Polyacrylamide Gel Electrophoresis (PAGE) refers as a biochemical technique used for separation of proteins, nucleic acids etc. according to their charge and size.
In this method, separation is done by migration of charged molecules through a gel matrix. The gel matrix is polyacrylamide gel. It act as a molecular sieve.
The principle is based on movement of charged molecules in electric field. Smaller molecules move faster than larger molecules. The rate of movement is controlled by pore size of gel.
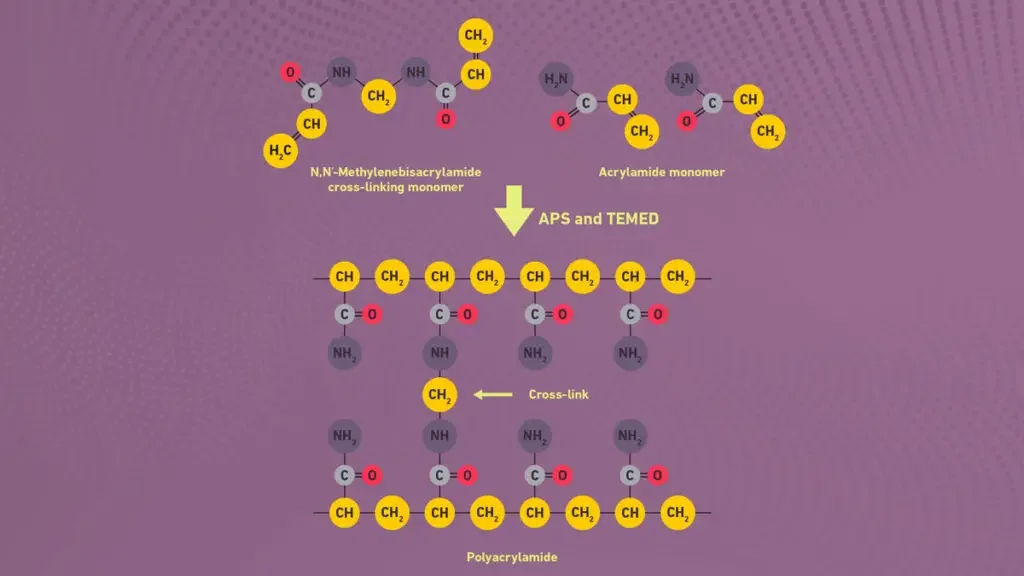
The polyacrylamide gel is formed by polymerization of acrylamide and bisacrylamide. Bisacrylamide act as cross linker. The reaction is done in presence of free radical initiator like ammonium persulfate (APS) and TEMED.
By changing the concentration of acrylamide and bisacrylamide, the pore size of gel can vary. This affect the resolution of protein bands. The gel should be polymerized properly, otherwise uneven band pattern may form.
The proteins carry different net charges. It depend on their amino acid composition and pH of buffer used. When electric current is applied, molecules move towards the electrode of opposite charge. Negatively charged proteins go to anode.
In PAGE, buffer maintain the pH and ionic strength. It prevent overheating and also diffusion of bands. The gel is generally placed vertically between glass plates and submerged by running buffer. Then electric current is applied from top to bottom.
Migration rate depends on molecule size, shape, net charge, gel concentration etc. The electrophoretic mobility is shown as μ = q/f, where q is charge and f is frictional ratio.
Two main types of PAGE are used. Native PAGE and SDS-PAGE. In Native PAGE, proteins retain their natural structure and charge. So separation occurs by charge mass ratio. In SDS-PAGE, proteins are denatured and coated with sodium dodecyl sulfate (SDS) giving uniform negative charge. So separation occurs only by size.
The movement of molecules continues until smaller fragments reach near the bottom. Bands are formed in gel. After electrophoresis, visualization is done by staining methods like Coomassie Brilliant Blue or Silver stain.
The migration pattern gives information about purity and approximate molecular weight of proteins. This method provide a sturdy and hardy analytical approach for biochemical and molecular biology studies.
Types of Polyacrylamide Gel Electrophoresis (PAGE)
Native PAGE – Proteins (or nucleic-acids) are separated under their natural (non-denatured) conditions, so the shape, charge and size all affect mobility, rather than just size.
Denaturing PAGE – The molecules are unfolded / linearised (for proteins by detergent like Sodium dodecyl sulfate (SDS) and sometimes reducing agents) so that separation is mostly by size (mass) rather than shape/charge.
SDS-PAGE – A specific form of denaturing PAGE for proteins, where SDS coats the polypeptides giving roughly uniform charge:mass ratio, and then they are separated by molecular weight (smaller travel faster).
Reducing SDS-PAGE – Similar to SDS-PAGE but with a reducing agent (eg. DTT or β-mercaptoethanol) added to break disulfide bonds, so subunits of multi-unit proteins separate.
Gradient-PAGE / 2D-PAGE – A gradient gel has varying acrylamide concentration (eg from low % to higher %) so a broad size-range of molecules can be resolved, or in 2-D PAGE the first dimension is by one property (like isoelectric point) and second dimension by molecular weight.
Other specialised PAGE types – There are special versions like native continuous PAGE systems, or variants for nucleic-acids (denaturing urea-PAGE) and gel systems for high resolution of small fragments.
Principle of Polyacrylamide Gel Electrophoresis (PAGE)
Principle of Polyacrylamide Gel Electrophoresis (PAGE) is based on the migration of charged biological macromolecules through polyacrylamide gel in electric field. The gel acts as a sieve like matrix.
The polyacrylamide gel has small pores. Molecules like proteins and nucleic acids passes through these pores during electrophoresis.
When electric current is applied, negatively charged molecules moves towards positive electrode called anode. The movement is different for different molecules.
Small molecules passes easily through the gel pores and moves faster. Large molecules are retained more by the gel matrix and moves slowly because of frictional resistance.
Thus molecules are separated in the form of bands. The separation depends on size, charge and shape of molecules.
In SDS-PAGE, sodium dodecyl sulphate (SDS) is added. It unfolds the protein and gives same negative charge to protein molecule. So the proteins are separated mainly on the basis of their molecular weight.
Types of Polyacrylamide Gel Electrophoresis (PAGE)
The following are the important types of Polyacrylamide Gel Electrophoresis (PAGE)–
- Native PAGE
Native PAGE is used to separate proteins in their natural form. In this method, protein is not denatured. The separation depends on charge, size and shape of protein. It also maintains folded structure, subunit interaction and biological activity. - SDS-PAGE
SDS-PAGE is a denaturing type of PAGE. In this method, sodium dodecyl sulphate (SDS) is used. It unfolds the protein and gives uniform negative charge. So the proteins are separated mainly according to molecular weight. - Reducing SDS-PAGE
Reducing SDS-PAGE is a modified type of SDS-PAGE. In this method, reducing agents like DTT or 2-mercaptoethanol are used. These agents break the disulphide bonds of proteins. Thus protein structure is converted into linear polypeptide chain. - Blue Native PAGE (BN-PAGE)
Blue Native PAGE (BN-PAGE) is used for separation of native protein complexes. In this method, Coomassie Brilliant Blue G-250 is used. It gives negative charge to protein surface without denaturing the protein. It is mainly useful for large membrane protein complexes. - Clear Native PAGE (CNE)
Clear Native PAGE (CNE) is a native type of PAGE without using dye. In this method, migration of protein depends mainly on its own charge. It is useful for catalytic and fluorescent assay because dye is not present. - High-Resolution Clear Native PAGE (hrCNE)
High-Resolution Clear Native PAGE (hrCNE) is an improved form of CNE. In this method, mild detergents like DDM and sodium deoxycholate are used. These form mixed micelles and gives negative charge shift. It gives better resolution and also keeps native conformation of protein. - Two-Dimensional PAGE (2D-PAGE)
Two-Dimensional PAGE (2D-PAGE) is used to separate complex protein mixture in two steps. In first step, proteins are separated by isoelectric point or native state. In second step, they are separated by molecular weight using SDS-PAGE. - Urea-PAGE
Urea-PAGE is a denaturing type of PAGE. In this method, high concentration of urea is used. Urea breaks hydrogen bonds and removes secondary structure of DNA or RNA. So they are separated according to molecular weight. - Zymogram PAGE
Zymogram PAGE is used to detect enzyme activity. In this method, substrate like gelatin or casein is mixed with polyacrylamide gel. Active enzyme breaks the substrate and clear zones are formed in the gel. - CTAB PAGE
CTAB PAGE uses a cationic detergent instead of SDS. It is useful for proteins which precipitate, aggregate or migrate abnormally with SDS. It can also help to preserve some enzymatic activity in mild condition. - Preparative Native PAGE
Preparative Native PAGE is used for separation and purification of intact native proteins. It is not only used for analysis. It is used to collect and isolate proteins from complex biological samples.
Requirements for Polyacrylamide Gel Electrophoresis (PAGE)
To perform Polyacrylamide Gel Electrophoresis (PAGE), several requirements and equipment are necessary. These include:
The following are the requirements for Polyacrylamide Gel Electrophoresis (PAGE)–
- PAGE rig
PAGE rig consists of glass plates, spacers, comb and clamps. Glass plates are used for gel casting. Spacer maintains the thickness of gel. Comb forms the wells where sample is loaded. - Casting stand
Casting stand is required for holding the glass plates during gel preparation. In this stand, liquid gel mixture is poured and allowed to polymerize. - Electrophoresis tank
Electrophoresis tank holds the gel and running buffer. The gel plate is fixed in this tank during electrophoresis. - Power supply
Power supply gives electric current to the apparatus. Due to this current, electric field is produced and charged molecules migrate through the gel. - Acrylamide and Bis-acrylamide
Acrylamide and Bis-acrylamide are required for preparation of gel matrix. Acrylamide forms long chain and Bis-acrylamide cross links the chain. So a porous polyacrylamide gel is formed. - Ammonium Persulphate (APS)
Ammonium Persulphate (APS) is required as free radical initiator. It starts polymerization reaction of acrylamide and bis-acrylamide. - TEMED
TEMED acts as catalyst. It helps APS in formation of free radicals and polymerization takes place faster. - Gel buffer
Gel buffer is added in the gel. It maintains the proper pH during electrophoresis. Tris buffer is commonly used for this purpose. - Sample
Sample may be protein or nucleic acid. This is the material which is separated by PAGE. - Sample loading buffer
Sample loading buffer is mixed with sample before loading into the well. It contains glycerol or sucrose which makes the sample heavy. So sample goes down properly into the well. - Tracking dye
Tracking dye is present in loading buffer. Bromophenol blue is commonly used. It shows the movement of sample during running of gel. - Molecular weight marker
Molecular weight marker is loaded with sample. It gives standard bands. These bands are compared with sample bands for finding approximate size. - Sodium Dodecyl Sulphate (SDS)
Sodium Dodecyl Sulphate (SDS) is required for SDS-PAGE. It unfolds the protein and gives negative charge on protein. Thus protein moves according to molecular weight. - Reducing agent
Reducing agent like DTT or 2-mercaptoethanol is used for breaking disulphide bonds. It separates protein structure into polypeptide chain. - Urea
Urea is used in denaturing PAGE for DNA and RNA. It breaks hydrogen bonds and removes secondary structure. - Running buffer
Running buffer is filled in the electrophoresis tank. It carries current and helps movement of molecules through gel. Tris-glycine buffer is commonly used. - Staining solution
Staining solution is used after electrophoresis. It makes the separated bands visible. For protein, Coomassie Brilliant Blue, silver stain and SYPRO Ruby are used. For nucleic acids, ethidium bromide or safer dye is used and bands are seen under UV light.
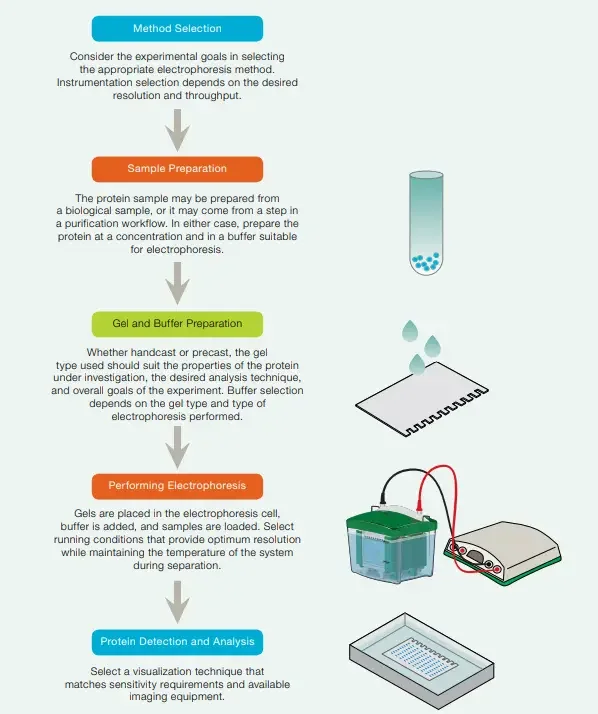
Steps Involved in Polyacrylamide Gel Electrophoresis (PAGE)
1. Sample preparation
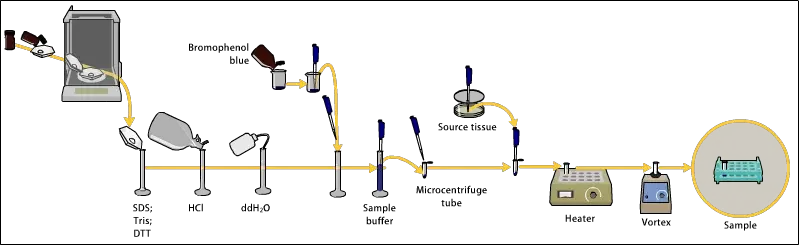
The following are the steps for sample preparation of Polyacrylamide Gel Electrophoresis (PAGE)–
- The sample is collected from protein or nucleic acid source which is needed to be separated by PAGE technique.
- The concentration of sample is adjusted in proper range so that clear and visible bands are obtained after electrophoresis.
- Sample buffer is added to the sample. It contains Tris-HCl, SDS, glycerol, bromophenol blue etc. It maintains pH and gives density to the sample.
- The protein sample is heated at 95°C for 3-5 minutes. This heating helps in breaking secondary and tertiary structure of protein.
- Sodium dodecyl sulphate (SDS) present in the buffer gives uniform negative charge to all protein molecules.
- Sometimes β-mercaptoethanol (β-ME) or DTT (dithiothreitol) is added. These reducing agents breaks the disulphide bonds in protein structure.
- The sample and buffer are mixed properly so that they are completely homogenized. Bubbles should not be formed during mixing.
- The sample volume is kept small, usually 10-20 µL, depending on the well size of gel.
- After denaturation, the sample is cooled on ice for few minutes to prevent re-aggregation of protein molecules.
- The loading dye or tracking dye like bromophenol blue helps in monitoring the migration of sample during electrophoresis run.
- Before loading, the sample may be centrifuged shortly for about 30 seconds. It removes insoluble particles or bubbles.
- The prepared sample is carefully loaded into the wells of polyacrylamide gel by using micropipette.
- Molecular weight marker is also loaded in one separate well for comparison of protein sizes.
- In Native PAGE, sample is prepared without SDS and reducing agents. In this method, protein is kept in native folded form.
- The prepared sample should be used freshly or stored at -20°C if delay occur before electrophoresis.
- All these steps ensure that protein molecules migrate properly according to their molecular weight or charge during PAGE run.
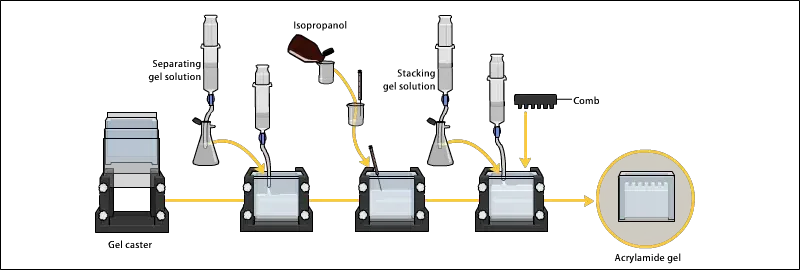
2. Preparing acrylamide gels
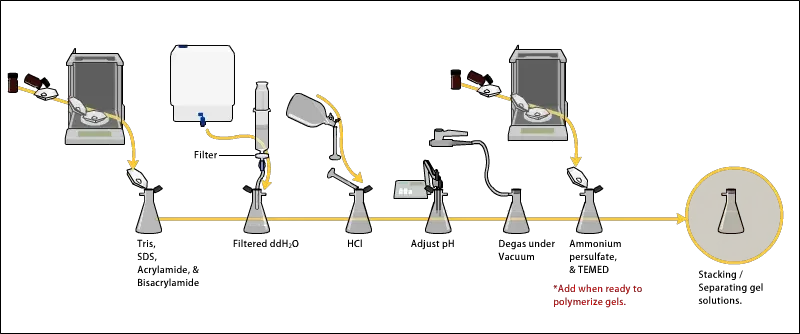
The following are the steps for preparing acrylamide gels–
- The gel solution is prepared by mixing acrylamide and bis-acrylamide solution in fixed ratio. This ratio decide the pore size of gel. Usually 30:1 ratio is used for protein separation.
- Tris-HCl buffer is added in the mixture for maintaining pH. Mostly pH 8.8 is used for resolving gel and pH 6.8 is used for stacking gel.
- Then distilled water is added to make the final volume of gel solution. The volume depends on the size and thickness of gel required.
- Before polymerization, SDS (sodium dodecyl sulfate) is added when SDS-PAGE is performed. Usually 0.1% SDS is used for denaturing protein.
- The gel solution is de-gassed by mild vacuum or by nitrogen bubbling. This is done to remove oxygen, because oxygen inhibits the polymerization reaction.
- Polymerization is started by adding ammonium persulfate (APS) and TEMED (tetramethylethylenediamine). These two produces free radicals and starts the chain reaction.
- The resolving gel is poured first between the glass plates up to the required height. Usually it is poured about 3/4th of the plate.
- After pouring resolving gel, a thin layer of water or butanol is added over it. This makes the surface level and also prevent contact of oxygen with gel solution.
- The resolving gel is allowed to polymerize for about 20-30 minutes. After polymerization, water or butanol is removed gently by pipette.
- The surface of resolving gel is washed carefully. This removes remaining water or butanol from upper surface.
- Then stacking gel solution is prepared separately. It contains lower acrylamide concentration, usually around 4%, and lower pH buffer of 6.8.
- APS and TEMED are also added in stacking gel solution for polymerization.
- The stacking gel is poured over the polymerized resolving gel. A comb is inserted immediately to form wells for sample loading.
- The stacking gel is allowed to polymerize for about 10-20 minutes. After complete polymerization, the comb is removed carefully.
- The wells are rinsed with running buffer. This removes unpolymerized acrylamide residues from the wells.
- The prepared gel is now ready for fitting in electrophoresis apparatus. It can be used immediately or kept in moist condition for short time.
- Proper polymerization, acrylamide/bis-acrylamide ratio and pH adjustment affect the quality of bands during electrophoresis.
3. Electrophoresis
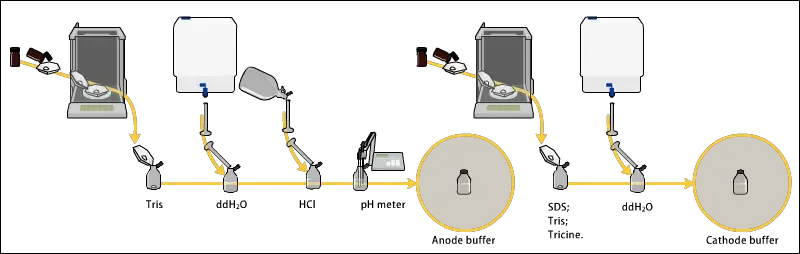
The following are the steps of electrophoresis–
- The gel assembly is placed properly in the electrophoresis apparatus. It is tightened carefully so that running buffer do not leak out.
- Running buffer like Tris-Glycine or Tris-Tricine is poured into upper and lower tank. The buffer is added till the electrodes are fully immersed.
- The wells of gel are rinsed gently with buffer. This removes unpolymerized residues and bubbles which may disturb sample loading.
- The prepared protein samples are loaded carefully into the wells by using micropipette. Molecular weight marker is also loaded in one separate well.
- The samples should settle properly in the wells. Overflow or mixing of sample between adjacent wells should not occur.
- The power supply is connected with correct polarity. In SDS-PAGE, negatively charged protein-SDS complexes migrate towards the positive electrode, anode.
- The voltage is applied usually 80-120 V during stacking part. Then it is increased to 120-200 V during resolving stage depending on the gel type.
- During the run, bromophenol blue tracking dye is observed. It moves ahead of protein bands and shows the progress of electrophoresis.
- The electrophoresis is continued until the dye front reaches near the bottom of gel. Normally it takes about 1-2 hours.
- Overheating should be avoided during electrophoresis. So the apparatus may be kept in cooling condition or circulating buffer is used.
- After completion of run, the power supply is turned off. The gel plates are carefully dismantled by using spatula or gel knife.
- The gel is separated gently from glass plate. It is transferred to staining tray without tearing.
- The staining step is done immediately for visualization of separated protein bands. Coomassie Brilliant Blue or Silver stain is commonly used.
- When bands are fixed and visible, the gel is photographed or analyzed for molecular weight estimation etc.
4. Detection
The following are the steps for detection after Polyacrylamide Gel Electrophoresis (PAGE)–
- After electrophoresis, the gel is removed gently from the glass plates. It is then placed in a tray for staining process.
- Coomassie Brilliant Blue (CBB) stain is usually used for detection of proteins. It binds with amino acid residues, mainly arginine and aromatic amino acids.
- The gel is immersed in staining solution. The solution contains CBB R-250 dissolved in methanol: acetic acid: water mixture. Usually 5:1:4 ratio is used.
- The staining is done by slow agitation at room temperature for 1-2 hours. It is continued until blue colour appears clearly in protein bands.
- Sometimes heating is used for quick staining. But too high temperature can deform the gel or diffuse the bands.
- After staining, the gel is transferred to destaining solution. It contains methanol, acetic acid and water in different ratio.
- The destaining is done by changing the solution several times. It removes extra dye from background and only protein bands remain blue.
- For higher sensitivity, Silver staining is used. It can detect proteins in nanogram range. But this method is more complex and sensitive to contamination.
- In Silver staining, fixation is done first in ethanol and acetic acid. Then the gel is treated with silver nitrate solution and developed with formaldehyde solution.
- Sometimes fluorescent dyes or radioactive labelling are also used. Fluorescent dyes are detected under UV light and radioactive bands are detected by autoradiography.
- After completion of detection step, the gel is washed with distilled water. Then it is preserved by drying or storing in plastic sheet for record purpose.
- The clarity of bands, contrast and sharpness depends on staining efficiency and gel handling during the process etc.
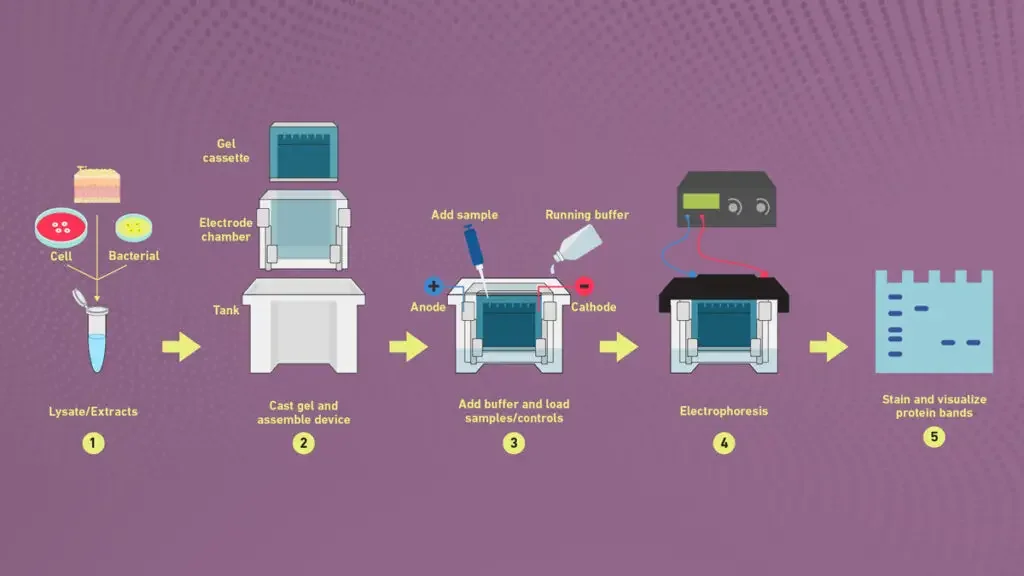
SDS PAGE vs native PAGE
| Basis | SDS-PAGE | Native PAGE |
|---|---|---|
| Basis of separation | It separates proteins mainly on the basis of molecular weight. | It separates proteins on the basis of size, shape and natural charge. |
| Protein condition | It is carried out in denaturing condition. | It is carried out in non-denaturing condition. |
| Protein structure | SDS unfolds the protein and destroys secondary, tertiary and quaternary structure. | Protein remains in its natural folded form and higher structure is maintained. |
| Electrical charge | SDS masks the natural charge and gives almost same negative charge to all proteins. | Natural charge of protein remain as it is and affects migration through gel. |
| Protein form | Proteins are converted into linear polypeptide chain. | Proteins remain in native form with subunit interaction. |
| Biological activity | Biological activity is lost due to denaturation. | Biological and enzymatic activity is maintained. |
| Recovery | Protein cannot be recovered in stable active form after separation. | Protein can be recovered after separation in active form. |
| Uses | It is used for molecular weight determination, sample purity and analysis of protein subunits. | It is used for protein-protein interaction, intact protein complex and functional protein study. |

Applications of Polyacrylamide Gel Electrophoresis (PAGE)
The following are the applications of Polyacrylamide Gel Electrophoresis (PAGE)–
- PAGE is used for separation and analysis of biological macromolecules like proteins and nucleic acids. It helps in fractionation, identification and characterization of these molecules on the basis of size, charge and conformation.
- PAGE is used for estimation of molecular weight of unknown proteins and DNA/RNA fragments. The migration distance of unknown sample is compared with standard marker ladder.
- SDS-PAGE is used to check the purity of protein sample. Preparative Native PAGE is also used for physical isolation and purification of intact proteins like metalloproteins and membrane proteins from complex biological mixtures.
- Native PAGE and Blue Native PAGE are used for studying protein complexes and protein-protein interactions. These methods helps in preserving enzymatic activity and also analyze assembly status and subunit composition of large structures like mitochondrial respiratory chains.
- In clinical pathology, PAGE is used in serum protein electrophoresis (SPEP) and urine protein electrophoresis (UPEP). It helps to detect pathological changes like monoclonal M-spikes, which are related with multiple myeloma, lymphoma, leukemia and kidney disease.
- In forensic science, PAGE is used for high resolution sizing of short tandem repeats (STRs) and mitochondrial DNA fragments. It is useful in DNA fingerprinting, paternity testing and identification of human remains.
- Denaturing Urea-PAGE is used for nucleic acid analysis. It is used in DNA sequencing, finding composition of RNA species, checking quality of RNA preparation, analyzing ribozyme activity and purification of synthetic oligonucleotides.
- Two-Dimensional PAGE (2D-PAGE) is used for global proteomic profiling. It helps to separate thousands of individual proteins in one gel, map whole proteome, detect post-translational modifications, identify protein isoforms and observe changes in protein expression.
- Zymogram PAGE is used for detection of enzyme activity inside the gel matrix. It is useful for enzymes like proteases and collagenases.
Advantages of Polyacrylamide Gel Electrophoresis (PAGE)
The following are the advantages of Polyacrylamide Gel Electrophoresis (PAGE)–
- PAGE gives very high resolution for separating biological macromolecules like proteins and nucleic acids. So close sized molecules can also be separated properly in the gel.
- The pore size of polyacrylamide gel can be controlled by changing the concentration of gel chemicals. This helps to prepare gel according to the size of molecule which is required to be separated.
- Polyacrylamide gel forms a strong and synthetic matrix. It is thermo-stable, transparent, electrically neutral and chemically inert, so it is suitable for electrophoretic separation.
- PAGE can separate molecules on the basis of size, electrical charge or combination of both. This depends on the gel and buffer system which are used.
- The polyacrylamide matrix can withstand high voltage gradient during electrophoresis. So it is durable during the running process.
- The gel can be used with different staining and destaining methods after electrophoresis. It can also be dried for permanent record like autoradiography or digested to extract separated fractions.
- In some variants like Native PAGE, the intact structure of protein complexes is maintained. So it is useful for studying protein-protein interactions.
- PAGE is used for different types of samples. It is applicable for both soluble proteins and membrane protein complexes.
Limitations of Polyacrylamide Gel Electrophoresis (PAGE)
The following are the limitations of Polyacrylamide Gel Electrophoresis (PAGE)–
- Unpolymerized acrylamide is toxic and it can easily penetrate through skin. It may act as neurotoxin, suspected carcinogen and reproductive toxin. So strict safety protocol, gloves, lab coat and proper waste disposal as hazardous chemical waste are required during handling.
- Traditional PAGE is a labor-intensive and time consuming process. Handcasting of gel takes more time, needs careful optimization and sometimes the gel is less uniform and less reproducible than precast gels or automated systems.
- Some proteins shows abnormal migration in PAGE. Highly hydrophobic membrane proteins and heavily glycosylated proteins can bind SDS differently, so they may not separate properly according to size. Very large proteins of 250,000 to 600,000 Da are also difficult to resolve in standard gel system.
- PAGE is prone to artifacts and distortions. Smiling effect may occur due to uneven Joule heating, vertical streaking may occur from overloaded sample and skewed bands may occur due to air bubbles or uneven gel casting.
- Native PAGE has some limitation because proteins are kept in non-denaturing condition. Membrane proteins may aggregate without detergents, Clear Native PAGE (CNE) is mostly limited to acidic proteins having pI < 7, Blue Native PAGE (BN-PAGE) uses Coomassie dye which can reduce fluorescence in labelled samples, and reliable molecular weight markers for native folded proteins are not always available.
- Advanced variations like Two-Dimensional PAGE (2D-PAGE) need high technical skill, special equipment and careful control of experimental conditions. So the method becomes more difficult and costly.
References
- Abcam. (n.d.). Comprehensive guide to RNA purification.
- Abcam. (n.d.). SDS-PAGE guide: Sample preparation to analysis.
- Academia.edu. (n.d.). (PDF) Acrylamide polymerization bulletin.
- Agilent Technologies. (n.d.). TapeStation automated electrophoresis for DNA & RNA quality control.
- Asmita, B. (2025, February 17). Microfluidics market analysis (2024-2035): Trends, innovations, and future outlook. PharmiWeb.com.
- Bio-Rad Laboratories. (n.d.). Introduction to polyacrylamide gels.
- Bio-Rad Laboratories. (n.d.). Protein electrophoresis methods.
- Bio-Rad Laboratories. (n.d.). Protein staining.
- Bio-Techne. (n.d.). Jess automated western blot system (004-650) by ProteinSimple, part of Bio-Techne.
- Bio-Techne. (n.d.). Jess ProteinSimple – Learn more about automated western blots.
- BYJU’S. (n.d.). SDS PAGE vs native PAGE.
- Chakravarty, P. R. (n.d.). Troubleshooting SDS-PAGE gel running issues. GoldBio.
- Chakravarty, P. R. (n.d.). Troubleshooting SDS-PAGE sample preparation issues. GoldBio.
- Chromatography Today. (n.d.). 200 years of electrophoresis.
- Conference Series. (2026). Microfluidics 2026 | August 27-28, 2026 | Berlin, Germany.
- Ervin. (2026, March 2). Protein Simple Jess a guide to automated western blotting. Neurodex.
- Ezsias, B., Goessweiner-Mohr, N., Siligan, C., Horner, A., Vargas, C., Keller, S., & Pohl, P. (2025, August 1). Clear native gel electrophoresis for the purification of fluorescently labeled membrane proteins in native nanodiscs. Analytical Chemistry, 97(31), 16796–16804.
- Georgia Tech. (2001, January 5). SDS PAGE gel electrophoresis PAGE.
- Grand View Research. (n.d.). Microfluidics market size and share | Industry report, 2033.
- Hycult Biotech. (n.d.). Troubleshooting sodium dodecyl sulfate-polyacrylamide gel electrophoresis.
- King, J. (n.d.). Ulrich Laemmli’s development of SDS polyacrylamide gel electrophoresis. MIT Faculty Newsletter.
- LICORbio. (2020, January 10). What are the different types of western blot gels?
- MBL Life Science. (n.d.). The principle and method of polyacrylamide gel electrophoresis (SDS-PAGE).
- Menter, P. (2000). Acrylamide polymerization: A practical approach. Bio-Rad Laboratories.
- MilliporeSigma. (n.d.). Sample prep & gel electrophoresis troubleshooting.
- MtoZ Biolabs. (2018). Advantages and disadvantages of 2D blue native/SDS-PAGE protein complex analysis.
- New England Biolabs. (n.d.). Denaturing PAGE/urea or denaturing agarose gel (NEB #B0363).
- PatSnap Eureka. (2025, June 26). Why gel electrophoresis still matters in 2025?
- PatSnap Eureka. (2025, June 30). How to select optimal staining methods in gel electrophoresis?
- PMC – NIH. (n.d.). Checking your browser – reCAPTCHA.
- PMC – NIH. (n.d.). Comparison of automated and traditional western blotting methods.
- PMC – NIH. (n.d.). Denaturing urea polyacrylamide gel electrophoresis (urea PAGE).
- PMC – NIH. (n.d.). Polyacrylamide gel electrophoresis: A versatile tool for the separation of nanoclusters.
- PMC – NIH. (n.d.). Protein detection in gels using fixation UNIT 10.5.
- PMC – NIH. (n.d.). Titanium dioxide photocatalytic polymerization of acrylamide for gel…
- R&D Systems. (n.d.). Jess automated western blot system.
- Research and Markets. (2026, January). Microfluidics market report 2026.
- ResearchGate. (n.d.). (PDF) REVIEW: AN APPROACH TO POLYACRYLAMIDE GEL ELECTROPHORESIS.
- Rubin, M. (2012, June/July). Case Western Reserve University – Environmental health and safety. Case Western Reserve University.
- Technology Networks. (n.d.). Polyacrylamide gel electrophoresis, how it works, technique variants and its applications.
- Thermo Fisher Scientific. (n.d.). Protein detection on gels, blots and arrays—Section 9.3.
- Thermo Fisher Scientific. (n.d.). Protein gel staining methods.
- Unknown Author. (n.d.). Capillary electrophoresis vs gel electrophoresis: Comparing separation techniques.
- Unknown Author. (n.d.). The foundations, methodological evolutions, and future trajectories of polyacrylamide gel electrophoresis in molecular analytics.
- Wikipedia contributors. (2026, February 2). Polyacrylamide gel electrophoresis. Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (2026, March 30). History of electrophoresis. Wikipedia, The Free Encyclopedia.