Molisch’s test is a classic chemical test used for detection of carbohydrate in a given sample. It is a highly sensitive test and it is considered as a general screening test for almost all types of carbohydrates. It is named after the Austrian botanist Hans Molisch.
This test can detect monosaccharides, disaccharides and polysaccharides. It can also give positive result for complex compounds like glycoproteins and nucleic acids because the strong acids used in this test can hydrolyze them into simple monosaccharides. However the sugars having less than five carbon atoms like trioses and tetroses generally are not detected.
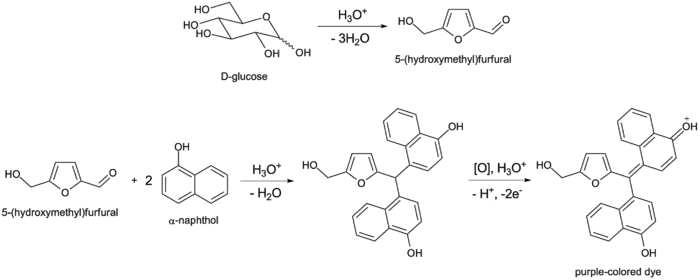
The test works on the principle of acid catalysed dehydration. When carbohydrate is treated with concentrated mineral acid (H2SO4 or HCl) it loses water molecules and aldehyde compounds are formed. Pentoses forms furfural and hexoses forms 5-hydroxymethylfurfural. These aldehydes then undergo condensation reaction with Molisch’s reagent which is α-naphthol dissolved in ethanol.
If carbohydrates are present then a purple or violet or purplish-red ring is formed at the junction of acid layer and aqueous solution. If ring is not formed then it indicates negative result and absence of carbohydrate.
Objectives of Molisch Test
Objectives of Molisch test are as follows–
- To detect presence or absence of carbohydrate in a given sample as a rapid screening test.
- To confirm that sugar backbone is present with at least five carbon atoms and it helps in ruling out trioses and tetroses.
- To differentiate carbohydrates from other biomolecules like proteins or lipids and to check sample purity.
- To detect sugars that are bound with complex compounds like glycoproteins and nucleic acids (DNA and RNA).
Molisch’s Test Principle
Molisch’s test is a general test used for detection of carbohydrates in a given sample. It is based on acid catalysed dehydration of carbohydrate followed by condensation reaction with a phenolic compound. It gives a characteristic purple or reddish-purple ring which confirms the presence of carbohydrate.
In this principle the carbohydrate is treated with strong mineral acid (concentrated H2SO4 or HCl). Due to action of acid the carbohydrates are dehydrated and cyclic aldehydes are formed. Pentoses are dehydrated to form furfural and hexoses are dehydrated to form 5-hydroxymethylfurfural.
If disaccharides and polysaccharides are present then the glycosidic bond is first hydrolyzed by acid and simple monosaccharides are formed and then dehydration occurs. In next step these aldehyde compounds condenses with two molecules of α-naphthol (Molisch’s reagent) forming a coloured complex. This coloured complex is seen as purple ring at the junction of two liquids.

Molisch’s Test Reaction
Requirements
Requirements for Molisch test are as follows–
- Molisch’s reagent– α-naphthol dissolved in ethanol (95% or 99%) usually 2% to 5% solution.
- Concentrated sulphuric acid (H2SO4) (strong mineral acid) used for dehydration of sugars. Concentrated HCl can also be used in some procedure.
- Test sample solution (aqueous sample). If sample is solid then it must be dissolved completely in water.
- Distilled water– for preparing solutions and for negative control.
- Clean and dry test tubes with test tube stand/rack.
- Pipette or dropper– to add Molisch reagent and to carefully add concentrated acid along side of test tube to form separate layer.
Procedure of Molisch Test
Procedure of Molisch test are as follows–
- Take a clean and dry test tube and add about 2 ml of the aqueous test sample (or standard carbohydrate solution). A control tube with 2 ml distilled water can also be taken.
- Add 2 to 3 drops of freshly prepared Molisch’s reagent into the test tube.
- The solution is mixed gently by shaking so that reagent mixes properly with sample.
- The test tube is kept tilted at about 45 degree and 1 to 2 ml of concentrated H2SO4 is added slowly along the inner wall of the test tube by a pipette.
- The acid forms a separate lower layer due to its high density. The test tube should not be shaken at this stage because charring can occur and test will be disturbed.
- The tube is kept undisturbed for few minutes in rack and junction of two layers is observed for violet/purple or reddish-purple ring formation.
Result Interpretation of Molisch’s Test
Result interpretation of Molisch’s test is as follows–
Positive result (Rapid)
A vivid purple or dark purple or violet ring is formed immediately at the junction of two layers. It indicates presence of monosaccharides (glucose or fructose) because no prior hydrolysis is required.
Positive result (Delayed)
A light purple or reddish-purple ring is formed slowly after some time. It indicates presence of disaccharides or polysaccharides (sucrose or starch) as acid takes extra time to hydrolyze glycosidic bonds before dehydration and condensation occurs.
Negative result
No colour ring is formed at the interface and junction remains colourless. It confirms absence of carbohydrates having five or more carbon atoms.
Charred or dark ring
A blackish or dark brown ring is formed instead of purple ring. It occurs when sugar concentration is very high or excess acid is added or tube is shaken and rapid charring of sugars is produced.
Greenish ring
A faint green ring or green tint can be seen at junction. It usually indicates impurities in reagent or background reaction with strong acid.
False positive
Sometimes a purple ring is formed due to certain organic acids (citric acid, lactic acid, oxalic acid, formic acid, malic acid) under strong acidic condition and it can be taken as carbohydrate positive.
False negative
Some carbohydrates like trioses and tetroses or certain amino sugars cannot form cyclic aldehydes and ring is not formed. In complex mixtures strong nucleophiles can also react with intermediates and result becomes negative even when carbohydrate is present.


Uses of Molisch Test
Uses of Molisch test are as follows–
- It is used to detect carbohydrates as a general screening test for monosaccharides disaccharides and polysaccharides in a given sample.
- It is used to differentiate carbohydrate from other biomolecules like proteins and lipids.
- It is used to detect sugars present in complex compounds like nucleic acids (DNA and RNA) glycolipids and glycoproteins.
- It is utilised in industries to check sugar contamination in products and to verify sugar-free labelled items.
- It helps to detect carbohydrate formation as by-products in different chemical reactions.
- It gives a preliminary idea about complexity of carbohydrate based on rate of colour ring development (simple sugars reacts faster than complex sugars).
- It is used in school and college laboratory for teaching basic carbohydrate testing and biochemical analysis.
Limitations of Molisch Test
Limitations of Molisch test are as follows–
- It cannot detect carbohydrates having less than five carbon atoms like trioses and tetroses because required furfural type intermediates are not formed.
- It is a general group test and it only indicates presence of carbohydrate but it cannot differentiate between different types of sugars (monosaccharide or polysaccharide).
- False positive result can be obtained with some non-carbohydrate substances like organic acids (citric acid lactic acid formic acid oxalic acid) which can form similar intermediates.
- False negative result can occur when strong nucleophiles are present in mixture and condensation reaction is interfered. Some amino sugars also do not dehydrate properly and positive ring is not obtained.
- Impurities in reagents can interfere and sometimes greenish ring is formed instead of purple ring.
- Concentrated H2SO4 is required which is hazardous and it can cause burns. If acid is added in excess or tube is shaken then sample can get charred and result becomes unclear.
Advantages of Molisch Test
Advantages of Molisch test are as follows–
- It is a simple test and procedure is easy and fast with minimal apparatus.
- It is highly sensitive and even trace amount of carbohydrates can be detected.
- It is a universal screening (group) test and it gives positive reaction for most classes of carbohydrates like monosaccharides disaccharides trisaccharides and polysaccharides.
- It is selective for carbohydrates and pure proteins or lipids generally do not give positive result which helps in checking sample purity.
- The purple ring formation is distinct and visual observation is clear for confirming carbohydrate presence.
- By observing thickness and darkness of ring a semi-quantitative idea about carbohydrate concentration can be obtained.
References
- Aakash Educational Services Limited. (n.d.). Molischs test in chemistry: Definition, types and importance.
- academics.su.edu.krd. (n.d.). Molisch’s test is a general test for all carbohydrates. In this test, carbohydrates when reacted with conc. H2SO4 g.
- A-Level Biology. (n.d.). Tests for carbohydrates | Types, principles, apparatus.
- AL-Khafaji, I. H. (2024). Review methods of detecting sugar in a sample. The American Journal of Interdisciplinary Innovations and Research, 6(1), 50-59.
- Analytical biochemistry of the Molisch reaction: A comprehensive examination of principles, methodology, and diagnostic applications in carbohydrate characterization. (n.d.).
- AquaPhoenix Scientific, Inc. (2017). Safety data sheet: Molisch reagent.
- AquaPhoenix Scientific, Inc. (2020). Safety data sheet: Molisch reagent (MO2900SS).
- BYJU’S. (n.d.). Molisch’s test principle.
- Carbohydrates – The Molisch test. (n.d.).
- Carumba, G. M. S. (n.d.). Laboratory experiment 3 test for carbohydrates. Scribd.
- Central Drug House (P) Ltd. (n.d.). Molisch’s reagent material safety data sheet SDS/MSDS.
- Chemistry Stack Exchange. (2018). Organic chemistry – Clarification in the mechanism for Molisch’s test for glucose.
- Collegedunia. (n.d.). Molisch test: Carbohydrates, procedure & observations.
- Environmental Health and Safety, UC Merced. (n.d.). Standard operating procedure: Sulfuric acid.
- Flinn Scientific, Inc. (2014). Safety data sheet (SDS): Molisch reagent.
- Gest, H. (1991). The legacy of Hans Molisch (1856-1937), photosynthesis savant. Photosynthesis Research, 30(1), 49-59.
- Hamburg, B. (2014). Sulfuric acid safety tips – Sulfuric acid MSDS information. VelocityEHS.
- Kiddle. (2026). Hans Molisch facts for kids.
- King Saud University. (n.d.). General color tests for carbohydrates (Experiment 4).
- Koracademy. (n.d.). General reactions of carbohydrates: Molisch’s reaction.
- Loba Chemie Pvt. Ltd. (2018). Molisch’s reagent safety data sheet.
- Maglangit, J. A. (2012). Is it possible for Molisch test to have a false negative result? [Online forum post]. ResearchGate.
- Mwafi, N. (n.d.). Qualitative tests for carbohydrates. Mutah University.
- Narwal, S. S., & Jain, S. K. (1994). Hans Molisch (1856-1937): The father of allelopathy. Allelopathy Journal, 1(1), 1-5.
- Setiawan, D. (2024). Basic biochemical analysis: Monosaccharide detection by Molish reaction. Universitas Negeri Semarang.
- Singla, R. (n.d.). Molisch test: Definition, procedure, and results. Vedantu.
- Testbook. (n.d.). Molischs test learn its principle, reagents, procedure and observations.
- Unacademy. (n.d.). Notes on Molisch’s test.
- Wikipedia contributors. (2025). Molisch’s test. In Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (2026). Hans Molisch. In Wikipedia, The Free Encyclopedia.