Analytical balance is a sensitive laboratory instrument used for measuring very small mass with high accuracy. It is commonly used in chemistry, biology, pharmaceutical and research laboratory.
It can measure mass up to 0.1 mg or 0.01 mg level. So it is used when accurate weighing of sample is needed. Small change in air, dust or vibration can affect the reading.
The weighing pan of analytical balance is enclosed by a glass chamber. This chamber is called draft shield. It protects the pan from air current, dust and other outside disturbance.
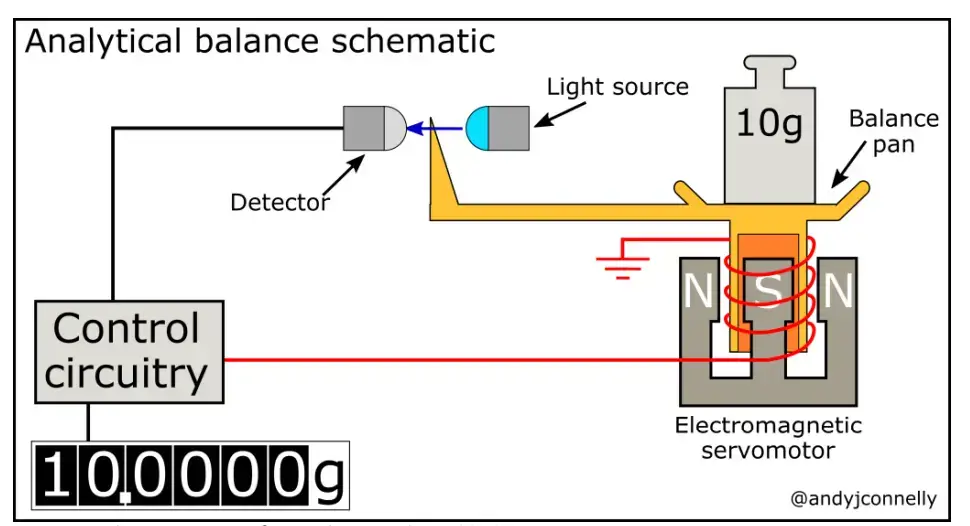
Modern analytical balance is mostly electronic. It does not use ordinary physical weights for measuring the mass. It works by Electromagnetic Force Restoration (EMFR) principle.
In this system, when sample is placed on the pan, downward force is produced by the weight of sample. An electromagnet produces upward force to balance this weight. The electric current needed for this balancing is measured and converted into mass value.
The first analytical balance was developed by Joseph Black around 1750. It was a two pan balance with a light beam and central fulcrum. Unknown sample was kept in one pan and known weights were kept in another pan.
Later single pan balance was developed by Erhard Mettler in 1946. This balance used internal weights and gave easier weighing. After that electronic analytical balances replaced the mechanical balances in many laboratories.
At present analytical balance gives fast digital reading. It is used for weighing chemicals, preparing standard solutions, weighing pharmaceutical samples and other quantitative laboratory work.
Principle of Analytical Balance
Principle of Analytical Balance is based on Electromagnetic Force Restoration (EMFR). In this principle the mass is not measured by comparing with physical weights. It is measured by the electric force needed to balance the sample weight.
When the sample is placed on the weighing pan, it produces a downward force. This force causes a very small displacement of the pan. The displacement is detected by an optical sensor present inside the balance.
After this, electric current is passed through a coil placed in a magnetic field. This produces an upward electromagnetic force. This force balances the downward force of the sample.
The pan is brought back to its original zero position. This position is called null position. The balance always tries to maintain this position during weighing.
The amount of current needed to restore the pan is directly proportional to the mass of the sample. This current is measured by the instrument and converted into digital mass reading. Thus analytical balance gives accurate weight of very small amount of sample.
Types of analytical balance

The following are the types of Analytical Balance–
- Two-pan analytical balance
It is a traditional mechanical balance. It has two pans and a beam supported on a central pivot. Unknown sample is kept in one pan and known standard weights are kept in another pan. - Equal-arm balance
It is also called two-pan balance. Both arms of the beam are equal in length. The mass is measured by balancing the sample with known weights. - Single-pan mechanical balance
It has only one weighing pan. It uses built-in internal weights and a counterweight. The mass is found by removing internal weights to balance the loaded sample. - Unequal-arm balance
It is a type of single-pan mechanical balance. One arm is short and holds the weighing pan. Another arm is long and carries constant counterweight. - Electronic single-pan balance
It is the most commonly used analytical balance in modern laboratory. It measures mass by producing electromagnetic force against the load placed on the pan. - Semi-micro balance
It is more sensitive than ordinary analytical balance. It can measure very small mass, generally up to 0.01 mg or 10 µg readability. - Microbalance
It is used for weighing very small quantity of sample. It can measure mass in microgram level, usually about 1 µg or 0.001 mg. - Ultra-microbalance
It is more sensitive than microbalance. It is used when extremely small amount of sample is to be weighed with very high accuracy.




Parts of Analytical Balance with Functions

The following are the main parts of Analytical Balance–
- Draft shield
It is a glass enclosure present around the weighing pan. It protects the sample from air current, dust and outside disturbance. - Weighing pan
It is the plate where sample or container is placed for weighing. It is usually made up of non-magnetic stainless steel. - Display panel
It shows the weight of sample. It also shows unit, error message and other function of the balance. - Power button
It is used to switch on and switch off the balance. - Tare button
It is used to make the display zero. It removes the weight of empty container from the final reading. - Mode button
It is used to change the weighing unit. Gram, milligram and other units can be selected by this button. - Level indicator
It shows whether the balance is placed in horizontal position or not. It may be a spirit bubble or digital level sensor. - Level adjustment feet
These are screw type legs present at the base of the balance. They are rotated to bring the bubble in centre position. - Internal calibration weight
These are built-in weights present inside the balance. The balance uses these weights for automatic calibration. - Data port
It is used for transferring balance data to computer, printer or laboratory system. USB, RS-232, Ethernet or Bluetooth may be present. - Drip tray
It is present below the weighing pan. It collects spilled sample and helps in cleaning of the balance. - Electromagnetic weighing cell
It is the internal weighing mechanism of modern analytical balance. It contains magnet, coil, optical sensor and electronic control system for measuring mass.

Operating Procedure of Analytical Balance
The following are the operating procedure of Analytical Balance–
- The balance is placed on a stable and vibration free table. It should be kept away from air draft, direct sunlight and sudden temperature change.
- The balance is switched on before use. It is allowed to warm up for 30 to 60 minutes, so the internal system becomes stable.
- The level indicator is checked. The leveling feet are adjusted until the bubble comes in centre position.
- The balance is calibrated before weighing. Internal calibration or external calibration may be done according to the balance type.
- The weighing pan is kept empty and the draft shield doors are closed. Then Zero or Tare button is pressed.
- A clean empty weighing container is placed at the centre of the pan. The draft shield doors are closed after placing the container.
- The reading is allowed to become stable. Then Tare button is pressed to make the display zero.
- The container is removed from the balance for adding sample. Sample should not be added directly inside the balance chamber.
- The sample or container should be handled with forceps or gloves. Fingerprint, moisture and body heat may change the weight.
- The container with sample is again placed at the centre of the weighing pan. The draft shield doors are closed completely.
- The display is observed until the reading becomes stable. The stability mark should appear before taking the reading.
- The final mass shown on the display is recorded carefully. The unit of measurement should also be noted.
- After weighing, the sample and container are removed from the pan. No material should be left on the balance.
- Spilled powder or dust is cleaned with soft brush or lint free cloth. The weighing pan and chamber should be kept clean.
- The draft shield doors are closed after cleaning. The balance may be kept in standby mode for next use.
Calibration Procedure of Analytical Balance
The following are the calibration procedure of Analytical Balance–
- The balance is kept in stable room. Vibration, air current, direct sunlight and sudden temperature change should be avoided.
- Temperature and humidity should be controlled. These changes may affect the weighing result.
- The level indicator is checked before calibration. The leveling feet are adjusted until the bubble comes at centre.
- The balance is switched on and allowed to warm up for 30 to 60 minutes. This makes the internal electronic parts stable.
- The weighing pan is cleaned and kept empty. No dust, powder or old sample should remain on the pan.
- For internal calibration, all draft shield doors are closed first. This prevents air current during calibration.
- The CAL button is pressed or internal calibration option is selected from menu. The balance then starts automatic calibration.
- The balance places its internal calibration weight by motor system. It takes about 60 to 120 seconds for adjustment.
- The display is observed until calibration message appears. Cal OK or adjustment completed message shows that calibration is finished.
- The accuracy is checked by weighing a certified external check weight. The reading should come within the acceptable limit.
- For external calibration, the empty balance is first made zero. The chamber doors are closed and Zero or Tare button is pressed.
- External calibration mode is selected from display menu. The balance gives command for placing the required calibration weight.
- The certified calibration weight is placed at the centre of pan. Clean forceps or lint free gloves should be used, bare hand should not be used.
- The draft shield doors are closed and reading is allowed to stabilize. The balance registers the mass and completes the adjustment.
- The calibration weight is removed after completion. Another known reference weight may be weighed for checking the accuracy.
- The calibration record is written in log book. Date, time, type of calibration, weight used, operator name and result should be recorded.
Precautions For Analytical Balances
The following are the precautions for Analytical Balance–
- The balance should be placed on stable and vibration free table. The table should be horizontal and not moved during weighing.
- The balance should be kept away from air current. Windows, fans and air conditioner should not directly face the balance.
- Direct sunlight should be avoided. Sudden temperature change may affect the reading.
- High humidity and magnetic field should be avoided near the balance. These may give unstable reading.
- The balance should be warmed up before use. Usually 30 to 60 minutes warm up is required according to the model.
- The level indicator should be checked before weighing. The leveling feet are adjusted until the bubble comes at centre.
- Sample, weighing vessel and calibration weight should not be touched by bare hand. Moisture, oil and body heat may change the weight.
- Clean forceps, tongs or gloves should be used for handling sample container and standard weight.
- The sample and container should be at room temperature before weighing. Hot or cold material may produce air current inside the chamber.
- The sample should be placed at the centre of weighing pan. Off-centre loading may give wrong reading.
- The draft shield doors should be closed before taking the reading. Doors should be opened slowly to avoid air turbulence.
- The reading should not be taken quickly. The stability mark should appear on display before recording the final weight.
- The balance should not be overloaded beyond its maximum capacity. Overloading may damage the weighing cell.
- Sample or container should not be dropped on the pan. It may damage the sensitive internal mechanism.
- Pointed or sharp object should not be used on touch screen display. It may scratch or damage the screen.
- Calibration should be done regularly. Internal calibration or certified external weights are used for this purpose.
- The weighing pan and chamber should be cleaned after use. Soft brush, lint free cloth and mild cleaning solution may be used.
- Harsh chemicals, abrasive powder and organic solvent should not be used for cleaning. These may damage the balance surface and chamber.
Factors that affect readings on analytical balances
The following are the factors that affect readings on Analytical Balances–
- Temperature fluctuation affects the weighing result. Hot or cold sample may create air current inside the weighing chamber and the reading may become low or unstable.
- Air current affects the weighing pan. Air from window, door, fan, air conditioner or breathing may cause fluctuating reading.
- Vibration gives unstable reading. Foot movement, nearby machine, refrigerator and centrifuge may disturb the balance.
- Static electricity affects the weighing of plastic container and dry powder. It may attract or repel the pan and gives false reading.
- Humidity affects many samples. Hygroscopic sample absorbs water from air and its mass increases.
- Low humidity increases static charge. So drifting and poor repeatability may occur.
- Magnetism affects the balance reading. Magnetic sample may interact with internal magnet or sensor coil of the balance.
- Air buoyancy changes the apparent mass of object. The object displaces air and the measured mass may become slightly lower than true mass.
- Improper handling gives error in weight. Fingerprint, oil, moisture and body heat from hand may change the mass of sample or container.
- Unlevel balance gives wrong reading. If the balance is not horizontal, systematic error occurs during weighing.
- Inadequate warm up affects the reading. If the balance is not warmed up properly, internal electronics are not stable and reading may drift.
- Chemical reaction may change the sample mass. Some reactive samples absorb gas, lose volatile material or react with air during weighing.
- Evaporation gives low reading. Volatile sample may lose mass while kept on the pan.
- Condensation gives high reading. Cold sample may collect moisture from air and the weight may increase.
Use of analytical balance
The following are the uses of Analytical Balance–
- It is used for accurate weighing of sample in laboratory. Powder, liquid and other materials are weighed for analysis and research work.
- It is used for sample preparation. Small amount of chemical or biological sample can be measured accurately.
- It is used in quantitative analysis. Reagents and chemicals are weighed for experiment and calculation of molar concentration.
- It is used for preparation of standard solution. Exact amount of solute is weighed before dissolving in solvent.
- It is used in formulation work. Different components are weighed in exact quantity for drug preparation and chemical manufacturing.
- It is used in pharmaceutical laboratory. Tablets, capsules and drug ingredients are weighed and checked.
- It is used for density determination. Density or specific gravity of solid and liquid can be measured by using density kit.
- It is used for pipette calibration. The water delivered by pipette is weighed and the pipette accuracy is checked.
- It is used for piece counting. The number of same type of small items like pills or electronic parts can be calculated from weight.
- It is used for interval weighing. Increase or decrease in sample weight is measured after fixed time.
- It is used in evaporation and absorption study. Loss or gain of weight of sample is recorded.
- It is used for check weighing. Packaged materials and pharmaceutical dosage forms are checked whether their weight is within limit or not.
- It is used for animal weighing. Dynamic weighing mode is used for moving small animals or live specimens.
- It is used for tare weighing. Weight of container is removed and only the sample weight is measured.
- It is used for percent weighing and total weighing. It helps in routine filling, dosing and average weight measurement.
Advantages of Analytical Balance
The following are the advantages of Analytical Balance–
- It gives very accurate weighing result. Very small change in mass can be detected by this instrument.
- It gives high precision. Same sample gives almost same reading when proper condition is maintained.
- It can measure mass in sub-milligram level. So it is useful for very small quantity of sample.
- It gives reading quickly. Modern analytical balance stabilizes within few seconds.
- It has internal calibration system in many models. The balance can adjust itself according to environmental change.
- It has draft shield around the weighing pan. This protects the reading from air current, dust and outside disturbance.
- It is easy to use. Digital display shows the mass directly.
- It has different weighing functions. Formulation, percentage weighing, piece counting and density determination can be done.
- It can transfer data to computer or printer. USB, RS-232 or other data ports may be present.
- It helps in proper record keeping. The weighing data can be stored or printed for laboratory record.
- It gives reproducible result. So it is useful in Good Laboratory Practices (GLP) and quality control work.
- It reduces manual error. Digital reading and automatic functions make weighing more reliable.
Limitations of Analytical Balance
The following are the limitations of Analytical Balance–
- It is costly instrument. Analytical balance is more expensive than ordinary laboratory balance.
- It is highly sensitive to surrounding condition. Small change in temperature, air current and vibration may change the reading.
- Static electricity may affect the weighing result. Plastic container and dry sample may give unstable reading.
- It needs controlled place for use. The balance should be kept on stable table and away from wind, heat and movement.
- It has limited weighing capacity. Most analytical balances are used for small mass only, generally about 100 g to 300 g.
- It depends on electricity. Modern electronic balance cannot work without proper power supply.
- It is not suitable for rough field use. Remote place without stable power and proper table may give wrong result.
- It needs warm up time before use. Usually 30 to 60 minutes are needed for stable reading.
- It is delicate in construction. Rough handling or heavy load may damage the internal weighing cell.
- The weighing pan should not be overloaded. Overload may permanently affect the balance accuracy.
- The display and touch screen may be damaged if handled with sharp object. So it should be used carefully.
- It needs regular cleaning and maintenance. Dust, spilled chemical and moisture may affect the performance.
- It needs calibration from time to time. Without calibration, the result may not be accurate.
Video guide of Analytical Balance
References
- Adam Equipment. (n.d.). Difference between analytical & precision balances?
- Adam Equipment. (n.d.). Understanding the different types of laboratory balances and their uses.
- Agriculture Notes. (n.d.). Types of analytical balances: Mechanical and electronic. Agriculture.Institute.
- American Pharmaceutical Review. (n.d.). Advanced weigh cell technology for healthcare packaging applications.
- Choudhary, A. (2024, April 17). SOP for operation and calibration of Mettler Toledo analytical balance. Pharmaguideline.
- Cox, A. (2025, September 2). What is an analytical balance. Triton Store.
- DSCBalances. (2025, November 23). What is analytical balance understanding its importance.
- Eagle Scales. (2024, May 27). Analytical balances: Definition, principle, types, uses & parts.
- Fisher Scientific. (2023). The effects of static electricity on analytical weighing. Lab Reporter, Issue 3.
- GNW Instrumentation. (n.d.). Precision vs analytical vs semi-micro balances: A complete lab guide.
- Hawach Scientific. (n.d.). Types of electronic balances in the laboratory.
- Hinotek. (2025). What is an analytical balance? The ultimate guide to principle, uses & accuracy (2025).
- Hinotek. (n.d.). The principle of electromagnetic force compensation in analytical balance.
- Hinotek. (n.d.). Tips for analytical balance: Installation, calibration, and standard operating procedures.
- Hogan, R. (2021, September 7). How to calculate air buoyancy correction with examples. ISOBudgets.
- Karki, P. (n.d.). Analytical balance- Definition, principle, parts, types, examples. Microbe Notes.
- Lab Manager. (n.d.). External weights vs. internal calibration: Which lab balance system is right for your lab.
- Lab Manager. (n.d.). How static electricity disrupts lab balance accuracy — and how to eliminate it.
- Laboratory Supply Network. (n.d.). Density measurements with a balance.
- Lee, H.-H., Yoon, K.-T., & Choi, Y.-M. (2021). Checkweigher using an EMFC weighing cell with magnetic springs and air-bearings. Metrology and Measurement Systems, 28(3), 465–478. https://doi.org/10.24425/mms.2021.137135
- Mettler Toledo. (n.d.). Electrostatic charges during weighing.
- Mettler-Toledo AG. (n.d.). Operating instructions Excellence analytical balances XS models – Part 1.
- Mettler-Toledo GmbH. (2016). Analytical balances XSE models: User manual.
- Mettler-Toledo GmbH. (2018). Precision and analytical balances ME: User manual.
- Mettler-Toledo. (n.d.). English quick guide MS-TS precision and analytical balances.
- Mettler-Toledo. (n.d.). User manual precision and analytical balances ME.
- MRC Laboratory Equipment. (n.d.). Analytical balances operating instructions.
- National Institute of Standards and Technology. (2019). SOP 2: Recommended standard operating procedure for applying air buoyancy corrections (NIST IR 6969-2019).
- Precisa Gravimetrics AG. (n.d.). Hybrid weighing cell | Weighing scales and balances | Load cell.
- Prime Lab Med. (2025, January 13). What is an analytical balance? Its components & working principle. Precisa analytical balances.
- PT Hyprowira Adhitama. (2017). Parts of the analytical balance and their use.
- ResearchGate. (n.d.). INTRODUCTION OF ANALYTICAL BALANCE.
- San Diego Scale. (n.d.). How to calibrate an analytical balance for accurate results.
- Sartorius AG. (1999, July). Sartorius analytical balances with the monolithic weighing system service manual for BP, GP, and LA models.
- Sartorius AG. (2026). Analytical balances.
- Sartorius. (n.d.). Effects of static electricity on analytical weighing.
- Schoonover, R. M., & Jones, F. E. (1981). Air buoyancy correction in high-accuracy weighing on analytical balances. Analytical Chemistry, 53(6), 900-902.
- Sullivan, C. R. (2022). Joseph Black. EBSCO Research Starters.
- Tovatech. (n.d.). A comprehensive guide to analytical balance calibration.
- Unknown. (n.d.). Buoyancy correction and air density measurement.
- Unknown. (n.d.). Differences between semi-micro analytical, analytical and precision balances [Video]. YouTube.
- Unknown. (n.d.). Technical metrology and the analytical balance: A comprehensive analysis of principles, structural engineering, and laboratory application.
- Wikipedia. (2026, March 1). Analytical balance. In Wikipedia.
- Wikipedia. (2026, April 15). Joseph Black. In Wikipedia.