Complement system is a group of blood proteins that takes part in innate immune response. It helps in killing microbes, inflammation and phagocytosis. It also increases the action of antibodies and immune cells.
Complement system is an important part of innate immune system. It acts as the first line defense against invading pathogens like bacteria, viruses and also damaged host cells.
It is made up of more than 50 proteins and protein fragments. These proteins are mainly synthesized in the liver and circulate in blood as inactive precursor form. They are also present on cell surface and sometimes inside the cells.
The name complement is given because it complements or increases the action of antibodies and phagocytic cells. It helps in removing microbes, dead cells and cellular debris from the body.
When any foreign material enters into the body, the complement system becomes activated. It is activated by a series of protein cleavage reaction. This reaction occurs in cascade form, where one activated protein activates another protein.
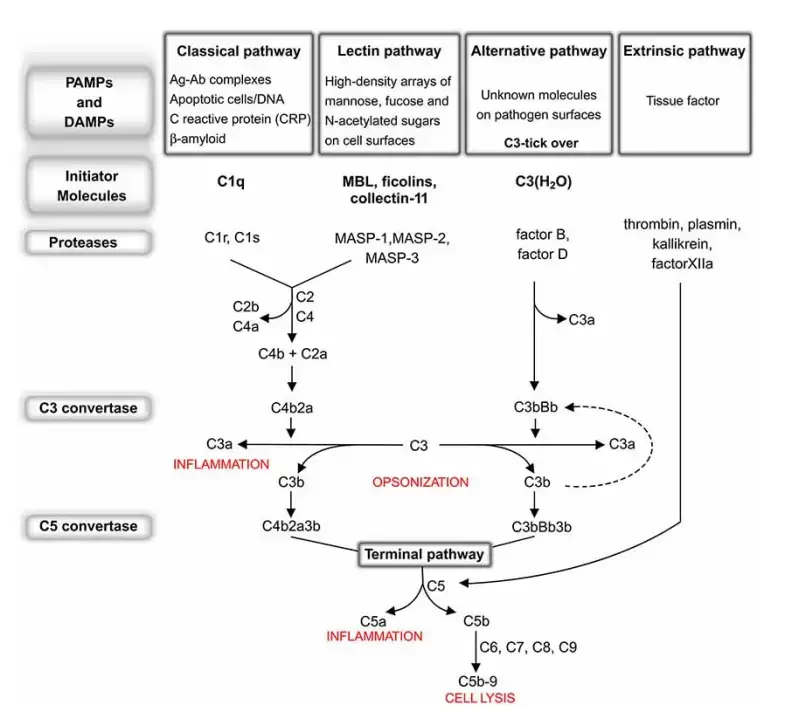
There are three main pathways of complement activation. These are classical pathway, alternative pathway and lectin pathway. The classical pathway is mainly started by antibody attached with antigen. The alternative pathway is activated directly by foreign surface. The lectin pathway is activated when lectin proteins recognize sugar present on microbial surface.
All these three pathways finally meet at the activation of C3 protein. C3 is the central component of complement system. After its activation, many other reactions are started which help in destruction of pathogen.
The complement system has three major functions. First, it produces inflammation by releasing small protein fragments. These fragments attract immune cells like neutrophils and macrophages at the site of infection.
Second, it helps in opsonization. In this process, microbes and dead cells are coated by complement proteins. This makes them easy to be recognized and engulfed by phagocytic cells.
Third, it causes direct killing of pathogen by forming membrane attack complex (MAC). MAC forms pore like structure on the cell membrane of pathogen. As a result, water enters into the cell, the cell swells and finally ruptures.
The complement system is very powerful. So, it is controlled by regulatory proteins of the host body. This regulation is necessary to prevent damage of normal healthy tissues.
History and Discovery of the Complement System
- In 1888, scientist George Nuttall first observed that sheep blood serum had mild killing action against anthrax bacterium. But this activity was lost when the serum was heated.
- In 1891, Hans Ernst August Buchner also observed this heat-sensitive killing property of serum. He named this substance alexin, which means “to ward off” in Greek.
- During 1894-1896, Jules Bordet studied this immune reaction in cholera. He showed that immune principle has two components.
- One component was heat-stable and it was responsible for specific immunity against microorganisms. The other component was heat-sensitive and it was responsible for non-specific antimicrobial action.
- The heat-sensitive component was the alexin. For this important discovery, Jules Bordet later received the Nobel Prize in Physiology or Medicine in 1919.
- In 1899, Paul Ehrlich renamed the heat-sensitive substance as complement. He used this name because this substance complements the action of immune cells and helps antibodies in destroying invading bacteria.
- In the early 1900s, there was a debate between Paul Ehrlich and Jules Bordet. Ehrlich believed that every antigen-specific antibody has its own specific complement.
- Bordet believed that there is only one general type of complement. This view was later found to be correct.
- In 1907, Adolfo Ferrata and Erwin Brand discovered that complement system was not a single substance. It was actually made up of two separate substances.
- In the 1920s, a third complement substance was identified. This showed that complement was more complex than earlier thought.
- From 1920s to 1960s, improvement of laboratory instruments helped in more discovery of complement proteins. Scientists found that complement is not a simple serum factor.
- It is a complex protein system made up of many proteins. Later it was understood as a cascade system of more than 40 proteins, which act together in immune defense.
Components of the Complement System
- Central component
- C3 is the central protein of the complement system. All three pathways finally come to this protein and after its cleavage the further complement reactions are started.
- Classical pathway components
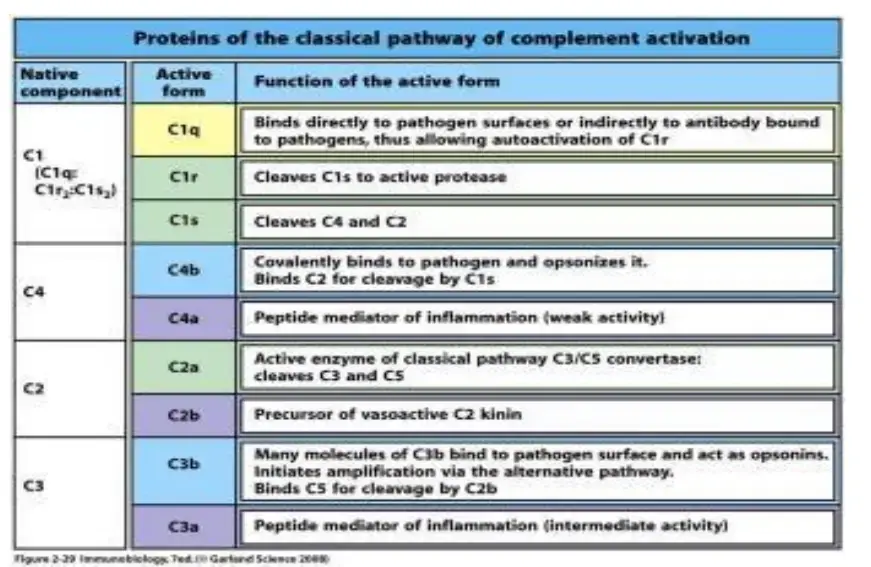
- C1 complex is made up of C1q, C1r and C1s. C1q is used for recognition of target and C1r and C1s are serine proteases which take part in activation of other complement proteins.
- C4 and C2 are also important proteins of the classical pathway. These are cleaved after the activation of C1 complex and form the classical pathway C3 convertase.
- Lectin pathway components
- Mannose-binding lectin (MBL) and ficolins are target recognition molecules of the lectin pathway. They bind with sugar molecules present on the surface of microorganisms.
- MASP-1, MASP-2 and MASP-3 are the enzymes related with MBL. These are called MBL-associated serine proteases and help in cleavage of complement proteins in this pathway.
- Alternative pathway components
- Factor B and Factor D are used in the alternative pathway. These proteins help in formation of alternative pathway C3 convertase on the foreign surface.
- Properdin (Factor P) is a stabilizing protein. It stabilizes the alternative pathway C3 convertase and helps to continue the complement reaction for more time.
- Terminal pathway components
- C5, C6, C7, C8 and C9 are terminal components of the complement system. These proteins join one by one and form membrane attack complex (MAC). MAC makes pore on the target cell membrane and causes lysis of the cell.
- Fluid-phase regulators and inhibitors
- C1 inhibitor (C1-INH) is a regulatory protein present in fluid phase. It stops excess activation of classical pathway and lectin pathway.
- Factor H and Factor I are used for regulation of C3b. They inactivate C3b and stop the cascade when the reaction is not required.
- C4b-binding protein (C4BP) inhibits the classical pathway C3 convertase. It controls the reaction by acting on C4b.
- Carboxypeptidase N (CPN) and Carboxypeptidase B2 (CPB2) are enzymes that inactivate inflammatory proteins like C3a and C5a. They remove the terminal basic residues and reduce their activity.
- Clusterin and Vitronectin are regulators of terminal pathway. They prevent the insertion of MAC into host cell membrane.
- Membrane-bound regulators
- Decay accelerating factor (DAF / CD55) is present on the host cell membrane. It dissociates C3 convertase and C5 convertase, so complement activation is stopped on normal body cell.
- Membrane cofactor protein (MCP / CD46) acts as a cofactor for Factor I. It helps in cleavage of C3b and C4b on host cell surface.
- Protectin (CD59) prevents the final formation of MAC. It protects the host cell membrane from complement mediated lysis.
- Complement receptors
- C3aR, C5aR1 (CD88) and C5aR2 (C5L2/GPR77) are anaphylatoxin receptors. These receptors bind with C3a and C5a and take part in inflammatory reaction.
- CR1 (CD35), CR2 (CD21), CR3, CR4 and CRIg are opsonin receptors. These are mainly present on immune cells and recognize complement coated microbes, which helps in phagocytosis.
Properties and Characteristics of Complement Proteins
- Inactive form – Complement proteins mainly circulate in the blood plasma and tissue fluids as inactive precursors. These inactive proteins are called zymogens or proenzymes and they are activated only after any infection or foreign surface is present.
- Site of synthesis – Most of the complement proteins are synthesized by the liver cells, called hepatocytes. Some amount are also produced by tissue macrophages, blood monocytes and epithelial cells of gastrointestinal and genitourinary tract.
- Serum abundance – Complement proteins are present in serum in large amount. They form nearly 10% of the globulin fraction of human blood serum.
- Heat sensitivity – Complement proteins are heat-labile proteins. Their activity is destroyed by heating the serum and this property helped in the early discovery of complement system.
- Activation by cleavage – Complement proteins are activated by proteolytic cleavage. In this process, one activated protein cleaves another protein and this again activates the next protein in the sequence. Thus, the reaction goes in cascade form.
- Enzymatic activity – Many activated complement proteins work as enzymes. Some important complement enzymes are C1r, C1s, Factor D and Factor I. These are serine proteases and they cut other complement proteins during activation.
- Fragment name – When a complement protein is cleaved, usually two fragments are formed. The larger fragment is generally called b fragment, such as C3b and C4b, and it remains attached with target surface. The smaller fragment is called a fragment, such as C3a and C4a, and it diffuses away in fluid and may act as inflammatory mediator.
- Covalent binding ability – Some complement proteins like C3 and C4 contain a reactive internal thioester bond. After cleavage this bond becomes exposed and C3b and C4b can bind covalently with the surface of pathogen or target cell.
- Short life and instability – Some active complement intermediates are highly unstable. The exposed thioester bond of C3b remains active only for very short time and C3bBb also has short half-life. It becomes more stable when properdin is attached with it.
- Strict regulation – Complement proteins are very powerful in their action. So, they are controlled by soluble regulators and membrane-bound regulatory proteins. This regulation is needed to protect the normal host tissues from complement mediated damage.
- Destructive nature – Activated complement proteins can produce inflammation, opsonization and lysis of target cell. This property is useful for killing microbes but it can become harmful if the reaction occurs on own healthy cells.
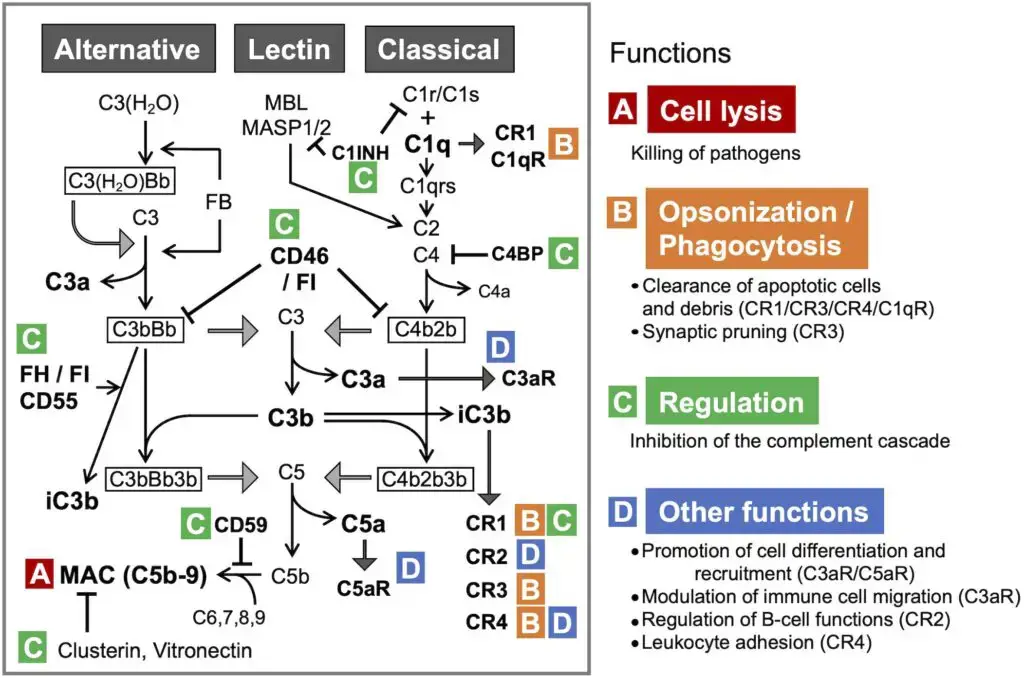
Activation Pathways of the Complement System
- Classical pathway
- Activation trigger – The classical pathway is mainly started when C1 complex binds with antibody attached antigen. Here C1q binds with IgG or IgM present on the foreign antigen surface. It may also start when C1q directly binds with pathogen surface or altered self materials like apoptotic cells.
- Mechanism – After binding of C1q, C1r and C1s become activated. Then C1s cleaves C4 and C2. The larger fragments of these proteins remain on the target surface and form classical pathway C3 convertase.
- Lectin pathway
- Activation trigger – The lectin pathway is activated without antibody. In this pathway mannose-binding lectin (MBL) or ficolins bind with specific sugar molecules like mannose residues present on microbial surface.
- Mechanism – After binding of MBL or ficolins, MASP-1 and MASP-2 become activated. These enzymes act like C1r and C1s of classical pathway. They cleave C4 and C2 and form the same C3 convertase as formed in classical pathway.
- Alternative pathway
- Activation trigger – The alternative pathway does not require antibody and also does not require special recognition molecule. It is continuously active at low level in blood. This is called tick-over, where C3 is spontaneously hydrolysed.
- Mechanism – By tick-over, a C3b-like molecule is formed. If C3b binds with host cell, it is inactivated by regulatory proteins. But if it binds with foreign surface, then Factor B attaches with it. After that Factor D cleaves Factor B and forms C3bBb, which is the alternative pathway C3 convertase. This convertase is stabilized by Properdin (Factor P) and then it starts amplification of complement reaction.
Mechanism of Complement Activation Cascade
Phase 1: Initiation and Target Recognition
Step 1: Classical pathway recognition
In classical pathway, the complement cascade starts when C1q binds with antigen-antibody complex. The antibody may be IgG or IgM attached with foreign antigen. Sometimes C1q also binds directly with pathogen surface or altered self cells.
Step 2: Activation of C1 enzymes
After binding of C1q, change occurs in C1 complex. Then C1r and C1s become activated. These are serine proteases and they start the next cleavage reaction.
Step 3: Cleavage of C4 and C2
Active C1s cleaves C4 into C4a and C4b. It also cleaves C2 into C2b and C2a. C4b remains on the target surface and C2a later joins with it.
Step 4: Lectin pathway recognition
In lectin pathway, mannose-binding lectin (MBL) or ficolins bind with carbohydrate present on microbial surface. This pathway does not need antibody for its activation.
Step 5: Activation of MASP enzymes
After binding of MBL or ficolins, MASP-1 and MASP-2 become activated. These enzymes act like C1s of classical pathway. They cleave C4 and C2 in the same way.
Step 6: Alternative pathway tick-over
In alternative pathway, C3 is continuously hydrolysed at low level in blood. This is called tick-over. By this process C3(H₂O) is formed.
Step 7: Initial C3b formation
C3(H₂O) binds with Factor B. Then Factor D cleaves Factor B and a fluid phase enzyme is formed. This enzyme produces the first C3b fragments.
Phase 2: C3 Convertase Assembly and Amplification
Step 1: Classical and lectin C3 convertase formation
In classical pathway and lectin pathway, C4b and C2a join together on the target cell membrane. This forms C4b2a. It is the C3 convertase of both classical and lectin pathway.
Step 2: Alternative C3 convertase formation
In alternative pathway, surface bound C3b binds with Factor B. Then Factor D cleaves this bound Factor B. As a result C3bBb is formed, which is the alternative pathway C3 convertase.
Step 3: Stabilization by properdin
The C3bBb complex is not very stable. Properdin (Factor P) binds with this complex and makes it stable. So, the alternative pathway reaction continues for more time.
Step 4: Cleavage of C3
Both C3 convertases cleave many molecules of C3 into C3a and C3b. C3a goes away in the fluid and acts as an anaphylatoxin. It takes part in inflammatory reaction.
Step 5: Binding of C3b
The newly formed C3b binds with the target surface. It binds strongly by covalent attachment. This C3b also helps in opsonization, so phagocytic cells can recognize the microbe easily.
Step 6: Amplification loop
New C3b molecules again bind with Factor B and form more C3bBb. This makes more and more cleavage of C3. This repeated reaction is called amplification loop.
Phase 3: C5 Convertase Assembly
Step 1: Binding of extra C3b
When extra C3b binds with the already formed C3 convertase, the activity of enzyme changes. Now it starts acting on C5. This step starts the next part of complement cascade.
Step 2: Classical and lectin C5 convertase formation
In classical pathway and lectin pathway, C4b2a combines with C3b. This forms C4b2a3b. It is the C5 convertase of classical and lectin pathway.
Step 3: Alternative C5 convertase formation
In alternative pathway, C3bBb combines with another C3b. This forms C3bBb3b. It is the C5 convertase of alternative pathway.
Step 4: Cleavage of C5
The formed C5 convertase cleaves C5 into C5a and C5b. C5a goes away and takes part in inflammatory reaction. C5b remains on the surface and starts the final attack pathway.
Phase 4: Terminal Pathway or Membrane Attack Complex
Step 1: Binding of C6 and C7
In this phase, C5b first binds with C6. After that C7 also binds with it. This forms C5b-7 complex.
Step 2: Membrane insertion
The C5b-7 complex is lipophilic in nature. So, it enters into the lipid layer of target pathogen membrane. This is the first membrane attached complex of terminal pathway.
Step 3: Binding of C8
After membrane insertion, C8 binds with C5b-7 complex. This forms C5b-8. C8 enters deeper in the membrane and makes the complex ready for C9 binding.
Step 4: Binding of C9
Many molecules of C9 bind with C5b-8 complex. Usually 12 to 18 molecules of C9 join together. They arrange in ring like form.
Step 5: Formation of MAC
The complete ring like pore is called membrane attack complex (MAC) or C5b-9. It is formed on the membrane of target cell. This pore damages the normal structure of the membrane.
Step 6: Cell lysis
Through MAC, water and ions move freely across the membrane. The cell cannot control this movement. So, the cell swells, then ruptures and finally dies. This is called complement mediated cell lysis.
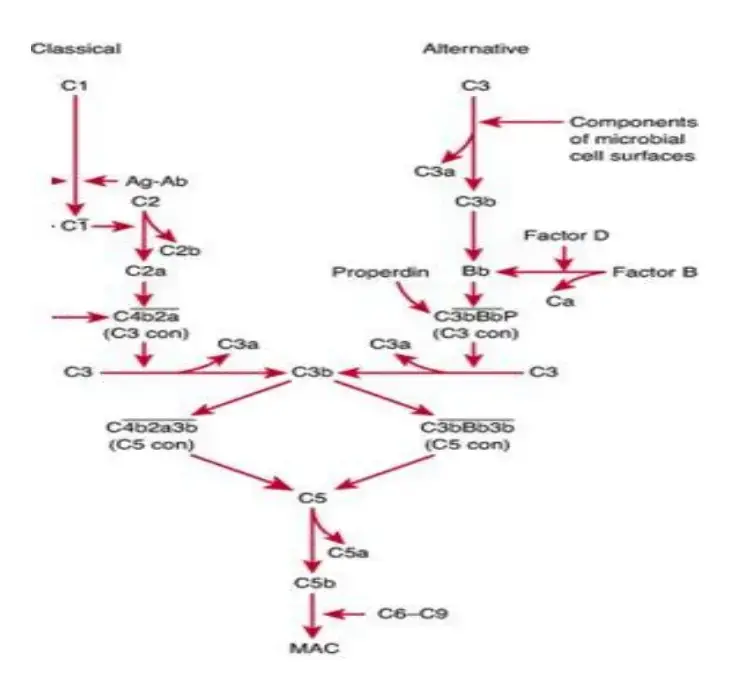
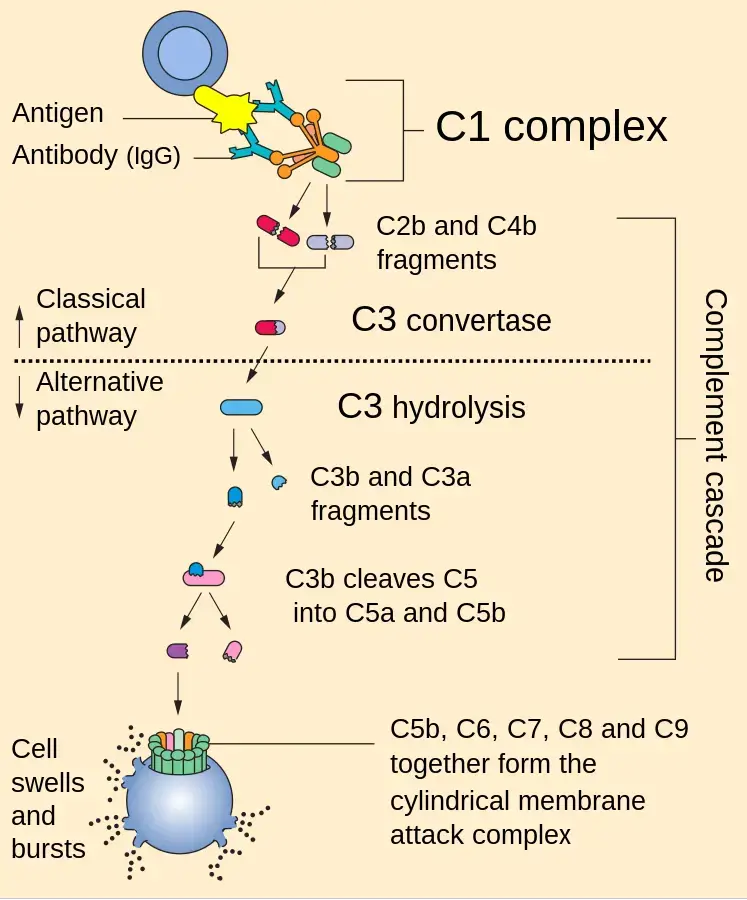
Biological Functions of the Complement System
- Opsonization and phagocytosis – C3b and other complement proteins coat the surface of microbes. This coating makes the microbe easy to recognize by macrophages and neutrophils. Then these phagocytic cells engulf the microbe and destroy it.
- Cell lysis – The terminal complement proteins form membrane attack complex (MAC) on the membrane of target cell. This MAC makes pore in the membrane. Water enters into the cell, the cell swells and finally ruptures.
- Inflammation – Small fragments like C3a and C5a are formed during complement activation. These are called anaphylatoxins. They take part in inflammatory reaction and increase the response at the site of infection.
- Chemotaxis – C5a attracts inflammatory cells towards the infected area. Mainly neutrophils and macrophages move to that place. This is useful for removal of foreign material.
- Histamine release – C3a and C5a stimulate mast cells. Then mast cells release histamine. Due to this, blood vessels become dilated and more permeable.
- Clearance of immune complexes – The complement system helps in removing antigen-antibody complexes from blood. These complexes are coated by complement proteins and then cleared by phagocytic cells.
- Removal of dead cells – Complement proteins also help in removal of dead cells, damaged cells and apoptotic cells. This removal occurs in safe way and usually does not produce unnecessary inflammation.
- Activation of B cells – Some complement fragments like C3d bind with receptors on B-cells. This helps in activation of B-cells. As a result, antibody production becomes increased.
- Connection with adaptive immunity – The complement system is mainly part of innate immunity, but it also helps adaptive immunity. It helps in better antibody response and memory cell formation.
- Protection of body – The complement system protects the body against bacteria, viruses and altered cells. It acts by coating, inflammation, phagocytosis and direct membrane attack.
Membrane Attack Complex (MAC)
Membrane Attack Complex (MAC) is the final cell killing structure of the complement system. It is also called terminal complement complex or C5b-9. It is formed at the end of complement cascade and causes lysis of target cell.
The formation of MAC starts when C5 convertase cleaves C5 into C5a and C5b. C5a goes away in fluid and takes part in inflammatory reaction. C5b remains on the target surface and starts the formation of terminal complex.
After this, C5b binds with C6 and C7. This forms C5b-7 complex. The complex is lipophilic in nature, so it attaches with the outer surface of target cell membrane.
Then C8 binds with C5b-7 complex and forms C5b-8. C8 enters deeper into the lipid bilayer of the membrane. It also helps in attachment of the final component C9.
In the next step, many molecules of C9 come and bind with C5b-8. Usually 12 to 18 molecules of C9 polymerize together. They form a tubular ring like pore in the membrane. This complete pore is called membrane attack complex (MAC) or C5b-9.
The MAC destroys the normal structure of the target cell membrane. It makes transmembrane holes through the membrane. Due to this, water, ions and small molecules move inside the cell without control.
As a result, osmotic balance of the cell is lost. Water enters into the cell, the cell becomes swollen and finally ruptures. This process is called osmotic lysis and in this way MAC kills the target pathogen.
The body also has some regulatory proteins which protect normal host cells from MAC formation. This regulation is important because MAC can damage own healthy tissues if it is not controlled.
Protectin (CD59) is a membrane-bound regulatory protein present on host cells. It binds with C5b-8 complex and stops the binding of C9. So, final pore formation does not occur on host cell membrane.
Vitronectin and Clusterin are soluble regulatory proteins present in fluid phase. They bind with free C5b-7 or C5b-8 complexes. This prevents their accidental insertion into the membrane of nearby host cells.
Formation Process of Membrane Attack Complex (MAC)
- Initiation – The formation of membrane attack complex (MAC) starts when C5 convertase cleaves C5 protein. By this cleavage C5a and C5b are formed. C5b remains near the target membrane and starts the terminal complement pathway.
- Initial assembly – C5b then binds one after another with C6 and C7. This forms C5b-7 complex. The complex becomes lipophilic in nature, so it can attach with lipid membrane of the target cell.
- Membrane insertion – The formed C5b-7 complex inserts into the lipid bilayer of target cell membrane. This is the first step where terminal complement proteins become attached with the membrane.
- C8 penetration – After this, C8 binds with C5b-7 complex and forms C5b-8. C8 is made up of α, β and γ subunits. This complex penetrates deeper into the membrane and it also acts as receptor for C9.
- C9 recruitment – The C5b-8 complex now attracts C9 molecules. Usually 12 to 18 molecules of C9 come to this site and start joining with each other.
- C9 polymerization – The C9 molecules polymerize around the membrane attached complex. They arrange in a ring like form. This ring structure gradually makes a complete pore through the target cell membrane.
- Pore formation – The complete pore formed by C5b-9 is called membrane attack complex (MAC). It is a stable transmembrane pore. It passes across the membrane and damages the normal membrane barrier.
- Cell destruction – Through the MAC pore, water, ions and small molecules move without control. The cell loses its osmotic balance. Water enters into the cell, the cell swells and finally ruptures. This death of target cell is called complement mediated lysis.
Regulation of the Complement System
Regulation of complement system is required because complement proteins can damage the own host cells also. So, many regulatory proteins are present in plasma and on cell membrane. These proteins stop extra activation of complement cascade.
- Classical and lectin pathway regulators
- C1 inhibitor (C1-INH) – C1 inhibitor is a soluble regulatory protein. It binds with C1r and C1s and makes them inactive. So, the classical pathway is stopped. It also binds with MASP-1 and MASP-2 and stops the lectin pathway.
- MASP-3, MAp44 and MAp19 – These proteins are related with lectin pathway. They compete with MASP-1 and MASP-2 for binding with lectin recognition molecules. But they cannot cleave C4 and C2. So, the next reaction of lectin pathway does not occur.
- C3 and C5 convertase regulators
- Factor I (FI) – Factor I is a plasma serine protease. It cleaves C3b and C4b into inactive fragments like iC3b and C4d. Due to this, C3b and C4b cannot make new convertase enzymes. Factor I needs other cofactor proteins for its work.
- Factor H (FH) – Factor H is the main soluble regulator of alternative pathway. It binds with C3b and competes with Factor B. It also works as cofactor for Factor I and helps in cleavage of C3b. It breaks the alternative pathway C3 convertase, so the reaction becomes stopped.
- C4b-binding protein (C4BP) – C4BP is a fluid phase regulator. It breaks the classical pathway C3 convertase. It also works as cofactor for Factor I and helps in inactivation of C4b.
- Membrane cofactor protein (MCP / CD46) – MCP is a membrane bound protein present on almost all host cells. It works as cofactor for Factor I. It helps in cleavage of C3b and C4b on host cell surface, so complement activation is controlled.
- Decay accelerating factor (DAF / CD55) – DAF is present on the host cell membrane. It breaks C3 convertase and C5 convertase complex. So, the complement reaction cannot continue on normal body cell.
- Complement receptor 1 (CR1 / CD35) – CR1 is present on leukocytes and erythrocytes. It has two regulatory functions. It helps Factor I in cleavage of C3b and C4b, and it also breaks C3 convertase and C5 convertase.
- Terminal pathway regulators
- Protectin (CD59) – Protectin is a membrane bound glycoprotein of host cell. It binds with C5b-8 complex and blocks the binding of C9. So, C9 cannot join and membrane attack complex (MAC) pore is not formed on host cell.
- Clusterin and Vitronectin – Clusterin and Vitronectin are soluble proteins present in fluid phase. They bind with free sC5b-7, sC5b-8 and sC5b-9 complexes. So, these complexes cannot enter into the membrane of nearby healthy cells.
- Anaphylatoxin inactivators
- Carboxypeptidase N (CPN) and Carboxypeptidase B2 (CPB2 / TAFI) – These are plasma enzymes. They act on C3a, C4a and C5a, which are inflammatory anaphylatoxins. They remove the C-terminal arginine residues and convert them into desArg forms. Due to this, these fragments become weak in their inflammatory action.
Biological Consequences of Complement Activation
Complement activation produces many biological effects in the body. These effects help in killing of microbes, removal of immune complexes and activation of immune cells. But excess activation may also damage the host tissue.
- Pathogen and cell lysis – The final result of complement cascade is the formation of membrane attack complex (MAC) or C5b-9. This complex enters into the membrane of target bacteria or pathogen and makes pore. Due to this, water and ions move inside the cell, the cell swells and finally ruptures by osmotic lysis.
- Opsonization – Complement fragments mainly C3b bind with the surface of pathogen and foreign antigen. This coating process is called opsonization. By this, the microbe becomes marked for destruction.
- Enhanced phagocytosis – After opsonization, phagocytic cells like macrophages and neutrophils can recognize the coated microbe easily. Then they engulf the pathogen and destroy it inside the cell.
- Inflammation – During complement activation small fragments are released. Mainly C3a and C5a are important in this reaction. These are called anaphylatoxins and they increase inflammatory reaction at the site of infection.
- Immune cell recruitment – C5a attracts immune cells towards the infected area. This movement of cells is called chemotaxis. Due to this, neutrophils, macrophages and other inflammatory cells come near the pathogen.
- Histamine release – C3a and C5a stimulate mast cells and basophils. These cells release histamine. As a result, blood vessels become dilated and their permeability is increased.
- Smooth muscle contraction – Anaphylatoxins may also induce contraction of smooth muscle. This is one of the inflammatory effects of complement activation.
- Clearance of immune complexes – The complement system helps in removal of soluble antigen-antibody complexes from circulation. These complexes are coated by complement proteins and then removed safely from blood.
- Removal of cellular debris – Complement proteins also bind with damaged cells, dead cells and apoptotic cells. These cells are then removed by phagocytes in a silent way. So, unnecessary inflammation against own cell material is prevented.
- Stimulation of adaptive immunity – Complement activation also helps in adaptive immunity. C3d binds with receptors present on B-cells and helps in their activation. By this, antibody production becomes increased.
- B-cell activation – C3d acts like a molecular adjuvant. It lowers the activation requirement of B-cells. So, B-cells become activated more easily and the adaptive immune response starts quickly.
Role of Complement System in Innate and Adaptive Immunity
Complement system takes part in both innate immunity and adaptive immunity. It first acts against foreign pathogens without waiting for antibody formation. Later it also helps B-cells and T-cells for better immune response.
- Role in innate immunity
- First line defense – Complement system acts as an early defense system of the body. It recognizes foreign pathogens and danger signals from damaged or dying host cells. These foreign patterns are called PAMPs and danger signals are called DAMPs.
- Opsonization and phagocytosis – Complement fragments, mainly C3b, coat the surface of invading microbes. This coating marks the microbe for destruction. After this, macrophages and neutrophils recognize the coated microbe easily and engulf it by phagocytosis.
- Inflammation and cell recruitment – During complement activation, C3a and C5a are formed. These are anaphylatoxins. They increase local inflammation, increase permeability of blood vessels and bring innate immune cells to the site of infection or injury.
- Direct pathogen lysis – Terminal complement proteins form membrane attack complex (MAC) or C5b-9 on the pathogen surface. This complex makes holes in the cell membrane. Due to this, water enters into the cell and the pathogen dies by osmotic lysis.
- Clearance of cellular debris – Complement proteins also bind with dead cells and apoptotic cells. These cells are then removed by phagocytes in a silent way. So, unnecessary inflammation is not produced against own cell materials.
- Role in adaptive immunity
- B-cell activation – Complement fragment C3d helps in activation of B-cells. It binds with CR2 (CD21) receptor on B-cells and reduces the amount of signal needed for B-cell activation. So, B-cells become activated more easily.
- Antibody production – When B-cells are activated with help of C3d, antibody production becomes increased. It also helps in formation of B-cell memory, so the body can give better response during next infection.
- T-cell survival – Complement also works inside T-cells by an intracellular complement system, called complosome. Resting T-cells produce internal C3a, which helps in their normal survival.
- T-cell activation and differentiation – After activation of T-cells, internal C3 and C5 fragments bind with receptors like CD46, C3aR and C5aR1. These signals help in multiplication of T-cells and their change into active effector cells like Th1 cells.
- Control of T-cell response – Complement signaling also helps to stop excess T-cell reaction. Signals through CD46 and C5aR2 make T-cells produce IL-10. IL-10 is an anti-inflammatory cytokine and it helps the immune response to come into control phase.
Complement Deficiencies and Related Disorders
Complement deficiency is the absence or defective function of any complement protein or regulatory protein. In this condition the complement system may not work properly. Some patients suffer from repeated infection and some patients suffer from damage of own cells due to uncontrolled complement activity.
- Terminal pathway deficiency
- C5-C9 deficiency – The deficiency of C5, C6, C7, C8 and C9 affects the formation of membrane attack complex (MAC). As a result, pore is not formed properly on the bacterial membrane. So, the killing of some bacteria by complement system becomes weak.
- Neisseria infection – In terminal pathway deficiency, patient is commonly affected by Neisseria species. The important bacteria are Neisseria meningitidis and Neisseria gonorrhoeae. Repeated infection may occur and meningitis may become severe and life threatening.
- C1 inhibitor deficiency
- Hereditary angioedema – C1 inhibitor (C1-INH) deficiency is caused by mutation in SERPING1 gene. In this condition C1-INH protein is either less or not working properly. This disorder is known as hereditary angioedema (HAE).
- Bradykinin accumulation – When C1-INH is not present in proper amount, the kallikrein-kinin system becomes uncontrolled. Due to this, bradykinin is produced in excess amount. It causes repeated swelling of skin, gastrointestinal tract and upper airway. Swelling in upper airway is dangerous and may cause death.
- Complement Factor I deficiency
- C3 depletion and infection – Complement Factor I (CFI) is used to control C3b and C4b. In its deficiency, C3b and C4b are not regulated properly and complement activation continues. Due to continuous activation, C3 level becomes low. Patient may suffer from repeated severe bacterial infections like pneumonia and sepsis.
- Autoimmune disease – Uncontrolled complement activation may start autoimmune reaction in the body. It may be found with Systemic Lupus Erythematosus (SLE) and Rheumatoid Arthritis. In these diseases, immune reaction occurs against own tissues.
- Organ specific damage – CFI mutation is also related with kidney and eye disorders. Kidney diseases include atypical hemolytic uremic syndrome (aHUS) and C3 glomerulopathy (C3G). Eye disease includes Age-Related Macular Degeneration (AMD).
- Complement regulator deficiency
- Factor H mutation – Factor H is the important regulator of alternative pathway. When Factor H is defective, host cells are not protected properly from complement attack. This may cause atypical hemolytic uremic syndrome (aHUS), C3 glomerulopathy and Age-Related Macular Degeneration (AMD).
- Paroxysmal nocturnal hemoglobinuria – Paroxysmal Nocturnal Hemoglobinuria (PNH) is an acquired genetic disorder. In this disease, blood cells do not have proper CD55 and CD59. These two are protective membrane regulators. So, complement attacks own red blood cells and causes their destruction, which is called hemolysis.
- CHAPLE disease – CHAPLE disease occurs due to absence of CD55 regulator. It is also called CD55-deficient protein-losing enteropathy. In this disease complement system becomes overactive and causes damage of digestive tract.
- Complement overactivation disorders
- Nervous and muscular diseases – Abnormal complement activity is found in some nervous system and muscular diseases. These include generalized myasthenia gravis (gMG), neuromyelitis optica spectrum disorder (NMOSD), spinal cord injury and Alzheimer’s disease.
- Ocular disease – Local overactivation of C3 protein in the eye may cause Geographic Atrophy. It is an advanced form of Age-Related Macular Degeneration and may affect vision.
- Autoimmune and inflammatory diseases – Dysregulated complement activity may cause tissue damage in different inflammatory diseases. Some examples are cold agglutinin disease, ANCA-associated vasculitis, Barraquer-Simons syndrome, asthma and inflammatory bowel disease.
Clinical Significance of the Complement System
Complement system is clinically important because it is involved in infection, inflammation and many immune diseases. When complement is deficient, the body cannot remove microbes properly. When complement is overactive, it may damage own blood cells, kidney, eye and other tissues.
- Therapeutic target and drugs – Complement system is used as a target for treatment of many autoimmune and inflammatory diseases. Some drugs block C5, such as eculizumab, ravulizumab and crovalimab. Some drugs block C3, such as pegcetacoplan. Iptacopan and danicopan act on Factor B or Factor D. These drugs are useful in diseases which were very difficult to manage before.
- Blood and kidney diseases – Uncontrolled complement activation is found in serious blood and kidney diseases. In Paroxysmal Nocturnal Hemoglobinuria (PNH), complement attacks own red blood cells. Due to this, red blood cells are destroyed and hemolysis occurs. In atypical hemolytic uremic syndrome (aHUS), complement damages endothelial cells and small clots are formed in blood vessels. It may finally cause kidney failure.
- Hereditary angioedema – Hereditary Angioedema (HAE) occurs due to deficiency or abnormal function of C1 esterase inhibitor (C1-INH). When C1-INH is not working, bradykinin is produced in excess amount. Due to this, sudden swelling occurs in skin, gastrointestinal tract and upper airway. Upper airway swelling is dangerous because it may block breathing.
- Diagnostic markers – Complement proteins are used in diagnosis of some diseases. Low level of C4 for long time is an important marker for HAE. Other complement products like sC5b-9, C3dg and iC3b are measured to know whether complement activation is high or treatment is working or not.
- Eye diseases – Excess local activation of complement is found in eye disease. Geographic Atrophy is an advanced form of Age-Related Macular Degeneration. In this disease, complement activation damages the retinal tissue. Drugs like pegcetacoplan and avacincaptad pegol are used in its treatment.
- Nervous system diseases – Abnormal complement activity is found in some nervous system diseases. These include Alzheimer’s disease, Huntington’s disease and spinal cord injury. In these conditions, complement may take part in tissue damage.
- Complosome and autoimmunity – Complosome is the complement system present inside the cells. It helps in cell metabolism, autophagy and T-cell function. When this intracellular complement system is disturbed, autoimmune diseases may occur. It is seen in Rheumatoid Arthritis (RA) and Systemic Lupus Erythematosus (SLE). It is also found in severe inflammation during viral infection like COVID-19.
- Coagulation and thrombosis – Complement system is connected with blood clotting system. C5a can increase tissue factor and activate platelets. Due to this, unwanted clot formation may occur. This is important in DIC, sepsis and severe COVID-19.
- Infection susceptibility – Terminal complement proteins are needed for formation of membrane attack complex (MAC). If these proteins are deficient, patient becomes more susceptible to severe infection. The most important infection is by Neisseria meningitidis.
- Risk of complement inhibitor therapy – Complement blocking drugs are useful, but they also increase the risk of infection. Meningococcal and encapsulated bacterial infections may occur. So, vaccination is needed before starting these drugs.
References
- Abcam. (n.d.). Complement cascade and its inhibitors.
- Arbore, G., Kemper, C., & Kolev, M. (2017). Intracellular complement − the complosome − in immune cell regulation. Molecular Immunology, 89, 2-9. https://doi.org/10.1016/j.molimm.2017.05.012
- AstraZeneca. (2024). Voydeya approved in the US as add-on therapy to ravulizumab or eculizumab for treatment of extravascular haemolysis in adults with the rare disease PNH.
- AstraZeneca US. (2024). VOYDEYA™ approved in the US as add-on therapy to ravulizumab or eculizumab for treatment of extravascular hemolysis in adults with the rare disease PNH.
- C5aR-dependent cell activation by physiological concentrations of C5adesArg: Insights from a novel label-free cellular assay – PMC. (n.d.).
- Comparative Effectiveness of Pegcetacoplan Versus Ravulizumab and Eculizumab in Complement Inhibitor-Naïve Patients with Paroxysmal Nocturnal Hemoglobinuria: A Matching-Adjusted Indirect Comparison – PMC. (n.d.).
- COMPLEMENT: AN OVERVIEW FOR THE CLINICIAN – PMC – NIH. (n.d.).
- Complement Technology. (n.d.). Factor I Catalog Number: A138 Sizes Available: 100 µg/vial Concentration: 1.0 mg/mL.
- Complement Technology. (n.d.). Name: C3a desArg Anaphylatoxin (Not Recombinant) Catalog Number: A119 Sizes Available: 50 µg Concentration: 0.5 mg/mL.
- Complement and coagulation cascades – Cusabio. (n.d.).
- Complement regulator CD46: genetic variants and disease associations – PMC – NIH. (n.d.).
- Complement System Part I – Molecular Mechanisms of Activation and Regulation – PMC. (n.d.).
- Complement-Coagulation Cross-Talk: A Potential Mediator of the Physiological Activation of Complement by Low pH – PMC. (n.d.).
- Complosome — the intracellular complement system – PMC. (n.d.).
- Creative Biolabs. (n.d.). Complement Component Factor I/CFI.
- Creative Biolabs. (n.d.). Complement Regulator of Complement System.
- Discover HAE. (n.d.). Hereditary Angioedema Mechanism of Disease & Pathogenesis.
- FDA approval of crovalimab: a milestone in paroxysmal nocturnal hemoglobinuria treatment. (n.d.).
- Freiwald, T., & Afzali, B. (2025). The secret life of complement: challenges and opportunities in exploring functions of the complosome in disease. Journal of Clinical Investigation, 135(12), e188350. https://doi.org/10.1172/JCI188350
- Human C3a and C3a desArg anaphylatoxins have conserved structures, in contrast to C5a … – PMC. (n.d.).
- Işık, B., & Erbaş, O. (2023). Diagnosis and treatment modalities for hereditary angioedema in emergency medicine. Demiroglu Science University Florence Nightingale Journal of Medicine, 9(3), 133-141. https://doi.org/10.5606/fng.btd.2023.134
- Jupe, S. (2016). CPN, CPB2 cleave C3a, C5a. Reactome Pathway Database.
- Kang, Y.-H., Varghese, P. M., Aiyan, A. A., Pondman, K., Kishore, U., & Sim, R. B. (2024). Complement-Coagulation Cross-talk: Factor H-mediated regulation of the Complement Classical Pathway activation by fibrin clots. Frontiers in Immunology, 15, 1368852. https://doi.org/10.3389/fimmu.2024.1368852
- Kolev, M., & Kemper, C. (2017). Keeping It All Going—Complement Meets Metabolism. Frontiers in Immunology, 8, 1. https://doi.org/10.3389/fimmu.2017.00001
- Liv Hospital. (n.d.). Next-Generation Complement Inhibitors.
- Mechanisms involved in hereditary angioedema with normal C1-inhibitor activity – PMC. (n.d.).
- Merle, N. S., Church, S. E., Fremeaux-Bacchi, V., & Roumenina, L. T. (2015). Complement System Part I – Molecular Mechanisms of Activation and Regulation. Frontiers in Immunology, 6, 262. https://doi.org/10.3389/fimmu.2015.00262
- Molecular Intercommunication between the Complement and Coagulation Systems – PMC. (n.d.).
- National Center for Biotechnology Information (NCBI). (n.d.). 27202 – Gene ResultC5AR2 complement C5a receptor 2 [ (human)].
- Overview of Complement Activation and Regulation – PMC. (n.d.).
- Patsnap Synapse. (2025). What diseases does Pegcetacoplan treat?
- Rodríguez de Córdoba, S., Reparaz Suevos, A., González Sanz, S., Fernández, F. J., Vega, M. C., Varela, E. C., Lada Colunga, A., Vallejo Llamas, J. C., & Gonzalez-Rodríguez, A. P. (2025). C3 mutations and poor pegcetacoplan response in paroxysmal nocturnal hemoglobinuria. Frontiers in Immunology, 16, 1723596. https://doi.org/10.3389/fimmu.2025.1723596
- Seiler, D. L., Kähler, K. H., Kleingarn, M., Sadik, C. D., Bieber, K., Köhl, J., Ludwig, R. J., & Karsten, C. M. (2023). The complement receptor C5aR2 regulates neutrophil activation and function contributing to neutrophil-driven epidermolysis bullosa acquisita. Frontiers in Immunology, 14, 1197709. https://doi.org/10.3389/fimmu.2023.1197709
- Seiler, D. L., Kleingarn, M., Gilani, S. Z., Reichel, P. E., Köhl, J., & Karsten, C. M. (2022). The Importance of C5aR2 in Neutrophil Function and Its Impact on Neutrophil-mediated Diseases. Journal of Cellular Immunology, 4(6), 194-201. https://doi.org/10.33696/immunology.4.150
- Sino Biological. (n.d.). Complement Activation Pathways.
- Sino Biological. (n.d.). Complement System and Coagulation.
- The Complement Cascade: Molecular Mechanisms of Activation, Regulatory Networks, the Complosome, and Next-Generation Therapeutics. (n.d.).
- The Controversial C5a Receptor C5aR2: Its Role in Health and Disease – PMC. (n.d.).
- The Lectin Pathway of the Complement System—Activation … (n.d.).
- The National PNH Service. (n.d.). Complement Inhibition.
- The complement system: history, pathways, cascade and inhibitors – PMC. (n.d.).
- Trageser, J. (2026). Complement Inhibitors as Therapeutic Agents for Autoimmune Disease. BioSupply Trends Quarterly.
- Vincent, D., Parsopoulou, F., Martin, L., Gaboriaud, C., Loules, G., Cichon, S., Germenis, A. E., Ghannam, A., & Drouet, C. (2024). Hereditary Carboxypeptidase N deficiency, a clinical situation presenting with urticaria and angioedema. Authorea. https://doi.org/10.22541/au.170668305.57822603/v1
- Wikipedia. (n.d.). Complement system.
- Woodward, L. (2024). Why the kidney – blame the complosome? aHUS Alliance Action.
- Zhang, T., Wu, K., Ma, N., Wei, L., Garstka, M., Zhou, W., & Li, K. (2020). The C5a/C5aR2 axis promotes renal inflammation and tissue damage. JCI Insight, 5(7), e134081. https://doi.org/10.1172/jci.insight.134081