Chromatography is a laboratory technique used for separation of components from a mixture. It is also used for identification and purification of the separated components. It is used in both analytical and preparative work.
In analytical purpose, chromatography is used to determine the chemical composition and concentration of a sample. In preparative purpose, it is used to isolate and collect a particular component for further use.
Chromatography is based on the movement of mixture through two different phases. These are stationary phase and mobile phase. The stationary phase remains fixed in one position, while the mobile phase moves over or through it.
The stationary phase may be a solid material or a liquid present on a solid support. It may be present as porous beads inside a column or as a thin layer on a plate. The mobile phase is a fluid such as solvent, liquid mixture or an inert gas.
During chromatography, the sample is dissolved in the mobile phase and carried through the stationary phase. Different components of the mixture show different attraction for the two phases. Some components remain attached more with stationary phase and move slowly.
The components which have more affinity towards mobile phase move faster. This creates different rate of movement of the components. As a result, the components are separated from each other.
The separation in chromatography may occur by partition or adsorption. After separation, the individual components can be detected, measured and analysed. Thus chromatography is useful for study of mixture and purification of substances.
Definition of Chromatography
Chromatography is a laboratory technique for the separation of a mixture by passing it in a solution or suspension or as a vapor (as in gas chromatography) through a medium in which the components move at different rates.
Types of Chromatography
Chromatography are classified into following types-
- Based on physical state of mobile phase
- Liquid Chromatography (LC)
- Gas Chromatography (GC)
- Supercritical Fluid Chromatography (SFC)
- Based on chromatographic bed shape
- Column Chromatography
- Planar Chromatography
- Paper Chromatography
- Thin Layer Chromatography (TLC)
- Based on separation mechanism
- Adsorption Chromatography
- Partition Chromatography
- Ion Exchange Chromatography
- Size Exclusion Chromatography (SEC)
- Affinity Chromatography
- Hydrophobic Interaction Chromatography (HIC)
- Hydrophilic Interaction Chromatography (HILIC)
- Mixed Mode Chromatography
- Advanced and specialized techniques
- High Performance Liquid Chromatography (HPLC)
- Ultra High Performance Liquid Chromatography (UHPLC)
- High Performance Thin Layer Chromatography (HPTLC)
- Fast Protein Liquid Chromatography (FPLC)
1. Gas chromatography
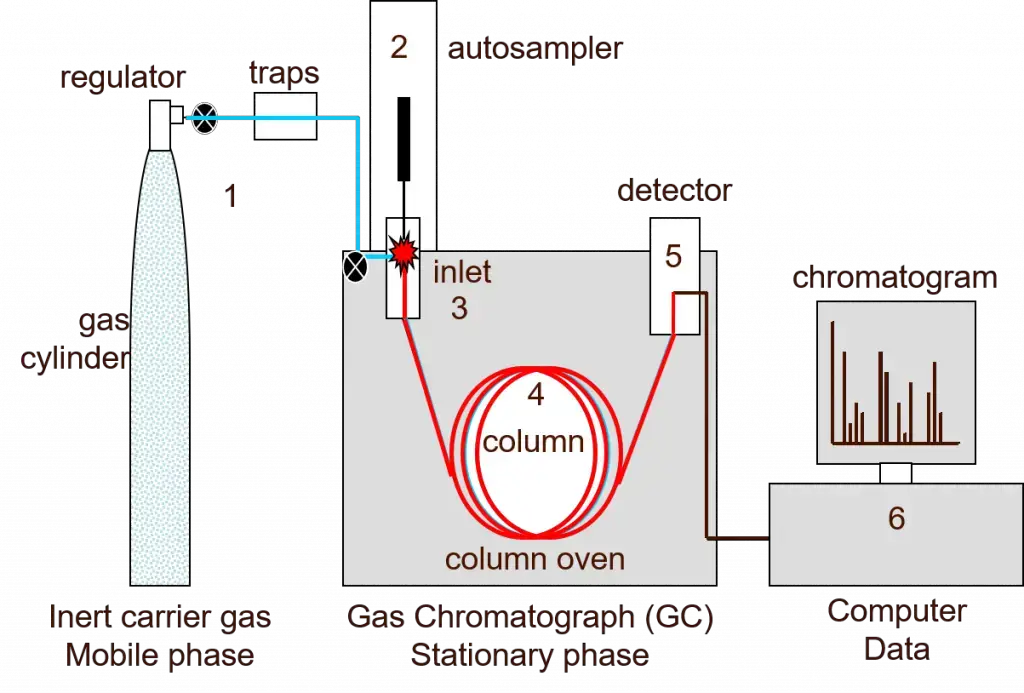
Principle of Gas Chromatography (GC)
Gas Chromatography is based on separation of volatile substances by using gas as mobile phase. The gas is inert carrier gas. It may be helium, nitrogen or hydrogen. The sample is changed into vapour and then passed through the column.
- The carrier gas carries the vapour sample through the column having stationary phase inside it.
- The stationary phase is present as thin liquid layer or solid polymer inside glass or metal column.
- The components are separated because all compounds do not have same boiling point and same attraction for stationary phase.
- The volatile compounds move faster with the carrier gas and come out first.
- The compounds which remain more with stationary phase move slowly and come out later.
- This method is used only when the compound can be vapourized and does not break at high temperature.
Uses of Gas Chromatography (GC)
- Gas Chromatography is used in pharmaceutical industries for quality control of drugs, stability study, impurity study and residual solvent estimation.
- It is used in environmental analysis for pollutants, pesticides, herbicides and volatile organic compounds in air, water and soil.
- It is used in forensic science for toxic substances, poisons, abused drugs and explosives.
- It is used in petrochemical industries for hydrocarbon mixtures present in gasoline, diesel and natural gas.
- It is used in food and beverage industries for flavours, fragrances, food additives and agricultural residues.
- It is used for natural products, especially essential oils and other complex extracts.
2. Supercritical Fluid Chromatography (SFC)
Principle of Supercritical Fluid Chromatography (SFC)
Supercritical Fluid Chromatography is based on use of supercritical fluid as mobile phase. Carbon dioxide is generally used. It is present above critical temperature and pressure.
- Carbon dioxide is kept above its critical temperature and pressure. In this condition it becomes supercritical fluid.
- It has low viscosity like gas and it can move fast through the column. It also has dissolving capacity like liquid.
- The sample is injected in the supercritical fluid. Then it passes through the column having stationary phase.
- Different substances do not move in same way. Some remain attached more with stationary phase and some are carried more by mobile phase.
- Separation is due to difference in polarity, size and affinity of the components.
- Temperature and pressure are changed to change the solvent strength of mobile phase.
- Methanol or ethanol is added with carbon dioxide for polar compounds.
Uses of Supercritical Fluid Chromatography (SFC)
- It is used in pharmaceutical analysis for drug discovery and quality control.
- It is used for separation of enantiomers.
- It is used for complex, lipophilic and heat sensitive drugs.
- It is used in food and beverage analysis for contaminants, nutritional additives and flavour compounds.
- It is used for decaffeination of coffee and extraction of hops.
- It is used in environmental analysis for pollutants, pesticides and hydrocarbons in air, water and soil.
- It is used for plant extracts and essential oils.
- It is used for polymers, waxes, fossil fuels and complex chemical mixtures.
3. Affinity chromatography

Principle of Affinity Chromatography
Affinity Chromatography is based on specific binding between target molecule and ligand. It is a selective method. The ligand is attached with solid stationary phase.
- In this chromatography, a specific ligand is fixed on the stationary phase matrix. The ligand may be antibody, enzyme substrate, inhibitor or metal ion.
- The sample mixture is passed through the column. Only the molecule having affinity for the ligand binds with it.
- Other unwanted molecules do not bind with the ligand. They pass through the column and are washed away.
- The bound molecule is then released from the column by changing the condition of mobile phase.
- Elution is done by changing pH, using high salt concentration or adding a competing ligand.
- In this method, separation is very specific because only the target molecule binds with the ligand.
Uses of Affinity Chromatography
- It is used for purification of specific proteins and enzymes from complex biological mixture.
- It is used for recombinant protein isolation having affinity tags like His-tag or GST-tag.
- Immobilized Metal Ion Affinity Chromatography is used for histidine tagged proteins. Nickel or zinc ions are used for binding.
- It is used in immunoaffinity separation for capturing antigen by antibody attached with matrix.
- It is also used for capturing antibodies by using immobilized proteins.
- It is used for removal of unwanted contaminants from protein preparations.
- It is used in biological and clinical studies for drug-protein and hormone-protein interaction.
- It is used in immunoassays.
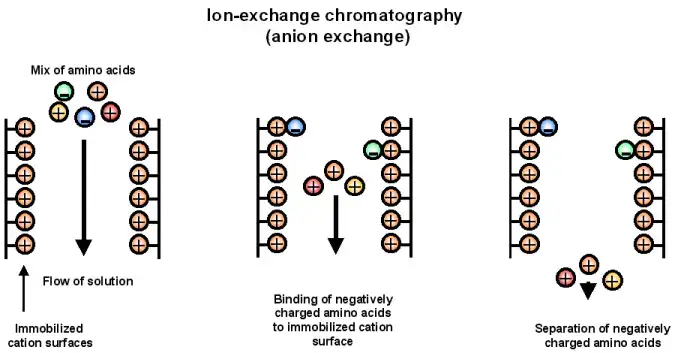
4. Ion exchange chromatography
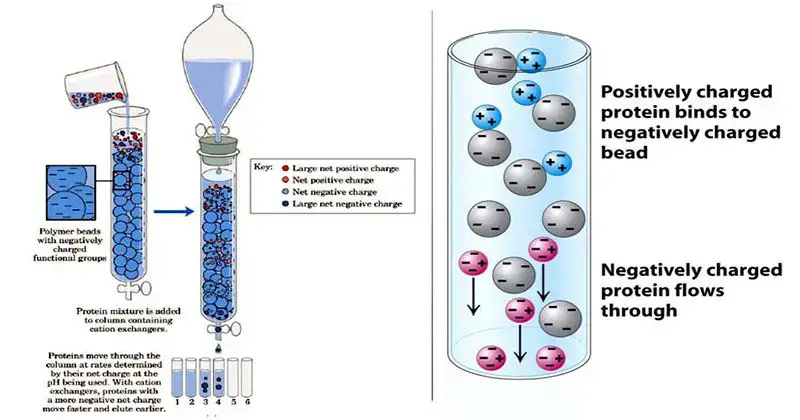
Principle of Ion Exchange Chromatography
Ion Exchange Chromatography is based on electrostatic attraction between charged molecules and oppositely charged stationary phase. The stationary phase has fixed ionic groups. The mobile phase is liquid buffer or electrolyte solution.
- In this chromatography, the stationary phase is an insoluble matrix. It is generally made of porous cross-linked resin beads having fixed ionic groups.
- The sample is carried by aqueous buffer or electrolyte solution through the column.
- The charged molecules in the sample bind with oppositely charged groups present on the resin.
- During binding, the analyte ions replace the mobile counter ions present on the stationary phase.
- The molecules which bind strongly remain in the column for longer time and move slowly.
- The bound molecules are released by increasing salt concentration or by changing the pH of mobile phase.
- When ionic strength is increased, the electrostatic attraction becomes weak and the molecules come out from the column.
- In cation exchange chromatography, negatively charged stationary phase is used and positively charged molecules are separated.
- In anion exchange chromatography, positively charged stationary phase is used and negatively charged molecules are separated.
Uses of Ion Exchange Chromatography
- It is used for separation and purification of charged biological molecules like proteins, amino acids and peptides.
- It is used for separation of nucleotides and DNA or RNA fragments.
- It is used in biotechnology for purification of enzymes, antibodies and hormones.
- It is used for charge variant analysis of monoclonal antibodies.
- It is used in environmental analysis for inorganic anions, cations, heavy metals and pollutants in water, soil and sewage sludge.
- It is used in clinical diagnosis for analysis of blood and urine samples.
- It is used for pharmaceuticals and drugs of abuse in toxicology work.
- It is used in water softening for removal of calcium, magnesium and iron ions.
- It is used in food and consumer product testing for food additives, sugars and other product checking.
5. Hydrophobic Interaction Chromatography (HIC)
Principle of Hydrophobic Interaction Chromatography (HIC)
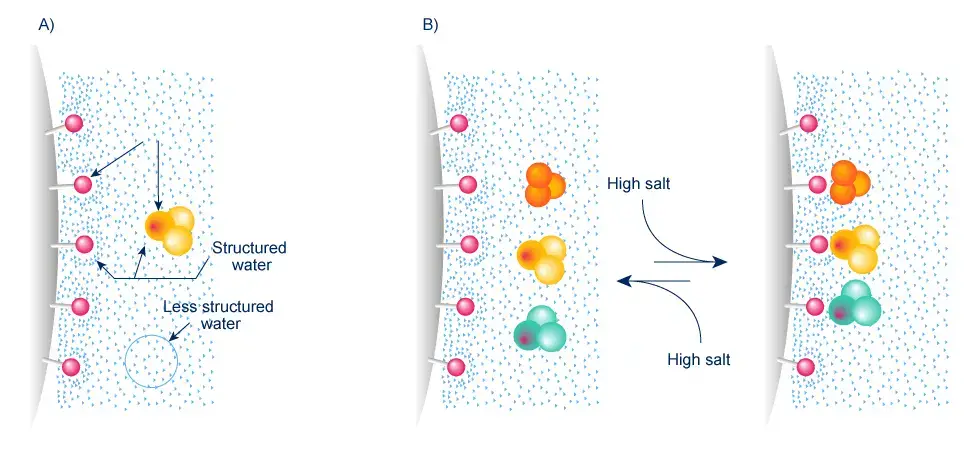
Hydrophobic Interaction Chromatography is based on hydrophobic nature of molecules. It is mainly used for proteins. The hydrophobic parts present on protein surface bind with hydrophobic stationary phase.
- In this chromatography, the stationary phase has mild hydrophobic groups attached with the matrix. Methyl, ethyl, propyl, butyl, octyl or phenyl groups are used.
- The sample is loaded in aqueous buffer having high salt concentration. Ammonium sulfate is commonly used.
- High salt condition makes hydrophobic parts of protein bind with the hydrophobic groups of stationary phase.
- The binding occurs by hydrophobic interaction and Van der Waals interaction.
- The proteins are separated because all proteins do not have same hydrophobic nature.
- The proteins are eluted by decreasing the salt concentration slowly.
- Elution may also be done by changing pH or by adding organic solvent or detergent.
- The proteins come out according to their hydrophobicity. More hydrophobic proteins are retained for longer time.
Uses of Hydrophobic Interaction Chromatography (HIC)
- It is used for separation and purification of proteins and enzymes.
- It is used for large biomolecules which have hydrophobic patches on their surface.
- It is a non-denaturing method because aqueous solution is used.
- It keeps the native three dimensional structure and biological activity of delicate proteins.
- It is used in capture step, intermediate purification and final polishing step.
- It is used after ammonium sulfate precipitation because sample already has high salt concentration.
- It is also used after ion exchange chromatography when high ionic strength condition is present.
- It is useful for samples which are sensitive to pH change or organic solvent but can tolerate high salt.
- Salt independent HIC methods are used for delicate samples like human antibodies from serum.
6. Hydrophilic Interaction Chromatography (HILIC)
Principle of Hydrophilic Interaction Chromatography (HILIC)
Hydrophilic Interaction Chromatography is based on separation of polar substances. It uses polar stationary phase and high organic mobile phase. A water rich layer is formed on the surface of stationary phase.
- In this chromatography, hydrophilic stationary phase is used and the mobile phase contains high amount of organic solvent.
- The substances are separated by partition between organic rich mobile phase and water enriched layer present on polar stationary phase.
- Hydrogen bonding and electrostatic interaction also take part in separation.
- The hydrophobic substances come out first from the column.
- The highly polar substances remain more with the stationary phase and come out later.
- The elution order is generally opposite to reverse phase chromatography.
- HILIC can be done by isocratic method, but gradient method is commonly used.
- In gradient elution, aqueous content of the buffer is increased and more hydrophilic substances come out.
Uses of Hydrophilic Interaction Chromatography (HILIC)
- It is used in metabolomics study for separation of polar metabolites.
- It is used for analysis of hydrophilic metabolites.
- It is used for simultaneous analysis of different hydrophilic metabolites.
- It does not need ion-pairing reagents for many hydrophilic metabolites.
- It is used with reverse phase chromatography as a complementary method.
- It gives wider coverage of analytes when used with reverse phase chromatography.
7. Mixed-Mode Chromatography
Principle of Mixed-Mode Chromatography
Mixed-Mode Chromatography is based on more than one type of separation in same resin or same column. It is also called multimodal chromatography. In this method, two or more interactions work together.
- In this chromatography, the stationary phase has more than one type of binding site.
- It may combine reversed phase type hydrophobic interaction and ion exchange type electrostatic interaction in same resin.
- The molecules are separated by more than one property at same time, such as charge and hydrophobicity.
- This gives better separation when mixture is complex.
- It is useful when single mode chromatography cannot separate closely related compounds properly.
Uses of Mixed-Mode Chromatography
- It is used for separation of complex mixtures where normal single mode chromatography gives poor resolution.
- It is used for separation of closely related compounds.
- It is used in protein and biomolecule purification.
- It is used in capture step, intermediate purification and final polishing step.
- It is used for analysis of hydrophilic metabolites.
- It can analyse many hydrophilic metabolites without using complex ion-pairing reagents in mobile phase.
8. Size-Exclusion Chromatography (SEC)
Principle of Size-Exclusion Chromatography (SEC)
Size-Exclusion Chromatography is based on separation of molecules according to their size or hydrodynamic volume. It is not based on chemical attraction with stationary phase. It is also called gel filtration chromatography or gel permeation chromatography.
- In this chromatography, the column is packed with porous gel or polymer beads. Dextran, agarose or cross-linked polyacrylamide are commonly used.
- The beads contain many pores of uniform size. The mobile phase carries the sample through these porous beads.
- The molecules are separated according to their ability to enter into the pores of stationary phase.
- Large molecules cannot enter the pores. So they pass through the space between beads and come out first.
- Small molecules enter inside the pores. So they take longer path and come out later.
- In this method, separation occurs only by size difference and not by adsorption or charge interaction.
- When aqueous solution is used as mobile phase, it is generally called gel filtration chromatography.
- When organic solvent is used as mobile phase, it is generally called gel permeation chromatography.
Uses of Size-Exclusion Chromatography (SEC)
- It is used for determination of molecular weight of proteins, peptides, polysaccharides and synthetic polymers.
- It is used to know the size distribution of polymers and biomolecules.
- It is used for separation and study of large biological molecules like proteins, nucleic acids, enzymes and viruses.
- It is used for desalting of protein solution.
- It is used for buffer exchange, because small salts and buffer molecules come out separately from large proteins.
- It is used as final purification step for removal of aggregates, protein fragments and unbound components.
- It is used for structural study of purified proteins.
- It can be done under native and non-denaturing condition, so protein structure is not damaged easily.
9. Paper Chromatography
Principle of Paper Chromatography
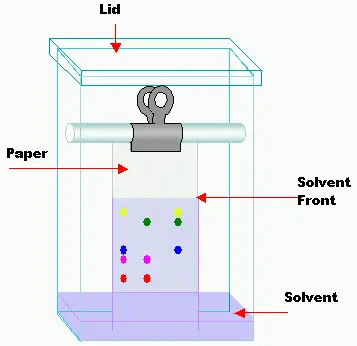
Paper Chromatography is mainly based on partition of substances between stationary phase and mobile phase. Some adsorption may also take part. The paper is cellulose paper and water held on cellulose acts as stationary phase.
- In this chromatography, moisture or water molecules present on cellulose fibres of filter paper act as stationary phase.
- The mobile phase is organic solvent or mixture of solvents.
- The sample is placed on paper as a small spot or line.
- The paper is kept in a closed chamber having small amount of mobile phase.
- The solvent moves upward through the pores of paper by capillary action and carries the sample components with it.
- The components separate because their solubility in water phase and organic mobile phase are not same.
- More polar substances remain more with polar cellulose paper and move slowly.
- Less polar substances remain more with mobile phase and travel more distance.
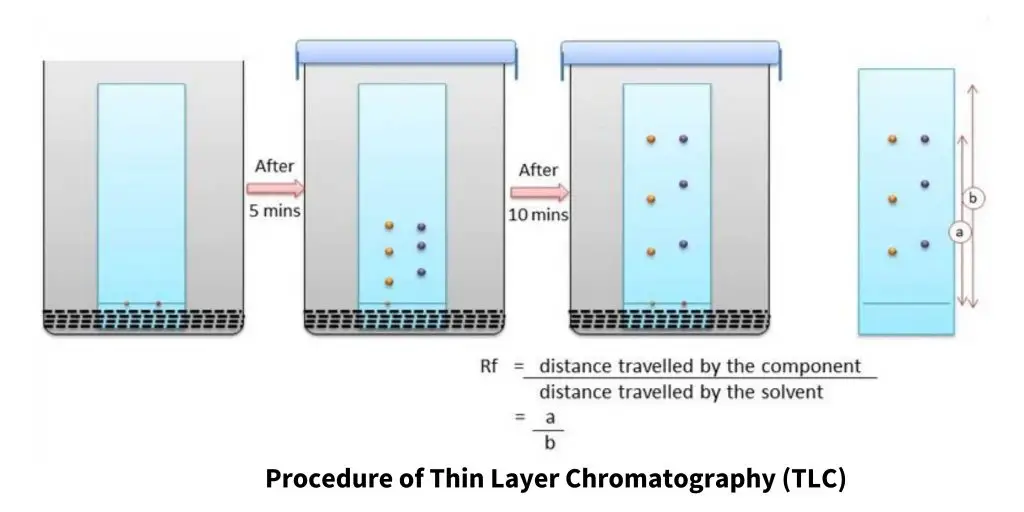
- The separation is measured by Rf value. It is the distance travelled by compound divided by distance travelled by solvent front.
Uses of Paper Chromatography
- It is used for separation of polar and non-polar substances present in complex mixtures.
- It is used for separation and identification of inks and dyes.
- It is used for separation of coloured plant pigments.
- It is used in biochemical analysis for amino acids, hormones and biological substances present in urine.
- It is used in medical and pharmaceutical work for detection of medicines and illicit drugs.
- It is used in forensic laboratory for fingerprinting, DNA and RNA analysis.
- It is used in inorganic chemistry for separation of salts and complexes.
10. Flash chromatography
Principle of Flash Chromatography
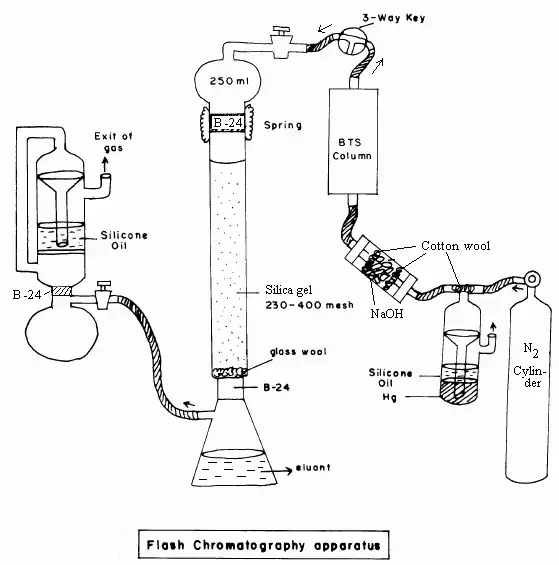
Flash Chromatography is a modified form of column chromatography. It is faster than ordinary column chromatography. In this method, pressure is used for movement of mobile phase through the column.
- Flash chromatography does not depend only on gravity for movement of solvent.
- Positive pressure is applied to push the liquid mobile phase through the column.
- The pressure used is low pressure, but it makes the solvent flow faster than gravity method.
- The column generally contains pre-packed plastic cartridge in modern flash chromatography.
- The sample components are separated in the column according to their affinity for stationary phase and mobile phase.
- Modern flash system may have gradient pumps, detectors and fraction collectors.
- Due to pressure and automation, the separation becomes faster and solvent use is also reduced.
Uses of Flash Chromatography
- It is used for fast preparative purification of compounds.
- It is used when separation is needed in short time.
- Most separations can be completed in less than 20 minutes.
- It gives better separation quality than old gravity column method.
- It is used in research and chemistry laboratories.
- It is used for separation, isolation and purification of small organic molecules.
- It is used for natural products, peptides and proteins.
11. Liquid chromatography
Principle of Liquid Chromatography (LC)
Liquid Chromatography is based on distribution of components between a liquid mobile phase and a stationary phase. The stationary phase may be solid or liquid coated on solid support. The mobile phase is liquid and it moves the sample through the column.
- The sample mixture is dissolved in liquid mobile phase and introduced into the column containing stationary phase.
- The liquid mobile phase passes through the column and carries the components of the mixture with it.
- The components are not retained equally in the column. Some components remain more with stationary phase and some remain more with mobile phase.
- The retention depends on nature of the compound such as polarity, size and charge.
- The components having more attraction for stationary phase are retained for longer time. They move slowly.
- The components having more attraction for liquid mobile phase move faster and come out earlier.
- Thus the separation takes place due to different movement of components in the column.
- In HPLC, high pressure pump is used to push the liquid mobile phase through fine packed column.
- Due to high pressure and fine particles, separation becomes faster and more clear.
Uses of Liquid Chromatography (LC)
- Liquid Chromatography is used in pharmaceutical analysis for drug development and quality control.
- It is used for estimation of active pharmaceutical ingredients (APIs), impurities and degradation products.
- It is used for dissolution testing, pharmacokinetic study and bioequivalence study.
- It is used in environmental monitoring for detection of pollutants in water, soil and air.
- It is used for analysis of pesticides, herbicides and pharmaceutical residues.
- It is used in food and beverage industries for checking nutrients, additives and preservatives.
- It is used for detection of harmful contaminants in food materials.
- It is used in biochemistry for separation of non-volatile and heat sensitive compounds.
- It is used for purification and analysis of proteins, peptides, amino acids and nucleic acids.
- It is used in clinical diagnosis for measuring drugs, biomarkers and hormones in blood and urine.
- It is used in forensic science for analysis of drugs of abuse, toxins and chemical evidences.
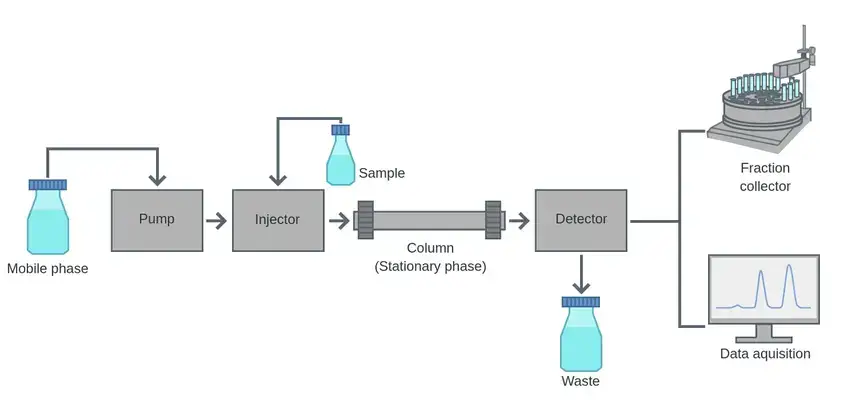
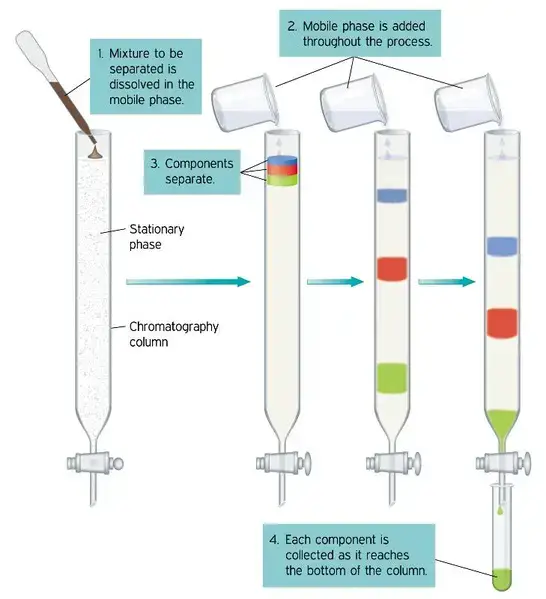
12. Column chromatography
Principle of Column Chromatography
Column Chromatography is based on separation of components in a packed column. The stationary phase is packed inside a cylindrical tube. Silica gel or alumina is commonly used.
- The stationary phase may be solid material or liquid coated on solid support. It remains fixed inside the column.
- The sample mixture is dissolved in mobile phase. The mobile phase may be liquid or gas.
- The mobile phase passes through the packed column by gravity or by external pressure.
- Different components do not move equally in the column. Some remain more with stationary phase and some move more with mobile phase.
- The components which are held more by stationary phase move slowly down the column.
- The components which have less attraction for stationary phase move faster and come out first.
- Separation may occur by adsorption, partition, size-exclusion, ion-exchange or biological affinity according to the column material used.
Uses of Column Chromatography
- It is used for preparative purification of compounds from a complex mixture.
- It is used to isolate and collect a required compound in larger amount.
- It is used for isolation of natural products like plant pigments, essential oils and small organic molecules.
- It is used for separation and purification of proteins, enzymes, amino acids, viruses and nucleic acids.
- It is used as basic column system in HPLC and Gas Chromatography.
- It is used in pharmaceutical industries for drug development and checking purity of products.
- It is used for detection of trace impurities in chemical and pharmaceutical samples.
- It is used in chemical industries and environmental analysis for monitoring pollutants.
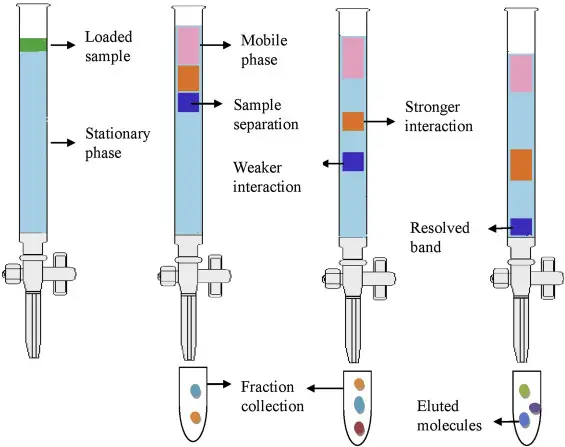
13. High-performance liquid chromatography (HPLC)
Principle of High-Performance Liquid Chromatography (HPLC)
High-Performance Liquid Chromatography is based on distribution of components between liquid mobile phase and stationary phase. The stationary phase may be solid or liquid coated on solid support. The mobile phase is liquid and it is forced through the column by high pressure.
- In HPLC, high pressure pump is used to push the sample and solvent through a tightly packed column.
- The pressure is generally about 4000 to 6000 psi. So the mobile phase moves fast through the column.
- The column is packed with very small particles. These particles are generally 3 to 5 microns in size.
- Small particles give large surface area for interaction between sample components and stationary phase.
- Different components of the mixture do not move in same way. Some remain more with stationary phase and some remain more with mobile phase.
- The compounds which interact more with stationary phase are retained for longer time and move slowly.
- The compounds which remain more with mobile phase move faster and come out earlier.
- Due to high pressure and fine particles, HPLC gives faster separation, better sensitivity and good resolution.
- Reversed Phase HPLC is the common type. It uses non-polar stationary phase and polar mobile phase.
- Normal Phase HPLC uses polar stationary phase and non-polar mobile phase. It is used according to the polarity of analytes.
Uses of High-Performance Liquid Chromatography (HPLC)
- HPLC is used in pharmaceutical industries for drug development and quality control.
- It is used for estimation of active pharmaceutical ingredients, trace impurities and degradation products.
- It is used for dissolution test and pharmacokinetic testing of drugs.
- It is used in environmental analysis for pesticides, herbicides and pharmaceutical residues in water, soil and air.
- It is used in food and beverage analysis for nutrients, vitamins, food additives and preservatives.
- It is used for detection of harmful contaminants in food products.
- It is used in clinical diagnosis for hormones, biomarkers and therapeutic drugs in blood and urine.
- It is used in forensic laboratories for toxins, poisons and drugs of abuse.
- It is used in biochemistry and proteomics for proteins, peptides and metabolites.
- It is useful for non-volatile and heat sensitive compounds.
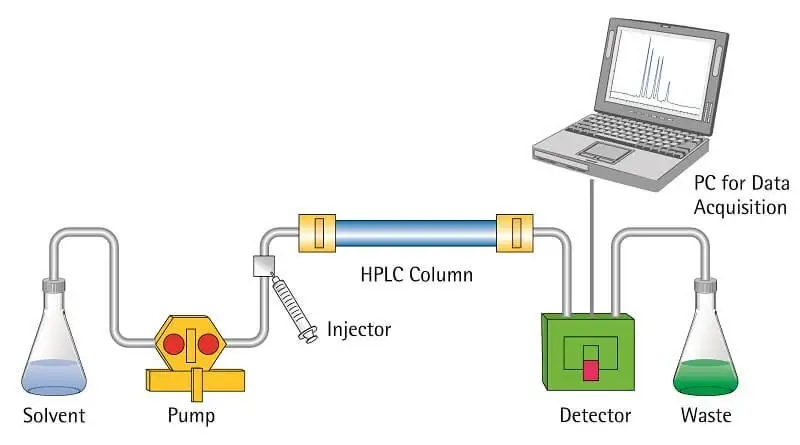
14. Ultra-High-Performance Liquid Chromatography (UHPLC)
Principle of Ultra-High-Performance Liquid Chromatography (UHPLC)
Ultra-High-Performance Liquid Chromatography works on same principle as HPLC. It separates components between liquid mobile phase and solid stationary phase. The main difference is that very small particles and very high pressure are used.
- In UHPLC, the column is packed with very fine stationary phase particles. These particles are generally less than 2 microns in size.
- The liquid mobile phase is forced through the tightly packed column by very high pressure.
- The pressure used in UHPLC is very high and it may be more than 15000 psi.
- Different components of the mixture move differently in the column. Some remain more with stationary phase and some move more with mobile phase.
- Small particles reduce band broadening. So the peaks become sharp and separation becomes better.
- Due to high pressure, the mobile phase can move at faster flow rate and analysis is completed in less time.
- UHPLC gives higher resolution, better efficiency and more sensitivity than ordinary HPLC.
Uses of Ultra-High-Performance Liquid Chromatography (UHPLC)
- UHPLC is used in pharmaceutical testing where large number of samples are analysed in short time.
- It is used in bioequivalence study, batch release testing and stability study.
- It is used for analysis of complex mixtures where compounds come out very close to each other.
- It is used for impurity profiling and detection of degradation products.
- It is used in pharmacokinetic and toxicology work for drugs and their metabolites.
- UHPLC is often used with mass spectrometry for trace level detection in plasma, serum and tissue samples.
- It is used in environmental and food safety analysis for trace contaminants and pollutants.
- It reduces analysis time and uses less organic solvent.
- It produces less hazardous waste, so it is useful in green analytical chemistry.
15. High-Performance Thin-Layer Chromatography (HPTLC)
Principle of High-Performance Thin-Layer Chromatography (HPTLC)
High-Performance Thin-Layer Chromatography is an advanced form of TLC. It uses a thin layer of stationary phase on glass or aluminium plate. The mobile phase moves over the plate by capillary action.
- In HPTLC, separation depends on the affinity of components for stationary phase and solubility in mobile phase.
- The components which remain more with stationary phase move slowly on the plate.
- The components which dissolve more in mobile phase move faster with the solvent.
- HPTLC plates have fine and uniform particles. These particles are generally 5 to 6 µm in size.
- Due to fine particles and compact packing, diffusion of sample is less and bands become sharp.
- It gives better resolution than ordinary TLC.
- In HPTLC, sample application is done by automated method. The sample is placed on plate as proper narrow band.
- The separated bands are detected by scanner. UV absorption, fluorescence or mass spectrometry may be used.
Uses of High-Performance Thin-Layer Chromatography (HPTLC)
- HPTLC is used in pharmaceutical industries for quality control, purity checking and estimation of active ingredients in drug formulations.
- It is used for fingerprinting of medicinal plants and herbal products.
- It is used for identification of active phytochemicals and detection of adulteration in herbal supplements.
- It is used in clinical and biomedical study for metabolism tests and drug level estimation in blood serum and tissues.
- It is used for separation of complex lipid subclasses.
- It is used in forensic science for toxicology study and detection of illicit drugs, poisons and misuse substances.
- It is used in food and environmental testing for pesticides, pollutants, food additives and vitamins.
- It is used in food, beverages and agricultural products analysis.
- It is used in lichenology for rapid screening and identification of lichen compounds.
16. Reverse-phase chromatography
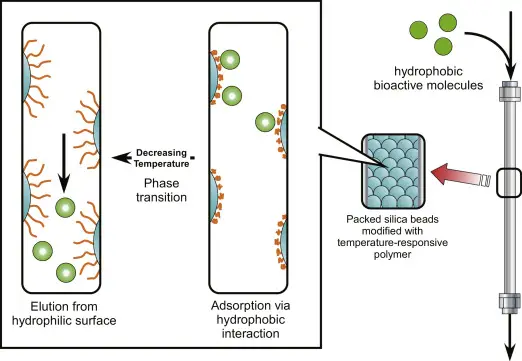
Principle of Reversed-Phase Chromatography
Reversed-Phase Chromatography is opposite to normal phase chromatography. It is based on hydrophobic interaction. The stationary phase is non-polar and mobile phase is polar.
- In this chromatography, the column contains non-polar stationary phase.
- Silica particles are generally bonded with non-polar hydrocarbon chain.
- C8 or C18 groups are commonly used as stationary phase.
- The mobile phase is polar. It is usually water mixed with methanol or acetonitrile.
- Polar substances remain more with polar mobile phase and move faster through the column.
- Non-polar substances bind more with non-polar stationary phase and move slowly.
- The substances come out according to their hydrophobic nature.
- More hydrophobic substances are retained for longer time and come out later.
Uses of Reversed-Phase Chromatography
- It is commonly used in modern HPLC methods.
- It is used in pharmaceutical analysis for drug testing and assay of drugs.
- It is used for checking purity of active ingredients.
- It is used for separation of non-polar and moderately polar compounds from complex mixtures.
- It is used in chemical laboratories for identification and estimation of analytes.
- It is used in environmental analysis and food safety work.
- It is used in routine quality control and research work.
17. Fast Protein Liquid Chromatography (FPLC)
Principle of Fast Protein Liquid Chromatography (FPLC)
Fast Protein Liquid Chromatography is based on separation of biomolecules by using liquid mobile phase and porous stationary phase. It is mainly used for proteins and nucleic acids. The condition is kept gentle so that the biomolecules are not damaged.
- In FPLC, aqueous solution or buffer is used as mobile phase.
- The mobile phase is passed through the column by positive displacement pump. It keeps the flow rate constant.
- The buffer composition can be changed by mixing fluids from different reservoirs.
- The stationary phase is resin beads packed inside glass or plastic cylindrical column.
- Cross-linked agarose is commonly used as the bead material.
- FPLC works at low pressure. The pressure is generally less than 600 psi.
- The flow rate may vary from 1 to 150 mL/min.
- The components are separated because they have different affinity for mobile phase and stationary phase.
- Aqueous buffer and low pressure make the process mild. So delicate proteins and other biomolecules do not denature easily.
Uses of Fast Protein Liquid Chromatography (FPLC)
- FPLC is used for purification of large and delicate biomolecules.
- It is mainly used for proteins and nucleic acids.
- It is used for analysis and separation of complex protein mixtures.
- It is used in biotechnology laboratories.
- It is used in protein research laboratories for sensitive biological samples.
- It is used with different separation methods such as ion exchange chromatography.
- It is used when protein should remain active and native during purification.
18. Planar Chromatography
Principle of Planar Chromatography
Planar Chromatography is based on separation of substances on a flat surface. The stationary phase is not packed in a column. It is present as paper or as a thin layer on plate.
- In this chromatography, the stationary phase is arranged as a flat plane or surface.
- The stationary phase may be cellulose paper in paper chromatography.
- It may also be a thin layer of adsorbent like silica gel, alumina or cellulose coated on glass, plastic or aluminium plate.
- The sample is applied on the flat surface as small spot or band.
- The plate or paper is placed in liquid mobile phase and the solvent moves through it by capillary action.
- The components move different distances on the surface because their attraction for stationary phase and mobile phase are not same.
- The components which remain more with stationary phase move slowly.
- The components which remain more with mobile phase move farther with the solvent.
- The separation is measured by Rf value. It is the distance travelled by solute divided by distance travelled by solvent front.
- Colourless compounds are detected by UV light, radioactivity or by spraying chemical reagents which produce visible colour.
Uses of Planar Chromatography
- It is used for identification of unknown substances by comparing Rf value with known standard.
- It is used for analysis of many samples on same plate.
- It is simple, fast and low cost method.
- Paper chromatography is used for separation of plant pigments, amino acids and dyes.
- High Performance TLC is used for fingerprinting of herbal medicines.
- It is used for identification of active phytochemicals and detection of adulterants in supplements.
- It is used in pharmaceutical industries for quality control and purity testing.
- It is used in clinical chemistry for screening drug levels in biological fluids like serum.
- It is used for water purity testing and environmental pollutants.
- It is used for detection of pesticides and trace contaminants in food and agricultural products.
- It is used in forensic science for identification of poisons, illicit drugs and misuse substances in biological media.
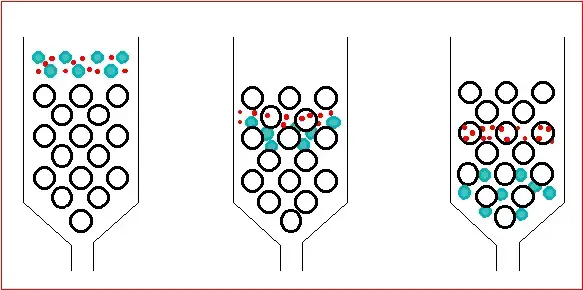
19. Adsorption Chromatography
Adsorption Chromatography is a type of chromatography in which separation takes place by adsorption of substances on solid stationary phase. The stationary phase has large surface area. Silica gel, alumina and activated carbon are commonly used.
- In this chromatography, the components are separated according to their adsorption strength on the surface of solid stationary phase.
- The mobile phase carries the sample over the solid surface. The mobile phase may be liquid or gas.
- Different components do not attach on the solid surface in same amount. Some are adsorbed strongly and some are weakly adsorbed.
- The components which are strongly adsorbed on stationary phase move slowly.
- The components which are weakly adsorbed remain more with mobile phase and move faster.
- Separation mainly depends on polarity of compounds. Polar compounds are generally adsorbed more strongly on the stationary phase.
- Non-polar compounds are less adsorbed and move faster with the mobile phase.
- Hydrogen bonding, van der Waals force and dipole-dipole interaction also take part in adsorption.
Uses of Adsorption Chromatography
- It is used for isolation of natural products from plant and other natural sources.
- It is used for separation of plant pigments.
- It is used for purification of small organic molecules and drugs.
- It is used in pharmaceutical and industrial laboratories for rapid purification.
- It is used in preparative chromatography when compounds have different polarity.
- It is used for large scale separation of compounds from mixture.
- It is used for separation of isomers having same chemical formula but different structure.
20. Partition Chromatography
Principle of Partition Chromatography
Partition Chromatography is based on distribution of substances between two immiscible phases. It is not based on adsorption on surface. The separation occurs due to different solubility of components in the two phases.
- In this chromatography, stationary phase is a liquid film coated on inert solid support or bonded with it.
- The mobile phase carries the sample through the system. It may be liquid or gas.
- The components of mixture divide between stationary liquid phase and mobile phase.
- The separation depends on partition coefficient of the component. It is the ratio of concentration in stationary phase and mobile phase.
- The components which are more soluble in stationary phase remain there for longer time and move slowly.
- The components which are more soluble in mobile phase move faster and come out earlier.
- This method generally gives symmetrical peaks and better resolution than adsorption chromatography.
Uses of Partition Chromatography
- It is used in pharmaceutical and biomedical research for separation and purification of active pharmaceutical ingredients.
- It is used for detection of trace impurities and drug metabolites.
- It is used in biochemical analysis for separation of amino acids, peptides and nucleotides.
- It is used in environmental and food safety work for detection of pesticides, pollutants and organic contaminants.
- It is used for analysis of water, soil and food samples.
- It is used in DNA and RNA sequencing work for isolation and purification of nucleic acid fragments.
- It is the basic principle of gas-liquid chromatography, liquid-liquid chromatography, HPLC and paper chromatography.
References
- A comparative review on High-Performance Liquid Chromatography HPLC Ultra Performance Liquid Chromatography UPLC High-Performance Thin Layer Chromatography HPTLC with current updates. (n.d.). ResearchGate.
- Harvey, D. (2021). 12.6: Other forms of chromatography. Chemistry LibreTexts.
- ALWSCI. (2025). A comparative analysis of adsorption chromatography and partition chromatography.
- Khan Academy. (n.d.). A closer look at types of chromatography.
- Shimadzu. (n.d.). Advanced analysis with supercritical fluids chromatography.
- Chaudhari, R., Suryawanshi, S., Chitkalwar, S., Patil, J., Gawali, U., Biradar, S., Sanap, S., Jengthe, P., Hole, P., & Deore, H. (2025). Advanced chromatographic techniques in pharmaceutical analysis. International Journal of Pharmaceutical Sciences, 3(7), 299-319. https://doi.org/10.5281/zenodo.15788069
- Advances in chromatography: Contemporary techniques and applications. (n.d.). Taylor & Francis.
- Kannan, R. (n.d.). Basics of chromatographic techniques course 1. C-CAMP.
- Biochromatography size-exclusion and ion-exchange techniques. (n.d.). LCGC International.
- Shelke, A., Yede, P., & Deokar, R. (n.d.). Chromatography: Types, basic principles and uses. International Journal of Scientific Research and Engineering Development, 7(4), 257-264.
- Premnath, S. M., & Zubair, M. (2024). Chromatography. In StatPearls. StatPearls Publishing.
- Chromatography. (2026). In Wikipedia.
- Jack Westin. (n.d.). Chromatography basic principles involved in separation process.
- BCC Research. (2025). Chromatography technologies powering the future of biopharmaceuticals.
- Sartorius. (2022). Chromatography – A journey through time.
- BYJU’S. (n.d.). Classification of chromatography based on the physical state of the mobile phase.
- PatSnap Eureka. (2025). Comparing HPLC and GC: Retention time and effectiveness.
- Comparison of sfc, gc and hplc. (n.d.). SlideShare.
- Danaher Life Sciences. (n.d.). Difference between UHPLC and traditional HPLC.
- Different chromatographic techniques and recent advancements for biomedical and pharmaceutical applications. (n.d.). PMC.
- Future-proofing chromatography: AI and automation at the helm. (n.d.). Lab Manager.
- 24 Market Reports. (2026). Gas chromatograph system market 2026 forecast to 2032.
- Research and Markets. (2025). Gas chromatography market – application trends & forecast 2025-2033.
- Meher, A. K., & Zarouri, A. (2025). Green analytical chemistry—recent innovations. Analytica, 6(1), 10. https://doi.org/10.3390/analytica6010010
- Waters Corporation. (n.d.). HPLC separation modes – stationary phase in HPLC.
- Boag, M. (2025). HPLC vs UHPLC: Key differences & applications. Phenomenex.
- Chromatography Today. (n.d.). HPLC vs UPLC – What’s the difference?
- PatSnap Eureka. (2025). HPLC vs UPLC: Resolution and throughput compared.
- Sigma-Aldrich. (n.d.). High-performance thin layer chromatography (HPTLC) plates.
- Kulkarni, R. N., Pandhare, R. B., Deshmukh, V. K., Mohite, P. B., & Pawar, A. R. (2021). High-performance thin layer chromatography: A powerful analytical technique in pharmaceutical drug discovery. Journal of Pharmaceutical and Biological Sciences, 9(1), 7-14. https://doi.org/10.18231/j.jpbs.2021.002
- High-performance thin layer chromatography: A powerful analytical technique in pharmaceutical drug discovery. (n.d.). PMC.
- High-performance thin-layer chromatography. (2025). In Wikipedia.
- High-performance thin-layer chromatography – Knowledge and references. (n.d.). Taylor & Francis.
- Chrom Tech, Inc. (2025). History of chromatography: From past to present.
- History of chromatography. (2025). In Wikipedia.
- Innovations in liquid chromatography: 2025 HPLC column and accessories review. (n.d.). LCGC International.
- American Laboratory Trading. (2024). Liquid chromatography comparison: HPLC vs UPLC vs FPLC.
- Dong, M. W. (2025). New HPLC, MS, and CDS products from 2024–2025: A brief review. LCGC International. https://doi.org/10.56530/lcgc.int.lq3471r4
- Interchim. (2019). Origins, evolution & principle of preparative and analytical liquid chromatography.
- Agriculture.Institute. (2025). Principles of chromatographic separation: Adsorption and partitioning.
- Recent advancements in high-performance liquid chromatography: A comparative approach. (n.d.). ResearchGate.
- Separation techniques: Chromatography. (n.d.). PMC.
- Wong, S. H. (1989). Supercritical fluid chromatography and microbore liquid chromatography for drug analysis. Clinical Chemistry, 35(7), 1293-1298.
- Marty, M. (2023). Supercritical fluid chromatography and its advantages. Pharmaceutical Analytical Chemistry, 8, 206. https://doi.org/10.35248/2471-2698.23.8.206
- The analytical architecture of modern chromatography: A comprehensive report on classifications, mechanisms, and evolutionary trajectories. (n.d.).
- Najafi, R. (2025). The evolution of gas chromatography and later GC-MS and LC-MS. One man’s journey from student to CEO. Emery Pharma.
- Teledyne LABS. (n.d.). The science of separation: Understanding high-performance liquid chromatography.
- Bio-Rad. (n.d.). Types of chromatography.
- Sehar, R. (n.d.). Types of ion exchange chromatography. Scribd.
- Chrom Tech, Inc. (2025). Understanding partition chromatography: Key principles and uses.
- Voices of separation science: Expert insights, emerging trends… (n.d.). LCGC International.
- Teledyne LABS. (n.d.). What is supercritical fluid chromatography.
- Teledyne LABS. (n.d.). The science of separation: Understanding high-performance liquid chromatography (Highlighted textlink).