Immunoglobulin E (IgE) is a class of antibody found only in mammals. It is present in very small amount in blood but it is highly active. It mainly takes part in allergic reactions and defence against parasitic infections.
Immunoglobulin E (IgE) is a type of antibody that is present only in mammals. It plays an important role in immune system. It is the least abundant antibody in blood circulation but it is very powerful in biological action.
IgE is present in very small amount in human blood, about 0.002% of total immunoglobulins. It has very short half-life in blood, generally less than one day. But it can remain in body tissues for weeks or months because it attaches strongly with some immune cells.
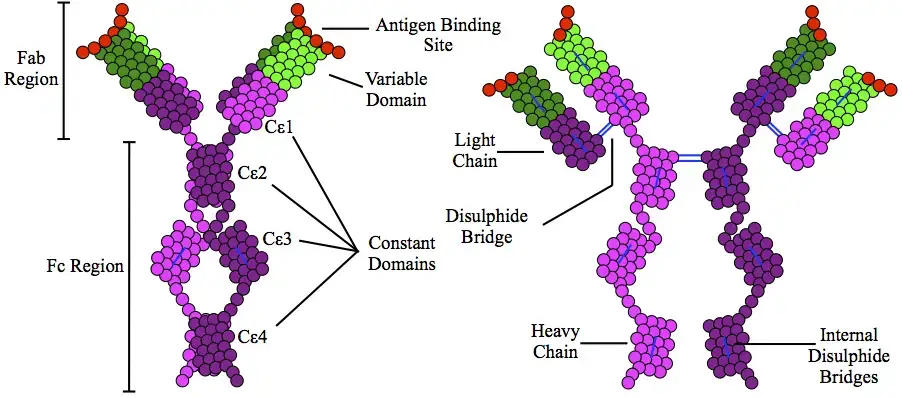
Structurally, IgE is a monomeric glycoprotein. It is made up of two identical heavy chains called epsilon chains and two identical light chains. Unlike IgG, it has no flexible hinge region. In place of hinge region, it contains a fourth constant heavy chain domain called Cε2.
This structure makes the IgE molecule compact, bent and asymmetrical. IgE is also highly glycosylated. It means sugar chains are attached with the molecule. These sugar chains form about 12% of its molecular weight and help in maintaining stability and receptor binding.
IgE mainly acts by binding with two receptors. These are high affinity receptor FcεRI and low affinity receptor CD23. FcεRI is present mainly on mast cells and basophils. CD23 is present on B cells and some other immune cells.
Due to strong affinity for FcεRI, most of the IgE produced in the body becomes attached to these cells. When any specific antigen or allergen enters the body, it binds with cell attached IgE molecules. This cross-linking activates the cell and causes degranulation.
During this process, inflammatory mediators like histamine, prostaglandins and leukotrienes are released into the surrounding tissue. These substances produce allergic and inflammatory reactions. This reaction is very rapid and strong.
Originally, IgE developed as a protective immune factor against parasitic infections like helminth worms and protozoa. It also helps in protection against environmental toxins and animal venoms. But in human disease, IgE is mainly known for allergic reactions.
IgE is the major antibody involved in Type I immediate hypersensitivity reaction. It causes conditions like allergic asthma, allergic rhinitis, food allergy and severe anaphylaxis. In some cases, self-reactive IgE is also associated with autoimmune diseases like lupus and bullous pemphigoid.
Some studies also indicate that IgE may have a protective role in cancer immune surveillance. Its strong inflammatory response may help in detection and destruction of cancer cells. Thus IgE is both protective and pathological antibody depending on the condition.
History and Discovery of IgE
- IgE is an antibody that evolved early during mammalian evolution. It is thought to be formed from IgY gene duplication event. It is unique because it is found in all mammals.
- The basic components for IgE-mediated allergic reaction were present about 360 million years ago. The first allergens were probably derived from pollens, spores and insects which were present in the diet of ancestral mammals.
- The original function of IgE was mainly protective. It developed as a defence mechanism against parasitic infections such as helminth worms. It also helped in protection against environmental toxins.
- IgE was the last antibody class discovered among the five main classes of human antibodies. Before this, other immunoglobulins like IgG, IgA, IgM and IgD were already known.
- In 1966, the research group of Ishizaka in Japan identified a new type of immunoglobulin. This antibody was able to produce allergic skin reaction. It was first named as γE antibody.
- In 1968, this newly identified antibody class was officially named as Immunoglobulin E (IgE). After this naming, it was accepted as the fifth major class of human immunoglobulin.
- In 1981, Margie Profet proposed the toxin hypothesis. It was later expanded in 1991. According to this hypothesis, IgE-mediated acute allergic reactions evolved to remove harmful toxins and animal venoms quickly from the body.
- This reaction also helps the host to avoid the same harmful substances again. Thus, allergy was not only a disease condition in early evolution, but also a protective response against dangerous environmental materials.
General Characteristics of IgE Antibodies
- Immunoglobulin E (IgE) is an antibody isotype which is found only in mammals. It is not found in other groups of animals.
- IgE is a monomeric glycoprotein. Its total molecular weight is about 190 kDa.
- IgE is composed of two identical heavy chains and two identical light chains. The heavy chains are called epsilon chains and each heavy chain is about 70 kDa.
- The light chains of IgE may be kappa or lambda type. Each light chain is about 23 kDa.
- IgE has two identical antigen binding sites. So, its valency is 2.
- Unlike many other antibodies, IgE does not contain a flexible hinge region. Instead of hinge region, its heavy chain has one variable domain and four constant domains.
- The constant domains of IgE heavy chain are Cε1, Cε2, Cε3 and Cε4. The presence of Cε2 domain gives a special structure to the molecule.
- Due to the Cε2 domain, the IgE molecule bends backward on itself. Thus it becomes compact, bent and asymmetrical in shape.
- IgE is highly glycosylated antibody. Complex sugar chains form about 12% of its total molecular weight.
- Each heavy chain of IgE contains seven N-linked glycosylation sites. These sugar chains are important for structural stability and binding with receptors.
- IgE is the least abundant antibody isotype in human blood. It forms only about 0.002% to 0.05% of total circulating immunoglobulins.
- In healthy individuals, the normal concentration of IgE is generally about 50-200 ng/mL. Even in low amount, it is biologically very active.
- IgE has very short half-life in blood circulation. It may remain in blood for less than one day to about 3 days.
- In tissues, IgE can persist for weeks or even months. This occurs because it binds very tightly with cellular receptors.
- IgE mainly binds with two receptors. These are high-affinity receptor FcεRI and low-affinity receptor CD23 or FcεRII.
- IgE shows allosteric receptor binding. When it binds with one receptor, its shape is changed and it prevents binding with another receptor.
- IgE does not activate the classical complement pathway. This is different from IgG and IgM, which can activate complement system.
Detail Structure of Immunoglobulin E (IgE)
- Immunoglobulin E (IgE) is a monomeric glycoprotein antibody. It has total molecular weight of about 190 kDa.
- IgE is made up of four polypeptide chains. These are two identical epsilon (ε) heavy chains and two identical light chains.
- Each epsilon heavy chain has molecular weight of about 70 kDa. The light chains may be kappa (κ) or lambda (λ) type and each is about 23 kDa.
- The heavy chains and light chains are joined together by disulfide bonds. These bonds are present between the chains and also within the chains.
- The disulfide bonds help in giving proper folded structure to the molecule. They also divide the chains into separate folded regions called domains.
- Each light chain of IgE has two domains. These are one variable domain (VL) and one constant domain (CL).
- Each heavy chain of IgE has five domains. These are one variable domain (VH) and four constant domains called Cε1, Cε2, Cε3 and Cε4.
- The VH and VL domains are present at the N-terminal end of the molecule. These two domains together form the antigen binding region.
- IgE has two identical antigen binding sites. So, the valency of IgE is 2.
- The main structural feature of IgE is absence of hinge region. It does not contain the flexible proline rich hinge region which is present in antibodies like IgG.
- In IgE, the hinge region is replaced by an extra constant domain called Cε2. This makes the structure different from other antibody classes.
- Due to the presence of Cε2 domain, the IgE molecule folds backward on itself. The Cε2 domains bend back about 40° to 50° towards the Cε3 and Cε4 domains.
- This backfolding gives IgE a compact and bent structure. It is not a very flexible extended Y-shaped molecule like some other antibodies.
- The shape of IgE is compact, asymmetric and rigidly bent. This special shape is important for its receptor binding and biological action.
- IgE is highly glycosylated antibody. Complex carbohydrate chains are attached with the molecule and they form about 12% of its total molecular weight.
- Each heavy chain has seven N-linked glycosylation sites. These sugar chains help in maintaining the stability of IgE structure.
- Some carbohydrate groups are very important for receptor binding. The oligomannosidic glycans at Asn394 in the Cε3 domain are needed for structural stability and binding with effector receptors.
- The tail region of IgE is called the Fc region. It is formed by the constant domains Cε2-Cε4 and it is mainly responsible for receptor binding.
- The high affinity receptor FcεRI binds mainly with the Cε3 domain. This binding occurs when the Cε3 region is in open conformation.
- The low affinity receptor CD23 binds to a different site. This site is present between the Cε3 and Cε4 domains and it is related with closed conformation.
- The Cε2 domain also has allosteric function. It helps to stabilize the Cε3 domain and prevents the antibody from detaching from FcεRI receptor.
Step by Step Process of Synthesis and Production of IgE
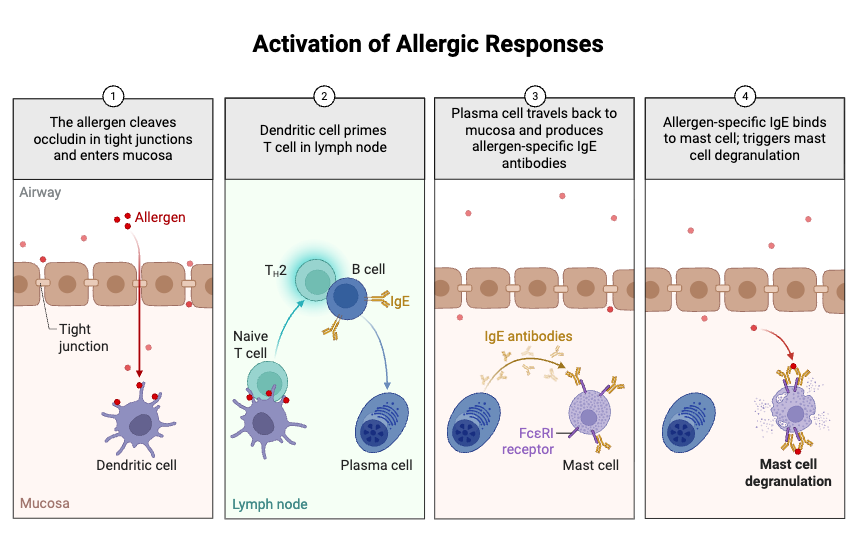
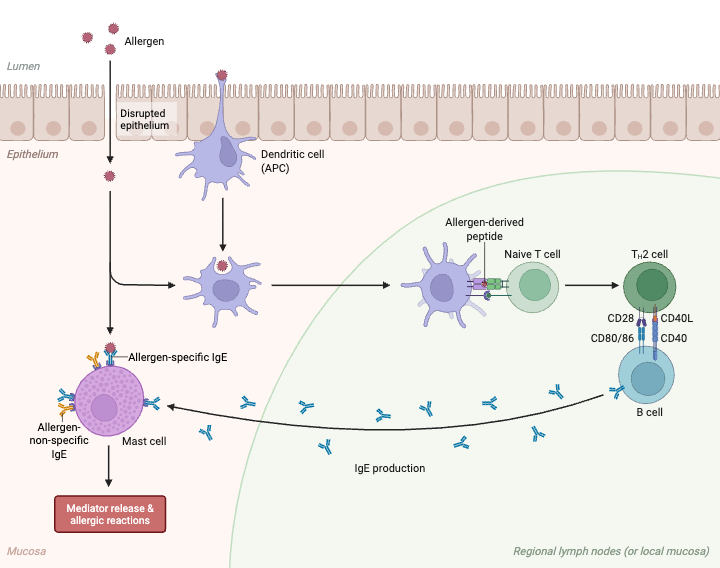
- Allergen enters the body and comes in contact with epithelial cells. The epithelial cells release signalling molecules called alarmins.
- Dendritic cells take up the allergen and process it. The processed allergen is then presented to naive T cells.
- Naive T cells become activated after antigen presentation. They mainly form T helper 2 (Th2) cells and T follicular helper (Tfh) cells.
- Th2 cells and Tfh cells move to the germinal centres of lymphoid organs. Here they interact with B cells.
- Activated Th2 and Tfh cells secrete cytokines. The main cytokines are Interleukin-4 (IL-4) and Interleukin-13 (IL-13).
- IL-4 and IL-13 bind with their receptors on naive B cells. This binding starts internal signalling inside the B cell.
- The JAK1/JAK3 and STAT6 pathways are activated inside the B cell. STAT6 enters the nucleus and starts transcription of germline constant epsilon (Cϵ) transcripts.
- The transcription of Cϵ transcripts opens the chromatin at the IgE gene locus. This makes the epsilon constant region ready for class switching.
- A second signal is given by direct contact between CD40 on B cell and CD40 ligand (CD40L) on activated T cell.
- The CD40-CD40L interaction activates the enzyme Activation-Induced Cytidine Deaminase (AID) in the B cell.
- AID acts on exposed single stranded DNA of switch regions. It changes cytosine residues into uracil.
- The DNA repair system removes these uracil residues. This produces double strand breaks in the DNA.
- During class-switch recombination, the DNA loop carrying other constant region genes such as Cμ, Cδ, Cγ and Cα is removed.
- The antibody variable region is joined directly with the Cϵ gene. This joining takes place by non-homologous end joining (NHEJ).
- The B cell is now permanently switched to produce IgE. The antigen specificity remains same but the antibody class becomes epsilon type.
- Mature Cϵ transcription and alternative splicing occurs. Proper mRNA for IgE synthesis is formed.
- The activated B cell differentiates into a plasma cell. Plasma cell synthesizes large amount of Immunoglobulin E (IgE).
- Final IgE antibodies are secreted into the blood. These antibodies then bind with FcεRI receptors on mast cells and basophils.
Distribution Process of IgE in the Body
- Immunoglobulin E (IgE) is first produced by activated plasma cells. After production, it is secreted in soluble form into the blood circulation.
- In blood, IgE is present in very low amount. In healthy person, its level is generally about 50 to 200 ng/mL.
- IgE forms only about 0.002% of total circulating antibodies. So, it is the least abundant antibody in human blood.
- Free IgE does not stay for long time in blood. Its half-life is very short, from less than one day to about 2 to 3 days.
- The rapid clearance of IgE occurs because free circulating IgE is removed from blood. Some amount is destroyed inside endosomes.
- Most of the IgE produced in the body is quickly captured from the blood. This occurs due to very strong affinity of IgE for FcεRI receptors.
- FcεRI receptors are mainly present on mast cells and basophils. Mast cells are mostly present in tissues and basophils are present in blood.
- After binding with FcεRI, IgE becomes firmly attached to these immune effector cells. This is called cellular sequestration of IgE.
- Bound IgE separates very slowly from its receptor. Due to this slow dissociation, it remains attached on the cell surface for long time.
- In tissues, receptor-bound IgE may persist for several weeks or even months. This helps in producing long lasting sensitization of the body.
- This tissue-bound IgE is important in allergic reaction. When same allergen enters again, it binds with cell attached IgE and activates mast cells and basophils.
- IgE is also present in membrane-bound form on the surface of B cells. This form is called membrane IgE (mIgE).
- mIgE acts as a receptor on B cells. It helps in antigen uptake and antigen presentation.
- Thus, IgE distribution is mainly of two types. One is soluble IgE in blood and another is receptor-bound IgE on immune cells.
- The main amount of IgE is not free in blood but remains attached with mast cells, basophils and some B cells. This makes IgE a very active antibody even when its blood concentration is very low.
IgE Receptors and Their Types
- FcεRI- FcεRI is the high-affinity receptor of Immunoglobulin E (IgE). It binds with IgE with very high affinity and locks it on immune cells for weeks or months.
- Tetrameric FcεRI (αβγ₂) is a four part form of the receptor. It is found almost only on mast cells and basophils. It triggers immediate allergic reactions, degranulation and anaphylaxis.
- Trimeric FcεRI (αγ₂) is a three part form of the receptor. It lacks the beta subunit and is found on antigen presenting cells like dendritic cells, macrophages, monocytes, also on eosinophils and some epithelial cells.
- Soluble FcεRI (sFcεRI) is a free-floating form present in blood serum. It acts as endogenous decoy by binding free IgE and prevents IgE from attaching to cellular receptors.
- CD23 or FcεRII- CD23 is the low-affinity receptor of IgE. It binds with IgE with much lower affinity and regulates IgE production. It requires IgE to fold into closed shape, so it cannot bind to same IgE molecule at the same time with FcεRI.
- CD23a is normally expressed on the surface of B cells. It helps in antigen presentation and regulates the synthesis of new IgE.
- CD23b is induced by inflammatory cytokines. It is present on macrophages, monocytes and epithelial cells and helps in phagocytosis and cytokine release.
- Soluble CD23 (sCD23) is formed when membrane receptor is cleaved off. It circulates in the body and further modulates and enhances IgE production by B cells.
- Galectin-3- Galectin-3 is formerly known as ε-binding protein. It is a soluble lectin and unlike other receptors, it does not bind with the protein backbone of IgE. It attaches with complex carbohydrate or sugar chains present on the IgE molecule.
- Galectin-3 cross-links IgE complexes and increases activation of mast cells and basophils.
- It also acts as adhesion molecule and helps to recruit eosinophils into inflamed tissues.
Mechanism of IgE-Mediated Immune Response
- Immunoglobulin E (IgE) antibodies are first produced by the immune system and they attach firmly with high-affinity FcεRI receptors present on the surface of mast cells and basophils.
- The mast cells are mainly present in tissues and basophils are present in blood circulation. After attachment with FcεRI, IgE remains fixed on these cells for long time.
- When the same allergen enters the body again, it binds with the cell attached IgE molecules. This allergen generally has more than one binding site.
- The allergen joins two or more nearby IgE antibodies and their FcεRI receptors together. This is called cross-linking.
- The cross-linking of IgE-FcεRI starts internal signaling inside the mast cell or basophil.
- During this process, enzymes like Lyn kinase are activated. The intracellular calcium level also increases rapidly.
- The signaling reaction causes secretory granules of the cell to fuse with the cell membrane. This produces immediate degranulation.
- During degranulation, stored inflammatory mediators are released into surrounding tissues. These include histamine, heparin, tryptase and chymase.
- At the same time, the activated cell starts new synthesis of lipid mediators. The major lipid mediators are prostaglandin D2 (PGD2) and leukotriene C4 (LTC4).
- The signaling pathway later reaches the nucleus of the cell. It activates gene transcription for delayed inflammatory mediators.
- After this, pro-inflammatory cytokines and chemokines are produced and released. The important cytokines are IL-4, IL-5, IL-13 and TNF-α.
- These released chemicals cause smooth muscle contraction, vasodilation and increased vascular permeability.
- They also recruit other inflammatory immune cells to the affected area. This increases local inflammation and allergic symptoms.
- Thus, IgE-mediated immune response occurs by IgE attachment, allergen cross-linking, intracellular signaling, degranulation and release of inflammatory mediators.
- This reaction is commonly called Type I allergic reaction and it is responsible for rapid allergic response in the body.
Role of IgE in Allergic Reactions
- Type I hypersensitivity – Immunoglobulin E (IgE) is the antibody which mainly takes part in Type I immediate hypersensitivity. This is seen in allergic asthma, allergic rhinitis, food allergy, atopic dermatitis and anaphylaxis.
- Cell sensitization – In first exposure, allergen enters the body and IgE is formed. These IgE molecules are fixed on FcεRI receptors of mast cells and basophils. The person then become sensitized for that allergen.
- Long stay – The attached IgE does not leave the receptor quickly. It remains on mast cells for weeks or months. Because of this, mast cell remains ready even when allergen is not present.
- Second exposure – When same allergen again enters, it binds with the fixed IgE on the cell. One allergen molecule may join two nearby IgE molecules. This joining of IgE and receptor is called cross-linking.
- Cell activation – Cross-linking starts activation of mast cells and basophils. The cell granules come near the membrane. Then the granules open and their materials are released outside.
- Chemical release – The released substances are histamine, tryptase, heparin, leukotrienes and prostaglandins. Some are already stored in granules and some are newly formed after activation.
- Tissue effect – These chemicals act on blood vessels, smooth muscles and mucous membrane. Smooth muscles contract. Blood vessels dilate and become more permeable. Swelling and redness are produced.
- Asthmatic effect – In respiratory tract, these reactions cause bronchospasm. Mucus secretion also increases. So breathing difficulty is developed in allergic asthma.
- Late reaction – The activated cells also release IL-4 and IL-5. These bring eosinophils and other inflammatory cells to the site. This makes late phase allergy and long lasting inflammation.
Role of IgE in Parasitic Infections
- Evolutionary defence – Immunoglobulin E (IgE) was mainly formed as defence antibody against large parasites. It works with mast cells and basophils. Mostly it acts against helminth worms like Schistosoma mansoni and Trichinella spiralis and also protozoan parasite like Plasmodium falciparum.
- Parasite marking – During infection, parasite specific IgE is produced. These IgE molecules attach with antigen present on the parasite surface. The parasite then become marked for attack.
- Cell attachment – The IgE attached on parasite now binds with FcεRI receptors of immune cells. Mainly mast cells and eosinophils take part here. The cell comes near the parasite.
- Granule release – After this binding, the immune cells are activated. The granules open and their materials are released outside. Histamine is released by mast cells and toxic granule proteins are released by eosinophils.
- Parasite damage – These toxic substances act on the outer covering of parasite. The cuticle is damaged. The parasite become weak and destruction of parasite is started.
- Worm expulsion – Histamine also acts on intestinal smooth muscle. The muscle contracts strongly. Due to this, worms present in intestine are pushed out from the gastrointestinal tract.
- Tick protection – IgE also gives some protection against tick bite. It is seen in some ticks like Ixodid ticks. These ticks may carry Lyme disease organism.
- Evasion by parasite – Some parasites escape this IgE response. They induce large amount of non-specific polyclonal IgE. This fills the mast cell receptors and parasite specific IgE cannot act properly.
- Use of inflammation – Some helminths use IgE reaction for their own benefit. Local allergy causes vasodilation and more vascular permeability. So more nutrients may come and parasite eggs can move through tissue more easily.
Step by Step Detail Mechanism of Action of IgE
- Cell sensitization – Free Immunoglobulin E (IgE) is produced by immune system. These IgE molecules get fixed on FcεRI receptors of tissue mast cells and blood basophils. The binding is very strong. So the cell remains sensitized for weeks or months.
- Allergen entry again – When same allergen enters the body again, it binds with the Fab arms of fixed IgE. One allergen can bind more than one IgE molecule. So two nearby IgE are joined together.
- Receptor clustering – Due to joining of IgE, the FcεRI receptors also come close to each other. This is called cross-linking. This is the main starting point of the reaction.
- Lyn activation – After receptor clustering, Lyn kinase becomes active. It is attached with β-subunit of FcεRI. Fyn kinase also takes part in this early reaction.
- ITAM phosphorylation – Lyn and Fyn phosphorylate ITAMs. These ITAMs are present in the intracellular part of β and γ subunits. After phosphorylation, these sites become active docking places.
- Syk binding – Spleen Tyrosine Kinase (Syk) comes from cytosol. It binds with phosphorylated ITAMs of γ-chain. After this binding, Syk becomes activated.
- Signal spreading – Activated Syk starts more reactions inside the cell. PLC-γ is phosphorylated. Calcium level rises fast inside the cell. Protein Kinase C (PKC) is also activated.
- Granule movement – The rise of calcium and PKC activation acts on secretory granules. The granules move towards plasma membrane. Then they fuse with the membrane.
- Degranulation – After fusion, stored materials of granules are released outside. This is called immediate degranulation. The reaction occurs rapidly after allergen binding.
- Ready mediator release – The released ready-made mediators are histamine, heparin, tryptase, chymase and carboxypeptidase A3 (CPA3). These act directly on nearby tissues.
- Arachidonic acid release – At the same time, cytosolic phospholipase A₂ (cPLA₂) becomes active. It acts on membrane phospholipids. Arachidonic acid is released from the membrane.
- Lipid mediator formation – From arachidonic acid, new lipid mediators are formed. These include prostaglandin D₂ (PGD₂) and leukotriene C₄ (LTC₄). These are formed after activation, not stored before.
- Nuclear signaling – Some signals move up to the nucleus. MAPK, NF-κB and JAK/STAT pathways become active. These start transcription of inflammatory genes.
- Cytokine release – Later, cytokines and chemokines are produced. The main cytokines are IL-4, IL-5, IL-13 and TNF-α. These come late than histamine.
- Tissue effect – The mediators act on blood vessels, smooth muscles and immune cells. Smooth muscle contracts. Blood vessels dilate and become leaky. Other inflammatory cells come to that place.
- Allergic effect – Swelling, redness, itching, mucus secretion and bronchospasm may occur. This is the action of IgE through mast cells and basophils.
Regulation of IgE Production
- IL-4 and IL-13 – Production of Immunoglobulin E (IgE) is mainly started by Interleukin-4 (IL-4) and Interleukin-13 (IL-13). These are secreted by T helper 2 (Th2) cells. They bind with receptors of naive B cells. Then STAT-6 pathway is activated and IgE gene transcription starts.
- CD40 signal – Cytokines alone are not sufficient for complete IgE production. A direct contact is needed. CD40 receptor of B cell binds with CD40 Ligand (CD40L) of activated T cell. This signal helps the B cell to switch toward IgE production.
- Tfh13 cells – In lymphoid organs, T follicular helper 13 (Tfh13) cells control high-affinity IgE formation. This IgE is related with severe allergic reaction. These cells help the B cells to produce more active allergic type IgE.
- Tfr cells – T follicular regulatory (Tfr) cells usually suppress immune response. But here they may promote food antigen-specific IgE production. They release IL-4 and IL-10 and help in this process.
- CD23 feedback – CD23 is the low-affinity receptor of IgE and maintains IgE balance. When IgE binds with membrane CD23 (mCD23) on B cells, it gives negative feedback and suppress more IgE synthesis. But when CD23 is cleaved into soluble CD23 (sCD23), it acts oppositely and with CD21 receptors it increases new IgE production.
- Inhibitory cytokines – Some cytokines stop IgE production. Interferon-gamma (IFN-γ), Transforming Growth Factor-beta (TGF-β) and high level of IL-21 act against IL-4 signaling. They suppress IgE gene transcription and shift B cells more towards IgG production.
- B-cell inhibition – B cells which express IgE on their surface can be directly controlled. Molecules like soluble FcεRIα (sFcεRIα) or special anti-IgE antibodies bind with these cells. This reduces IgE amplification signal or may induce apoptosis to remove the IgE-producing cells.
Factors Affecting IgE Levels
- Stimulatory cytokines – Immunoglobulin E (IgE) level is increased mainly by Interleukin-4 (IL-4) and Interleukin-13 (IL-13). These cytokines act on B cells. Then the B cells are directed to switch into IgE producing cells.
- Inhibitory cytokines – Some cytokines reduce IgE formation. Interferon-gamma (IFN-γ), Transforming Growth Factor-beta (TGF-β) and Interleukin-21 (IL-21) suppress IgE production. These cytokines push B cells more towards IgG production. High level of IL-10 can also decrease IgE response.
- CD40 signal – The contact between CD40 receptor on B cell and CD40 Ligand (CD40L) on T cell is needed for IgE synthesis. Without this contact, the B cell cannot properly start IgE class switching.
- CD23 feedback – CD23 is the low-affinity receptor of IgE. When IgE binds with membrane CD23 (mCD23) on B cells, it gives negative feedback and stops more IgE synthesis. But when CD23 is cut from cell surface and becomes soluble CD23 (sCD23), it moves in circulation and increases new IgE production.
- Age – IgE level changes with age. It is lowest in neonates. Then it rises slowly and reaches high level around 10 years of age. After this, it decreases and becomes more stable in adult life.
- Allergic diseases – In allergic or atopic diseases, IgE level becomes high. This is commonly seen in allergic asthma, allergic rhinitis, food allergy and atopic dermatitis.
- Parasitic infection – Large extracellular parasites increase IgE level strongly. Mainly helminth worms and protozoa produce very high total serum IgE. Here, many types of IgE are formed, so it is called polyclonal IgE response.
- Autoimmune diseases – In some autoimmune diseases, IgE level may also increase. It is seen in Systemic Lupus Erythematosus (SLE), Bullous Pemphigoid (BP) and Chronic Spontaneous Urticaria (CSU). This high IgE works as a sign of immune imbalance.
- Genetic syndromes – Some genetic defects produce very high IgE level. These are called Human hyper-IgE syndromes (HIES). Examples are Job syndrome due to STAT3 mutation, DOCK8 deficiency and PGM3 deficiency. In these cases, IgE may become more than 2000 IU/ml.
- Low class switching – Some genetic condition may reduce IgE formation. Il4 gene haploinsufficiency gives low IL-4 signal. Then the signal is not enough for class switching and IgE production becomes restricted.
- Other diseases – Some other diseases can also increase IgE level. It may occur in cancers like IgE myeloma, Sézary’s syndrome and bronchogenic carcinoma. Viral and bacterial infections like HIV and leprosy may also increase IgE in some cases.
Functions of Immunoglobulin E (IgE)
- Allergic reaction – Immunoglobulin E (IgE) is the main antibody which takes part in Type I immediate hypersensitivity reaction. It is seen in allergic asthma, allergic rhinitis, food allergy and severe anaphylaxis. Here IgE activates mast cells and basophils and allergic symptoms are produced.
- Parasitic defence – IgE acts as defence antibody against large extracellular parasites. It acts mostly against helminth worms and protozoa. IgE binds with parasite antigen and then immune cells release toxic proteins on parasite surface. Muscle contraction also occurs and parasites may be expelled out from body.
- Venom and toxin protection – IgE also gives protection against harmful environmental toxins and animal venoms. It is seen in venoms of bees and vipers. After small exposure, venom-specific IgE is formed and mast cells become ready. During next exposure, proteases are released which degrade and neutralize the venom.
- Cancer defence – IgE may help in tumour immune surveillance. IgE antibodies can bind with tumour-associated antigens. Then immune cells like macrophages and monocytes are activated. These cells attack the tumour cells and help in destruction of solid tumour.
- Autoimmune damage – Sometimes self-reactive IgE is produced. These are IgE autoantibodies and they attack own body tissues. This type of IgE is involved in Bullous Pemphigoid (BP), Systemic Lupus Erythematosus (SLE) and Chronic Spontaneous Urticaria (CSU). Tissue damage and inflammation are produced.
- Immune regulation – IgE also takes part in immune regulation through CD23 receptor. CD23 is the low-affinity receptor present on B cells and other immune cells. It helps in controlling further IgE production, antigen presentation to T cells and transport of allergens across respiratory and intestinal barriers.
Diseases Associated with Elevated or Abnormal IgE Levels
- Atopic and allergic diseases – Elevated Immunoglobulin E (IgE) is commonly found in allergic diseases. These are allergic asthma, allergic rhinitis or hay fever, atopic dermatitis or eczema, allergic keratoconjunctivitis, food allergy and chronic urticaria. It is also seen in eosinophilic gastrointestinal disorders like eosinophilic esophagitis, eosinophilic gastroenteritis, eosinophilic colitis and in Allergic Bronchopulmonary Aspergillosis (ABPA) due to Aspergillus mold.
- Autoimmune diseases – Abnormal IgE level is seen in some autoimmune conditions. These include Bullous Pemphigoid (BP), pemphigus vulgaris, pemphigus foliaceus, Systemic Lupus Erythematosus (SLE) and lupus nephritis. It may also occur in rheumatoid arthritis (RA), systemic sclerosis, Graves’ disease, Hashimoto thyroiditis, multiple sclerosis, autoimmune uveitis, Chronic Spontaneous Urticaria (CSU) and autoimmune pancreatitis.
- Parasitic infections – High IgE is produced during parasitic infection. Mostly helminth worms and protozoan infections increase IgE as defence response. Some examples are paragonimiasis, Tropical Pulmonary Eosinophilia (TPE) due to filarial parasite like Wuchereria bancrofti, Loeffler syndrome and acute toxocariasis or visceral larva migrans.
- Hyper-IgE syndromes and immunodeficiency – Very high IgE is found in Human Hyper-IgE Syndromes (HIES). These include Job syndrome due to Autosomal Dominant STAT3 deficiency, DOCK8 deficiency, PGM3 deficiency and AD-CARD deficiency. Other combined immunodeficiency diseases also show abnormal IgE, such as Wiskott-Aldrich syndrome, DiGeorge syndrome, Omenn syndrome and Comel-Netherton syndrome.
- Neoplasms – Some cancers may show increased or abnormal IgE level. These include IgE myeloma, Sézary’s syndrome, bronchogenic carcinoma, B-cell chronic lymphocytic leukemia and Hodgkin’s disease. In these conditions, IgE rise is not always allergic type but related with abnormal cell growth.
- Other infections and pathologies – IgE may also increase in viral, bacterial and fungal infections. These include HIV, RSV, Epstein-Barr virus (EBV), Cytomegalovirus (CMV), leprosy and systemic candidiasis. Some systemic and organ specific diseases are also associated with abnormal IgE, such as nephrotic syndrome, nephritis, liver disease, cystic fibrosis, Kawasaki disease, Guillain-Barré syndrome and Kimura disease.
Laboratory Detection and Measurement of IgE
- Diagnostic methods – Immunoglobulin E (IgE) is detected and measured by ELISA, RAST and FAST. These are commonly used for specific IgE detection. Solid phase immunoassays like Phadia ImmunoCAP are also used in clinical laboratory.
- Standard units – Serum IgE is measured in International Units per milliliter (IU/mL) or kilo-International Units per liter (kU/L). These units are used for same type of clinical reporting.
- WHO standard – All standard IgE measurements are traceable to World Health Organization (WHO) reference standard. It helps to compare the result between laboratories.
- Mass value – 1 IU/mL is equal to 1 kU/L. This is nearly equal to 2.42 ng/mL in physical mass.
- Total and specific IgE – Laboratory can measure both total serum IgE and allergen-specific IgE. Total IgE is useful in hyper-IgE syndromes and parasitic infections. But normal total IgE does not rule out allergy. So specific IgE is measured to find the exact allergen trigger.
- Cross reaction – Specific IgE test may show cross-reactivity between different antigens. Tropomyosin of house dust mite may cross-react with cockroach and shrimp antigen. So positive test need to be matched with clinical history.
- Age range – Normal IgE level varies with age. In healthy adult, upper reference limit is usually ≤114 kU/L. A value of ≥200 kU/L is considered to have strong predictive value for Type I hypersensitivity or atopy.
References
- Dehlink, E., Platzer, B., Baker, A. H., LaRosa, J., Pardo, M., Dwyer, P., Yen, E. H., Szépfalusi, Z., Nurko, S., & Fiebiger, E. (2011). A soluble form of the high affinity IgE receptor, Fc-Epsilon-RI, circulates in human serum. PLoS ONE, 6(4), e19098.
- Dehlink, E., Platzer, B., Baker, A. H., LaRosa, J., Pardo, M., Dwyer, P., Yen, E. H., Szépfalusi, Z., Nurko, S., & Fiebiger, E. (2011). A soluble form of the high affinity IgE receptor, Fc-Epsilon-RI, circulates in human serum [PDF]. Harvard DASH.
- Reactome Pathway Database. (n.d.). Allergen:p-LYN:p-FCERI:IgE aggregate [plasma membrane].
- National Center for Biotechnology Information. (n.d.). Anti-allergic and anti-inflammatory signaling mechanisms of natural compounds/extracts in in vitro system of RBL-2H3 cell: A systematic review. PMC.
- Biophysical architecture, effector receptor axis, and clinical translation of immunoglobulin E. (n.d.).
- Gönül, M., Keseroglu, H. O., Ergin, C., Özcan, I., & Erdem, Ö. (2016). Bullous pemphigoid successfully treated with omalizumab. Indian Journal of Dermatology, Venereology and Leprology, 82(5), 577–579.
- Liu, J., Xiang, T., Wang, W., & Bu, Z. (2022). Case report: Omalizumab successfully treated recalcitrant bullous pemphigoid in an elderly patient with multiple comorbidities. Clinical, Cosmetic and Investigational Dermatology, 15, 1391–1396.
- Vedantu. (n.d.). Classes of immunoglobulins: Types, functions & quick guide.
- Phillips, B. (2024, February 25). Drug in OUtMATCH clinical trial FDA-approved for the reduction of allergic reactions from accidental food exposures. UNC Health.
- National Center for Biotechnology Information. (n.d.). Evolving mechanistic insights into galectin functions. PMC.
- Roche. (2024, February 15). FDA approves Xolair as first and only medicine for children and adults with one or more food allergies.
- National Center for Biotechnology Information. (n.d.). IL-4 haploinsufficiency specifically impairs IgE responses against allergens in mice. PMC.
- González, A. A. (2021). IgE diseases. Clinical Research Trials, 7.
- National Center for Biotechnology Information. (n.d.). IgE based therapeutics are an emerging modality in cancer treatment. PMC.
- National Center for Biotechnology Information. (n.d.). IgE-neutralizing UB-221 mAb, distinct from omalizumab and ligelizumab, exhibits CD23-mediated IgE downregulation and relieves urticaria symptoms. PMC.
- Slepak, N. (2025, November 20). IgE: The key therapeutic target for allergy treatment. KACTUS.
- MicrobiologyInfo.com. (n.d.). Immunoglobulin E (IgE) – Properties, structure and functions.
- National Center for Biotechnology Information. (n.d.). Immunoglobulin E in health and disease. PMC.
- National Center for Biotechnology Information. (n.d.). Increased IgE serum levels are unrelated to allergic and parasitic diseases in patients with juvenile systemic lupus erythematosus. PMC.
- Datta, S., Chakraborty, D., & De, A. (2023). Ligelizumab: A novel molecule in the management of chronic spontaneous urticaria. Indian Journal of Skin Allergy, 2(1), 14–17.
- Baba, S., Kondo, K., Toma-Hirano, M., Kanaya, K., Suzukawa, K., Ushio, M., Suzukawa, M., Ohta, K., & Yamasoba, T. (2014). Local increase in IgE and class switch recombination to IgE in nasal polyps in chronic rhinosinusitis. Clinical & Experimental Allergy, 44(5), 701–712.
- National Center for Biotechnology Information. (n.d.). Mast cell signaling: The role of protein tyrosine kinase Syk, its activation and screening methods for new pathway participants. PMC.
- National Center for Biotechnology Information. (n.d.). Mast cells and IgE can enhance survival during innate and…. PMC.
- ResearchGate. (n.d.). Mast cells and IgE can enhance survival during innate and acquired host responses to venoms.
- National Center for Biotechnology Information. (n.d.). Mast cells and IgE in defense against lethality of venoms: Possible “benefit” of allergy. PMC.
- Bloom, D. (2026, May 29). New biologic aims to provide long-lasting protection for people with food allergies. SnackSafely.com.
- Macy, E. (2024). Omalizumab & reduction of food allergy associated symptoms. American Academy of Allergy, Asthma & Immunology.
- Food Allergy Research & Education (FARE). (n.d.). Omalizumab (Xolair®) for food allergy: What you need to know.
- National Center for Biotechnology Information. (n.d.). Omalizumab therapy for bullous pemphigoid. PMC.
- Health Research Authority. (n.d.). Phase I study of MOv18 IgE. NHS.
- National Center for Biotechnology Information. (n.d.). Quantitative time-resolved phosphoproteomic analysis of mast cell signaling. PMC.
- ResearchGate. (n.d.). Quantitative measurement of allergen-specific immunoglobulin E levels in mass units (ng/mL): An interlaboratory comparison.
- Alexandre, M. A. M., Bohelay, G., Gille, T., Le Roux-Villet, C., Soued, I., Morin, F., Caux, F., Grootenboer-Mignot, S., & Prost-Squarcioni, C. (2022). Rapid disease control in first-line therapy-resistant mucous membrane pemphigoid and bullous pemphigoid with omalizumab as add-on therapy: A case series of 13 patients. Frontiers in Immunology, 13, 874108.
- Yanagihara, Y. (2003). Regulatory mechanisms of human IgE synthesis. Allergology International, 52, 1–12.
- ResearchGate. (n.d.). Riaz I. Zuberi’s research works | La Jolla Bioengineering Institute and other places.
- Cao, P., Xu, W., & Zhang, L. (2022). Rituximab, omalizumab, and dupilumab treatment outcomes in bullous pemphigoid: A systematic review. Frontiers in Immunology, 13, 928621.
- National Center for Biotechnology Information. (n.d.). Role of IgE receptors in IgE antibody-dependent cytotoxicity and phagocytosis of ovarian tumor cells by human monocytic cells. PMC.
- National Center for Biotechnology Information. (n.d.). Scrodentoid A inhibits mast cell–mediated allergic response by blocking the Lyn–FcεRIβ interaction. PMC.
- Sutton, B. J., & Davies, A. M. (2015). Structure and dynamics of IgE–receptor interactions: FcεRI and CD23/FcεRII. Immunological Reviews, 268, 222–235.
- King’s College London. (2025, April 10). Study shows how new antibody therapy works against ovarian cancer.
- ResearchGate. (n.d.). The 3rd International Standard for serum IgE: International collaborative study to evaluate a candidate preparation.
- Longdom Publishing. (n.d.). The essential role of B cell class switching in mediating allergic reactions.
- National Center for Biotechnology Information. (n.d.). The high affinity IgE receptor FcεRI is expressed by human intestinal epithelial cells. PMC.
- Thaçi, K., Gyorgypal, A., Anthony, R. M., & Conroy, M. E. (2026). The role of immunoglobulin E in non-atopic disorders. Frontiers in Immunology, 16, 1728940.
- National Center for Biotechnology Information. (n.d.). The role of immunoglobulin E in non-atopic disorders. PMC.
- ResearchGate. (n.d.). The role of immunoglobulin E in non-atopic disorders.
- ResearchGate. (n.d.). The structure of the IgE Ce2 domain and its role in stabilizing the complex with its high-affinity receptor FceRI.
- Abe, Y. (2026, May 21). Understanding ligelizumab vs. omalizumab for urticaria: Advanced science. Ubie Doctor’s Note.
- Spillner, E., Coline, S., Christensen, L. H., Andersen, P. S., Lund, G., Johansen, T., Jabs, F., Ballegaard, A.-S. R., & Kemter, A. M. (2024, January 4). Displacers of ige-fceri (Patent No. WO2024003376A1). World Intellectual Property Organization.
- Keown, M. B., Ghirlando, R., Young, R. J., Beavil, A. J., Owens, R. J., Perkins, S. J., Sutton, B. J., & Gould, H. J. (1995). Hydrodynamic studies of a complex between the Fc fragment of human IgE and a soluble fragment of the Fc epsilon RI alpha chain. Proceedings of the National Academy of Sciences, 92, 1841–1845.