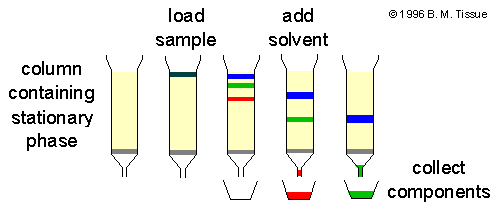
Liquid Chromatography is a separation technique used to separate, identify and measure the components of a liquid mixture. It is used in analytical chemistry for the analysis of complex samples. In this technique, the sample is passed through a column with the help of a liquid solvent.
The liquid solvent is called mobile phase. The column contains a solid material or liquid coated support which is called stationary phase. When the sample mixture moves through the column, different components interact differently with the mobile phase and stationary phase.
The compounds which are strongly attracted to the stationary phase move slowly through the column. The compounds which have more attraction towards the mobile phase move faster. Due to this different movement, the components of the mixture separate from each other and come out of the column at different time.
This process is referred to as differential migration. It helps in the detection and quantification of each component of a mixture. So, Liquid Chromatography is very important for separation of chemical substances from complex liquid samples.
The history of Liquid Chromatography started in the early 1900s by Russian botanist Mikhail S. Tswett. He separated plant leaf extract by passing it through open glass column containing powdered chalk (calcium carbonate) and alumina. He observed the formation of different coloured bands in the column.
The word chromatography was given by Tswett. It is derived from Greek words chroma meaning colour and graph meaning writing. At first, the process was slow because it was dependent on gravity and took long time to complete.
In 1941, Archer J. P. Martin and Richard L. M. Synge suggested that separation can be improved by using small particles in the stationary phase and by applying pressure to move the liquid through the column. This idea later became the basis of modern liquid chromatography.
In 1966, Csaba Horvath and Seymour Lipsky gave the first experimental report on High Pressure Liquid Chromatography. In 1967, the first commercial system was introduced by Waters Associates. Later, with the improvement of pumps, injectors and small column particles, it developed into High Performance Liquid Chromatography (HPLC).
Today, HPLC is one of the most widely used analytical techniques. It is used for separation of drugs, biomolecules, food components, environmental chemicals and many other substances. It gives fast, accurate and efficient separation of liquid samples.
Principle of Liquid Chromatography
Liquid Chromatography is based on the differential distribution of substances between two phases. One is stationary phase and another is mobile phase. The mobile phase is liquid which moves through the column with the sample.
When the sample is added into the column, the components of the sample starts distributing between the stationary phase and mobile phase. The stationary phase may be solid particles or a liquid coated on solid support. The mobile phase carries the substances forward through the column.
The substances which are strongly attracted by the stationary phase remains for more time in the column. So they move slowly. The substances which are more soluble or more attracted towards the mobile phase moves fast with the liquid.
Thus, different components move at different speed. This causes separation of the mixture into different bands. These separated substances come out from the column at different time and can be detected and measured.
Types of Liquid Chromatography
The following are the types of Liquid Chromatography–
A. Based on separation mode and mechanism
- Reversed-phase chromatography
In this type non-polar stationary phase and polar mobile phase is used. The separation occurs mainly due to hydrophobic interaction. - Normal-phase chromatography
In this type polar stationary phase and non-polar mobile phase is used. The compounds are separated according to their polarity. - Ion-exchange chromatography
This method is used for separation of charged molecules. Inorganic ions, organic acids, proteins and other charged substances are separated by electrostatic attraction. - Size-exclusion chromatography
In this method molecules are separated according to their size. Large molecules come out first and small molecules enter into pores and come out later. - Gel filtration chromatography
It is a type of size-exclusion chromatography used mostly for water soluble biomolecules. Proteins and polysaccharides can be separated by this method. - Gel permeation chromatography
It is also a type of size-exclusion chromatography. It is mostly used for polymers and organic soluble molecules. - Affinity chromatography
This method is based on specific biological binding. The target molecule binds with a specific ligand present on the stationary phase, such as antigen-antibody binding. - Hydrophilic interaction liquid chromatography
HILIC is used for separation of highly polar molecules. It uses polar stationary phase and organic-rich mobile phase. - Partition chromatography
In this method the solute is separated by distribution between liquid stationary phase and mobile phase. The separation depends on polarity and partition coefficient. - Chiral chromatography
This method is used for separation of chiral compounds. It separates enantiomers or optical isomers. - Displacement chromatography
It is mainly used as preparative separation method. In this method one strong component displaces the bound sample molecules from the stationary phase.
B. Based on system technology, pressure and scale
- High-performance liquid chromatography
HPLC uses high pressure to push the mobile phase through column. The column contains small particles and gives high resolution separation. - Ultra-high-performance liquid chromatography
UHPLC uses very high pressure and very small particles. It gives faster analysis and higher sensitivity than HPLC. - Fast protein liquid chromatography
FPLC is used for purification of sensitive biomolecules like proteins and nucleic acids. It works at lower pressure and uses biocompatible system. - Low-pressure liquid chromatography
This method works at low pressure. It is used for simple purification and preparative separation. - Miniaturized liquid chromatography
In this type the system is made in small scale. It uses less sample and less solvent. - Capillary liquid chromatography
It is a miniaturized type of liquid chromatography. It uses narrow capillary column for separation. - Nano liquid chromatography
It uses very low flow rate and very small sample amount. It is useful when sample is very limited. - Microchip liquid chromatography
In this method separation is done on small microchip device. It reduces solvent use and analysis time. - Flash chromatography
It is a rapid preparative liquid chromatography method. Pressure is used to pass solvent through a short column packed with adsorbent. - Simulated moving bed chromatography
It is a continuous chromatographic method. It is used for large scale separation of compounds.
Liquid Chromatography Instrumentation

The following are the main parts of Liquid Chromatography–
- Solvent reservoir
It is a container used for keeping the mobile phase. Glass bottles are commonly used for this purpose. - Solvent degasser
It is used for removing dissolved gases from the solvent. If gas remains in solvent, air bubble may form in the system. - Pump
It is used to push the mobile phase through the system. It gives constant flow of solvent under pressure. - Sample injector
It is used for adding the sample into the mobile phase. It may be a manual injector or automatic autosampler. - Column
It is the main part of liquid chromatography. Separation of sample components takes place inside the column. - Stationary phase
It is packed inside the column. The sample components interact with this phase and move at different rate. - Mobile phase
It is the liquid solvent which carries the sample through the column. The nature of mobile phase affects the separation. - Column oven
It is used to keep the column at fixed temperature. Temperature change may affect retention time and separation. - Detector
It is used to detect the compounds which come out from the column. UV-visible detector, fluorescence detector and mass spectrometer are common detectors. - Fraction collector
It is used for collecting separated fractions from the column. It is mostly used in preparative liquid chromatography. - Data system
It is used to record the detector signal. The result is shown as chromatogram.
Liquid Chromatography Protocol
The following are the steps of Liquid Chromatography–
- The mobile phase is prepared by using pure solvents. The solvent is filtered through 0.22 µm membrane.
- The filtered solvent is degassed before use. It removes air bubbles from the solvent.
- The sample is prepared by extraction, centrifugation and filtration. It removes dust, precipitate and interfering particles.
- The instrument and software are switched on. The solvent reservoir is connected with the mobile phase line.
- The purge valve is opened and mobile phase is passed at high flow rate. It removes old solvent and air from the tubing.
- The column is connected in the system. The direction of column flow should be checked before starting the run.
- Starting mobile phase is passed through the column. Usually 5 to 10 column volumes are passed until pressure and baseline become stable.
- The filtered sample is injected into the column. Injection valve or autosampler is used for this step.
- Too much sample should not be injected. Overloading gives broad peak and poor separation.
- The column is washed with starting buffer or mobile phase. Weakly bound impurities and non-specific materials are removed.
- The sample components are eluted by mobile phase. It may be done by same solvent system or by changing solvent composition.
- In isocratic elution, same mobile phase composition is used throughout the run. It is useful for simple mixture.
- In gradient elution, mobile phase composition is changed with time. It is used for mixture having compounds of different polarity.
- The eluted compounds are detected by detector. The detector signal is recorded by the data system.
- After elution, strong solvent is passed through the column. It removes strongly retained compounds from the stationary phase.
- The column is flushed with proper storage solvent. The flow rate is slowly reduced to zero.
- The instrument is switched off after washing and storage. The solvent bottle and waste line are checked properly.

Limitations of Liquid Chromatography
The following are the limitations of Liquid Chromatography–
- It is costly method. The instrument, column, solvents and maintenance need high cost.
- It needs trained person for operation. Pump, injector, column, detector and software must be handled properly.
- Method development may take long time. Proper mobile phase, column and flow rate must be selected.
- Some complex sample analysis may take more time. The run may continue for several minutes to hours.
- It generally needs small sample volume. Large sample loading may give broad peak and poor separation.
- Miniaturized liquid chromatography has low sample loading capacity. So only very small amount of sample can be used.
- The system may get contaminated by dirty sample. Proteins, lipids, surfactants and other organic matter may block the column.
- Column clogging may occur if sample and solvent are not filtered. This increases pressure and affects the result.
- The column may become saturated after repeated use. Then the separation becomes poor and column needs replacement.
- Some compounds may degrade during the run. High pressure, temperature or unsuitable solvent may affect sensitive compounds.
- High backpressure is produced when fine particles are used in the column. This puts stress on pump and tubing.
- Compounds having very similar adsorption property may be difficult to separate. They may elute together or very close to each other.
- It is not very suitable for volatile compounds. Gas chromatography (GC) is better for such compounds.
- Regular maintenance is needed. If pump, detector or column is not maintained properly, the result may become inaccurate.
Advantages of Liquid Chromatography
The following are the advantages of Liquid Chromatography–
- It gives good separation of mixture. Many components present in a complex sample can be separated in one run.
- It has high resolution. Closely related compounds can also be separated by selecting proper column and mobile phase.
- It has good sensitivity. Very small amount of compound can be detected by this method.
- It is used for different types of samples. Small molecules, proteins, DNA, drugs and environmental samples can be analysed.
- It is suitable for non-volatile compounds. The compound does not need to be converted into vapour form.
- It is useful for heat sensitive compounds. As high temperature is not needed, thermally unstable compounds can be separated.
- It is faster than simple column chromatography. Pump is used to pass the mobile phase through the column.
- It gives reproducible result when proper condition is maintained. Same flow rate, column and mobile phase gives similar retention time.
- It can be automated. Sample injection, solvent flow and data recording can be controlled by instrument.
- It is useful for routine laboratory analysis. Many samples can be analysed one after another with less manual work.
- It can be connected with different detectors. UV-Visible detector, fluorescence detector and mass spectrometry (MS) can be used.
- It is used for both qualitative and quantitative analysis. The compound can be identified and its amount can also be measured.
Applications of Liquid Chromatography
The following are the uses of Liquid Chromatography–
- Drug development is done by using liquid chromatography. New drug compounds are separated, purified and checked in different stages.
- Quality control of drugs is done by this method. Purity and potency of active pharmaceutical ingredients (APIs) are tested.
- Stability testing of drugs is done by liquid chromatography. Degradation products formed during storage, heat sterilization or processing can be detected.
- Chiral separation is done by this method. Enantiomers of drug are separated because one form may be more active or more safe than other form.
- Pharmacokinetic study is done by liquid chromatography. Drug and its metabolites are detected in blood, serum, urine and other biological fluids.
- Therapeutic drug monitoring is done by this method. The amount of drug in patient sample is measured for maintaining proper dose.
- Proteomics and metabolomics study is done by liquid chromatography. Proteins, peptides, nucleic acids and metabolites are separated and estimated.
- Disease diagnosis is helped by this method. Blood, serum and urine samples are analysed for detecting low amount of biomarker.
- Biomarker discovery is done by liquid chromatography. It helps to find disease related molecules in complex biological samples.
- Drug testing is done in forensic and toxicology work. Drugs of abuse, toxins and their metabolites are detected from hair, blood, sweat and urine.
- Legal investigation uses liquid chromatography. It is used in DUI, drug-impaired driving and probation monitoring cases.
- Doping control is done by this method. Performance enhancing drugs are detected in sports and veterinary samples.
- Food contaminant testing is done by liquid chromatography. Pesticides, veterinary drugs, antibiotics, allergens and toxins are detected in food.
- Food adulteration testing is done by this method. Products like olive oil, wine and other food materials are checked for authenticity.
- Nutritional analysis is done by liquid chromatography. Carbohydrates, proteins, fats, vitamins and minerals are identified and measured.
- Environmental monitoring is done by this method. Pollutants, pesticides, herbicides and PFAS are detected from water, soil and air.
- Cosmetic quality control is done by liquid chromatography. Active ingredients and harmful contaminants in cosmetic products are checked.
- Agrochemical and industrial chemical analysis is done by this method. Pesticide residues, organic chemicals and inorganic synthetic chemicals are analysed.
Precautions of Liquid Chromatography
The following are the precautions of Liquid Chromatography–
- High purity solvent should be used for mobile phase preparation. HPLC grade or LC-MS grade solvent is used, because impure solvent may damage the column and give unwanted peaks.
- Mobile phase and sample should be filtered before use. Usually 0.22 µm membrane filter is used to remove small particles, which may block the tubing, injector or column.
- Mobile phase should be degassed properly before running. Dissolved gases may form air bubble in the system and it causes pressure fluctuation and noisy baseline.
- Sample solvent should be matched with the starting mobile phase as much as possible. If strong or wrong solvent is used, peak fronting or sample precipitation may occur.
- Volatile buffer should be used when required. Unnecessary salts should be avoided because salt may precipitate and block the system.
- Flow rate and pressure should be increased slowly. Sudden pressure shock may disturb the column packing and decrease the column life.
- Column pressure limit should be followed during the run. The column should not be operated near its maximum pressure for long time.
- Sample overloading should be avoided. Excess sample volume or highly concentrated sample gives broad peak and poor separation.
- Injector should be handled gently. Syringe should be inserted slowly and injection valve should be rotated carefully to avoid damage.
- Solvent change should be done carefully. When two solvents are not miscible, an intermediate solvent should be passed first through the system.
- Column and injector should be washed after analysis. Strong cleaning solvent is used to remove buffer, salt and strongly retained impurities.
- Column storage should be done in proper storage solvent. The column ends should be closed tightly so that stationary phase bed does not become dry.
- Tubing and fittings should be checked regularly. Loose or damaged fitting may cause leakage under high pressure.
- Toxic and inflammable solvents should be handled in well ventilated place. Safety glasses, gloves and lab coat should be used during work.
- Solvent waste should be collected in properly labelled container. It should be disposed safely and not thrown directly into sink.
Troubleshoot for Liquid Chromatography
The following are the troubleshoot for Liquid Chromatography–
- High backpressure occurs due to blockage in column frit, tubing or precipitation of buffer salt. The pre-column filter should be replaced, sample and mobile phase should be filtered and the column may be back flushed if it is allowed.
- Low pressure occurs due to leakage in capillary connection or air in the pump head. The fittings should be tightened, pump should be purged and flow rate should be checked.
- Fluctuating pressure occurs due to trapped air in pump, dirty check valve or damaged piston seal. The solvent should be degassed, pump should be purged and check valve should be cleaned.
- Peak tailing occurs due to secondary interaction with stationary phase or column overloading. The mobile phase pH should be adjusted, injection volume should be reduced and triethylamine (TEA) may be added if required.
- Peak fronting occurs when sample is not dissolved properly or injection solvent is too strong. The sample should be dissolved in starting mobile phase and sample concentration should be reduced.
- Split or double peak occurs due to contamination at column inlet, worn rotor seal or strong injection solvent. Guard column should be replaced, main column should be washed and sample solvent should be matched with mobile phase.
- Broad peak occurs due to dead volume, slow flow rate or large detector cell volume. Short narrow capillary should be used and flow rate should be adjusted.
- Ghost peak occurs due to carryover from previous run, contaminated solvent or plastic vial. Blank injection should be run, sampler and column should be flushed and fresh high purity solvent should be used.
- Retention time shifting occurs due to poor column equilibration, mobile phase mixing problem or column degradation. The column should be equilibrated with 5 to 10 column volumes and pump line should be checked for leaks or bubbles.
- Baseline drift occurs due to air in pump, temperature change or old mobile phase. Solvent should be degassed, fresh mobile phase should be used and column oven may be used.
- No peak detected occurs when sample is not injected properly or data transfer fails. Injector, autosampler, injection loop and data system should be checked.
- Negative peak occurs when sample absorbance is lower than mobile phase background. Mobile phase with less background absorbance or different detection wavelength should be used.
References
- LCGC International. (n.d.). A well-written analytical procedure for regulated HPLC testing.
- Unknown Author. (n.d.). Advances in liquid chromatography: A comprehensive technical analysis of principles, methodologies, and industrial frameworks.
- Temwani, K., Pan, D., Wu, Z., Zhang, Y., & Lan, H. (2025). Advances in miniaturized liquid chromatography for the detection of organic pollutants in food, environmental, and biological samples. Separations, 12(11), 312. https://doi.org/10.3390/separations12110312
- Tittarelli, R., Stefani, L., Romani, L., Mineo, F., Vernich, F., Mannocchi, G., Pellecchia, M. R., Russo, C., & Marsella, L. T. (2024). Application of LC-MS/MS for the identification of drugs of abuse in driver’s license regranting procedures. Pharmaceuticals, 17(12), 1728. https://doi.org/10.3390/ph17121728
- PMC – NIH. (n.d.). Application of liquid chromatography in food analysis.
- Oszmianski, J., & Lachowicz-Wiśniewska, S. (Eds.). (2022). Applications of liquid chromatography in analysis of pharmaceuticals and natural products. MDPI.
- BUCHI. (n.d.). As easy as ABC: How to use the van Deemter equation to optimize your chromatography. Buchi.com.
- Separation Science. (n.d.). Back to basics #9: Fundamental resolution equation – Alpha.
- Waters Corporation. (n.d.). Beginner’s guide to high-performance liquid chromatography (HPLC).
- Li, B., & Haynie, D. T. (2006). Chiral drug separation. In Encyclopedia of chemical processing (pp. 449-458). Taylor & Francis.
- Phenomenex. (n.d.). Chiral HPLC column.
- Unknown Author. (n.d.). Chiral separation and enantiomeric analysis: Critical importance in pharmaceutical development.
- PMC. (n.d.). Chiral separations in preparative scale: A medicinal chemistry point of view.
- Waters Corporation. (2023, April 20). Choosing the correct column for chromatographic selectivity. Waters Blog.
- MAC-MOD Analytical. (n.d.). Chromatographic band broadening and the van Deemter equation.
- Unknown Author. (2025). Chromatography 2025: The trends transforming laboratories.
- Taylor, J. (2016, February 18). Comparison of selectivity and efficiency: Two key factors in determining chromatographic resolution of components of a mixture. Bioanalysis Zone.
- BOC Sciences. (n.d.). Enantiomeric purification (HPLC/SFC).
- PharmChek. (2025, February 19). Ensuring accurate drug test results: The role of LC-MS/MS confirmation.
- Shimadzu UK Ltd. (2020). Back to basics: Explaining resolution.
- Unknown Author. (n.d.). FPLC versus analytical HPLC: Two methods, one origin, many differences.
- ResearchGate. (n.d.). FPLC vs HPLC analysis comparison studies – are you aware of any?
- LabX. (n.d.). FPLC vs. HPLC: Key differences, applications, and how to choose the right system.
- ResearchGate. (n.d.). Green approaches in high-performance liquid chromatography for sustainable food analysis: Advances, challenges, and regulatory perspectives.
- Champion, B. (2011, February 8). HPLC separation fundamentals. Agilent Technologies, Inc.
- Washington State University. (2016, March 3). HPLC standard operating procedure.
- Thermo Fisher Scientific. (n.d.). HPLC troubleshooting.
- Tamang, S. (2026, April 5). HPLC: Principle, instrumentation, steps, types, uses, diagram. Microbe Notes.
- Unknown Author. (n.d.). High performance liquid chromatography 1. Switch ON the all the …
- Conduct Science. (2019, June 26). High performance liquid chromatography (HPLC) protocol.
- Ahmed, R. (2024). High-performance liquid chromatography (HPLC): Principles, applications, versatility, efficiency, innovation and comparative analysis in modern analytical chemistry and in pharmaceutical sciences. Clinical Investigation (Lond.), 14(9), 524-535.
- Wikipedia contributors. (n.d.). High-performance liquid chromatography. Wikipedia.
- Bio-Rad. (n.d.). Introduction to liquid chromatography principles.
- LCGC International. (n.d.). LC troubleshooting essentials: A guide to common problems and solutions for peak tailing, ghost peaks, and pressure spikes.
- Unknown Author. (n.d.). Lecture 3: General chromatography theory.
- American Laboratory Trading. (2024, March 15). Liquid chromatography comparison: HPLC vs UPLC vs FPLC.
- The Business Research Company. (2026). Liquid chromatography devices market report 2026.
- Waters Corporation. (n.d.). Liquid chromatography separation modes.
- Cytiva. (n.d.). Liquid chromatography – principles, application, type.
- PMC. (n.d.). Recent advances in liquid chromatography–mass spectrometry (LC–MS) applications in biological and applied sciences.
- Bates, M. N., Helm, A. E., & Barkholtz, H. M. (2024). Screening for forensically relevant drugs using data-independent high-resolution mass spectrometry. Chemical Research in Toxicology, 37(4), 571-579. https://doi.org/10.1021/acs.chemrestox.3c00379
- Foods. (n.d.). Special issue: Application of chromatography in food analysis, safety and quality control. MDPI.
- McNair, H. (n.d.). The van Deemter equation. Chromedia.
- Agilent Technologies. (n.d.). Tips and tricks of HPLC system troubleshooting.
- ALWSCI. (n.d.). Troubleshooting poor peak shape: A complete workflow from mobile phase to consumables.
- Unknown Author. (n.d.). Understanding chromatogram peaks – fronting, tailing, ghosting & rounded explained.
- Chrom Tech, Inc. (n.d.). Understanding liquid chromatography: Key principles and techniques.
- Wikipedia contributors. (n.d.). Van Deemter equation. Wikipedia.
- Agilent Technologies. (n.d.). What is high-performance liquid chromatography (HPLC)?
- Amy. (2012, March 7). What is the difference between HPLC and FPLC and why is FPLC preferable for protein purification? Biology Stack Exchange.
- IUPAC. (n.d.). Liquid chromatography (LC). The IUPAC Compendium of Chemical Terminology.
- Boag, M. (2025, June 18). Understanding the van Deemter equation in chromatography. Phenomenex.