HPLC (High-performance liquid chromatography) is a liquid chromatography technique used to separate, identify and quantify different components present in a liquid mixture. It is also called high-pressure liquid chromatography. It is used when accurate separation is needed in a mixture.
In this technique, a liquid solvent is used as mobile phase and the column is packed with solid porous particles which acts as stationary phase. The mobile phase with the sample is forced through the column by high-pressure pumps. During this process, different components do not behave same inside the column. Some components have stronger affinity towards stationary phase, so they move slowly. Some components are more attracted to the mobile phase, so they move fast and come out early. This gives separation into different bands.
After separation, the components exit from the column and pass through a detector. The detector measures the amount of each component and gives signal. A chromatogram is obtained in the form of peaks. HPLC is used in pharmaceutical testing, environmental testing, food safety analysis and clinical diagnostics for routine analysis and quality checking.
Principle of HPLC (High-performance liquid chromatography)
Principle of HPLC (High-performance liquid chromatography) is based on the separation of components due to their different interaction with a stationary phase and a mobile phase under high pressure. A liquid solvent is used as the mobile phase and it is forced through a column packed with solid adsorbent particles called stationary phase. The sample is injected into this continuous flow and allowed to pass through the column.
During this process, different molecules interact differently with the stationary phase. Molecules having stronger affinity towards stationary phase are retained for longer time and move slowly. Molecules having stronger affinity towards the mobile phase move fast and elute early. This difference may be due to polarity, electrical charge or molecular size and because of this, separation occurs as different components come out at different time.
After leaving the column, the separated components pass through a detector. The detector produces an electrical signal and a chromatogram is formed. The chromatogram peaks are used for identification and measurement of the sample components.
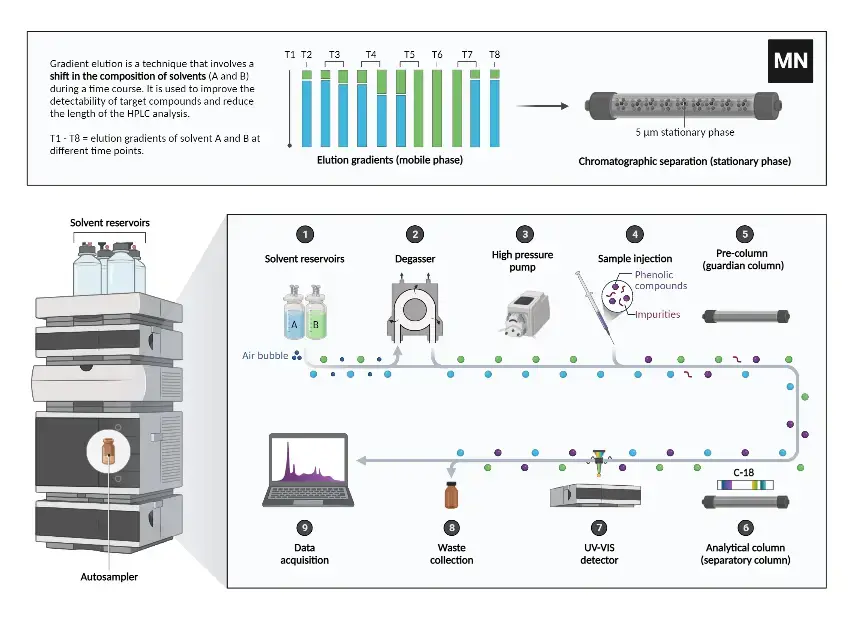
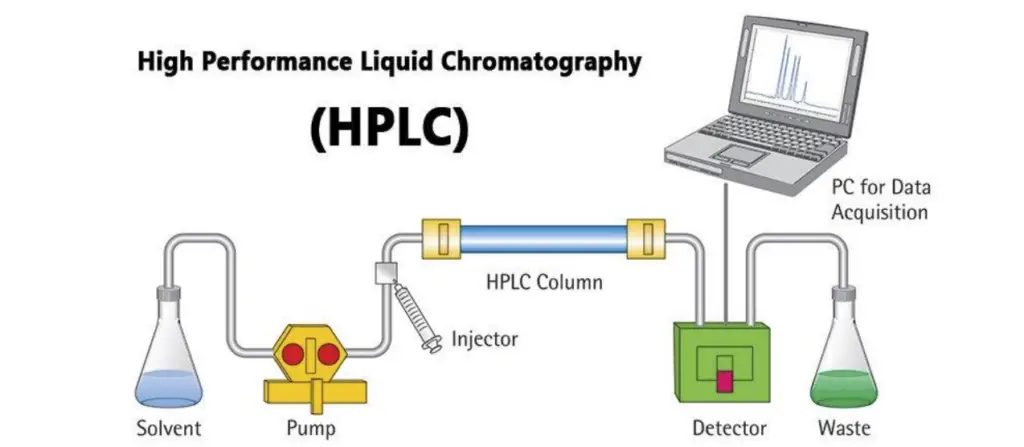
Instrumentation of HPLC

- Solvent reservoir- It is a container for mobile phase (eluent). The solvent is taken from here and used to carry the sample in whole system.
- Solvent degasser- It removes dissolved gases from the mobile phase. It avoids bubble formation. It reduces pressure fluctuation and baseline noise.
- High pressure pump (solvent delivery system)- It produces high pressure to push the mobile phase through the packed column. It maintains a constant flow of solvent and gives uniform delivery.
- Sample injector (manual or autosampler)- It is used to introduce a measured volume of sample into the flowing mobile phase. The injection is kept reproducible.
- HPLC column- It is a tube (generally stainless steel) packed with stationary phase. The separation of components takes place inside this column.
- Column oven- It maintains the column at a constant temperature. It prevents temperature changes during run and improves reproducibility of retention time and peak shape.
- Detector- It detects the components when they come out from the column. It converts the presence of component into an electrical signal. It may be UV/Vis, fluorescence, refractive index or mass spectrometry detector.
- Data system (recorder/workstation)- It records the detector signal and gives chromatogram in the form of peaks. It is used for identification and quantification of components.
Types of HPLC
1. Normal-phase HPLC (NP-HPLC)- Polar stationary phase is used and non-polar mobile phase is used. Components are separated mainly by polarity. Least polar compound comes out first.
2. Reversed-phase HPLC (RP-HPLC)- Non-polar (hydrophobic) stationary phase is used and mobile phase is polar (aqueous). The attraction is more towards hydrophobic surface, so retention differs. More polar component elute early and less polar component stays longer.
3. Size-exclusion chromatography (SEC)- It separates molecules by size. Large molecules do not enter the pores and they come out fast. Small molecules enter the porous particles and they are eluted late. Gel permeation chromatography (GPC) uses non-aqueous mobile phase. Gel filtration chromatography (GFC) uses aqueous buffer.
4. Ion-exchange chromatography (IEC)- It is used for charged components. Stationary phase contains ion exchanger with opposite charge. Components are retained according to electrical affinity and then eluted.
5. Hydrophilic-interaction chromatography (HILIC)- It is also referred to as aqueous normal-phase (ANP) in some notes. Highly polar stationary phase is used and mobile phase is mostly organic with water. It is used to retain highly polar and hydrophilic compounds.
6. Hydrophobic-interaction chromatography (HIC)- It is used mainly for large biomolecules like proteins. Separation occurs by surface hydrophobicity but the condition is kept aqueous.
7. Chiral chromatography- It is used to separate enantiomers (optical isomers). Chiral selector is present in stationary phase or sometimes in mobile phase, and due to this two enantiomers separate.
8. Affinity chromatography- It is based on specific biological binding. Only the target compound binds and retained, others pass through. Elution is done after breaking the binding.
9. Ligand-exchange chromatography (LEX)- Separation occurs by formation of temporary coordination complex. Metal ions are immobilized on stationary phase and analyte binds for short time and then moves.
10. Mixed-mode chromatography- More than one separation mechanism is present in one column. It is used when sample is complex and single mode is not enough.
11. Partition chromatography- It is an older type. Analyte distributes between liquid stationary phase and the eluent. Difference in polarity gives separation.
Protocols/Step by step procedure of HPLC
- Sample preparation- The sample is prepared in suitable liquid form. It is simplified and concentration is adjusted. Contaminants are removed by filtration, dilution, centrifugation, extraction or derivatization, so it does not interfere and system is not blocked.
- Mobile phase preparation- High purity solvents are selected. The solvents are filtered to remove particles. Degassing is done by sonication, vacuum or inline degasser to remove dissolved gases and avoid pressure problem. The mobile phase is then pumped through the system at controlled flow rate.
- Column selection- A suitable column is selected depending on sample. Column dimensions, particle size and stationary phase type is checked. The column should be compatible for proper separation.
- Sample injection- A fixed volume of prepared sample is injected into the mobile phase stream. Injection is done by manual injector or autosampler. The sample enters into the column with the mobile phase.
- Separation- The sample components separate inside the column due to different interaction with stationary phase. Components with weak interaction move fast and elute early. Components with strong interaction are retained longer and elute late. This separation may depend on polarity, charge or size.
- Detection- The eluted components pass through a detector. Detector may be UV detector or mass spectrometer. The detector converts the presence of compounds into electrical signal.
- Data analysis- The signals are processed by data system. A chromatogram is obtained as peaks. Retention time is used for identification and peak area is used to calculate concentration.
Factors Affecting HPLC
- Stationary phase properties- Particle size affects separation and back pressure. Surface area affects retention. Stationary phase chemistry decides interaction with analyte and resolution.
- Mobile phase composition- Solvent type and polarity changes elution. pH affects ionization of compounds. Buffer concentration and organic modifier affect peak shape and separation.
- Flow rate- Higher flow rate reduces retention time. Very high flow rate reduces resolution. Low flow rate increases run time and may improve separation.
- Column temperature- Temperature affects solvent viscosity and mass transfer. Controlled temperature can improve peak shape and reduce retention time. High temperature may damage stationary phase or degrade sample.
- Analyte properties- Polarity of analyte affects retention. Electrical charge and functional groups decide interaction with phases. Molecular size also affects movement and separation.
- Sample injection volume- High injection volume overloads column and peak becomes broad or distorted. Very low injection volume reduces signal and sensitivity.
- Elution mode- Isocratic elution keeps mobile phase constant. Gradient elution changes mobile phase composition gradually. Choice affects analysis time and separation of complex mixture.
Applications of HPLC
- Drug quality control- It is used to check purity of drugs. It is used to check peak purity. It is used for stability testing of finished product.
- Formulation and process work- It is used to monitor reaction changes. It is used to evaluate formulations. It is used during development work.
- Multiple component estimation- It is used for simultaneous estimation of more than one active ingredient. It is used to detect and measure impurities in tablets, syrups and vaccines.
- Clinical testing- It is used for analysis of blood and urine. It is used for therapeutic drug monitoring. It is used for neonatal screening.
- Disease diagnosis related testing- It is used in some cancer related testing. It is used in inherited metabolic disorder testing (Phenylketonuria). It is used for hemoglobin disorder testing (thalassemia, porphyria).
- Vitamins and hormone analysis- It is used to measure vitamins. It is used to measure hormones. It is used to measure amino acids in biological fluids.
- Environmental analysis- It is used to detect pollutants in water, soil and sludge. It is used for pesticide analysis. It is used to monitor industrial contaminants and toxic metal ions.
- Water quality monitoring- It is used for drinking water testing. It is used for wastewater quality checking. It is used to monitor endocrine disrupting chemicals.
- Forensic work- It is used to detect illicit drugs. It is used to detect doping agents. It is used to detect poisons and explosives in samples.
- Metabolite detection- It is used to detect drug glucuronides in human biological samples. It is done without enzymatic cleavage in many cases.
- Food and beverage testing- It is used to detect melamine. It is used to detect mycotoxins. It is used for veterinary drug residue analysis.
- Nutritional component analysis- It is used to quantify vitamins, carbohydrates, lipids, amino acids and purines in food samples.
- Cosmetics testing- It is used to quantify active compounds. It is used to check preservatives like parabens. It is used to detect toxic substances like formaldehyde and hydroquinone.
- Chemical and material testing- It is used to analyze synthetic polymers and additives. It is used for purity checking of raw materials. It is used for quality control of chemical catalysts and some nanomaterial surface chemistry.
Advantages of HPLC
- High speed analysis- It gives rapid separation because high pressure pump is used. The run time is less as compared to many traditional methods.
- High resolution- It gives better separation of closely related compounds. It gives sharp peaks and clear elution pattern in a single run.
- Accuracy and precision- It is used for accurate identification and quantification. The result is reproducible when same condition is maintained.
- High sensitivity- It can detect compounds in very low concentration. It is useful for trace level analysis.
- Automation facility- Modern system can be automated easily. Injection, run and data recording can be done in continuous routine work.
- Wide versatility- It can analyze many types of compounds. Small molecules, biomolecules, polar, ionic and high molecular weight compounds can be analyzed.
- Suitable for sensitive compounds- It can be used for non-volatile compounds. It can be used for thermally unstable compounds which cannot be analyzed by GC.
- Broad use in different fields- It is used in pharmaceuticals, environmental testing, clinical diagnostics, forensic testing and food analysis. It is used because method is simple and efficient.
Limitations of HPLC
- High cost- The instrument is costly. Maintenance cost is high. Solvents and reagents are also expensive.
- Skilled person required- Setup is not simple. Operation needs training. Troubleshooting is also difficult without experience.
- Method development takes time- New method development is time consuming. Many parameters are adjusted like flow rate, column type, temperature and mobile phase composition. Optimization needs patience.
- Poor for volatile compounds- It is not sensitive for highly volatile compounds. Such compounds are better analyzed by gas chromatography.
- Solvent waste problem- Large amount of organic solvents are used in many methods. Hazardous solvents produce chemical waste and disposal becomes a problem.
- Regulatory issues- In some fields method harmonization is not uniform. Compliance and standardization may become difficult in clinical type testing.
References
- Agilent Technologies. (2019). Retaining and separating polar molecules – A detailed investigation of when to use HILIC versus a reversed-phase LC column,.
- Agilent Technologies. (n.d.). Tips and tricks of HPLC system troubleshooting.
- Agilent Technologies. (n.d.). What is high-performance liquid chromatography (HPLC)?,.
- Allan Chemical Corporation. (n.d.). HPLC vs. UHPLC: Key differences.
- Avantor ACE. (n.d.). Chromatographic band broadening and the van Deemter equation (Knowledge Note #0010). MAC-MOD Analytical.
- Bailey, D. (2025, December 10). HPLC components overview: The 5 key parts explained. Timberline Instruments.
- Boag, M. (2025, April 1). Key applications of HPLC. Phenomenex.
- BÜCHI Labortechnik AG. (n.d.). As easy as ABC: How to use the van Deemter equation to optimize your chromatography.
- Chawla, G., & Chaudhary, K. K. (2019). A review of HPLC technique covering its pharmaceutical, environmental, forensic, clinical and other applications. International Journal of Pharmaceutical Chemistry and Analysis, 6(2), 27-39. https://doi.org/10.18231/j.ijpca.2019.006,.
- Chrom Tech, Inc. (n.d.). Understanding HPLC instrumentation: Principles & uses.
- Chrom Tech, Inc. (n.d.). What is the difference between UHPLC and HPLC?.
- Comprehensive technical analysis of high-performance liquid chromatography: Principles, instrumentation, and multidisciplinary applications in modern analytical science. (n.d.).
- Gritti, F., & Guiochon, G. (2006). General HETP equation for the study of mass-transfer mechanisms in RPLC. Analytical Chemistry, 78(15), 5329-5347. https://doi.org/10.1021/ac060203r.
- JASCO Inc. (n.d.). High-performance liquid chromatography (HPLC) system configuration.
- JASCO Inc. (n.d.). HPLC separation modes: Types, mechanisms & examples.
- Karasali, H., Kasiotis, K. M., Machera, K., & Ambrus, A. (2014). Case study to illustrate an approach for detecting contamination and impurities in pesticide formulations. Journal of Agricultural and Food Chemistry, 62(47), 11347-11352. https://doi.org/10.1021/jf504729g,.
- Keyfi, F., Alaei, A., Daryasari, H. M., Hakimi, A., & Gharavi, P. (2024). Utilizing high-performance liquid chromatography (HPLC) in clinical diagnostics. In O. Núñez (Ed.), Relevant applications of high-performance liquid chromatography in food, environmental, clinical and biological fields. IntechOpen. https://doi.org/10.5772/intechopen.1008238,.
- KNAUER. (n.d.). HPLC basics – Essential guide to chromatography principles.
- Organomation. (n.d.). Preparing samples for HPLC-MS/MS analysis.
- Phenomenex. (n.d.). Chiral HPLC column.
- Phenomenex. (n.d.). Complete guide to HPLC instrumentation.
- Phenomenex. (n.d.). HPLC troubleshooting mini guide – Peak issues.
- QxMD. (n.d.). Journal of Analytical Toxicology.
- SCION Instruments. (n.d.). HPLC troubleshooting guide.
- Shimadzu. (n.d.). What is HPLC (High performance liquid chromatography).
- Sigma-Aldrich. (n.d.). Basics of chiral HPLC.
- Sigma-Aldrich. (n.d.). Hydrophilic interaction liquid chromatography.
- Tamang, S. (2026, April 5). Chiral chromatography: Principle, components, steps, types, uses. Microbe Notes.
- Tamang, S. (2026, April 5). HPLC: Principle, instrumentation, steps, types, uses, diagram. Microbe Notes.
- Thermo Fisher Scientific. (n.d.). HPLC troubleshooting.
- Waters Corporation. (n.d.). HPLC separation modes – Stationary phase in HPLC.
- Wikipedia contributors. (2026, April 2). High-performance liquid chromatography. In Wikipedia, The Free Encyclopedia,.
- Wikipedia contributors. (2026, March 17). Van Deemter equation. In Wikipedia, The Free Encyclopedia,.