Fluorescence Spectroscopy is an analytical technique used to study the fluorescence emitted by a sample after excitation by light.
It is also known as fluorimetry or spectrofluorometry. In this method, UV or visible light of definite wavelength is allowed to fall on the sample. The electrons of molecules absorb this energy and go to excited state.
After a short time, the excited molecules return to lower energy state. During this process, light is emitted at longer wavelength and lower energy. This emitted light is measured by an instrument called spectrofluorometer.
The difference between excitation wavelength and emission wavelength is called Stokes shift. Due to this difference, the method gives more selectivity and less interference.
The efficiency of fluorescence is expressed by quantum yield. It is the ratio of photons emitted to photons absorbed. The decay time of fluorescence is called fluorescence lifetime, which can be measured by time-domain or frequency-domain method.
Fluorescence Spectroscopy is more sensitive than many absorption methods. Very low concentration and trace amount of substances can be detected by this method.
It is widely used in chemistry, biology and medicine for study of molecules, reaction, binding, structural change and dynamic process.
The phenomenon of fluorescence was observed long before its proper explanation. In 1845, Herschel observed blue emission from quinine solution under UV light. The term fluorescence was given by Sir George G. Stokes in 1852. Later many modern biological uses were developed by Gregorio Weber.
Principle of Fluorescence Spectroscopy
Fluorescence Spectroscopy is based on absorption and emission of light by fluorescent molecules.
In this method, light of suitable wavelength is allowed to fall on the sample. The molecule absorbs this light. The electron moves from ground state (S₀) to excited singlet state (S₁ or S₂).
The excitation takes place very fast. It occurs in about 10⁻¹⁵ seconds. The electron now has higher energy.
Some energy is lost after excitation. This is lost as heat by vibrational relaxation and internal conversion. The molecule comes to lowest vibrational level of excited singlet state.
Then the molecule returns back to the ground state. During this return, light is emitted. This emitted light is called fluorescence.
The emitted light has less energy than absorbed light. So its wavelength is longer. This difference is known as Stokes shift.
The intensity of fluorescence depends on concentration of fluorophore. Up to certain limit, more fluorophore gives more fluorescence. After that, quenching may occur.
The efficiency of the process is called quantum yield (Φf). It means the ratio of emitted photons to absorbed photons.
Fluorescence is mostly shown by rigid and planar molecules. These molecules generally contain conjugated π-system. Examples are anthracene, fluorescein and rhodamine.
Thus fluorescence occurs by excitation, loss of some energy and emission of fluorescent light.
Applications of Fluorescence Spectroscopy in inorganic chemistry
The following are the applications of Fluorescence Spectroscopy in inorganic chemistry-
- Fluorescence Spectroscopy is used for study of metal ions in solution, because metal ions may increase or decrease the fluorescence of ligands.
- Trace amount of metal ions such as Al³⁺, Zn²⁺, Cd²⁺, Hg²⁺, Pb²⁺ and Fe³⁺ can be detected by using fluorescent reagents.
- Complex formation between metal ion and ligand can be studied by change in fluorescence intensity.
- It is useful in study of stability of coordination compounds, because binding of metal ion with ligand changes the emission.
- The structure and surrounding of metal complexes can be studied, mainly when the ligand is fluorescent.
- Lanthanide ions such as Eu³⁺ and Tb³⁺ are studied by this method because they gives sharp emission bands.
- Quantum yield, fluorescence lifetime and excited state behaviour of inorganic compounds can be measured.
- In inorganic photochemistry, excited state reaction and photo-induced electron transfer of metal complexes are studied by fluorescence method.
- Heavy metal ion quenching is studied by this method, which gives idea about interaction between metal ion and fluorophore.
- Toxic metal ions present in water, soil extract and industrial sample can be detected by fluorescence change.
- Inorganic pollutants can be detected in very small amount due to high sensitivity of the method.
- Fluorescent chemosensors are used for selective detection of metal ions and anions such as F⁻, CN⁻, NO₃⁻ and PO₄³⁻.
- Metal-organic frameworks (MOFs) are studied by fluorescence spectroscopy for guest molecule detection and metal ion interaction.
- Inorganic nanoparticles and quantum dots are studied by this method because their emission depends on size, surface and composition.
- It is a sensitive and non-destructive method for analysis of inorganic species in solution and solid state.
Special fluorimetric applications
The following are the special fluorimetric applications of Fluorescence Spectroscopy–
- Fluorescence quenching study is used to determine molecular interaction between fluorophore and quencher molecule, which may be static or dynamic type.
- The quenching mechanism can be studied by observing change in fluorescence intensity or fluorescence lifetime after interaction of quencher with excited molecule.
- Fluorescence lifetime measurement is used to study excited state decay process, and it gives information about the environment around fluorophore molecule.
- The lifetime value may change with solvent polarity, temperature or binding of analyte, which indicates change in microenvironment.
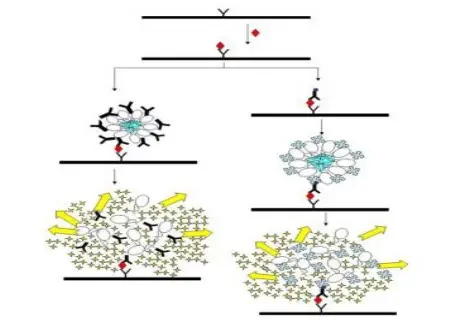
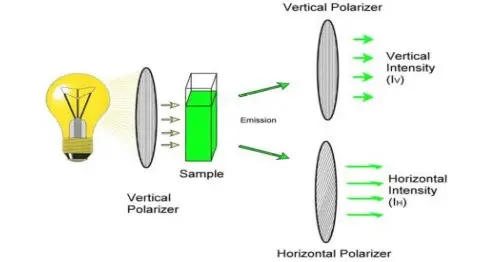
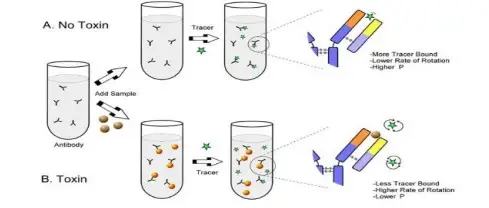
- Fluorescence polarization (FP) is used for determination of rotational diffusion and molecular size in solution, where larger molecules show higher polarization value.
- It is applied in binding study, because change in polarization shows formation of complex between small and large molecule.
- Fluorescence Resonance Energy Transfer (FRET) is used for measuring distance between donor and acceptor chromophores, generally in 10–100 Å range.
- FRET is useful to study protein folding, conformational change and interaction of biological macromolecules because it is very sensitive to spatial change.
- Synchronous Fluorescence Spectroscopy (SFS) is used for mixture analysis, where excitation and emission wavelengths are scanned together to improve spectral resolution.
- It is useful for determination of overlapping fluorescent compounds, such as aromatic hydrocarbons in complex sample.
- Time-resolved Fluorescence Spectroscopy is used to observe transient excited state process, reaction kinetics and photophysical behaviour.
- Anisotropy measurement is used to study rotational motion, viscosity and molecular binding in different media.
- Front-face fluorescence technique is used for solid or turbid samples, where normal right angle method gives scattering or absorption problem.
- Phase-modulation fluorimetry is used for high frequency modulation study to determine lifetime and phase shift parameters.
- Single molecule fluorescence is used for detection of individual fluorophore, and helps to study molecular heterogeneity and dynamic fluctuation.
- Quantum yield determination is used to compare fluorescence efficiency of different compounds.
- These special methods are used in biochemistry, material science, environmental analysis and design of optical sensors.
- These techniques give information about molecular interaction and microenvironment, but careful calibration and control are needed for accurate result.
References
- Libretexts. (2022, August 28). 1.11: Fluorescence spectroscopy. Chemistry LibreTexts. https://chem.libretexts.org/Bookshelves/Analytical_Chemistry/Physical_Methods_in_Chemistry_and_Nano_Science_%28Barron%29/01%3A_Elemental_Analysis/1.11%3A_Fluorescence_Spectroscopy
- PerkinElmer Ltd. (n.d.). An introduction to fluorescence spectroscopy. https://www.chem.uci.edu/~dmitryf/manuals/Fundamentals/Fluorescence%20Spectroscopy.pdf
- Wilson, M. (2017, November 15). The fundamentals and history of fluorescence and quantum dots. Learn & Share | Leica Microsystems. https://www.leica-microsystems.com/science-lab/life-science/the-fundamentals-and-history-of-fluorescence-and-quantum-dots/
- ZEISS Microscopy Online Campus | Microscopy Basics | Fluorescence Microscopy. (n.d.). https://zeiss-campus.magnet.fsu.edu/articles/basics/fluorescence.html
- News-Medical. (2018, November 21). The history of fluorescence microscopy. https://www.news-medical.net/life-sciences/The-History-of-Fluorescence-Microscopy.aspx
- Povrozin, Y., & Barbieri, B. (2016). Fluorescence spectroscopy. In Myer Kurtz (Ed.), Handbook of Measurement in Science and Engineering (Vol. 3). John Wiley & Sons. https://iss.com/media/Fluorescence_Spectroscopy.pdf
- Barbieri, B., Prof. David Jameson, Nicolás Monardes, Charles de L’Écluse, Galileo Galilei, Vincenzo Casciarolo, Robert Boyle, David Brewster, John Herschel, Edmond Becquerel, George Gabriel Stokes, E. Nichols, E. Merrit, Stern, Volmer, S.J. Vavilov, F. Perrin, E. Gaviola, A. Jablonski, . . . F. Goppelsröder. (n.d.). A short history of fluorescence. https://diverdi.colostate.edu/C372/experiments/steady%20state%20fluorescence%20of%20quinine%20and%20quenching%20by%20chloride/references/short%20history%20of%20fluorescence.pdf
- Wikipedia contributors. (2025, September 16). Fluorescence spectroscopy. Wikipedia. https://en.wikipedia.org/wiki/Fluorescence_spectroscopy
- Settele, S., Schrage, C. A., Jung, S., Michel, E., Li, H., Flavel, B. S., Hashmi, A. S. K., Kruss, S., & Zaumseil, J. (2024). Ratiometric fluorescent sensing of pyrophosphate with sp3-functionalized single-walled carbon nanotubes. Nature Communications, 15(1). https://doi.org/10.1038/s41467-024-45052-1
- AZoM. (2024, February 16). Applications of advanced fluorescence spectroscopy. https://www.azom.com/article.aspx?ArticleID=13958
- Wikipedia contributors. (2025, May 8). Lanthanide probes. Wikipedia. https://en.wikipedia.org/wiki/Lanthanide_probes