ONPG test is a rapid biochemical test used in microbiology to detect the presence of β-galactosidase enzyme in bacteria. It is used to identify organisms which can produce β-galactosidase but may not ferment lactose in routine lactose tests.
Normally lactose fermentation needs two enzymes. One is lactose permease which transports lactose inside the cell. Another is β-galactosidase which breaks lactose into glucose and galactose. Some bacteria have β-galactosidase but they lack permease so they appear as late lactose fermenter (cryptic) or may look like non lactose fermenter in normal media.
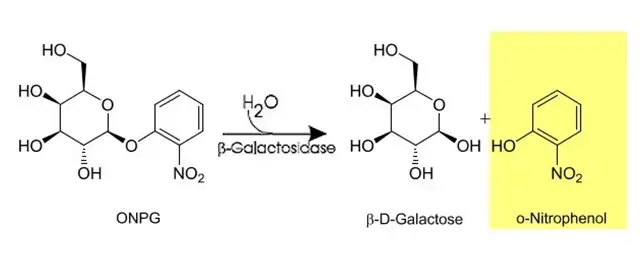
In ONPG test, ONPG (Ortho-nitrophenyl-β-D-galactopyranoside) is used which is a colourless synthetic lactose analogue and it can enter the cell without permease. If β-galactosidase is produced, ONPG is hydrolysed and o-nitrophenol is released. o-nitrophenol gives yellow colour (in alkaline condition) and this yellow colour is taken as positive ONPG test.
Objectives of ONPG Test
- To detect β-galactosidase enzyme activity in the organism.
- To differentiate rapid lactose fermenters, late (slow) lactose fermenters and true non lactose fermenters.
- To identify late lactose fermenters (cryptic lactose fermenters) which have β-galactosidase but lack lactose permease.
- To help in identification of gram negative bacteria mainly Enterobacteriaceae group (like Shigella, Salmonella) and also some other organisms (Neisseria spp.).
Principle of ONPG Test
Principle of ONPG Test is based on showing β-galactosidase enzyme in the organism by using ONPG substrate. In usual lactose fermentation, lactose permease is needed to carry lactose inside the cell and then β-galactosidase breaks it. So permease negative organisms may look as late lactose fermenter or non lactose fermenter in routine lactose test.
In this test, ONPG (Ortho-nitrophenyl-β-D-galactopyranoside) is used which is colourless and it can enter the bacterial cell even without permease. If β-galactosidase is produced, ONPG is split by the enzyme and o-nitrophenol is formed. o-nitrophenol gives yellow colour. This yellow colour formation is taken as positive ONPG and it confirms β-galactosidase activity and helps to separate late lactose fermenters from true non lactose fermenters.
Requirements of ONPG Test
- Pure bacterial culture (18-24 hours) grown on lactose containing media like TSI agar, KIA or MacConkey agar.
- ONPG substrate (ONPG broth) or ONPG differentiation disk.
- Sterile normal saline (0.85% to 0.9% NaCl) for making bacterial suspension.
- McFarland standard (2 or 3) to match turbidity of suspension.
- Sterile test tubes (small size) with caps (example 13×100 mm).
- Sterile inoculating loop and sterile pipette with pipump.
- Incubator (aerobic condition) at 35°C to 37°C.
- Optional reagents in some methods like toluene (1 drop) and phosphate buffer (1.0 M monosodium phosphate buffer, pH 7.0).
- Control strains (positive and negative) for quality control (E. coli / Neisseria lactamica as positive, Proteus mirabilis / Salmonella Typhimurium as negative).
ONPG broth Composition and preparation
Composition (per litre)
- Casein peptone (Casitone) – 7.5 g.
- ONPG (Ortho-nitrophenyl-β-D-galactopyranoside) – 1.5 g to 2.0 g.
- Sodium chloride (NaCl) – 3.75 g.
- Disodium hydrogen phosphate (Na2HPO4) – 0.35 g to 9.46 g (as buffer).
- Phenylalanine – 4.0 g (in some formulations).
- Potassium dihydrogen phosphate (KH2PO4) – 0.907 g (in some formulations).
- Final pH – 7.5 ± 0.2 at 25°C.
Preparation
- Take 1000 ml sterile distilled water in a flask.
- Add the required quantity of all ingredients and mix properly.
- Gentle heating is done just to dissolve the components completely.
- Do not autoclave the medium because ONPG is heat labile and it gets destroyed.
- Sterility is maintained by using sterile water and sterile glasswares or filtration can be done.
- Dispense into sterile test tubes (commonly 5 ml per tube).
- Store in dark place or amber bottle because ONPG is light sensitive and may hydrolyse.
- Store at 2°C to 8°C (refrigerator) or as per lab protocol.
ONPG Test Procedure
Pre induction
- Test organism is grown on lactose containing media (TSI agar / MacConkey agar) for 18 to 24 hours. This is done for inducing β-galactosidase.
Tube (ONPG broth) method
- ONPG broth is brought to room temperature.
- ONPG broth is heavily inoculated with pure culture using sterile loop. Dense suspension is made.
- Tube is incubated aerobically at 35°C to 37°C with loose cap.
- Tube is checked for yellow colour at 1 hour.
- If no colour, incubation is continued and checked up to 24 hours (late lactose fermenters).
Disk method
- Sterile test tube is taken and 0.2 ml to 0.5 ml sterile normal saline is added.
- Heavy inoculum is emulsified in saline to make dense bacterial suspension.
- One ONPG disk is added aseptically and tube is gently shaken. Disk should remain submerged.
- Incubate at 35°C to 37°C.
- Observe yellow colour at 20 minutes, 1 hour and then hourly up to 6 hours.
- If negative at 6 hours, re incubate and check again at 24 hours.
Optional (toluene assisted)
- One drop of toluene is added to bacterial suspension for permeabilizing cell wall.
- Keep for 5 minutes at 37°C.
- Then ONPG solution / ONPG disk is added and incubation and reading is done in same way.
ONPG Test Result
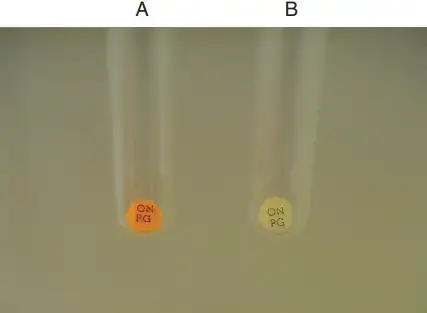
Positive result
- Development of yellow colour in the tube broth or on ONPG disk (any shade of yellow) is seen.
- It indicates β-galactosidase is produced by the organism and ONPG is hydrolysed.
- o-nitrophenol is formed which gives yellow colour.
- Rapid lactose fermenters give yellow colour within minutes to 1 hour.
- Late lactose fermenters may take few hours and can be positive even at 24 hours.
Negative result
- No colour change is seen. Tube or disk remains colourless.
- It indicates β-galactosidase is not produced and ONPG is not hydrolysed.
False positive / invalid
- Yellow pigment producing organisms cannot be tested properly (example Flavobacterium).
- Already yellow colour will be present from beginning and it may look like positive result.
ONPG Test – List of Organisms
Here is a list of ONPG positive and ONPG negative organisms:
ONPG Positive Organisms
- Escherichia coli
- Citrobacter species (including Citrobacter freundii)
- Shigella sonnei
- Klebsiella species (including Klebsiella pneumoniae)
- Serratia marcescens
- Enterobacter aerogenes
- Salmonella arizonae (Salmonella enterica subsp. arizonae)
- Salmonella bongori
- Salmonella enterica subsp. diazonae
- Salmonella Choleraesuis
- Neisseria lactamica
- Vibrio cholerae
- Burkholderia cepacia
- Stenotrophomonas maltophilia
- Shigella dysenteriae (Note: There is a discrepancy in the microbiological literature regarding this organism. Some sources list it as ONPG positive, while others broadly classify it as ONPG negative alongside S. flexneri and S. boydii).
ONPG Negative Organisms
- Proteus species (including Proteus mirabilis and Proteus vulgaris)
- Most Salmonella species (including Salmonella Typhi and Salmonella Typhimurium)
- Other Shigella species (specifically Shigella flexneri and Shigella boydii)
- Providencia species
- Most Pseudomonas species (including Pseudomonas aeruginosa)
- Vibrio parahaemolyticus
- Other Neisseria species (including Neisseria gonorrhoeae and Neisseria meningitidis)
Quality Control of ONPG Test
Positive Control Strains (Produce a yellow color change):
- Escherichia coli (e.g., ATCC 25922)
- Neisseria lactamica (e.g., ATCC 23971)
- Citrobacter freundii (e.g., ATCC 8090)
- Enterobacter aerogenes / Klebsiella aerogenes (e.g., ATCC 13048)
- Salmonella Choleraesuis (e.g., ATCC 12011)
Negative Control Strains (Remain colorless):
- Proteus mirabilis (e.g., ATCC 12453)
- Salmonella Typhimurium (e.g., ATCC 14028)
- Neisseria gonorrhoeae (e.g., ATCC 43069)
- Proteus vulgaris / Proteus hauseri (e.g., ATCC 13315)
Precautions of ONPG Test
- ONPG reagent should be protected from light. ONPG is photosensitive and it may hydrolyse and yellow colour can develop giving false positive. Store ONPG broth / solution / disk in dark place or amber container.
- Before use, ONPG disk or broth should be checked. If yellow discoloration is already present, it should be discarded.
- Test organism should be grown on lactose containing media (MacConkey / TSI) before doing ONPG. This step is needed for inducing β-galactosidase, otherwise false negative may come.
- Pigment producing bacteria should not be used (yellow pigment like Flavobacterium). Natural yellow colour will interfere and gives false positive reading.
- Heavy inoculum should be used. If light inoculum is taken, reaction becomes slow and result may not come properly.
- ONPG broth should not be autoclaved. ONPG is heat labile and it gets destroyed by high temperature. Sterile water and sterile materials are used or filtration is done.
- Stainless steel or nichrome wire loop should be avoided. Oxidation products after flaming may cause false positive reaction.
- In tube method, small volume tubes should be covered properly to prevent drying during incubation.
- o-nitrophenyl compound is phenolic and it should be handled carefully. Inoculated tubes are biohazard and aseptic technique should be followed.
- Positive and negative control should be run along with test organism (E. coli as positive, Proteus mirabilis as negative) to check reagent working.
Uses of ONPG Test
- It is used to identify late lactose fermenters (slow / cryptic lactose fermenters) and to differentiate rapid lactose fermenters, late lactose fermenters and true non lactose fermenters.
- It is used for differentiation of Enterobacteriaceae members. It helps in separating closely related organisms (example Shigella sonnei from other Shigella species, Citrobacter from Salmonella).
- It is used for identification of some non enteric gram negative bacteria. It helps to differentiate Neisseria lactamica from Neisseria gonorrhoeae and Neisseria meningitidis. It is also used to differentiate Vibrio cholerae from Vibrio parahaemolyticus.
- It is used as indicator test for detection of coliforms in drinking water and food samples, and it indicates fecal contamination.
- It is used along with lactose fermentation test to know permease activity of organism (permease presence or absence).
- In clinical chemistry, ONPG reaction is used for quantitative estimation of serum sodium (by sodium dependent β-galactosidase method) and it helps in electrolyte disorder like hyponatremia and hypernatremia.
- ONPG cleavage reaction is used in some diagnostic and research works as reporter enzyme (ELISA / lateral flow). It is also used as marker in senescence studies and in some lysosomal storage disease diagnosis (example GM1 gangliosidosis).
Advantages of ONPG Test
- It gives rapid result. Result may come within 20 minutes to few hours. In lactose fermentation test, late fermenters may take 18 hours to many days.
- It is more sensitive because it detects intracellular β-galactosidase activity directly. So more positive results are obtained than routine lactose tests.
- ONPG substrate can enter the cell without β-galactoside permease. So organisms lacking permease can also be detected and late (cryptic) lactose fermenters are identified easily.
- It gives direct confirmation of functional enzyme. It confirms the organism is producing active β-galactosidase, not only gene presence.
- Clear visual colour reaction is seen. Yellow colour due to o-nitrophenol formation is directly observed and it is easy to interpret than acid pH change in lactose media.
- It is cost effective test. ONPG broth and disks are cheaper than molecular tests and it can be used as screening test.
- ONPG disks are convenient to use. It reduces reagent wastage and gives standard concentration of substrate in each test.
- Principle of ONPG reaction can be used in automated biochemical identification systems, so it is adaptable for automation.
Limitations of ONPG Test
- ONPG test cannot be used properly for yellow pigment producing organisms (Flavobacterium, Cronobacter, Serratia). Natural yellow colour may look like positive and false positive can come.
- Pre induction is required. Organism should be grown on lactose containing media before test. If grown on glucose media, β-galactosidase induction becomes less and false negative may occur.
- ONPG reagent is light sensitive. Exposure to sunlight or long room light can hydrolyse ONPG and yellow tint can develop even without organism, giving false positive.
- It detects only β-galactosidase enzyme. It does not show complete lactose fermentation pathway and actual lactose fermentation in media.
- ONPG test is not a single confirmatory identification test. It should be used along with other biochemical tests and culture media results (MacConkey, TSI etc.) for final identification.
References
- Alfa Chemistry. (2026). β-Galactosidase in in vitro diagnostics: Precision enzymology for clinical sodium detection..
- American Society for Microbiology. (n.d.). Development of a sensitive chemiluminometric assay. ASM Journals..
- A structural view of the action of Escherichia coli (lacZ) beta-galactosidase. (n.d.). PubMed – NIH..
- Beta-galactosidase and lactose fermentation in the identification of enterobacteria including salmonellae. (n.d.). PubMed Central (PMC) – NIH..
- CABI Digital Library. (2014, October 29). Isolation and identification of a new yeast isolate with high beta-galactosidase activity from Syrian dairy products..
- Center for Food Safety and Applied Nutrition. (2025, February 28). BAM R53: ONPG test. U.S. Food and Drug Administration..
- Comprehensive analysis of the ONPG test: Principle, enzymology, clinical application, and diagnostic significance in microbiology. (n.d.)..
- Dahal, P. (2023, March 31). ONPG test- Principle, procedure, results, uses. Microbe Notes..
- Dalynn Biologicals. (2014, October). oNPG disks..
- Direct and indirect roles of His-418 in metal binding and in the activity of β-galactosidase (E. coli). (n.d.). PubMed Central (PMC) – NIH..
- Genera: Shigella and Salmonella. (n.d.)..
- Hardy Diagnostics. (1996). ONPG rapid test broth..
- Hardy Diagnostics. (2020). ONPG differentiation disks..
- Held, P. (n.d.). Kinetic analysis of ß-galactosidase activity using the PowerWave™ HT and Gen5™ data analysis software. BioTek / Agilent..
- HiMedia Laboratories Pvt. Ltd. (2022, January). ONPG technical data (DD008)..
- Juers, D. H., Heightman, T. D., Vasella, A., McCarter, J. D., Mackenzie, L., Withers, S. G., & Matthews, B. W. (2001). A structural view of the action of Escherichia coli (lacZ) β-Galactosidase. Biochemistry, 40(49), 14781–14794. https://doi.org/10.1021/bi011727i.
- Králová, S., Busse, H.-J., Bezdíček, M., Sandoval-Powers, M., Nykrýnová, M., Staňková, E., Krsek, D., & Sedláček, I. (2021). Flavobacterium flabelliforme sp. nov. and Flavobacterium geliluteum sp. nov., two multidrug-resistant psychrotrophic species isolated from Antarctica. Frontiers in Microbiology, 12, 729977. https://doi.org/10.3389/fmicb.2021.729977.
- Measuring lactase enzymatic activity in the teaching lab. (n.d.). PubMed Central (PMC) – NIH..
- Measuring lactase enzymatic activity in the teaching lab. (n.d.). ResearchGate..
- Microxpress. (n.d.). ONPG disc intended use..
- Probiotic potential of β‑galactosidase‑producing lactic acid bacteria from fermented milk and their molecular characterization. (n.d.). PubMed Central (PMC) – NIH..
- Proprep. (2025). Conduct an ONPG test to detect enzyme activity, elucidating its applications in microbiology and biotechnology..
- Remel. (2010, January 11). ONPG broth. Thermo Fisher Scientific..
- Reynolds, J. (2021, August 1). 42: Beta-galactosidase test (ONPG). Biology LibreTexts..
- Richard, J. P., Huber, R. E., Heo, C., Amyes, T. L., & Lin, S. (1996). Structure−reactivity relationships for β-galactosidase (Escherichia coli, lac Z). 4. Mechanism for reaction of nucleophiles with the galactosyl-enzyme intermediates of E461G and E461Q β-galactosidases. Biochemistry, 35(38), 12387–12401. https://doi.org/10.1021/bi961029b.
- Rosco Diagnostica. (n.d.). Screening for Salmonella and Shigella in faecal samples..
- Shigenobu, K., & Aoyama, N. (1998, June 16). Method of quantitative determination of sodium ions (U.S. Patent No. 5,766,870). U.S. Patent and Trademark Office..
- Taylor & Francis. (n.d.). ONPG – Knowledge and references..
- TM Media. (2019, November 8). TM 1918 – ONPG broth..
- UK Health Security Agency. (2025, July 31). Identification of Shigella species (UK SMI ID 20, Issue 4.1)..