Dark field microscopy is a special illumination technique used in light microscope to increase contrast of transparent, unstained or low contrast specimens. The specimen looks bright, and the background looks dark or almost black. It is useful when normal bright background observation is not giving clear contrast.
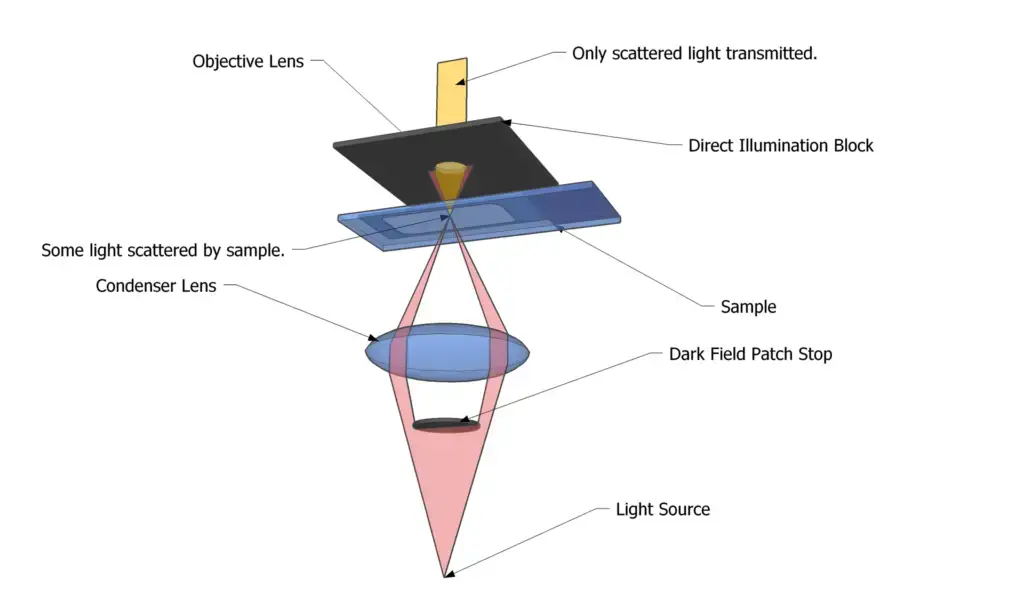
It is done by using a modified condenser with a central opaque stop. This stop blocks the direct light from passing straight through the specimen. The condenser sends an oblique and hollow cone of light on the specimen at a steep angle.
Because of this steep illumination, the direct light does not enter into the objective lens. Only the light which is diffracted, refracted or reflected by the specimen structures enters the objective. This scattered faint light forms the final image, so the object appears bright on dark background.
Dark field microscopy is preferred because staining is not required. Complex sample preparation is also not needed in many cases. It helps to observe live aquatic organisms, unstained bacteria, tissue culture cells, and very small particles in their natural living state.
Principle of Dark Field Microscope
Principle of Dark Field Microscope is based on blocking the direct central light and allowing only the scattered light from the specimen to enter the objective lens. An opaque disc (stop) is placed below the condenser lens. This stop blocks the straight light rays moving upward through the microscope.
In this method, the light is made to form an inverted hollow cone. The cone of light falls on the specimen from the sides at a highly oblique angle. If the slide is empty, the light rays cross and completely miss the objective lens. So the background remains pitch black.
When the specimen is present in the focal plane, its structures interact with this oblique light. The rays are diffracted, reflected or refracted by the specimen parts. Only this scattered light is redirected upward and collected by the objective. The image is seen as bright specimen against dark background, giving high contrast for transparent and unstained living samples.
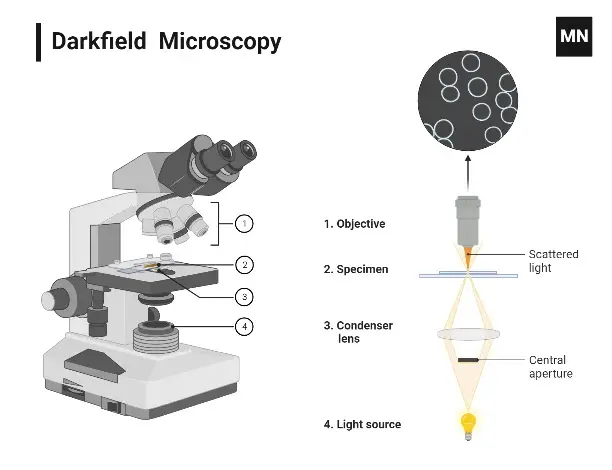
Parts of Dark Field Microscope
The parts of a darkfield microscope include the following:
- Light source– A very bright high intensity light source is required. Because only a small amount of scattered light finally forms the image and most of the direct light is blocked. It is usually placed below the stage and provides illumination to the condenser system.
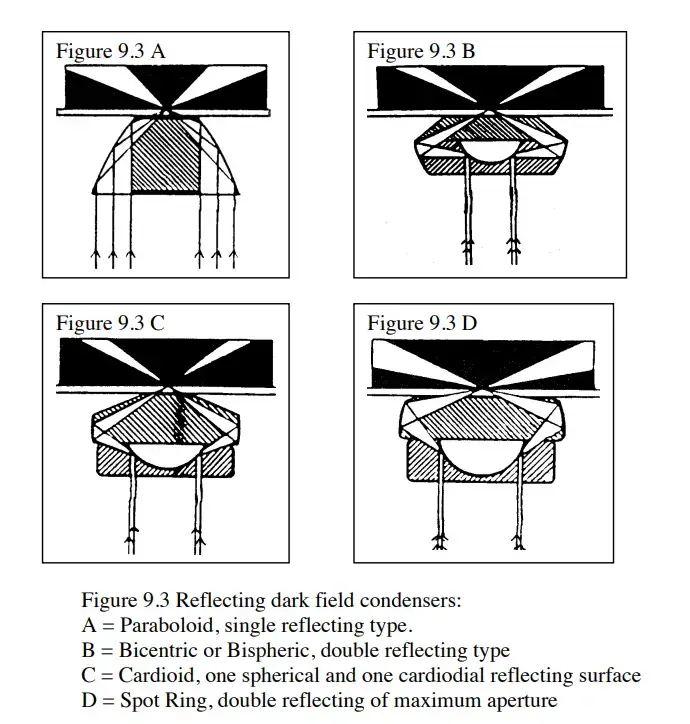
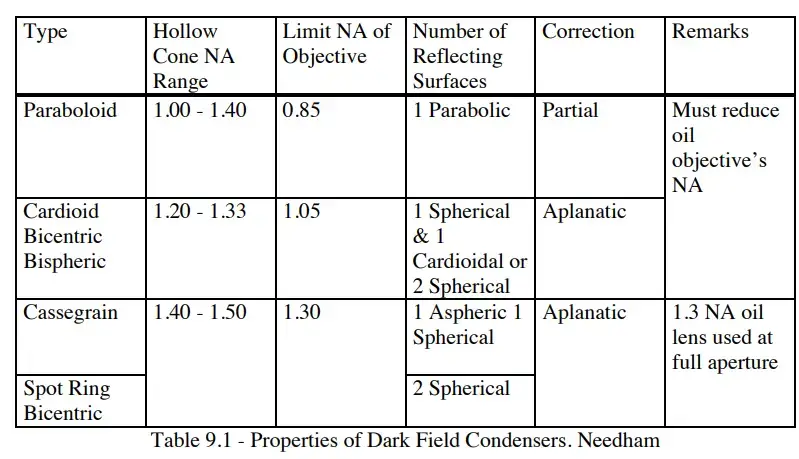
- Dark field condenser– This is the most essential component. It produces a hollow cone of oblique light that strikes the specimen at high angles. It prevents direct light from entering the objective and ensures only scattered light is collected.
- Opaque stop (patch stop or central disc)– It is a solid circular stop placed inside or below the condenser. It blocks the central beam of light. Due to this only peripheral rays pass and dark background effect is produced.
- Objective lens– It collects only the scattered light coming from the specimen. Direct light misses the lens due to oblique illumination. This selective collection forms a bright image on a dark background.
- Eyepiece (ocular lens)– It magnifies the image formed by the objective lens. The final enlarged image of the specimen is seen through this lens.
- Specimen stage (glass slide)– It holds the specimen in position for observation. Usually thin and transparent specimens are used. Proper positioning is needed for correct illumination and image formation.
- Mechanical stage and adjustment knobs– It helps in precise movement of the slide and focusing of the specimen. Coarse adjustment and fine adjustment are used to get a sharp image.
- Body parts (arm, base, nosepiece)– These are the supporting parts of the microscope. They provide stability during observation and allow changing of objective lenses. Most of these parts are same like compound microscope, but dark field condenser and central stop system is the main difference.
The light’s path in Dark Field Microscope
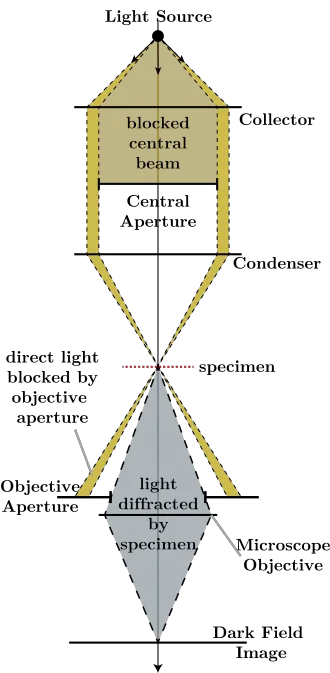
- Light from the source enters the microscope and moves towards the condenser to illuminate the sample.
- An opaque disc (patch stop) blocks the direct central light rays. Only the outer ring of light is allowed to pass.
- The dark field condenser focuses this peripheral light. It forms an inverted hollow cone of light.
- This hollow cone is directed to strike the specimen at a very oblique angle.
- Due to this steep angle, the directly transmitted light crosses the specimen and diverges strongly. It completely misses the objective lens, so background remains dark.
- When the oblique light hits the specimen, some part of light is scattered. It may be reflected, refracted or diffracted by the specimen structures.
- Only this scattered light enters the objective lens and forms the image. The specimen is seen bright against black background.
Operating Procedure of Darkfield Microscope
- Place the specimen slide on the stage. Select a low power objective (10X). Focus the specimen first in normal brightfield illumination.
- Set up proper illumination (Köhler illumination). The light source and diaphragms are adjusted and aligned. This gives proper even illumination on the specimen.
- Increase the light intensity to maximum. Darkfield needs very bright light because most of the direct light is blocked by the stop.
- Condenser setting– For low magnification. Open the field diaphragm and condenser aperture diaphragm fully. Insert the central opaque stop (patch stop) in the light path. For high magnification. Remove the normal brightfield condenser and place a high NA darkfield condenser. Raise the condenser till it is just below the slide.
- Center the condenser. Remove the eyepiece and look down the tube (or use phase telescope). The rear focal plane is viewed. Use the condenser centering screws and adjust till the opaque stop blocks the bright disc of direct light completely.
- Apply immersion oil (for high magnification). Lower the condenser slightly. Put a drop of immersion oil on the top lens of condenser. Raise the condenser slowly till oil touches the underside of the slide. Put one more drop of oil on the coverslip. Swing the oil immersion objective into position. Air bubbles should not be present, otherwise light will scatter and image will be spoiled.
- Adjust objective aperture if needed. If the oil objective has iris diaphragm, reduce the objective NA. It should remain below condenser NA, and background light is blocked.
- Final focusing. Look through eyepiece and focus slowly. The field changes from bright to grey and then to pitch black. At correct focus, background is black and specimen structures shine bright.
Application of Dark Field Microscope
- Clinical diagnostics– It is used for rapid detection of Treponema pallidum (spirochete) in clinical samples. It helps in diagnosis of syphilis by direct observation of organism.
- Live biological observation– It is used to view living and unstained aquatic organisms. Algae, plankton, diatoms, protozoa and small invertebrates can be observed in natural condition.
- Cellular analysis– It is used to examine living blood cells, sperm and tissue culture cells. Yeast cells can also be seen. Motility of flagellated bacteria is observed without using harmful chemical stains.
- Metallurgy and failure analysis– It is used to observe surface topography of metals. Grain boundaries, surface defects and hairline cracks are identified in metal samples.
- Gemology and mineralogy– It is used to study diamonds and other precious stones. Internal inclusions, flaws and refractive index gradients can be detected.
- Semiconductor manufacturing– It is used to inspect semiconductor wafers. Random defects like scratches, dust and particles are detected. Pattern anomalies due to photolithography error can also be seen.
- Nanotechnology– It is used to visualize sub-resolution particles and nanomaterials. Carbon nanotubes and nanoparticles (gold or silver) can be tracked by dark field observation.
- Materials science– It is used to study thin sections of polymers, fibers, hairs, ceramics and colloids. Sharp edges, outlines and structural discontinuities are observed clearly.
Types of specimens used for Dark Field Microscope
Types of specimens used for Dark Field Microscope
- Minute living aquatic organisms
Transparent and living aquatic forms are used. Diatoms, algae, plankton, small insects and small invertebrates like shrimp can be observed in wet mount. - Unstained microorganisms
Unstained bacteria, yeast and protozoa are suitable. Spirochetes like Treponema pallidum are also seen well because they scatter light and show bright outline. - Biological fluids and single cells
Fresh wet mounts are used. Blood cells, sperm and tissue culture cells can be examined. Biological fluids from plants and animals are also used for dark field observation. - Very thin histological and tissue sections
Very thin sections are required, and they should be unstained or very lightly stained. Bone, hair and plant sections can be used in thin preparation. - Sub-microscopic particles
Very small structures are detected by dark field. Bacterial flagella, carbon nanotubes and nanoparticles (gold or silver) can be visualized because they scatter light strongly. - Crystals and minerals
Chemical crystals and mineral particles can be observed. Diamonds and other precious stones are used to see inclusions and refractile defects. - Industrial and material science samples
Thin sections of polymers, ceramics, colloids, fibres and glass showing porosity can be examined. Metals with hairline fractures and surface defects are also suitable. - Specialized biological preparations
Some special preparations like autoradiography and gold labelling preparations are also observed in dark field.
Advantages of Dark Field Microscope
- No staining required– No chemical staining is needed to increase contrast. Living and unstained cells can be observed safely without killing the specimen.
- Minimal sample preparation– The sample preparation is very less. Complex steps are not required, so time and resources are saved.
- Good for transparent specimens– It is very effective for transparent specimens which absorb little or no light. Aquatic organisms, yeast, tissue culture cells and bacteria can be studied easily.
- Outlines and external details are clear– The edges, boundaries and outlines are seen clearly. Refractive index differences can also be noticed in the specimen.
- Detection of very small particles– The resolution limit is similar to brightfield, but very small suspended particles can be detected. Particles around 20 to 40 nm like bacterial flagella can be seen because they scatter light on pitch black background.
- Simple and low cost method– It is a simple technique and cost effective. A standard brightfield microscope can be converted to dark field by adding dark field condenser or stop with less cost.
- Less artifacts in image– Images are mostly free from halo and relief type artifacts. It is less disturbed compared to phase contrast or DIC in many cases.
- High diagnostic utility– It gives rapid and onsite detection of some pathogens. Non-culturable bacteria like Treponema pallidum (syphilis organism) can be detected by direct viewing.
Limitations of Dark Field Microscope
- High light intensity requirement– Most of the direct light is blocked in dark field. So the image becomes low light (light starvation). A very bright high intensity light source is required to make specimen visible.
- Risk of specimen damage- Because intense illumination is used, heat may be produced. Live and delicate specimens can get damaged during prolonged observation.
- Very sensitive to dust and contaminants
Dust, dirt, smudges and air bubbles scatter light and shine bright. This produces glare and it can hide the specimen. Slides and optical surfaces must be kept very clean. - Specimen thickness limitation– Specimen should be very thin. If the sample is thick or dense, light scatters from many layers and background becomes grey. Contrast is reduced and dark background effect is lost.
- Not good for accurate size measurement– The image is formed by scattered light, so glare is produced. Objects may look slightly bigger than actual size. So it is not reliable for precise measurement.
- Poor internal detail- It mainly shows outline and edges. Internal structures of dense cells are not seen properly. So internal detail resolution is poor.
- Artifacts and false colours– Objects above or below the focus plane can also scatter light. This gives blurring and distortion of image. Pigmented specimens may show false colours because image depends on scattered light spectrum.
- Time sensitivity for live wet mount– Fast moving live organisms can disturb the image. Wet preparations should be examined quickly, otherwise movement affects image quality.
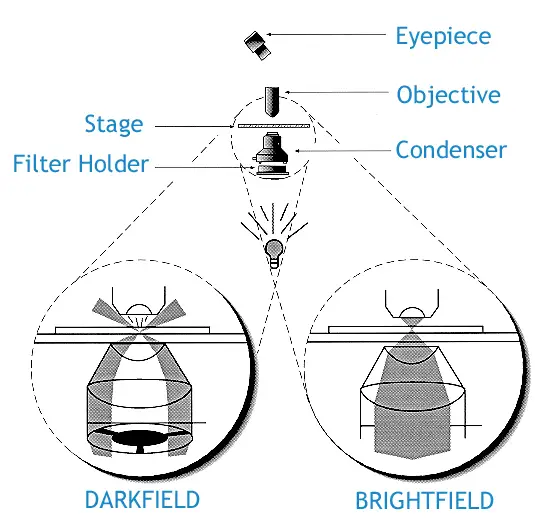
Difference between Dark field and Bright field microscopy – Bright Field vs Dark Field
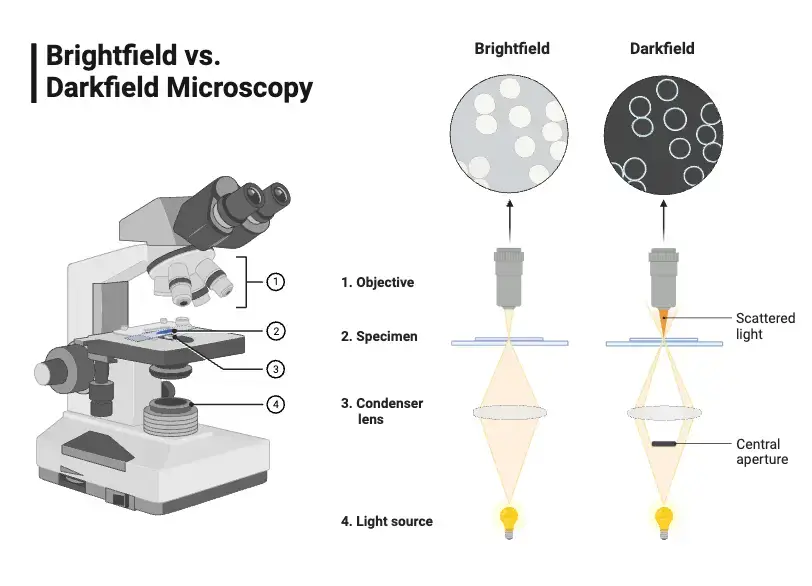
- Background appearance– Brightfield gives a dark or coloured specimen image on a bright background. Dark field gives a bright specimen on a dark or pitch black background.
- Illumination method– Brightfield illuminates the specimen directly with full cone of light. Dark field uses an opaque stop to block central direct light. It sends an oblique hollow cone of light, and only scattered light enters the objective lens.
- Staining requirement– Brightfield often needs staining to give contrast for transparent samples. Many times living cells get killed by stains. Dark field does not require staining for contrast and it is used for live and unstained samples.
- Visible details– Brightfield is more useful for internal structures, mainly after staining. Dark field is best for outlines, edges, boundaries and surface details. Internal cell detail is not seen properly in dark field.
- Particle detection– Brightfield is limited by normal optical resolution. Dark field has same formal resolution, but it can detect very small suspended particles and fibres. Around 20 to 40 nm particles like bacterial flagella can be detected due to strong scattering on black background.
- Specimen thickness– Brightfield can tolerate different thickness of specimen. Dark field needs very thin specimen. Thick and dense sample scatters too much light and the background becomes grey, so contrast is lost.
- Sensitivity to contaminants– Brightfield is somewhat forgiving with minor slide imperfections. Dark field is very sensitive to dust, dirt, fingerprints and air bubbles. These contaminants scatter light and appear bright, creating glare and degrading the final image.
Dark Field Microscope Images

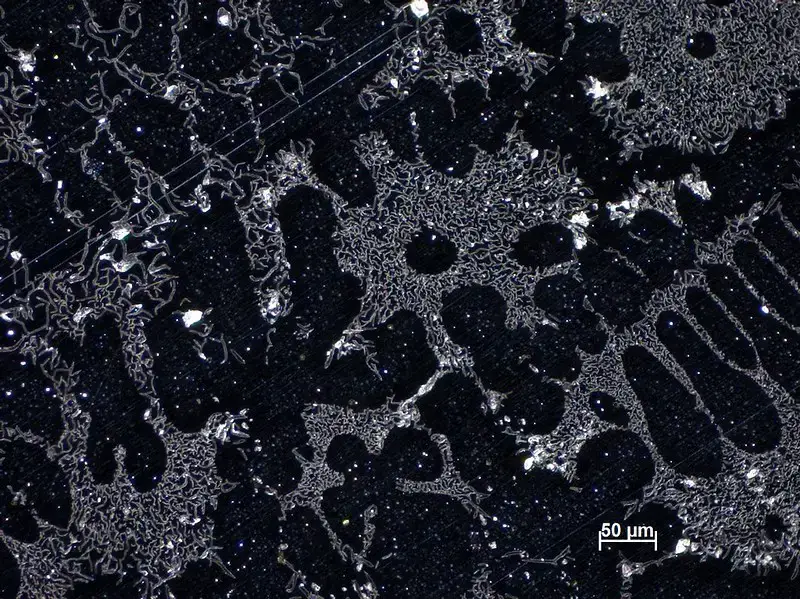

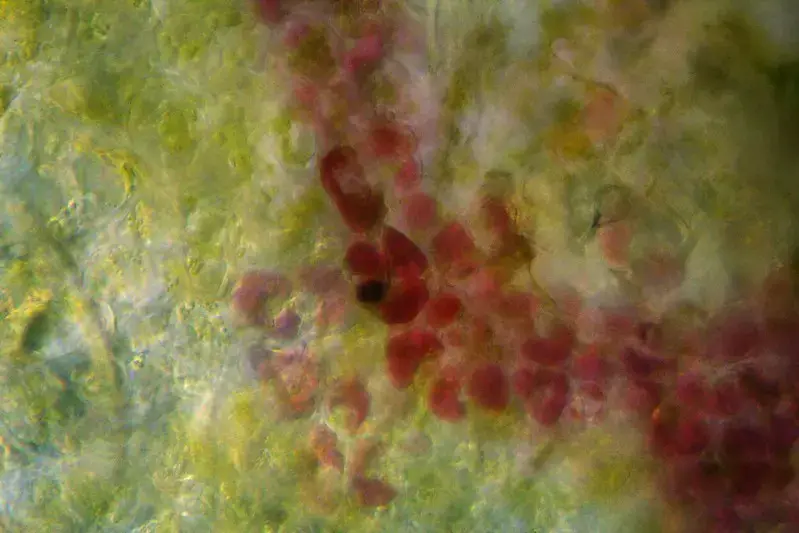
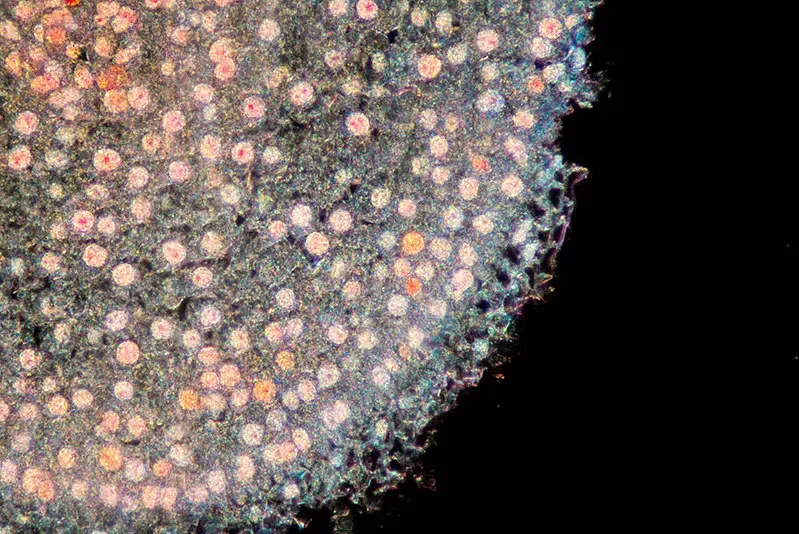

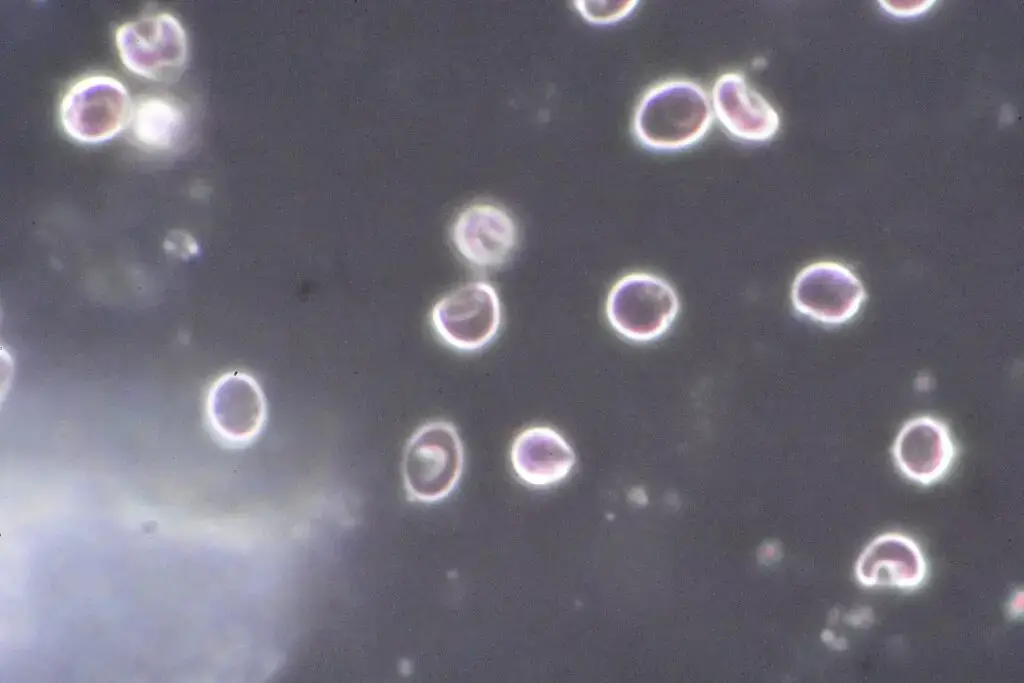
References
- Abramowitz, M., & Davidson, M. W. (2015). Specialized microscopy techniques: Darkfield illumination. Molecular Expressions Microscopy Primer. https://micro.magnet.fsu.edu/primer/techniques/darkfield.html
- Abramowitz, M., & Davidson, M. W. (2015). Specialized microscopy techniques: Darkfield microscope configuration. Molecular Expressions Microscopy Primer. https://micro.magnet.fsu.edu/primer/techniques/darkfieldsetup.html
- Abramowitz, M., & Davidson, M. W. (n.d.). Darkfield illumination. Evident Scientific. https://evidentscientific.com/en/microscope-resource/knowledge-hub/techniques/darkfield
- Abramowitz, M., & Davidson, M. W. (n.d.). Darkfield microscope configuration. Evident Scientific. https://evidentscientific.com/en/microscope-resource/knowledge-hub/techniques/darkfieldsetup
- Abramowitz, M., Herman, B., Murphy, D. B., & Davidson, M. W. (n.d.). Anatomy of the fluorescence microscope: Light path, filter cubes & detectors. Evident Scientific. https://evidentscientific.com/en/microscope-resource/knowledge-hub/techniques/fluorescence/anatomy/fluoromicroanatomy
- AccessEmergency Medicine. (n.d.). Dark-field examination for Treponema pallidum. In The Atlas of Emergency Medicine (5th ed.). McGraw-Hill. https://accessemergencymedicine.mhmedical.com/content.aspx?bookid=2969§ionid=250464364
- Ambooken, B., Binesh, V. G., Asokan, N., Sarin, A., Natarajan, B., & Subi, C. T. (2024). Dark ground microscopy for identification of Treponema pallidum. Journal of Skin and Sexually Transmitted Diseases, 6(2), 185–187. https://doi.org/10.25259/JSSTD_39_2022
- Aryal, S. (2022). Darkfield microscope- Definition, principle, uses, diagram. Microbe Notes. https://microbenotes.com/darkfield-microscopy/
- Bagnell, C. R., Jr. (2012). Chapter 9: Dark field microscopy. In Pathology 464 – Light microscopy. https://www.chem.uci.edu/~dmitryf/manuals/Fundamentals/Dark%20Field%20microscopy.pdf
- Berdan, R. (2022, July 19). Introduction to dark-field microscopy. Motic Microscopes. https://moticmicroscopes.com/blogs/articles/introduction-to-dark-field-microscopy
- Burbank, J. A., Jr. (2022). Petrographic microscopes. EBSCO Research Starters. https://www.ebsco.com/research-starters/science/petrographic-microscopes
- Carl Zeiss Microscopy GmbH. (2020). Light microscopy contrast methods for materials research [PDF]. https://asset-downloads.zeiss.com/catalogs/download/mic/7bea90f7-4078-4fae-a496-d50f2e5f1602/EN_poster_Contrast-Methods-for-Materials-Research.pdf
- Centers for Disease Control and Prevention. (1961). Treponema pallidum (ID# 10179). Public Health Image Library (PHIL). https://wwwn.cdc.gov/phil/Details.aspx?pid=10179
- De Grand, A. (2020, December 17). What is darkfield microscopy? Evident Scientific. https://evidentscientific.com/en/insights/what-is-darkfield-microscopy
- dorgman03. (n.d.). Dark field microscopy overview. Scribd. https://www.scribd.com/document/694662670/DARK-FIELD-MICROSCOPY
- Fellers, T. J., & Davidson, M. W. (2018). Anatomy of the MIC-D digital microscope: Darkfield illumination. Molecular Expressions. https://micro.magnet.fsu.edu/optics/olympusmicd/anatomy/micddarkfield.html
- Gao, P. F., Lei, G., & Huang, C. Z. (2021). Dark-field microscopy: Recent advances in accurate analysis and emerging applications. Analytical Chemistry, 93(11), 4707–4726. https://doi.org/10.1021/acs.analchem.0c04390
- Hayden, E. (2002). BioImaging: Adventures on the dark side: An introduction to darkfield microscopy. BioTechniques, 32(4), 756–760. https://www.tandfonline.com/doi/pdf/10.2144/02324bi01
- ioLight. (n.d.). Different types of light microscopy. https://iolight.co.uk/different-types-of-light-microscopy/
- Ishmukhametov, I., & Fakhrullin, R. (2021). Dark-field hyperspectral microscopy for carbon nanotubes bioimaging. Applied Sciences, 11(24), 12132. https://doi.org/10.3390/app112412132
- Microscope World. (n.d.). Darkfield microscopy. https://www.microscopeworld.com/darkfield-microscopy/
- Moon, G. (2025, January 21). Semiconductor defect detection with bright field microscopy and dark field microscopy. Deep Block. https://www.deepblock.net/blog/semiconductor-defect-detection-with-bright-field-microscopy-and-dark-field-microscopy
- Moore, S. (2022, October 19). The benefits and limitations of dark field microscopy. AZoOptics. https://www.azooptics.com/Article.aspx?ArticleID=2335
- Murphy, D. B., & Davidson, M. W. (n.d.). Comparison of phase contrast & DIC microscopy. Nikon’s MicroscopyU. https://www.microscopyu.com/tutorials/comparison-of-phase-contrast-and-dic-microscopy
- Murphy, D. B., Hinsch, J., Spring, K. R., & Davidson, M. W. (2015). Comparison of phase contrast and DIC microscopy. Molecular Expressions Microscopy Primer. https://micro.magnet.fsu.edu/primer/techniques/dic/dicphasecomparison.html
- Murphy, D. B., Spring, K. R., & Davidson, M. W. (n.d.). DIC vs phase contrast microscopy: Image quality & application guide. Evident Scientific. https://evidentscientific.com/en/microscope-resource/knowledge-hub/techniques/dic/dicphasecomparison
- Nikon Instruments Inc. (n.d.). Darkfield illumination. Nikon’s MicroscopyU. https://www.microscopyu.com/techniques/stereomicroscopy/darkfield-illumination
- Nikon Instruments Inc. (n.d.). Introduction to fluorescence microscopy. Nikon’s MicroscopyU. https://www.microscopyu.com/techniques/fluorescence/introduction-to-fluorescence-microscopy
- Nikon Instruments Inc. (n.d.). Polarized light microscopy. Nikon’s MicroscopyU. https://www.microscopyu.com/techniques/polarized-light/polarized-light-microscopy
- Ross, C. S. (1950). The dark-field stereoscopic microscope for mineralogic studies. American Mineralogist, 35, 906–910. https://msaweb.org/AmMin/AM35/AM35_906.pdf
- Theel, E. S., Katz, S. S., & Pillay, A. (2020). Molecular and direct detection tests for Treponema pallidum subspecies pallidum: A review of the literature, 1964–2017. Clinical Infectious Diseases, 71(Suppl 1), S4–S12. https://pmc.ncbi.nlm.nih.gov/articles/PMC7312206/
- Theoretical and applied foundations of dark field microscopy: A comprehensive technical analysis. (n.d.).
- Wikipedia contributors. (2026). Dark-field microscopy. In Wikipedia, The Free Encyclopedia. https://en.wikipedia.org/wiki/Dark-field_microscopy
- Wilkinson, S. (2022, March 26). How to centre a microscope darkfield condenser. Microscopes.com.au. https://microscopes.com.au/blogs/news/how-to-centre-a-microscope-darkfield-condenser
- ZEISS. (n.d.). Microscopy basics: Enhancing contrast in transmitted light. ZEISS Campus. https://zeiss-campus.magnet.fsu.edu/print/basics/contrast-print.html