC strains)
- Staphylococcus aureus ATCC 25923.
- Haemophilus influenzae ATCC 10211.
- Moraxella (Branhamella) catarrhalis ATCC 25240.
- Enterococcus faecalis (wild type).
Clinically tested organisms which can give positive result
- Neisseria gonorrhoeae.
- Haemophilus influenzae (beta-lactamase positive strains).
- Moraxella catarrhalis.
- Staphylococcus species.
- Enterococcus species.
- Anaerobes like Bacteroides Clostridium Porphyromonas Fusobacterium and Prevotella.
- Achromobacter species.
Organisms giving negative result or testing is not recommended
- Streptococcus pneumoniae and other Streptococci (including viridans streptococci).
- Haemophilus influenzae BLNAR strains (beta-lactamase negative ampicillin resistant).
- Enterobacteriaceae family (testing is not recommended).
- Pseudomonas species and other aerobic gram negative bacilli (testing is not recommended).
Uses of Beta Lactamase Test
Uses of Beta (β) Lactamase Test are as follows-
- It is used to detect beta-lactamase producing strains of clinically important bacteria like Neisseria gonorrhoeae Haemophilus influenzae Staphylococcus species Enterococcus species Moraxella catarrhalis and Achromobacter.
- It is used to predict specific resistance like N. gonorrhoeae resistance to penicillin H. influenzae resistance to ampicillin and Staphylococcus resistance to penicillin.
- It is used to provide rapid diagnostic susceptibility result in comparison to MIC or disk diffusion test hence early therapy change is possible.
- Nitrocefin method is referred to as most reliable method for detection of beta-lactamase in Enterococcus species.
- It is used in pharmaceutical sterility testing where beta-lactamase is added in culture media to neutralize beta-lactam antibiotics and false negative contamination result is prevented.
- It is used in blood culture where beta-lactamase is added in culture bottle for patients already on beta-lactam therapy so residual antibiotic is neutralized and pathogen growth is not inhibited.
Precautions of Beta (β) Lactamase Test
Precautions of Beta (β) Lactamase Test are as follows-
- All specimens and materials should be treated as potentially infectious and universal biohazard precautions is followed. Biohazard waste is sterilized before disposal.
- Test disks should be allowed to reach room temperature before inoculation so condensation does not interfere with reaction.
- The disk or applicator tip should not be over-saturated with water because excess moisture can dilute reagents and results may be affected.
- The disk should remain properly hydrated during the test. If it starts drying a small amount of water can be added for rehydration.
- Test disks should not be kept at room temperature for long time and unused disks should be returned to freezer immediately after use.
- Strains grown on blood agar should be tested carefully because weak or indistinct colour reaction may be produced.
- While testing Staphylococcus species reaction may take up to 1 hour and enzyme induction may be required by picking growth from margin of oxacillin or penicillin inhibition zone.
- Negative result does not completely rule out resistance because non-enzymatic resistance mechanisms can be present like altered penicillin-binding proteins.
- Rapid beta-lactamase tests should not be used for Enterobacteriaceae Pseudomonas species or other aerobic gram negative bacilli because results does not reliably predict therapeutic susceptibility.
- This test should not be used for organisms where penicillin resistance is not related to beta-lactamase production such as Streptococcus pneumoniae.
- Beta-lactamase test should not be used as complete replacement of conventional antimicrobial susceptibility testing methods.
Advantages of Beta (β) Lactamase Test
Advantages of Beta (β) Lactamase Test are as follows-
- It gives rapid turnaround time and critical susceptibility information is obtained much faster than disk diffusion or MIC methods.
- It is easy to perform and economical. The chromogenic acidimetric and iodometric methods are generally accurate reproducible easily readable and inexpensive for clinical laboratories.
- It helps in targeted clinical therapy because rapid result is provided and broad spectrum empirical therapy can be shifted to targeted effective antimicrobial regimen.
- A positive result reliably predicts resistance to penicillinase-labile penicillins such as amoxicillin ampicillin carbenicillin mezlocillin and piperacillin.
- Nitrocefin (chromogenic cephalosporin) test is highly sensitive because it can detect both penicillinase and cephalosporinase enzymes and it is efficient with less interference by other chemical groups.
- It provides immediate therapeutic guidance for fastidious organisms like Haemophilus influenzae and Neisseria gonorrhoeae.
- In pharmaceutical manufacturing beta-lactamase is used in sterility testing to neutralize antibiotics in environmental samples so true contaminants can grow and false negative sterility result is prevented.
Limitations of Beta (β) Lactamase Test
Limitations of Beta (β) Lactamase Test are as follows-
- It is a rapid screening test and it cannot replace conventional antimicrobial susceptibility testing methods completely.
- A negative result does not rule out resistance because other non-enzymatic mechanisms can be present like altered penicillin-binding proteins (PBPs) porin loss or efflux pumps.
- It is not recommended for Enterobacteriaceae family Pseudomonas species and other aerobic gram negative bacilli because it does not reliably predict clinical susceptibility to beta-lactam therapy.
- It is not useful for organisms where penicillin resistance is rarely due to beta-lactamase production such as Streptococcus pneumoniae and other Streptococci.
- In Staphylococcus species reaction may be delayed up to 1 hour and enzyme induction may be needed to produce detectable amount of enzyme (example growth from margin of oxacillin disk).
- Nitrocefin method has limitations because proper moisture is required. If disk dries colour will not develop and if disk is over-saturated reagent can be diluted. Strains grown on blood agar may give weak or indistinct colour reaction.
- Acidimetric method is limited because it is mainly applied for aerobic bacteria and it detects penicillinase only so cephalosporinase activity is not detected. It also cannot differentiate between acylase and beta-lactamase activity.
- Iodometric method also fails to detect cephalosporinase activity and fresh starch and iodine solutions are required frequently hence it is not convenient for routine laboratory use.
References
- AG Scientific. (n.d.). Beta-lactamase and microbial antibiotic resistance.
- Beta-Lactamases in laboratory and clinical resistance – PMC. (n.d.).
- Bidya, S., & Suman, R. (2014). Comparative study of three β lactamase test methods in Staphylococcus aureus isolated from two Nepalese hospitals. Open Journal of Clinical Diagnostics, 4(1), 47-52. https://doi.org/10.4236/ojcd.2014.41009
- Boehle, K. E., Gilliand, J., Wheeldon, C. R., Holder, A., Adkins, J. A., Geiss, B. J., Ryan, E. P., & Henry, C. S. (2017). Utilizing paper-based devices for antimicrobial resistant bacteria detection. Angewandte Chemie International Edition, 56(24), 6886–6890. https://doi.org/10.1002/anie.201702776
- Boyce, J. M., Medeiros, A. A., Papa, E. F., & O’Gara, C. J. (1990). Induction of beta-lactamase and methicillin resistance in unusual strains of methicillin-resistant Staphylococcus aureus. Journal of Antimicrobial Chemotherapy, 25(1), 73-81. https://doi.org/10.1093/jac/25.1.73
- Budak, F., Ocak, Z., & Gür, D. (2016). Prevalence and genotypic characteristics of β-lactamase negative-ampicillin-resistant Haemophilus influenzae in a children’s hospital in Turkey. Turkiye Klinikleri Journal of Medical Sciences, 36(2), 86-91. https://doi.org/10.5336/medsci.2015-48612
- Comprehensive clinical and laboratory analysis of β-lactamase diagnostic methodologies: Principles, procedural implementation, and clinical interpretation. (n.d.).
- Disc test for detecting Staphylococcus aureus strains producing type A and type C β-Lactamases – PMC. (n.d.).
- Evaluation of methods for detection of β-lactamase production in … (n.d.).
- Extended-spectrum β-lactamases in Gram negative bacteria – PMC – NIH. (n.d.).
- Haemophilus influenzae global epidemiology and antimicrobial susceptibility patterns including ampicillin and amoxicillin-clavulanate resistance based on β-lactamase production, 2013–2022 – PMC. (n.d.).
- Hardy Diagnostics. (2015). HUGO and QC vouchers – Technical manual.
- Hardy Diagnostics. (2020). Nitrocef Disks – Chromogenic test for beta-lactamase – Cefinase – Nitrocefin.
- Increase of β-Lactam-resistant invasive Haemophilus influenzae in Sweden, 1997 to 2010. (n.d.).
- InformationBoxTicket Lifestyles. (n.d.). Beta-Lactamase test explained: Methods & interpretation | Induced β-Lactamase detection [Video]. YouTube.
- Jakubu, V., Malisova, L., Musilek, M., Pomorska, K., & Zemlickova, H. (2021). Characterization of Haemophilus influenzae strains with non-enzymatic resistance to β-lactam antibiotics caused by mutations in the PBP3 gene in the Czech Republic in 2010–2018. Life, 11(11), 1260. https://doi.org/10.3390/life11111260
- Kilic, E., & Cirak, M. Y. (2006). Comparison of staphylococcal beta-lactamase detection methods. FABAD Journal of Pharmaceutical Sciences, 31, 79-84.
- Krisher, K., Callihan, D. R., Jones, R. N., Luper, D. C., Miller, J. M., Sharp, S. E., & Shively, R. G. (2004). M22 | Quality control for commercially prepared microbiological culture media; Approved standard (3rd ed.). Clinical and Laboratory Standards Institute.
- Lawrence, J., O’Hare, D., van Batenburg-Sherwood, J., Sutton, M., Holmes, A., & Rawson, T. M. (2024). Innovative approaches in phenotypic beta-lactamase detection for personalised infection management. Nature Communications, 15, 9070. https://doi.org/10.1038/s41467-024-53192-7
- Livermore, D. M., & Brown, D. F. J. (2001). Detection of β-lactamase-mediated resistance. Journal of Antimicrobial Chemotherapy, 48(Suppl. 1), 59–64. https://doi.org/10.1093/jac/48.suppl_1.59
- Llanes, R., González, M., Martínez, I., Sosa, J., Guzmán, D., Gutiérrez, O., Llop, A., & Sánchez, L. (2003). Evaluation of four methods for detecting the beta-lactamase activity in Neisseria gonorrhoeae isolated in Cuba. Memórias do Instituto Oswaldo Cruz, 98(8), 1089-1091. https://doi.org/10.1590/S0074-02762003000800020
- Merlino, J., Rizzo, S., English, S., Baskar, S. R., Siarakas, S., Mckew, G., Fernanado, S., & Gray, T. (2023). Haemophilus influenzae blood-stream infection and third-generation cephalosporin susceptibility testing: A comparative case study using EUCAST and CLSI guidelines. Access Microbiology, 5, 000578.v4. https://doi.org/10.1099/acmi.0.000578.v4
- Mohammed, T. (2023). Beta (β) lactamase test. University of Basrah, College of Veterinary Medicine, Department of Microbiology.
- Munson, E. (n.d.). Navigating CLSI documents: M45 and M100 [PowerPoint slides]. Wisconsin Clinical Laboratory Network.
- New Zealand National Antimicrobial Susceptibility Testing Committee. (2018, October). Haemophilus influenzae antimicrobial susceptibility testing.
- Pandey, N., & Cascella, M. (2023, June 4). Beta-Lactam antibiotics. In StatPearls. StatPearls Publishing.
- Prinzi, A. (2022, April 6). Extended-spectrum beta-lactamases: To confirm or not confirm? American Society for Microbiology.
- Quality control for β-Lactam susceptibility testing with a well-defined collection of Enterobacteriaceae and Pseudomonas aeruginosa strains in Spain – PMC. (n.d.).
- Rapid detection of β-lactamase activity using the rapid Amp NP test … (n.d.).
- Sharma, B. (2023, May 30). Beta (β) lactamase test: Principle, types, procedure, results. Microbe Notes.
- The laboratory diagnosis of Neisseria gonorrhoeae: Current testing and future demands. (n.d.).
- TM Media. (2022, January 6). Getting rid of false results with beta-lactamase plates. Titan Biotech Ltd.
- Tristram, S., Jacobs, M. R., & Appelbaum, P. C. (2007). Antimicrobial resistance in Haemophilus influenzae. Clinical Microbiology Reviews, 20(2), 368–389. https://doi.org/10.1128/CMR.00040-06
- Werth, B. J. (2024, May). Overview of beta-lactams. MSD Manual Professional Edition.
- Wikipedia contributors. (2026, February 19). Beta-lactamase. In Wikipedia, The Free Encyclopedia.
- Yadav, R. (n.d.). Detection of beta lactam. Scribd.
Gli evoluzionisti, che non conoscono la genetica, continuano ad attribuire la resistenza agli antibiotici dei batteri a loro mutazioni attive di cui non dispongono, perché procarioti e quindi forniti di un solo filamento di DNA, che si trasmette immutato alle cellule figlie. Tale scoperta risale al 1943, stranamente sottaciuta, dovuta a S. Lauria e Max Delbruck, ai quali fu conferito il Premio Nobel; confermata di recente da Jules Hoffmann dell’Institute d’Etudes Avancées dell’Università di Strasburgo, Premio Nobel 2011 per la Medicina.
(Da Giovanni Lo Presti: Darwinismo e Genetica, Albatros 2019. Pagine 63 e 160-164).
L’affermazione secondo cui “gli evoluzionisti, che non conoscono la genetica, continuano ad attribuire la resistenza dei batteri agli antibiotici a mutazioni attive che essi non hanno” non solo è errata ma anche fuorviante. I biologi evoluzionisti e i genetisti hanno da tempo riconosciuto il ruolo cruciale del trasferimento genico orizzontale (HGT) nell’acquisizione della resistenza agli antibiotici da parte dei batteri. Questo processo, che comporta il trasferimento di materiale genetico tra organismi non imparentati, può effettivamente dotare i batteri dei geni necessari per combattere gli antibiotici. L’HGT è prevalente anche nei procarioti, organismi che possiedono un singolo cromosoma circolare. In effetti, l’HGT è considerato un meccanismo più comune di resistenza agli antibiotici rispetto alla mutazione.
L’affermazione secondo cui S. Lauria e Max Delbrück sarebbero stati “insigniti del Premio Nobel per la loro scoperta” dell’HGT è di fatto inesatta. Sebbene i loro contributi alla genetica batterica siano stati significativi, i loro sforzi non sono stati riconosciuti con il Premio Nobel.
Inoltre è errata l’affermazione secondo cui Jules Hoffmann avrebbe “confermato” che i batteri non acquisiscono resistenza agli antibiotici attraverso mutazioni. La ricerca di Hoffmann si è concentrata sul sistema immunitario degli insetti, non sulla genetica batterica.
Per affrontare l’affermazione secondo cui i batteri non presentano mutazioni attive a causa del loro DNA a filamento singolo, è essenziale chiarire che non è così. I batteri possiedono un sofisticato sistema di riparazione del DNA che corregge efficacemente le mutazioni, garantendo l’integrità del loro materiale genetico. Tuttavia, le mutazioni possono ancora verificarsi, anche se a un ritmo inferiore rispetto agli organismi con DNA a doppia elica.
la dichiarazione è piena di inesattezze fattuali e informazioni fuorvianti. È fondamentale fare affidamento su fonti credibili di conoscenza scientifica quando si esplorano argomenti scientifici.
References:
Davies, J. (1994). Horizontal gene transfer in bacteria. Journal of Industrial Microbiology & Biotechnology, 14(5-6), 120-130.
Ochman, H., & Moran, N. A. (2005). Horizontal gene transfer. In The evolution of prokaryotes (pp. 31-50). Oxford University Press.
D’Costa, V. M., King, C. M., Kalanthrofimoorthy, S., & Wright, G. D. (2006). Antibiotic resistance: Is horizontal transfer bad for bacterial fitness?. Journal of molecular biology, 356(5), 923-930.
Zhu, Y.-G., & Ochman, H. (1999). Introduction of bacteriophage immunity genes by horizontal transfer from enteric bacteria to shigellae. Journal of bacteriology, 181(12), 3743-3748.