The most commonly used equipment is inoculation needles, transfer loops, inoculation, Bunsen burner, autoclave (or pressure cooker) incubators, hot air oven centrifuge, spectrophotometer magnetic stirrer electric shaker and rotary shaker heating plate, heating mantle distillation plant, UV-lamp carbon dioxide cylinder, water-bath and a single-pan balance that has weights (for general use) chemical balance, fine analytical balance pH meters, Quebec colony counter, Laminar air flow, camera lucida electrophoresis and a high-quality microscope and many more. To perform photomicrography, a photomicrographic camera mounted in a microscope equipped with every accessory is essential in the microbiology lab.
The other requirements: A large-sized container to store disposable items is essential in a laboratory for microbiology. Cotton rolls and forceps, scissors, blades, sealing films, cellotape, aluminum foil, parafilm, enamel tray, different kinds of containers including glass marker pen, brush, rubber, pencil measuring scale, Vernier’s Calliper, etc. are employed in laboratories for microbiology. A few of the tools and equipment are described in this document.

List of Instruments used in Microbiology Lab
- Inoculation needle and inoculation loop
- Bunsen burner (Spirit lamp)
- Waterbath
- Autoclave
- Laminar Air Flow
- Incubator
- Hot air oven
- Quebec colony counter
- The pH meter
- Balance
- Spectrophotometer (Colorimeter)
- Centrifuges
- Microscope
- Incinerator
- Deep Freezer (Laboratory Refrigerators and Freezers)
- Homogenizer
- Hot plate
- Magnetic Stirrer
- Vortex Mixture/ Vortexer
- Water Distiller
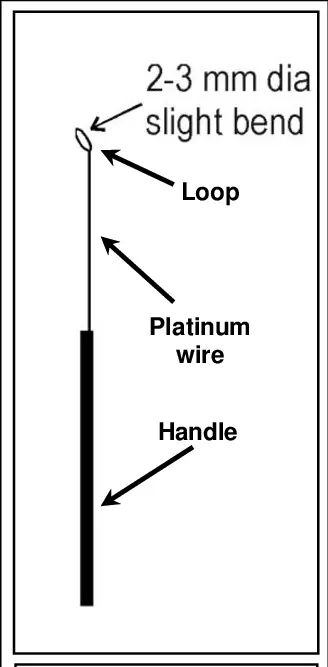
1. Inoculation needle and inoculation loop
- Inoculation needle and inoculation loop are microbiological instruments used to transfer microscopic samples of bacteria to culture medium for development and examination.
- Inoculation loop is a bent wire instrument with a tiny loop at the end, which is used for taking liquid or semi-liquid microbial sample and mostly used for streaking plates.
- During streaking, a small amount of bacterial culture is picked by the loop and spread over the surface of agar medium to isolate single bacterial colonies.
- Inoculation needle is a straight wire instrument with a pointed tip, which is used for transferring solid or dense microbial sample.
- It is mainly used for picking single colony and also used in stab culture, where the organism is inoculated deep into the agar medium.
- These instruments are generally made up of heat resistant materials such as platinum, tungsten or nichrome.
- Before and after use, inoculation loop and needle are sterilized by heating in flame until the wire becomes red hot.
- Sterilization is necessary to prevent contamination and cross-contamination between different cultures.
- Disposable inoculation loop and needle are also available, which are used once and then discarded.
- Disposable types reduce the need of repeated sterilization and helps to maintain aseptic condition in microbiology laboratory.
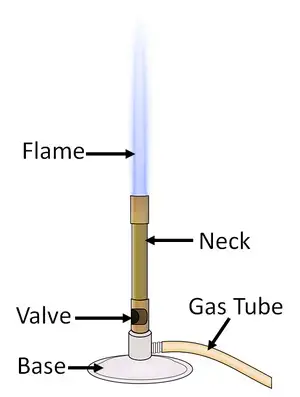
2. Bunsen burner (Spirit lamp)
- Bunsen burner or spirit lamp is a laboratory instrument used to produce flame by burning fuel in presence of oxygen.
- It is used for heating substances, boiling liquids, sterilizing instruments and also for different laboratory experiments.
- Bunsen burner works on the principle of combustion of fuel. In this process, fuel burns in presence of sufficient oxygen and produces heat with steady flame.
- In spirit lamp, ethanol or methanol is generally used as fuel. In Bunsen burner, coal gas or LPG is used as fuel.
- In spirit lamp, the fuel is stored in a small container and the wick absorbs the fuel. When the wick is ignited, the flame is produced.
- In Bunsen burner, the gas passes through the barrel and comes out from the top of burner. It is then lighted by match stick or spark.
- Air is mixed with the fuel before burning. This air mixing takes place through small holes or air inlets present at the lower part of burner.
- Proper mixing of air and fuel helps in complete combustion. It produces a hotter and cleaner flame.
- Bunsen burner has an adjustable collar which controls the amount of air entering into the burner.
- When more air is allowed, blue non-luminous flame is produced. This flame is hotter and suitable for heating and sterilization.
- When less air is allowed, yellow luminous flame is produced. This flame is less hot and may produce soot.
- The heat from flame is transferred to the object kept above the burner. It is used to heat test tubes, beakers, flasks and other laboratory materials.
- It is used to sterilize metal instruments like inoculating loop, inoculating needle, spatula and forceps by passing them through flame.
- It is also used for boiling water and chemical solutions during preparation and sterilization work.
- Bunsen burner or spirit lamp is used for evaporation of solvents by gentle heating, where liquid part is removed and solid residue remains.
- It is used in flame test. In this test, chemicals are introduced into flame and metal ions are identified by their characteristic flame colour.
- It is used to melt low melting point solids and to start some reactions which need initial heat for activation.
- In microbiology laboratory, Bunsen burner helps to maintain sterile field. The flame produces upward air current which reduces airborne contamination near working area.
- The flame must be handled carefully during use. Excess air intake may cause flame blowout and fuel spilling may cause fire.
- The efficiency of flame depends on proper fuel and air ratio. So, adjustment of air holes in Bunsen burner or wick length in spirit lamp changes the quality and heat of flame.
3. Water bath
- Water bath is a laboratory instrument used for heating samples gently by using heated water.
- It is used when direct flame may damage the chemical compound or biological sample.
- Water bath works on the principle of transfer of heat through water. The container with sample is placed inside heated water and the temperature remains uniform and stable.
- Water is heated by an electric coil or sometimes by gas burner present below the bath.
- Heat is first transferred to water by conduction and then it spreads in all directions through the water.
- A thermostat or regulator is used in water bath to control the temperature.
- The thermostat turns the heating on or off and keeps the water at a fixed set temperature.
- This prevents overheating of the sample and helps to get accurate result.
- During heating, convection currents are formed in water. These currents move the water continuously and keep all parts at same temperature.
- Water bath gives indirect heating. The sample is not in direct contact with flame or heating element.
- This prevents burning, damage or chemical breakdown of the sample due to high temperature.
- Water has high specific heat capacity, so it can hold temperature for long time.
- Small changes in heating does not affect the sample much because water gives thermal stability.
- Water bath is safer than direct flame because no open flame is generally involved.
- It reduces the risk of fire and also avoids sudden rise of temperature.
- Water bath is used for incubation of cultures in microbiology laboratory by maintaining required temperature for bacterial or fungal growth.
- It is used for melting substances like fats, waxes, agar and gelatin slowly without burning or decomposition.
- It is used for dissolving solids in liquids by maintaining warm temperature for longer time.
- It is used in enzyme reaction to keep enzyme solution at optimum temperature without denaturation.
- It is used for thawing frozen biological or chemical samples slowly to preserve their integrity.
- It is useful for maintaining constant temperature for long period in experiments like chemical rate studies.
- It is also used for slow evaporation of solvents, where solvent is removed without overheating the sensitive compounds.
4. Autoclave
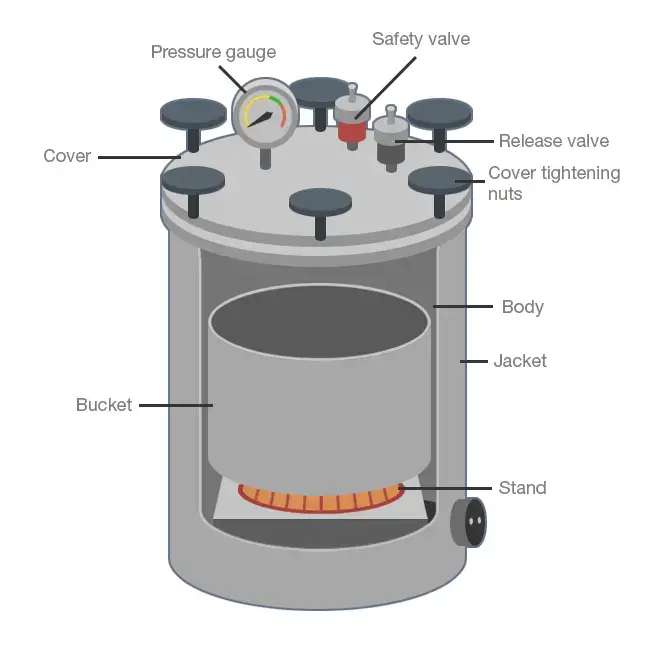
- Autoclave is a laboratory instrument used for sterilization of materials by using steam under high pressure.
- It works on the principle of moist heat sterilization. In this process, steam under pressure kills microorganisms by denaturing proteins and breaking the cell structures.
- Water present inside the chamber or in separate boiler is heated until it forms saturated steam.
- The steam fills the inner chamber and removes the air present inside it.
- When the steam is trapped inside the closed chamber, pressure is increased.
- The pressure is generally about 15 psi above atmospheric pressure, which helps the water to boil at higher temperature.
- Due to increased pressure, the temperature of steam reaches about 121°C or more in some autoclaves.
- Moist heat at this temperature is more effective than dry heat because steam can penetrate the materials properly.
- Steam condenses on the surface of instruments, glassware or media and releases large amount of latent heat.
- This rapid heat transfer helps in even sterilization of all exposed parts of the materials.
- The materials are kept at required temperature for fixed time, usually 15 to 20 minutes.
- Longer time is needed for large loads and for destruction of highly resistant spores.
- After sterilization, pressure is released slowly to avoid damage of materials and sudden boiling of liquids.
- The materials are allowed to cool down before opening the lid of autoclave.
- The effectiveness of autoclave depends on proper temperature, saturated steam, correct pressure and sufficient exposure time.
- Autoclave is used for sterilizing laboratory tools like glassware, pipettes, petri plates and metal forceps so that microbes and spores are removed.
- It is used for sterilizing culture media like nutrient agar, broth and selective media before use.
- It is used for treatment of biohazard waste like used culture plates, contaminated gloves and infectious laboratory waste before disposal.
- It is used for sterilizing surgical instruments like scalpels, scissors, clamps and other hospital tools to prevent infection.
- It is used for sterilizing dressings, gowns, bandages and drapes by steam under pressure.
- It is used for sterilizing laboratory liquids like buffers and water, so that microbial life is removed from them.
- Test tubes, flasks, bottles and other containers are sterilized in autoclave for safe reuse without contamination.
- It is essential in microbiology and molecular biology laboratory to keep culture work sterile and to prevent unwanted organisms in experiments.
5. Laminar Air Flow
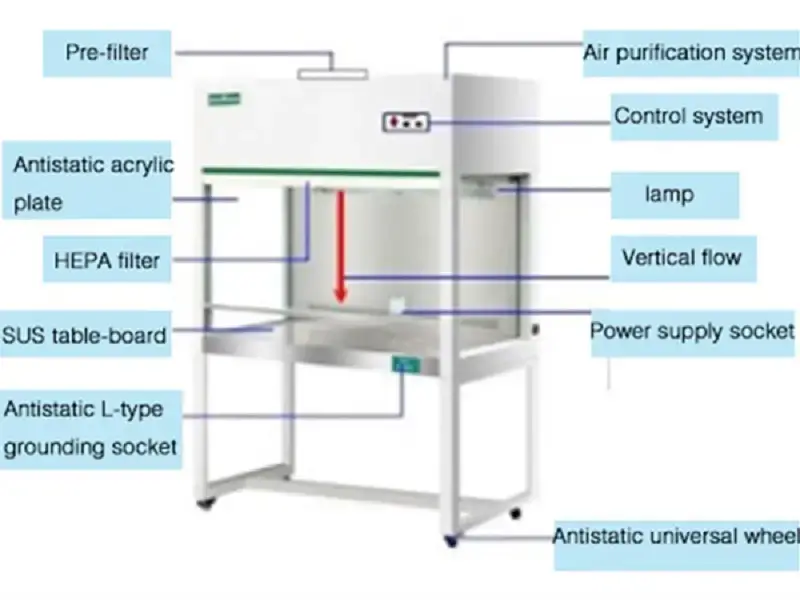
- Laminar Air Flow is a laboratory instrument used to provide clean and sterile working area by passing filtered air into the workspace.
- It is used in microbiology, molecular biology, plant tissue culture and other works where contamination should be avoided.
- Laminar Air Flow works on the principle of forcing HEPA filtered air into the working chamber in parallel and unidirectional stream.
- The air moves in one direction with low turbulence, so the contaminants are carried away from the working area.
- Room air is first drawn inside the cabinet by a blower.
- The large dust particles are removed by pre-filter before the air reaches the main filter.
- The air then passes through HEPA filter, which removes very small particles and many microorganisms from the air.
- HEPA filter can capture particles of about 0.3 µm with nearly 99.97% efficiency.
- The particles are trapped by impaction, interception and diffusion during filtration process.
- After filtration, clean air is passed through a perforated plenum or honeycomb surface.
- This helps to produce parallel air streams with controlled velocity and less turbulence.
- The direction of air flow may be horizontal or vertical depending on the type of laminar air flow.
- In horizontal laminar air flow, the air moves from back side to front side of the cabinet.
- It gives product protection but the operator is not properly protected, so it is not used for hazardous work.
- In vertical laminar air flow, the air moves from top to bottom over the working surface.
- It gives downward sweep of clean air and helps in better protection of the product in many laboratory works.
- A sterile field is created inside the cabinet by continuous flow of filtered air.
- The clean air curtain isolates the working zone and removes particles instead of mixing them with the sample.
- The blower runs continuously to maintain proper face velocity inside the cabinet.
- Pressure drop and air velocity are monitored and alarm may indicate when the air flow becomes poor or filter needs changing.
- The working efficiency depends on uniform air flow, intact HEPA filter, proper face velocity and regular maintenance.
- Laminar Air Flow is used for aseptic preparation of samples and handling microbial cultures without airborne contamination.
- It is used for pouring nutrient agar plates, filling broth tubes and preparation of other culture media in sterile condition.
- It is used in plant tissue culture for transfer and subculturing of plant explants, preventing fungal and bacterial infection.
- It is used in pharmaceutical work for preparation of sterile drugs, injections and IV fluids.
- It is used for handling cell cultures, where mammalian or bacterial cells are protected from dust and microorganisms.
- It is used for sterile packaging of small instruments, medical devices and research materials before sealing.
- It is used in diagnostic testing where clinical samples like blood, swabs or other fluids are processed in sterile condition.
- It is also used in research experiments like genetic engineering, cloning and PCR setup where particle free environment is required.
6. Incubator
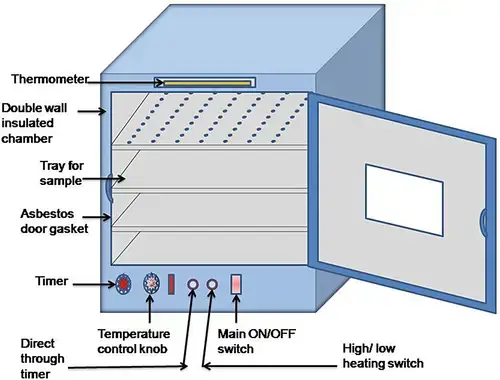
- Incubator is a laboratory instrument used to maintain controlled condition for growth of microorganisms and cells.
- It maintains proper temperature, humidity and sometimes CO₂ for supporting biological growth.
- Incubator works on the principle of keeping physical and chemical conditions steady inside the chamber.
- This stable condition helps the experiment to be repeated and gives proper growth of cultures.
- Heat is supplied by electric heater or water jacket present inside the incubator.
- Heat is transferred to the chamber air and shelves by conduction and convection.
- The temperature is raised up to the set point and then maintained at that level.
- Temperature is detected by probes like thermocouple or RTD.
- A thermostat or microprocessor controller controls the heating system by switching it on or off.
- This keeps the temperature within fixed limit and prevents overheating of the cultures.
- Humidity is maintained by water tray or built-in humidifier present inside the incubator.
- It adds water vapour into the chamber and prevents the culture media from drying.
- Air circulation is done by fans or by passive convection.
- The warmed air is distributed uniformly inside the chamber, so all shelves get same condition.
- In CO₂ incubator, carbon dioxide is supplied from cylinder or generator.
- CO₂ sensor measures the concentration and helps to maintain proper CO₂ level inside the chamber.
- CO₂ is generally kept at 5% for mammalian cell culture, which helps to maintain pH of bicarbonate buffered medium.
- Sterility is maintained by HEPA filtered air, UV lamp or periodic decontamination cycle.
- The inner surface is cleaned regularly and aseptic technique is followed to reduce contamination.
- The walls of incubator are insulated and the door has tight seal to reduce heat loss.
- Alarms and over-temperature cut off are present to protect the samples and also for safety.
- Incubator is used in microbiology laboratory for growing bacteria, fungi and yeast at optimum temperature.
- It is used in cell culture work for growth of mammalian or plant cells under controlled condition.
- It is used for egg hatching in poultry farms by maintaining proper temperature and humidity until chicks are formed.
- It is used in biochemical testing like enzyme assay, fermentation study and metabolic activity test at fixed temperature.
- It is used in genetic research for maintaining genetically modified organisms during transformation or gene expression work.
- It is used in pharmaceutical stability test, shelf-life study and microbial limit testing under controlled condition.
- It is used in food and dairy testing where milk, cheese, meat and other samples are incubated to detect microbial contamination.
- It is also used for environmental simulation to study microbial ecology, adaptation and stress response under controlled environment.
7. Hot air oven
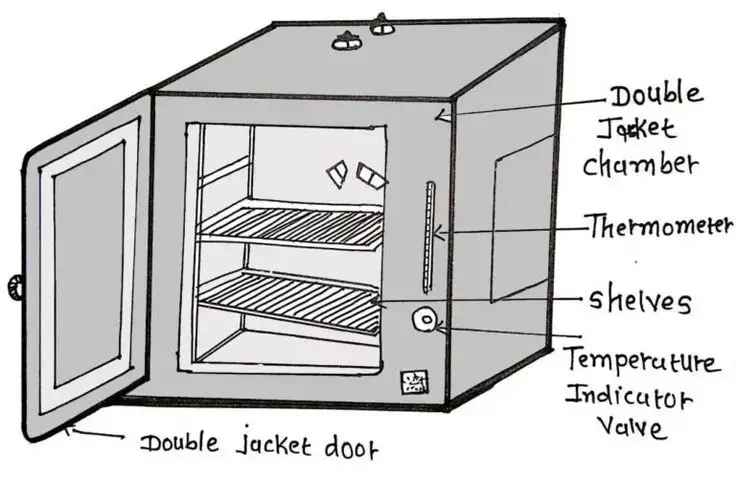
- Hot air oven is a laboratory instrument used for sterilization of materials by using dry heat.
- It works on the principle of dry heat sterilization, where hot air is circulated inside a closed chamber.
- The hot air kills microorganisms by oxidative damage, dehydration and denaturation of proteins.
- In this method, moisture is not present, so heat transfer is slow but it is very effective for dry materials.
- Heat is produced by electric heating elements present inside the oven.
- The electric energy is converted into heat energy and the chamber wall becomes hot.
- Heat is then transferred to the air inside the chamber and the materials are heated by conduction and convection.
- Fans or natural convection helps to move hot air around the shelves.
- Uniform distribution of hot air is necessary because uneven temperature may leave some microorganisms alive.
- The temperature is controlled by thermostat or digital controller.
- The temperature probe detects the chamber temperature and the heater is switched on or off automatically.
- If the door is opened during the cycle, temperature fluctuation may occur and sterilization may be affected.
- Sterilization is usually done at about 160°C for 2 hours or 170°C for 1 hour.
- The dry heat penetrates slowly into the materials and causes irreversible damage to microbial enzymes and structural proteins.
- Exposure time depends on load size, wrapping and type of materials.
- Heavy and insulated materials need longer time so that all parts can reach sterilizing temperature.
- After completion of cycle, the door should be kept closed until the temperature becomes low.
- Sudden opening of door may cause burn injury or breakage of glassware due to thermal shock.
- The efficiency of hot air oven depends on proper temperature, holding time and even circulation of hot air.
- Hot air oven is used for sterilizing glassware like test tubes, petri dishes, pipettes and flasks without moisture.
- It is used for sterilizing metal instruments like forceps, scalpels, scissors and other tools which can tolerate high dry heat.
- It is used for sterilizing powders such as starch, talc and some drugs which may clump or degrade in steam.
- It is used for sterilizing oils and fats like paraffin, glycerin and mineral oil, where moist heat may cause hydrolysis.
- It is used for sterilizing glass syringes, reusable syringes and glass ampoules to make them free from microorganisms.
- Empty glass containers are sterilized in hot air oven before filling with sterile media or chemicals.
- It is also used for sterilizing sharp instruments because dry heat reduces corrosion and dulling compared to steam sterilization.
- Hot air oven is used in pharmaceutical, cosmetic and food industries where moisture-free sterilization is required.
8. Quebec colony counter
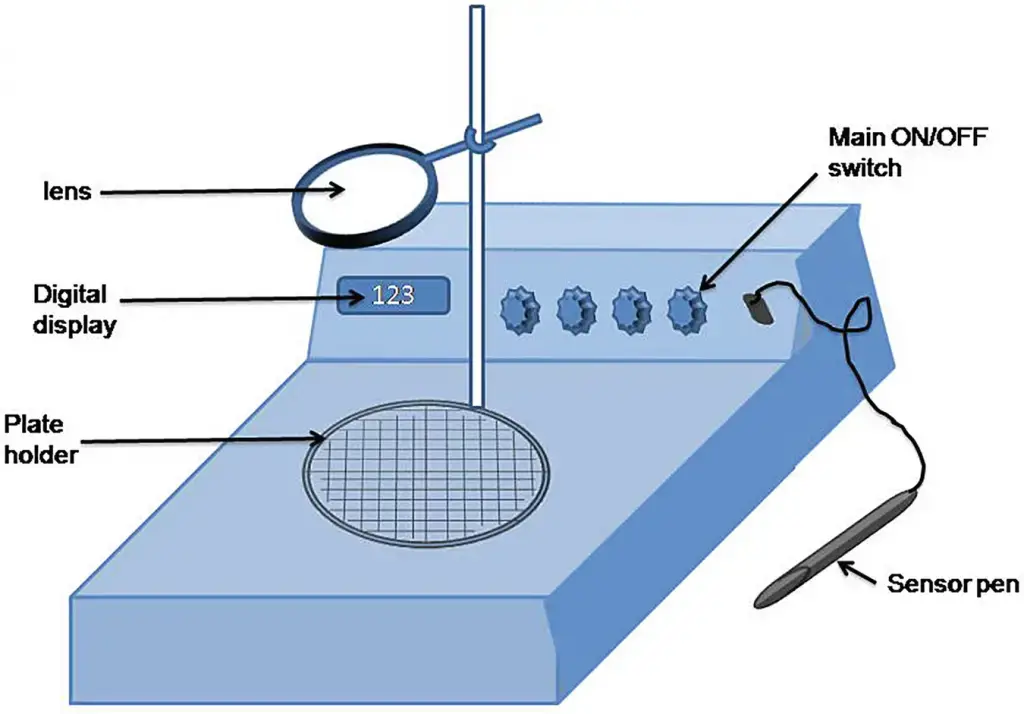
- Quebec colony counter is a laboratory device used for counting bacterial or fungal colonies on agar plates.
- It is widely used in microbiology laboratory because it makes colony counting faster, more accurate and less tiring for eyes.
- It works by using strong illumination from below the culture plate and a magnifying lens above the plate.
- Due to light and magnification, the colonies present on agar surface appear larger and more clear.
- This helps the operator to see each colony properly, even when the colonies are small or very close to each other.
- Light from LED or fluorescent lamp is passed through the translucent plate holder.
- The bright background makes the colony edges sharper and reduces the chance of missing colonies in dense culture.
- A fixed magnifying lens is placed above the plate, which generally magnifies the colonies about 1.5x to 3x.
- The magnifying lens reduces eye strain and helps in counting for long time without much fatigue.
- A marked grid pattern is present below the plate, which divides the agar plate into small sections.
- The grid helps the user to count colony section by section and then calculate the total number.
- This method reduces human error, double counting and skipping of colonies.
- In manual counting, the operator presses a digital or mechanical counter button each time a colony is seen.
- The counter keeps the running total displayed, so counting can be stopped and continued without losing the number.
- The plate is held stable during counting and it prevents slipping or movement of the plate.
- The accuracy of Quebec colony counter depends on proper illumination, magnification and use of grid system.
- Quebec colony counter is used for microbial enumeration in microbiology research, water testing, food safety analysis and pharmaceutical quality control.
- It is used in water quality testing to count microbes in drinking water, waste water and swimming pool samples.
- It is used in food and beverage industries like dairy, meat, bakery and beverage for checking microbial contamination.
- It is used in pharmaceutical analysis to test drugs, raw materials and medical devices for sterility and microbial limit.
- It is used in clinical diagnostic laboratory to count pathogens from patient samples like urine, sputum and wound swabs.
- It is used in environmental monitoring to check microbes from soil, air and surfaces in cleanrooms and biosafety laboratory.
- It is used in colleges and training institutes to teach microbiological counting technique to students.
- It is also used in research work like genetics, antibiotic testing and microbial ecology where accurate colony count is needed for data analysis.
9. The pH meter
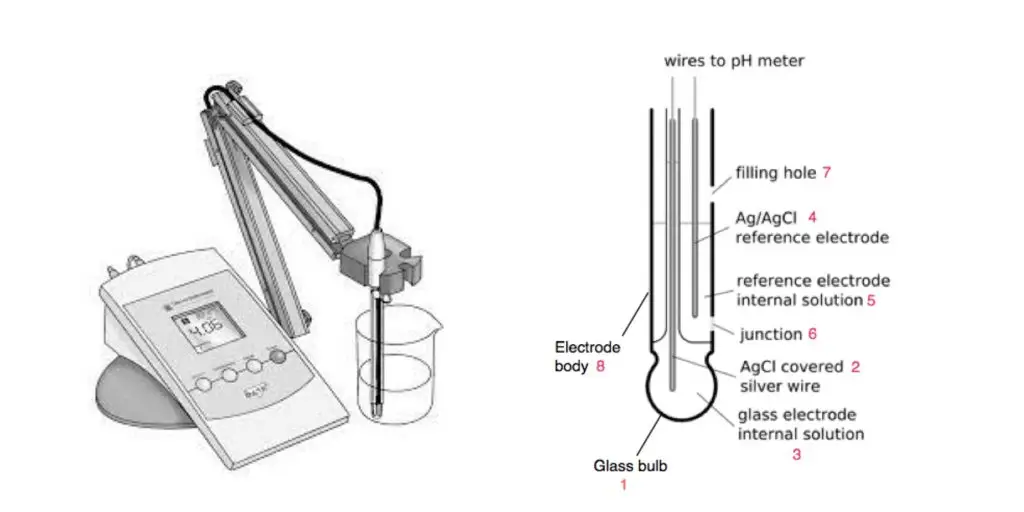
- The pH meter is a laboratory instrument used to measure the acidity or alkalinity of a solution.
- It is used to find the pH value of water, culture medium, chemical solutions and different liquid samples.
- The pH meter works on the principle of electrochemical potential difference between two electrodes.
- These two electrodes are glass electrode and reference electrode.
- The glass electrode is sensitive to hydrogen ion activity present in the solution.
- The reference electrode gives a constant and stable potential for comparison.
- When the glass electrode is dipped in the sample solution, hydrogen ions interact with the special glass membrane.
- Due to this interaction, a small potential difference or voltage is produced.
- This voltage depends on the concentration of hydrogen ions in the solution.
- The electronic circuit of pH meter measures this potential difference between glass electrode and reference electrode.
- The measured voltage is then converted into pH value by using the Nernst equation.
- In simple way, the pH meter senses small voltage change caused by hydrogen ion activity and shows it as pH reading.
- Before use, the pH meter is calibrated with standard buffer solutions of known pH.
- Calibration is necessary to get correct and accurate reading of the sample.
- The pH meter is used for checking quality of water in drinking water, effluent, swimming pool and aquarium.
- It is used in food and drink industry to maintain proper pH of dairy products, wine, beer, juices and sauces.
- It is used in soil and agriculture analysis to check soil pH for better crop growth and correct use of fertilizer.
- It is used in pharmaceutical industry to maintain pH of drug formulation, buffer solution and quality control samples.
- It is used in environmental monitoring to check acid rain effect, acidity of rivers, lakes and industrial waste.
- It is used in chemical and petrochemical industry for controlling pH of chemical reactions and process streams.
- In laboratory, it is used to measure pH in experiments, culture media and chemical solutions.
- It is also used in cosmetics industry to adjust pH of skin-care and hair-care products for safety and effectiveness.
10. Balance
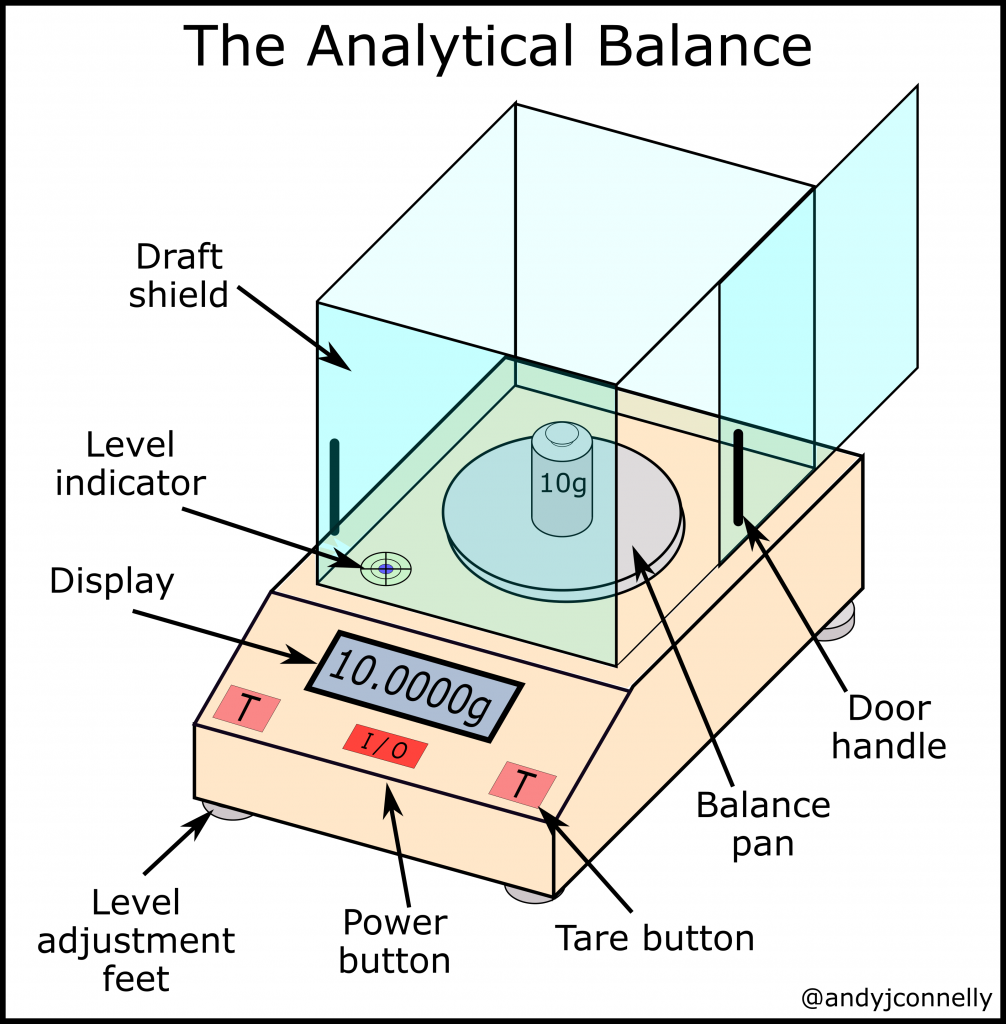
- Balance is a laboratory instrument used to measure the mass of chemicals, reagents and other materials.
- It is used in scientific laboratory, pharmaceutical work, food industry and different experimental works where exact mass is needed.
- Balance works on the principle of equilibrium of moments.
- When the turning forces on both sides of a pivot are equal, the beam remains horizontal and it indicates equal masses.
- In traditional mechanical balance, a beam is pivoted at the centre and two pans are present at equal distance from the pivot.
- The unknown mass is placed on one pan and standard weights are placed on the other pan.
- When the beam becomes level, the unknown mass and the standard weights are equal.
- This is because weight is proportional to mass under constant gravity.
- In electronic balance, load cell is used to measure the force produced by the weight of object.
- The load cell converts this force into an electrical signal.
- This signal is amplified and processed by the electronic system and the mass is shown digitally.
- Mechanical balance measures mass by comparing unknown mass with known standard masses.
- Electronic balance measures the force of gravity acting on the object and converts it into mass reading.
- Balance is used in scientific laboratories for weighing chemicals, reagents and biological samples accurately.
- It is used in pharmaceutical industry for weighing active chemicals and other substances during drug preparation.
- It is used in jewelry business for accurate measurement of precious metals and diamonds.
- It is used in food industry for checking quality, measuring ingredients and controlling portions.
- It is used in schools for teaching measurement of mass in biology, chemistry and physics laboratories.
- It is used in industrial work for measuring raw materials used in manufacturing process.
- It is used in environmental studies for weighing soil, plant and water samples during research.
- It is also used in forensic science for weighing fibers, powders, residues and other evidence samples.
11. Spectrophotometer (Colorimeter)
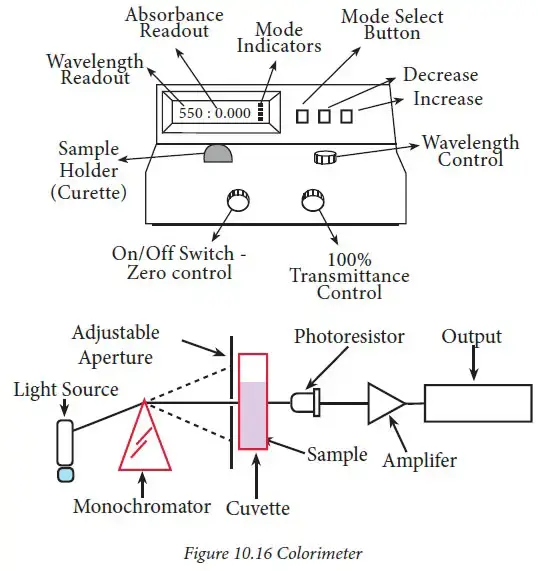
- Spectrophotometer or colorimeter is a laboratory instrument used to measure the amount of light absorbed by a coloured solution.
- It is used to determine the concentration of a substance present in the sample solution.
- Spectrophotometer or colorimeter works on the principle of Beer-Lambert’s law.
- According to this law, the absorbance of light by a solution is directly proportional to the concentration of absorbing substance and the path length of light through the solution.
- In this instrument, light of a specific wavelength is passed through the sample solution.
- The molecules present in the solution absorb some amount of light and the remaining light passes through the solution.
- The transmitted light then reaches the detector and the amount of absorbed light is measured.
- The absorbance is calculated by using Beer-Lambert’s law.
- The reaction is as follows- A = ε × c × l
- Here, A is absorbance, ε is molar absorptivity, c is concentration of solution and l is path length of cuvette.
- In simple way, the instrument measures how much light is absorbed by the sample and from this, concentration of the substance can be determined.
- A cuvette is used to hold the sample solution during measurement.
- The cuvette should be clean because dust, fingerprint or air bubble may affect the reading.
- Spectrophotometer is used in biochemistry and clinical laboratory for checking blood sugar, haemoglobin, cholesterol and enzyme activity.
- It is used in environmental monitoring to test water quality for nitrates, phosphates and heavy metals.
- It is used in pharmaceutical industry to find the amount of drug present in a sample and for quality checking of formulation.
- It is used in food and beverage industry for checking colour, additives and nutrient level of products.
- It is used in chemical research to study reaction rate and formation of products.
- It is used in microbiology to estimate microbial growth by measuring turbidity or optical density.
- It is used in forensic science for analysis of pigments, colours and toxic compounds present in evidence.
- It is also used in agriculture to find nutrient content in plant extracts and soil samples.
12. Centrifuges
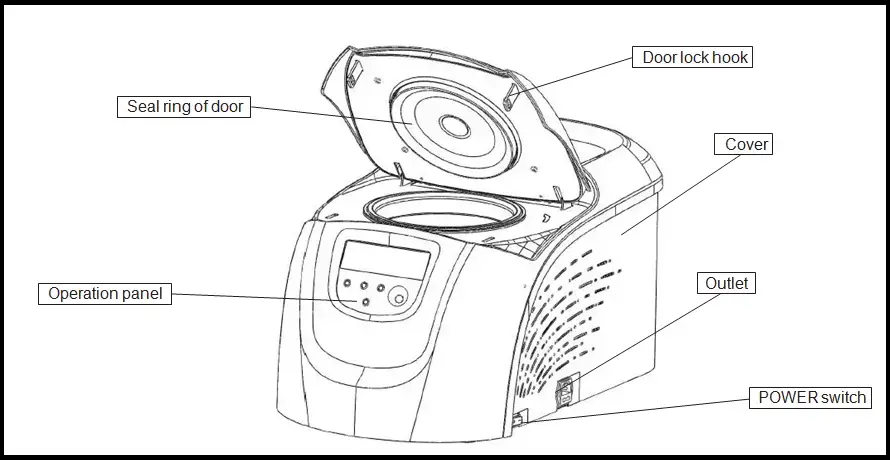
- Centrifuge is a laboratory instrument used to separate particles from a liquid mixture by rapid spinning.
- It is used for separation of blood cells, serum, plasma, cell organelles, precipitates and other suspended particles.
- Centrifuge works on the principle of centrifugal force.
- When a sample is rotated at high speed, the heavier particles move outward and settle at the bottom of the tube.
- The lighter liquid part remains above the settled particles and this is called supernatant.
- The solid or heavier material collected at the bottom is called pellet.
- The separation depends on size, shape, density of particles, viscosity of medium and speed of rotation.
- The sample tubes are placed inside the rotor of centrifuge.
- Rotor is the rotating part which holds the tubes and spins them at fixed speed.
- The tubes must be balanced properly before starting the centrifuge.
- Equal volume of sample or balance tube should be placed opposite to each other in the rotor.
- If tubes are not balanced, vibration occurs and it may damage the centrifuge or break the tubes.
- The speed of centrifuge is generally expressed in revolutions per minute (RPM).
- It may also be expressed as relative centrifugal force (RCF), which shows the actual force acting on the particles.
- During centrifugation, particles with higher density sediment faster than particles with lower density.
- After the spinning is completed, the centrifuge should be allowed to stop completely before opening the lid.
- Sudden opening or touching the rotor while moving is dangerous and may cause injury.
- Centrifuge is used in clinical laboratory for separation of serum and plasma from blood sample.
- It is used in microbiology for collecting bacterial cells, fungal spores and other microbial particles from suspension.
- It is used in biochemistry for separation of proteins, enzymes and precipitated materials.
- It is used in molecular biology for separation of DNA, RNA and cell components during different experiments.
- It is used in urine analysis to sediment cells, crystals and other particles present in urine.
- It is used in pharmaceutical laboratory for separation and purification of drug samples and suspensions.
- It is used in research work for isolation of cell organelles like mitochondria, chloroplast and nuclei by differential centrifugation.
- It is also used in food, dairy and environmental laboratories for separation of suspended particles from different samples.
13. Microscope
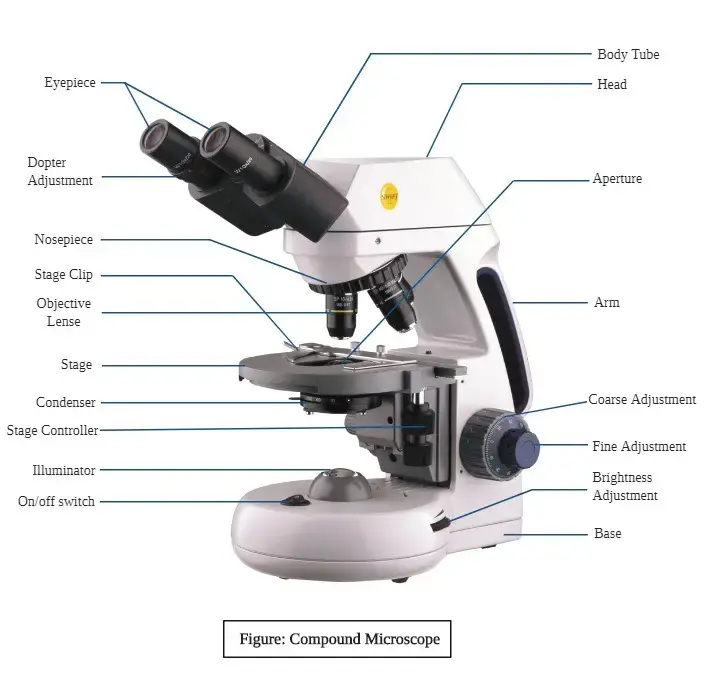
- Microscope is an instrument used to see small objects which are not seen by naked eye.
- It is used in laboratory for observing microorganisms, cells, tissues and other minute structures.
- It is based on the principle of magnification. In this process, light or electrons are focused through lenses and the object appears larger than its original size.
- Light microscope uses visible light. The light passes through the specimen or reflected from it and then lenses form the magnified image.
- In light microscope, total magnification is obtained by multiplying the magnification of objective lens and ocular lens.
- Electron microscope uses beam of electrons instead of light. As electrons have very short wavelength, it gives high resolving power and greater magnification.
- In microscope, light from the source passes through the condenser and falls on the specimen.
- The objective lens collects the light from specimen and forms a real image.
- This image is again magnified by the eyepiece and a virtual image is seen by the eye.
- The visibility of the specimen can be increased by staining, phase contrast or fluorescence method.
- These methods helps to observe fine details of cells, tissues and microorganisms.
- Resolution is the ability of microscope to distinguish two nearby points as separate points.
- It depends on wavelength of light and numerical aperture of the lens.
- Coarse adjustment and fine adjustment knobs are used for focusing the specimen.
- By focusing, the object is brought into sharp view and microscopic details can be observed.
- Microscope is used to study bacteria, fungi, protozoa and algae.
- It helps in identification and classification of microorganisms.
- It is used for study of cell structure such as nucleus, cytoplasm and cell organelles.
- It is used in medical diagnosis for examination of blood, tissue, parasite and other clinical samples.
- It is used in biology, microbiology and histology laboratory for teaching and research work.
- It is also used in forensic work for studying hair, fibres and body fluids.
- It is used in environmental study for observing plankton, soil microorganisms and water contaminants.
14. Incinerator
- Incinerator is an instrument used for burning waste materials at high temperature.
- It is used to convert waste into ash, gases and heat.
- It is based on the principle of thermal oxidation.
- In this process, waste materials are burned in presence of oxygen and the organic matter is oxidized.
- Due to burning, the volume and toxicity of waste are greatly reduced.
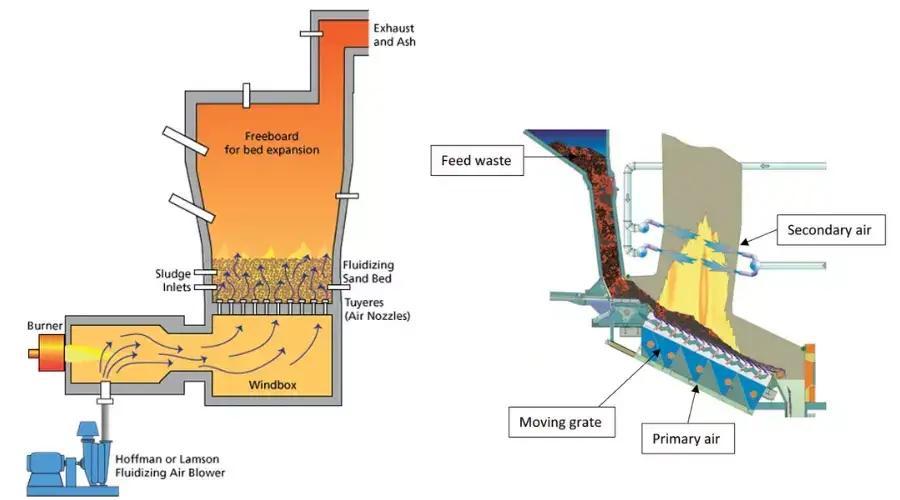
- The waste is first placed inside the primary combustion chamber.
- In this chamber, waste is exposed to high temperature flame and organic materials are changed into carbon dioxide, water vapour and heat.
- The gases and small particles formed in primary chamber then pass into the secondary combustion chamber.
- In secondary chamber, the incomplete combustion products are burned again at higher temperature.
- This helps to reduce smoke, bad smell and harmful pollutants.
- A controlled supply of air is needed for proper burning of waste.
- Primary and secondary air supply maintain oxygen level and temperature inside the incinerator.
- The heat produced during burning can be used for making steam, electricity or hot water in some incinerators.
- The gases produced from incinerator pass through filters, scrubbers or electrostatic precipitators before release.
- These devices remove dust particles, acidic gases and toxic compounds from the flue gas.
- The incombustible residue remains as ash after burning.
- This ash is collected from the bottom part and disposed safely.
- Incinerator is used for disposal of medical waste like syringes, bandages and pathological waste.
- It helps to destroy infectious materials and prevents spreading of diseases.
- It is used for disposal of hazardous wastes like chemical, pharmaceutical and toxic industrial wastes.
- It is used for reducing municipal solid waste and saving landfill space.
- It is used in farms and veterinary hospitals for disposal of dead animals.
- It is used in laboratory for burning biological and chemical waste to reduce contamination risk.
- It is also used in industrial waste management for disposal of manufacturing residues and by-products.
- Incinerator helps in environmental protection by reducing untreated waste and lowering soil, water and air pollution.
15. Deep Freezer (Laboratory Refrigerators and Freezers)

- Deep freezer is a laboratory instrument used to store biological samples, chemicals and reagents at very low temperature.
- It is used to preserve samples for long time and prevent their degradation.
- It is based on vapor compression refrigeration.
- In this process, heat is removed from the storage chamber and the temperature becomes much lower than room temperature.
- A refrigerant is circulated through compressor, condenser, expansion valve and evaporator.
- The refrigerant absorbs heat from inside the freezer and releases it outside.
- Compressor compresses the low pressure refrigerant vapour into high pressure and high temperature gas.
- This gas then passes through the condenser coil.
- In condenser, the high pressure gas releases heat to the surrounding environment and changes into liquid.
- The liquid refrigerant then passes through expansion valve.
- In expansion valve, the refrigerant expands and changes into low pressure cold vapour.
- Due to this expansion, the temperature of refrigerant drops very low before entering evaporator.
- In evaporator, the cold refrigerant absorbs heat from the freezer chamber.
- As heat is removed, the inner temperature decreases and stored materials are preserved.
- Thermostat is used to monitor and control the temperature of deep freezer.
- It regulates the working of compressor and helps to maintain constant low temperature.
- The walls of deep freezer are thick and insulated.
- This insulation prevents entry of heat from outside and keeps the temperature stable.
- Deep freezer is used for storage of biological samples like blood, plasma, serum and tissues.
- It maintains sample integrity and prevents degradation during research or clinical use.
- It is used for preservation of bacteria, viruses, fungi and other microbial cultures at very low temperature.
- This helps to maintain long term viability of microorganisms for experiments.
- It is used to store temperature sensitive chemicals, enzymes, antibodies and reagents.
- It helps to maintain their stability and activity.
- It is used for storage of vaccines, insulin and other drugs which need low temperature.
- It maintains their efficacy and shelf life.
- It is used in molecular biology for preservation of DNA, RNA and protein samples.
- It prevents enzymatic degradation and helps in reproducible experimental results.
- It is used in clinical laboratories for storing diagnostic kits and patient samples.
- It is also used in research and development for long term storage of experimental samples under controlled low temperature.
16. Homogenizer
- Homogenizer is a laboratory instrument used to break particles or droplets into smaller size and mix them uniformly in liquid medium.
- It is used to prepare stable and homogeneous mixture of solids, liquids or immiscible liquids.
- It is based on mechanical disruption and size reduction.
- In this process, large particles or droplets are broken down and dispersed evenly throughout the medium.
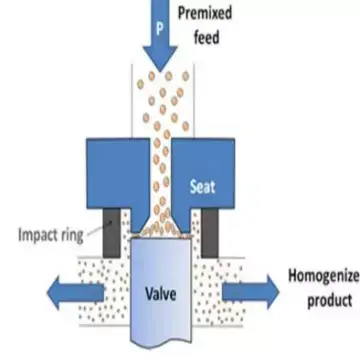
- The liquid sample is forced through narrow gaps or small orifices under high pressure.
- Due to this, strong shearing force is produced and larger particles are reduced into smaller particles.
- Particles also collide with each other or with the surface of homogenizer.
- Cavitation bubbles may also form and collapse during the process.
- The energy produced by impact and cavitation further helps in reducing the particle size.
- Homogenizer helps in mixing and emulsification of different components.
- It prevents settling of particles and separation of two phases.
- In high pressure homogenizer, very high pressure is applied, sometimes up to 2000 bar.
- This gives fine particle size and uniform consistency of the sample.
- The liquid passes continuously through the homogenizing valve or chamber.
- So, large amount of sample can be processed in efficient way.
- Cooling is often used during homogenization.
- It prevents denaturation or degradation of heat sensitive materials.
- Homogenizer is used in food industry for preparation of milk, dairy products, sauces and beverages.
- It improves texture, taste and shelf life of the food products.
- It is used in pharmaceutical industry for dispersing active ingredients in suspensions, emulsions and creams.
- This helps to maintain uniform dose and product consistency.
- It is used in biotechnology and research for breaking cells and releasing intracellular contents like proteins, enzymes and DNA.
- It is used in cosmetic industry for preparation of lotions, creams and gels with smooth and uniform texture.
- It is used in chemical industry for making paints, inks and adhesives with equal particle size.
- It is used in microbiology for homogenizing tissue or microbial samples before culturing or analysis.
- It is used in environmental analysis for preparation of soil, sludge and water samples for testing.
- It is also used in nanotechnology and emulsion preparation for producing nanoemulsions and fine dispersions.
17. Hot plate
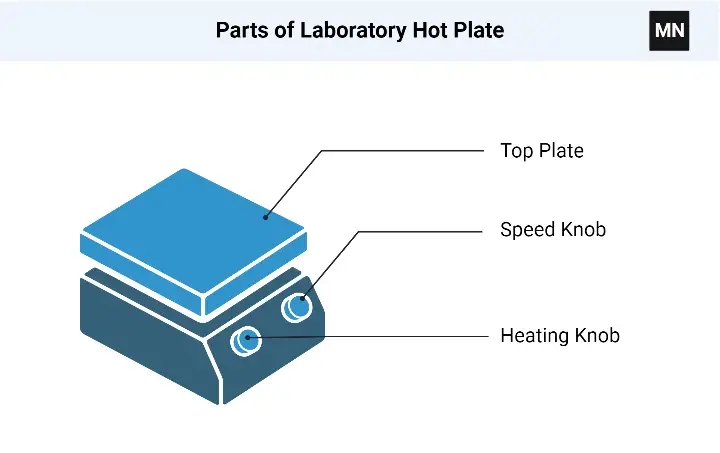
- Hot plate is a laboratory instrument used for heating liquids, solids and different samples.
- It is used in laboratory as a steady and controllable source of heat.
- It is based on electrical resistance heating.
- In this process, electric current passes through a resistive element and electrical energy is changed into heat energy.
- The heating element may be a coil or plate made up of high resistance material.
- When current flows through it, the element becomes hot and produces heat on the surface.
- Heat is transferred from the hot surface to the container or sample which is kept on it.
- This transfer of heat takes place mainly by conduction.
- The sample is heated in controlled way without using direct flame.
- Thermostat or digital controller is used to regulate the temperature of hot plate.
- It controls the current supply to heating element and maintains the required temperature.
- This prevents overheating or less heating of the sample.
- Some hot plates have magnetic stirrer with it.
- In this type, a magnetic bar is placed inside the container and it rotates during heating.
- This helps in uniform mixing of the solution and prevents local overheating.
- Hot plate may have safety features like thermal cut-off or overheat protection.
- These safety systems prevent damage of instrument and reduce chance of accident.
- Hot plate is used for heating solutions in beakers, flasks and test tubes.
- It is used to melt wax, paraffin and other low melting point solids.
- It is used for evaporation of solvents from solution to concentrate the sample.
- It is used to provide controlled heat for organic and inorganic chemical reactions.
- It is used in educational laboratory for practical work, demonstration and heating experiments.
- It is used for sample preparation, such as heating media, buffers and reagents before use.
- Hot plate with magnetic stirrer is used for heating with continuous mixing of solution.
- It is also used in research and small scale industrial work for synthesis, formulation and testing.
18. Magnetic Stirrer
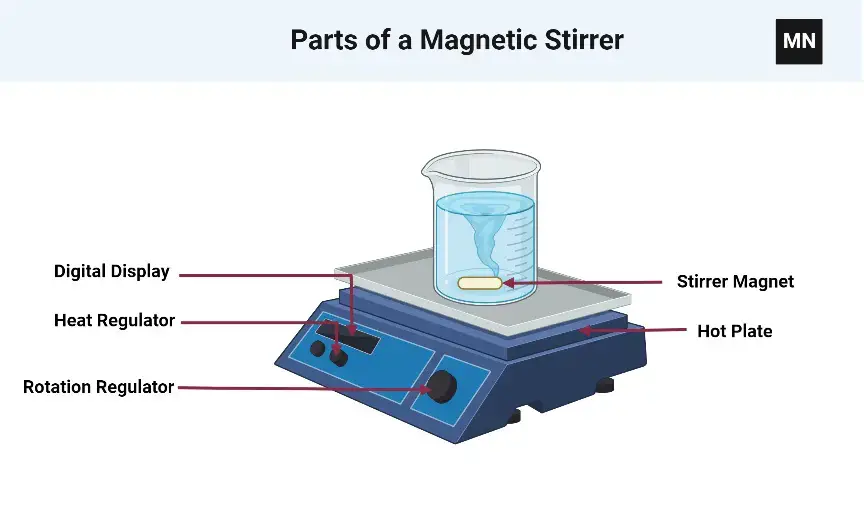
- Magnetic stirrer is a laboratory instrument used for mixing liquids and solutions uniformly.
- It is used for preparation of buffers, reagents, chemical solutions and other liquid samples.
- It is based on magnetic induction.
- In this process, a rotating magnetic field is produced below the platform and this field rotates the magnetic stir bar present inside the liquid.
- Electric current passes through coils or a permanent magnet rotates under the plate.
- This produces a rotating magnetic field below the container.
- A small magnetic bar coated with inert material is placed inside the beaker or flask.
- The stir bar aligns with the rotating magnetic field and starts rotating inside the liquid.
- Rotation of the stir bar mixes the liquid continuously and makes the solution uniform.
- The speed of stirring can be controlled by a speed controller.
- Slow or fast mixing can be done depending on the requirement of the sample.
- Some magnetic stirrers have heating element under the platform.
- This type is used when heating and stirring are needed at the same time.
- Magnetic stirrer gives non-contact mixing because the stir bar is not directly connected with the motor.
- This reduces chance of contamination and it is useful for sterile or closed system mixing.
- It can be used with beakers, flasks and different laboratory containers.
- Magnetic stirrer is used for uniform mixing of liquids, buffers and reagents.
- It is used during chemical reactions to give continuous stirring.
- Continuous stirring helps the reactants to mix properly and gives uniform reaction condition.
- It is used for dissolving powders, salts and other solutes in liquids.
- It helps in faster preparation of solutions.
- It is used for homogenization of suspensions, emulsions and colloids.
- It prevents settling of particles and separation of phases.
- Magnetic stirrer with hot plate is used for heating and stirring together during reaction work.
- It is used in educational laboratory for demonstration of mixing, solution preparation and chemical reactions.
- It is used in biological work for gentle stirring of cell cultures, microbial suspensions and enzyme solutions.
- It is also used in environmental and analytical studies for mixing soil, water and chemical samples before testing.
19. Vortex Mixture/ Vortexer
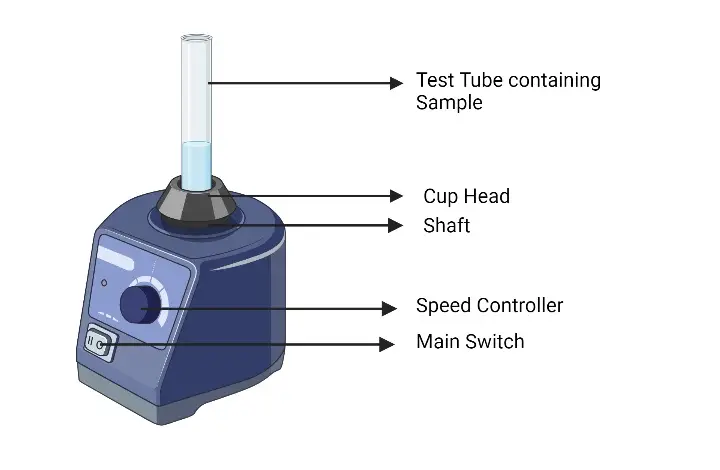
- Vortex mixer or vortexer is a laboratory instrument used for quick mixing of small amount of liquid samples.
- It is used for mixing samples in test tubes, microcentrifuge tubes, vials and small containers.
- It is based on rapid circular motion.
- In this process, a motor driven platform moves in circular or orbital motion and forms vortex inside the liquid.
- The platform or cup holder moves in small circular path.
- This movement gives kinetic energy to the tube or container placed on it.
- When the test tube touches the moving platform, the liquid inside the tube starts swirling.
- This swirling movement forms a vortex and mixes the contents properly.
- The speed of vortex mixer can be controlled by speed regulator.
- Gentle or vigorous mixing can be done according to the type of sample.
- Vortexer can work in touch mode or continuous mode.
- In touch mode, mixing occurs only when the tube is pressed on the platform.
- In continuous mode, the platform keeps moving continuously for longer mixing.
- It does not use any stirrer or direct mechanical part inside the liquid.
- So, chance of contamination is reduced and sample damage is also less.
- It is suitable for biological, chemical and clinical sample preparation.
- Vortex mixer is used for quick mixing of small volume liquids in test tubes, microcentrifuge tubes and vials.
- It is used for dissolving powders, salts and reagents in liquids.
- It helps in faster preparation of solution.
- It is used for homogenization of suspensions, emulsions and cell suspensions.
- This prevents settling of particles and phase separation.
- It is used in biological work for mixing bacterial cultures, enzymes and DNA or RNA samples.
- It helps to maintain sample integrity during experiment.
- It is used in small scale chemical reactions for rapid mixing of reactants.
- It is used in laboratory assays like immunoassay, ELISA and diagnostic tests for proper mixing of reagents.
- It is used before centrifugation to mix samples evenly and prevent stratification.
- It is also used in clinical and diagnostic laboratory for mixing blood, serum and other clinical specimens.
20. Water Distiller
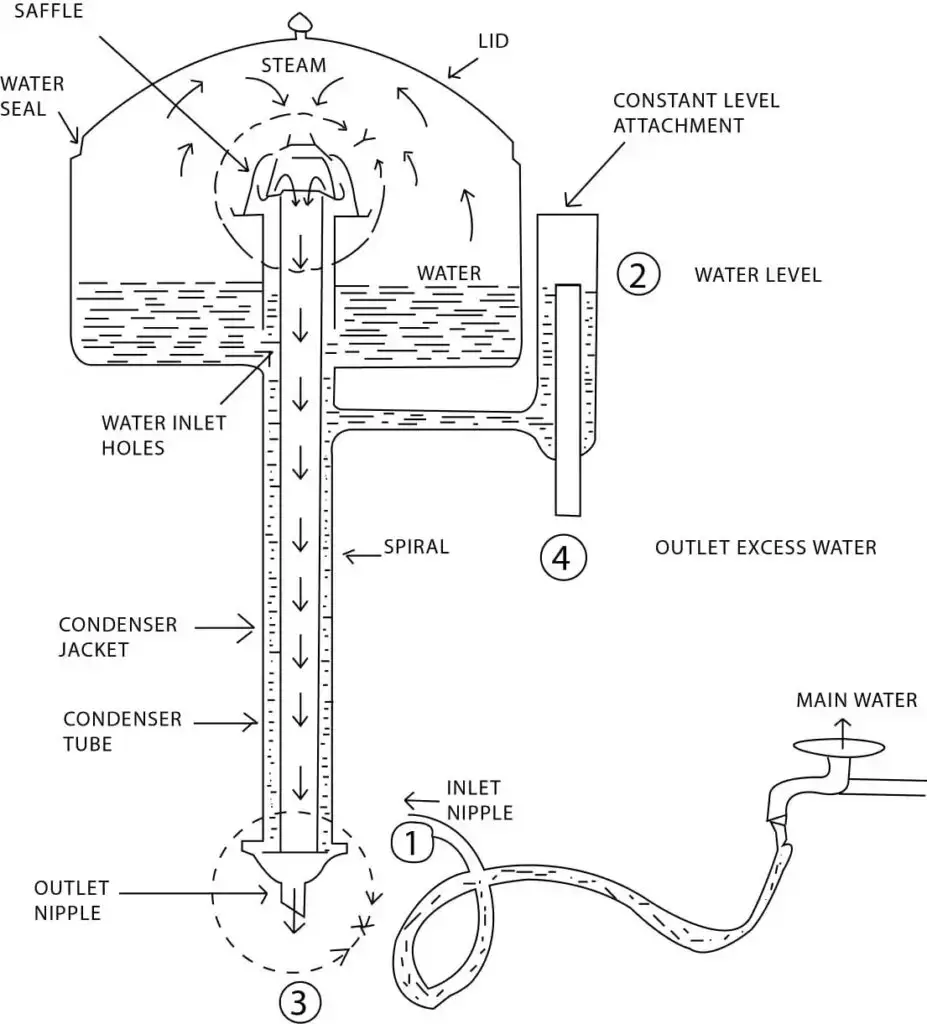
- Water distiller is an instrument used for making distilled water in laboratory.
- It is based on evaporation and condensation.
- The impure water is taken in boiling chamber and heat is supplied.
- On heating, water changes into vapour and salts, minerals and other impurities remain in the chamber.
- The vapour then passes into condenser or cooling coil.
- In condenser the vapour becomes cool and changes into water again.
- This water is collected in another vessel as distilled water.
- The heating is generally done by electric heater.
- In some distiller pre-filter is also present for removing large particles before boiling.
- Distilled water does not contain dissolved salts, minerals, heavy metals and many microorganisms.
- It is used for preparation of reagents, buffers, media and other solutions in laboratory.
- It is used in microbiology and chemistry works where pure water is required.
- It is used for washing glasswares and other laboratory equipments.
- It is used in medical and pharmaceutical laboratory for making sterile water and solutions.
- It is used in boilers and cooling system because scale formation is less.
- It is also used in food, beverage and cosmetic industry where clean water is needed.
References
- https://biologynotesonline.com/inoculating-loops-and-needles/
- https://biologynotesonline.com/bunsen-burner/
- https://biologynotesonline.com/laboratory-water-bath/
- https://biologynotesonline.com/autoclave-principle-parts/
- https://biologynotesonline.com/laminar-flow-hood-cabinet-parts-principle-application/
- https://biologynotesonline.com/incubator-principle-components-types-operating-procedure-use/
- https://biologynotesonline.com/hot-air-oven/
- https://biologynotesonline.com/colony-counter-types-principle-parts-uses-examples/
- https://biologynotesonline.com/ph-meter-principle-parts-procedure-types-uses/
- https://biologynotesonline.com/spectrophotometer/
- https://biologynotesonline.com/centrifuge-principle-parts-types-applications/
- https://biologynotesonline.com/light-microscope-labeled-diagram-definition-principle-types/
- https://biologynotesonline.com/incinerator/
- https://biologynotesonline.com/homogenizer/
- https://biologynotesonline.com/laboratory-hot-plate/
- https://biologynotesonline.com/magnetic-stirrer-definition-principle-types-parts-uses/
- https://biologynotesonline.com/vortex-mixer/
- https://biologynotesonline.com/water-distiller/