Paper Chromatography is a simple analytical technique used to separate and identify the components present in a chemical mixture. It is based on the principle of partition, where the substances are distributed between stationary phase and mobile phase. In this method, the stationary phase is usually a cellulose filter paper containing moisture within its fibers, whereas the mobile phase is a liquid solvent or mixture of solvents.
During this process, the lower edge of the paper is dipped into the solvent. The solvent then moves upward through the paper by capillary action carrying the dissolved sample components along with it. Different substances move at different speed because each component has different solubility and affinity towards stationary phase and mobile phase. The substances which are more soluble in solvent move faster and travel longer distance, while other substances move slowly on the paper.
The separated substances can be identified by measuring the (Rf) value (Retention factor). Paper chromatography is widely used in forensic science, pharmaceutical analysis, food testing and environmental studies because it is simple, inexpensive and requires very small amount of sample.
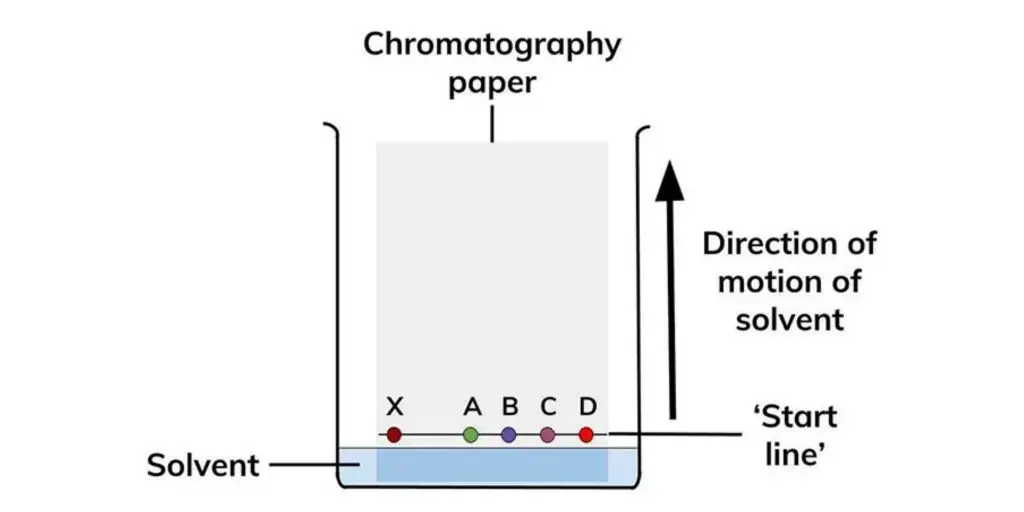
Principle of Paper chromatography
Paper chromatography is based on the principle of partition and also some adsorption between two phases. The two phases are stationary phase and mobile phase. In this method, the stationary phase is the thin layer of water or moisture held in the pores of cellulose paper.
The mobile phase is a solvent or mixture of solvents which moves through the paper by capillary action. When the solvent moves, it carries the applied sample along with it. The different components of the sample are distributed differently between the stationary moisture and moving solvent.
The components which are more soluble in the mobile phase move faster and travel more distance on the paper. The components which have more attraction towards the stationary phase remain behind and move slowly. In this way the mixture is separated into different components.
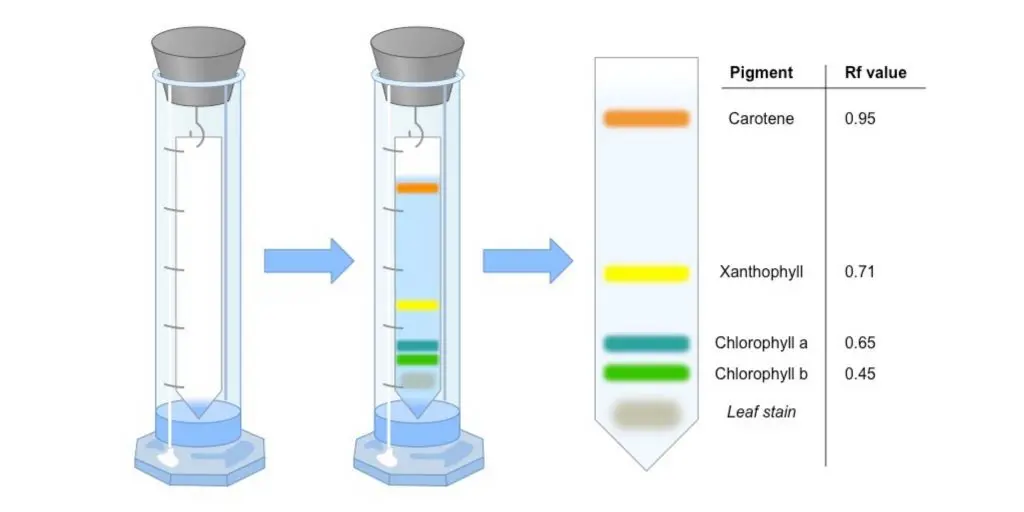
Types of Paper chromatography
The following are the types of Paper chromatography–
- Paper Adsorption Chromatography
In this type, the paper is impregnated with substances like silica or alumina which acts as stationary phase. The solvent acts as mobile phase and separation occurs by adsorption process. - Paper Partition Chromatography
It is the common type of Paper chromatography. In this method, the moisture present within the cellulose fibers acts as stationary phase and solvent acts as mobile phase. The separation occurs due to partition of substances between two liquid phases. - Ascending Chromatography
In this method, the solvent is placed at the lower part of chamber and it moves upward through the paper by capillary action against gravity. - Descending Chromatography
In this type, the solvent is kept at the upper side of chamber and it moves downward on the paper. The downward movement is helped by gravity. - Ascending-Descending Chromatography
It is a combined method of ascending and descending chromatography. The solvent first moves upward and then after crossing a rod it flows downward through the paper. - Circular or Radial Chromatography
In this method, the sample is applied at the center of circular filter paper. The solvent moves radially from the center towards outer side and separates the components into circular bands. - Two-Dimensional Chromatography
In this type, the sample is first separated in one direction on the paper. After drying, the paper is rotated at 90° angle and again developed in another solvent. It is mainly used for separation of complex mixtures.
Instrumentation of Paper chromatography
The following are the instruments and parts used in Paper chromatography–
- Chromatography paper
It acts as the stationary phase in Paper chromatography. It is made up of fine quality cellulose filter paper, such as Whatman filter paper. It has pores which hold a thin layer of moisture and help in separation of components. - Mobile phase
It is a solvent or mixture of solvents used to move through the paper. The solvent may be pure solvent, buffer solution or mixture of organic and inorganic solvents. It carries the sample components along with it by capillary action. - Sample applicator
It is used to apply a small amount of sample on the starting line of the paper. Capillary tube, micropipette or toothpick are commonly used for spotting the sample. - Chromatographic chamber
It is also called developing tank. It is a closed container made of glass, plastic or stainless steel in which the development of chromatogram takes place. The chamber is covered with lid so that solvent vapour remains saturated inside it and evaporation of solvent is reduced. - Detecting or visualizing agent
It is used to detect the separated components when they are colourless. UV light chamber, iodine vapour and chemical sprays such as ninhydrin are used for visualization of spots.
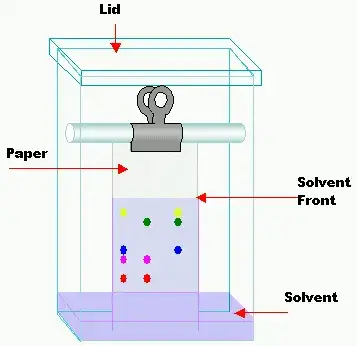
Steps in Paper Chromatography
The following are the steps in Paper chromatography–
- A good quality cellulose filter paper is selected and a suitable mobile phase or solvent mixture is also selected according to the nature of sample.
- The chromatography paper is cut into required size and a straight line is drawn with pencil about 1-2 cm above the lower edge of the paper. This line is called origin line.
- The solvent is poured at the bottom of developing chamber and the chamber is closed for some time. This makes the inside atmosphere saturated with solvent vapour.
- The sample is dissolved in suitable solvent if needed. A small spot of sample is placed on the pencil line with the help of capillary tube or micropipette.
- The paper is suspended in the chamber in such a way that lower end of paper touches the solvent but the sample spot remains above the solvent level.
- The solvent moves upward through the paper by capillary action. It carries the sample components along with it and different components move at different distance.
- When the solvent front reaches near the upper edge of the paper, the paper is taken out and the solvent front is marked immediately with pencil.
- The paper is dried and the separated spots are observed. If the spots are colourless, then iodine vapour, UV light or ninhydrin spray is used for detection.
Rf values – Retention factors
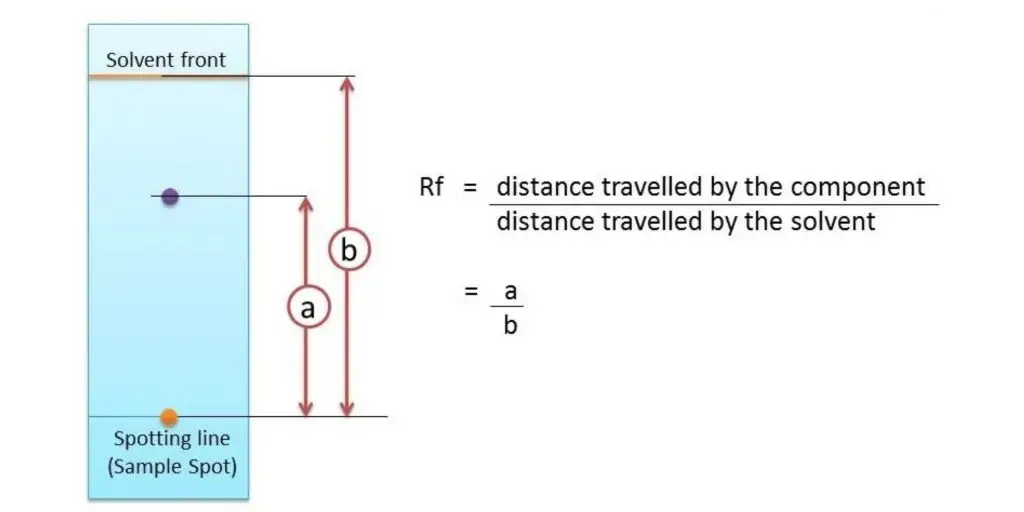
Rf value or Retention factor is a value used in chromatography to show the relative distance travelled by a separated component with respect to the solvent front. It is used for comparing and identifying the separated substances on the chromatogram.
It is calculated by dividing the distance travelled by the solute from the origin line to the centre of the spot by the distance travelled by the solvent front from the same origin line.
Rf value = Distance travelled by solute / Distance travelled by solvent front
The value of Rf is always between 0 and 1. If the value is 0, then the substance remains at the origin and does not move. If the value is 1, then the substance moves along with the solvent front.
The Rf value depends on polarity, solubility and attraction of the substance towards stationary phase and mobile phase. A lower Rf value shows more attraction towards stationary phase. A higher Rf value shows more solubility in mobile phase. It is not same for all conditions because it changes with solvent system, temperature, paper type and other experimental conditions.
Applications of Paper Chromatography
The following are the applications of Paper chromatography–
- In clinical diagnosis, Paper chromatography is used for detection of DNA, RNA, antigens and antibodies. Liver function, glucose level and cardiovascular markers can also be checked by this method.
- In forensic science, it is used for studying crime scene materials. Illegal drugs, toxins like cyanide and arsenic, explosive residue and ink of forged documents are detected.
- In environmental monitoring, this method helps in detecting pollutants from water, soil and air. Heavy metals, pesticides, nutrients, airborne pathogens and PFAS are separated and identified.
- In food testing, it is used to detect foodborne pathogens such as E. coli and Salmonella. Pesticides, heavy metals, artificial colours and spoilage products are also checked.
- In pharmaceutical analysis, purity of drugs and pharmaceutical products are tested. Active ingredients in the preparation are also followed during manufacturing.
- In clinical biochemistry, amino acids of urine or gastric juice are separated. This helps in studying some metabolic and genetic disorders.
- In biochemical research, complex reaction mixtures are separated by this method. It is also used in study of plant ripening and fermentation process.
- In cosmetic industry, chemical components and purity of cosmetic products are tested by Paper chromatography.
Advantages of Paper Chromatography
The following are the advantages of Paper chromatography–
- Paper chromatography is simple method and it can be performed easily in laboratory. The setup is also not complex and result is obtained in less time.
- It is an inexpensive technique as compared to other advanced chromatography methods. So it can be used in normal laboratory also.
- Very small amount of sample is required for analysis. Even minute quantity of sample can be separated by this method.
- It does not require much laboratory space. Only simple materials like chromatography paper, solvent and chamber are needed.
- The movement of solvent takes place by capillary action. So no external power supply or pumping system is needed.
- It can be used for separation and identification of different organic and inorganic compounds. Unknown compounds can also be studied by this method.
- It has good resolving power in qualitative analysis. The components of mixture can be separated clearly on the paper.
Limitations of Paper Chromatography
The following are the limitations of Paper chromatography–
- Only small quantity of sample can be applied on the paper. If more sample is used, then the spots may spread and separation is not proper.
- It is mainly useful for qualitative analysis. It is not very suitable for exact quantitative measurement.
- Complex mixture are difficult to separate by this method. All components may not form clear separate spots.
- It is less accurate than advanced methods like HPLC and HPTLC. So for more precise analysis these methods are preferred.
- The migration distance is limited because the paper has fixed length. Due to this, substances moving very fast or very slow may not separate properly.
References
- Adkins, J. A., Cate, D. M., Henry, C. S., & Mettakoonpitak, J. (2015). Recent developments in paper-based microfluidic devices. Analytical Chemistry, 87(1), 19-41.
- Advances in microfluidic paper-based analytical devices (µPADs): Design, fabrication, and applications. (n.d.). PubMed.
- Aryal, P., & Henry, C. S. (2024). Advancements and challenges in microfluidic paper-based analytical devices: Design, manufacturing, sustainability, and field applications. Frontiers in Lab on a Chip Technologies, 3, 1467423. https://doi.org/10.3389/frlct.2024.1467423.
- Aryal, S. (2022, April 21). Paper chromatography- Definition, types, principle, steps, uses. Microbe Notes.
- Biocompare. (n.d.). Chromatography solvents.
- Bruce, M., & the ICN Team. (2021). Paper chromatography: Separating and identifying metal ions. InterChemNet.
- Cellulose chromatography papers. (n.d.). Whatman.
- Chrom Tech, Inc. (2025, October 14). Journey through the history of paper chromatography.
- Chrom Tech, Inc. (2025, October 14). Understanding chromatography paper: Uses and methods.
- Chromatography. (n.d.). PubMed.
- Chromatography and Rf values (GCSE chemistry). (n.d.). Study Mind.
- Chromatography fundamentals, part III: Retention parameters of liquid chromatography. (n.d.). LCGC International.
- Chromatography helps free innocent man after 8 years on death row. (n.d.). Chromatography Today.
- Chromatography products. (n.d.). Whatman.
- Clark, J. (2023, August 29). E. Paper chromatography. Chemistry LibreTexts.
- Comprehensive analysis of paper chromatography: Theoretical foundations, instrumental design, and methodological evolution. (n.d.).
- Curry, A. S. (1954). The application of paper chromatography to forensic chemistry. Journal of Criminal Law and Criminology, 44(6), 787-794.
- Cytiva. (2026, April 9). Troubleshooting peak tailing and peak fronting in chromatography.
- Cytiva. (n.d.). Whatman™ Grade 3MM Chr cellulose chromatography papers.
- Delloyd’s Lab-Tech. (n.d.). Preparation of chromatography spray reagents.
- Environmental Studies (EVS) Institute. (2026, January 15). Chromatography in environmental monitoring: Techniques and their critical applications.
- EPFL. (n.d.). TLC visualization reagents.
- Fisher Scientific. (n.d.). Paper chromatography products.
- Han, Y., Tian, Y., Li, Q., Yao, T., Yao, J., Zhang, Z., & Wu, L. (2025). Advances in detection technologies for pesticide residues and heavy metals in rice: A comprehensive review of spectroscopy, chromatography, and biosensors. Foods, 14(6), 1070. https://doi.org/10.3390/foods14061070.
- Home Science Tools. (n.d.). Forensic chromatography activity.
- Juniata College. (n.d.). Forensic paper chromatography.
- LC troubleshooting essentials: A guide to common problems and solutions for peak tailing, ghost peaks, and pressure spikes. (n.d.). LCGC International.
- MilliporeSigma. (n.d.). Filter paper.
- Nobel Prize Outreach. (2026). The Nobel Prize in chemistry 1952.
- Noviana, E., Ozer, T., Carrell, C. S., Link, J. S., McMahon, C., Jang, I., & Henry, C. S. (2021). Microfluidic paper-based analytical devices: From design to applications. Chemical Reviews, 121(19), 11835-11885. https://doi.org/10.1021/acs.chemrev.0c01335.
- Organomation. (2025). Solvents in sample preparation for chromatography and mass spectrometry.
- Overload or minor peak? (n.d.). LCGC International.
- Paper chromatography. (n.d.).
- PubMed Central. (n.d.). Recent advances in microfluidic paper-based analytical devices toward high-throughput screening.
- Purushothaman, R. (n.d.). Paper chromatography/Radial paper chromatography (Principle, procedure, visualization & application) [Video]. YouTube.
- Restek Corporation. (n.d.). LC troubleshooting—All of my peaks are tailing! What should I do? [Video]. YouTube.
- Searle Separations Department. (n.d.). Thin layer chromatography (Procedure SEP-120).
- Shakashiri, B. (n.d.). Paper chromatography. Truman ChemLab.
- Shelke, A., Yede, P., & Deokar, R. (n.d.). Chromatography: Types, basic principles and uses. International Journal of Scientific Research and Engineering Development, 7(4), 257-264.
- Sigma-Aldrich. (n.d.). Whatman cellulose chromatography papers 3 CHR, W × L 460 mm × 570 mm, 100/pk Whatman paper.
- The role of Martin and Synge in the birth of modern chromatography. (n.d.). Chromatography Today.
- TutorChase. (n.d.). What solvents are used in paper chromatography?.
- University of Cambridge, Department of Biochemistry. (n.d.). Richard Laurence Millington Synge.
- Waykar, P. A. (2022). Paper chromatography.
- Wikipedia. (2026, January 1). Paper chromatography.