What are Peroxisomes?
Peroxisomes are small, single‑membrane organelles present in nearly all eukaryotic cells, they consist of a lipid bilayer that encloses enzymes specialized for oxidation reactions, especially those that break down fatty acids, amino acids and other toxic molecules, this process generates hydrogen peroxide which is then immediately converted into water and oxygen by catalase to prevent damage. They are known as oxidative organelles, they play key roles in lipid metabolism, including beta‑oxidation of very‑long‑chain fatty acids and biosynthesis of plasmalogens which are essential for brain and heart function. In plants and yeast, peroxisomes also conduct specialized functions such as the glyoxylate cycle and photorespiration, and they replicate by division without their own DNA, relying on protein import from the cytosol
Definition of Peroxisome
A peroxisome is a small, membrane-bound organelle found in the cytoplasm of eukaryotic cells. It contains enzymes that are primarily involved in the detoxification of harmful substances, the breakdown of fatty acids, and the metabolism of reactive oxygen species, particularly hydrogen peroxide.
Structure of Peroxisomes
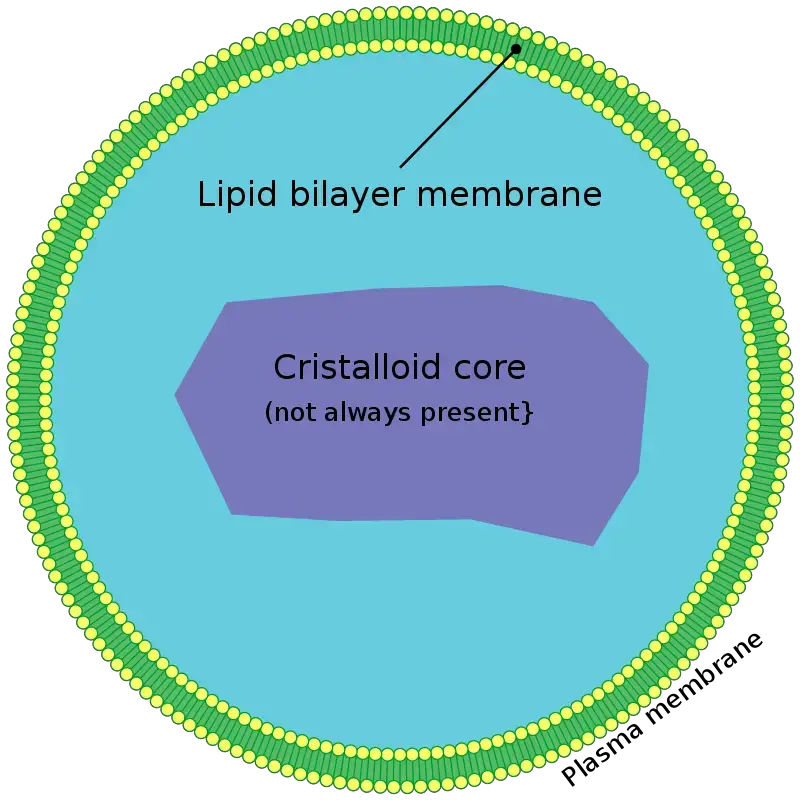
- Membrane – is determined by a single phospholipid bilayer, containing embedded peroxisomal membrane proteins such as PMP70 and PEX proteins that regulate import.
- Size & Shape – peroxisomes are small vesicles about 0.1–1.5 µm in diameter, spherical or tubular, and their size, number, shape varies with cell type and metabolic needs.
- Matrix – consists of a fine granular material or sometimes a crystalline/paracrystalline core (e.g., urate oxidase), where oxidative enzymes are concentrated.
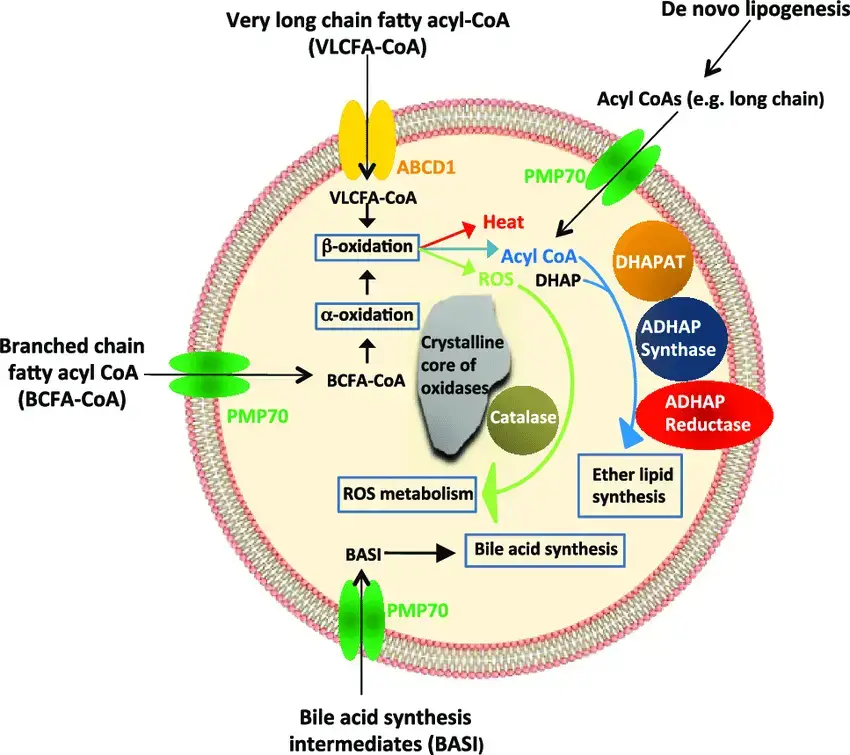
- Enzymes – matrix contains ~50‑60 enzymes, including catalase, oxidases (e.g., urate, D‑amino acid, fatty acid oxidases), supports beta‑oxidation of VLCFA, H₂O₂ detox, plasmalogen and bile acid synthesis.
- Protein Import – peroxisomes lack DNA; matrix proteins are translated in cytosol then imported via PTS1 (C‑term SKL motif) or PTS2 signals through PEX receptors and docking proteins (PEX13, PEX14) in membrane.
- Lipid Acquisition – membrane lipids are supplied by the ER via phospholipid transfer, enabling organelle growth and division.
- Dynamics – peroxisomes replicate by growth and fission, similar to mitochondria, depending on cell demands and environmental conditions
Location of Peroxisomes
- Peroxisomes are located in the cytoplasm of virtually all eukaryotic cells – they’re free‑floating organelles, not attached to cytoskeleton or membranes, and visible only under an electron microscope
- They’re especially abundant in metabolically active cells, such as those in the liver and kidney of animals
- In plant cells, they appear in seed, leaf, and root tissues; they can form specialized types like glyoxysomes for seed oil conversion or photorespiration domains in leaves
- Their numbers vary depending on cell type and conditions – like yeast cells that produce many peroxisomes when grown on fatty acids
So, peroxisomes are single‑membrane organelles dispersed in the cytosol of eukaryotic cells, with higher prevalence in cells with intense lipid metabolism (e.g., liver, kidney, germinating plant seeds).
Peroxisomal Enzymes
Here is the examples of enzymes which are present in Peroxisomes with their functions;
- Catalase is the major antioxidant enzyme in the peroxisomal matrix – it decomposes H₂O₂ into water and oxygen and prevents oxidative damage
- Oxidases transfer H atoms from substrates to O₂, forming H₂O₂ – includes D‑amino acid oxidase, urate oxidase (not in humans), polyamine oxidase, fatty acyl‑CoA oxidases, xanthine oxidoreductase; they generate H₂O₂ as by‑product
- Acyl‑CoA oxidase initiates β‑oxidation of very‑long‑chain fatty acids, yielding H₂O₂ and acetyl‑CoA
- Polyamine oxidase (PAO)/copper‑amine oxidase degrade polyamines, produce H₂O₂ and link to GABA biosynthesis in plants
- Superoxide dismutase (SOD) is present in matrix or bound to membrane in plant peroxisomes; it dismutates O₂•⁻ to H₂O₂
- Ascorbate‑glutathione cycle enzymes such as ascorbate peroxidase (APX), MDAR, DHAR, glutathione reductase are present especially in plants, detoxify ROS
- Xanthine oxidoreductase (XOR) catalyses purine breakdown, generating uric acid and reactive O₂ species
- Enoyl‑CoA isomerase, part of peroxisomal trifunctional enzyme, processes unsaturated fatty acyl‑CoAs in β‑oxidation
- Palmitoyl‑CoA hydrolase hydrolyzes long‑chain acyl‑CoAs, supports peroxisomal fatty acid metabolism
- 2‑Hydroxyphytanoyl‑CoA lyase is peroxisomal enzyme in α‑oxidation of branched fatty acids (e.g., phytanic acid)
- Malonyl‑CoA decarboxylase exists in peroxisomes to degrade malonyl‑CoA from odd‑chain FA oxidation, regulates lipid metabolism
Peroxisome 3D model
Peroxisome Front
Peroxisome Outer Surface
Peroxisome Biogenesis
Step 1 – Initiation from Endoplasmic Reticulum (ER)
- Peroxisome formation kicks off at the ER, where specialized membrane proteins like Pex3p begin accumulating.
- ER buds off vesicles enriched with peroxisomal membrane proteins (PMPs), forming pre-peroxisomal vesicles.
- These vesicles represent early structural units, carrying elements necessary for further maturation.
Step 2 – Vesicle Fusion into Pre-Peroxisomal Structures
- Vesicles emerging from ER fuse together, assembling a complete pre-peroxisomal structure.
- In yeast, vesicles containing Pex14 or ESCRT-III components merge, while in mammals, vesicles with Pex3, Pex14, or Pex16 fuse similarly.
- This fusion is coordinated by PEX gene products, which are essential at this stage.
Step 3 – Insertion of Translocon Components
- Translocon machinery proteins like Pex13 and Pex14 get incorporated into the peroxisomal membrane.
- This membrane complex forms the importomer system, preparing the organelle for matrix protein intake.
- The structure now becomes import-competent, enabling protein trafficking inside.
Step 4 – Import of Matrix Proteins from Cytosol
- Cytosolic proteins destined for peroxisomes carry PTS1 or PTS2 signals.
- Pex5 and Pex7 serve as receptors for PTS1 and PTS2, binding these cargos and docking at the peroxisomal surface.
- Once docked, the DTM (Docking and Translocation Machinery) enables translocation of cargo proteins into the matrix.
Step 5 – Receptor Recycling and Cargo Release
- After cargo is delivered, Pex5 gets mono-ubiquitinated and pulled back into the cytosol.
- This recycling depends on the REM complex, ensuring Pex5 availability for another round of import.
- Efficient turnover of import receptors prevents import blockages and maintains homeostasis.
Step 6 – Organelle Growth and Division
- Mature peroxisomes start to elongate, driven by Pex11β and other morphogenesis proteins.
- Division requires DLP1 and Mff, which constrict and sever elongated peroxisomes.
- This allows peroxisomes to multiply and match the cell’s metabolic needs.
Step 7 – Environmental Response and Proliferation
- Exposure to specific stimuli (e.g., ROS, fatty acids) triggers peroxisome proliferation.
- Increased expression of biogenesis and fission-related genes modulates this response.
- The process ensures rapid adjustment of peroxisome number under stress.
Step 8 – Quality Control via Pexophagy
- Damaged or obsolete peroxisomes are targeted by pexophagy, a selective form of autophagy.
- The import machinery may act as a surveillance system, sensing dysfunction and initiating degradation.
- Fusion of autophagosomes with lysosomes degrades peroxisomes, preventing oxidative stress buildup.
Step 9 – Balance and Homeostasis
- A dynamic balance between new peroxisome formation and degradation preserves cellular health.
- Disturbances in this cycle can cause metabolic disorders or oxidative damage.
- Thus, tight regulation of biogenesis, division, and pexophagy defines peroxisomal integrity.
Human Peroxisome-Related Disorders
- Peroxisome biogenesis disorders (PBDs) consist of defects in peroxins (PEX genes), causing absence or malfunction of peroxisomes and broad metabolic failure – they’re autosomal recessive
- Zellweger spectrum disorders (ZSD) include Zellweger syndrome (severe), neonatal adrenoleukodystrophy (intermediate), and infantile Refsum disease (milder) – all manifest with neuro‑hepato‑renal issues, hypotonia, seizures from birth
- Rhizomelic chondrodysplasia punctata (RCDP) type 1‑3 involve peroxisomal plasmalogen synthesis defects (from PEX7, GNPAT, or AGPS genes) – cause limb shortening, cataracts, intellectual disability
- Heimler syndrome arises from hypomorphic PEX1/PEX6 mutations – leads to hearing loss, enamel/nail defects, mild late‑onset retinal issues
- Single‑enzyme deficiencies occur with intact organelle but loss of one specific enzyme function
- X‑linked adrenoleukodystrophy (X‑ALD) from ABCD1 mutation – impaired VLCFA transport, leads to demyelination and adrenal insufficiency in males
- Refsum disease results from PHYH or PEX7 defects – accumulated phytanic acid causes neuropathy, retinitis, arrhythmias, hearing loss
- D‑bifunctional protein deficiency is due to HSD17B4 mutations – severe neonatal hypotonia, seizures, craniofacial dysmorphism, early death similar to Zellweger
- Acyl‑CoA oxidase deficiency, catalase deficiency (acatalasia), α‑oxidation defects, hyperoxaluria type 1, AMACR, and DHAP‑AT or AGPS deficiencies cause various metabolic and connective tissue effects
These disorders are grouped by whether they affect peroxisome assembly (PBDs) or single enzyme/transporter activity, and many have genetic and biochemical diagnosis options.
Functions of Peroxisomes
- Peroxisomes break down long-chain fatty acids into shorter ones, which makes acetyl-CoA. Acetyl-CoA then enters the citric acid cycle, where it is used to make energy. In yeast and plants, this only happens in peroxisomes.
- Detoxification of reactive oxygen species—peroxisomes use the enzyme catalase to turn hydrogen peroxide (H₂O₂), a byproduct of oxidative reactions, into water and oxygen, which stops cells from being damaged.
- Synthesis of plasmalogens—peroxisomes help make plasmalogens, which are ether phospholipids that are important for making myelin in nerve cells. Not making enough plasmalogen is linked to problems with the nervous system.
- Plants turn stored fatty acids into carbohydrates. For example, in germinating seeds, peroxisomes help turn fatty acids into carbohydrates through the glyoxylate cycle, which gives the seeds energy and building materials for growth.
- Plants’ photorespiration: peroxisomes help with photorespiration by breaking down phosphoglycolate, which is a byproduct of photosynthesis, into useful metabolites and recycling carbon.
- The oxidation of amino acids and polyamines: Peroxisomes oxidise some amino acids and polyamines, which helps with amino acid metabolism and keeping cellular polyamine levels in check.
- The liver makes bile acids from cholesterol with the help of peroxisomes. Bile acids are important for breaking down and absorbing fats in food.
- Peroxisomes do α-oxidation of phytanic acid, which is a branched-chain fatty acid, to make pristanic acid. This is an important step in the metabolism of some fatty acids.
- Biosynthesis of ether lipids- Peroxisomes help make ether lipids, which are important parts of cell membranes and help with cell signalling and keeping membranes stable.
What is the difference between lysosomes and peroxisomes?
Function –
- lysosomes degrade cellular waste, pathogens, damaged organelles via acid hydrolases in autophagy, phagocytosis, endocytosis
- peroxisomes oxidize fatty acids, detoxify reactive oxygen species notably H₂O₂ via catalase, metabolize very‑long‑chain fatty acids, biosynthesize plasmalogens
Enzyme content & pH –
- lysosomes contain >60 hydrolytic enzymes (proteases, lipases, nucleases) active at acidic pH (~4.5–5)
- peroxisomes have oxidative enzymes (oxidases, catalase) functioning at neutral pH, produce then breakdown H₂O₂
Origin & targeting –
- lysosomal enzymes are made in RER, modified in Golgi, tagged with mannose‑6‑phosphate, part of endomembrane system
- peroxisomal proteins are synthesized on free ribosomes, have PTS1/PTS2 targeting signals, import into peroxisomes independently of Golgi
Membrane & system placement –
- both single-membrane vesicles ~0.1–1.5 µm
- lysosomes are part of endomembrane network, peroxisomes are not, instead derived from ER and grow by division
Distribution & presence –
- lysosomes present mainly in animal cells (hundreds per cell), absent in red blood cells
- peroxisomes are found in virtually all eukaryotic cells, variable number based on cell type
Pathways & outcomes –
- lysosomal breakdown yields monomers (amino acids, sugars, fatty acids) that are recycled or used for energy
- peroxisomal oxidation produces medium‑chain fatty acids and detoxified products; supports energy metabolism and lipid synthesis
| Feature | Lysosome | Peroxisome |
|---|---|---|
| Function | Degrades cellular waste, pathogens, and damaged organelles via acid hydrolases in autophagy, phagocytosis, and endocytosis. | Oxidizes fatty acids, detoxifies reactive oxygen species (notably H₂O₂) via catalase, metabolizes very-long-chain fatty acids, and biosynthesizes plasmalogens. |
| Enzyme Content & pH | Contains >60 hydrolytic enzymes (proteases, lipases, nucleases) active at acidic pH (~4.5–5). | Contains oxidative enzymes (oxidases, catalase) functioning at neutral pH; produces then breaks down H₂O₂. |
| Origin & Targeting | Lysosomal enzymes are synthesized in the rough endoplasmic reticulum (RER), modified in the Golgi apparatus, tagged with mannose-6-phosphate, and part of the endomembrane system. | Peroxisomal proteins are synthesized on free ribosomes, have peroxisomal targeting signals (PTS1/PTS2), and import into peroxisomes independently of the Golgi apparatus. |
| Membrane & System Placement | Single-membrane vesicles ~0.1–1.5 µm; part of the endomembrane network. | Single-membrane vesicles ~0.1–1.5 µm; not part of the endomembrane system, instead derived from the endoplasmic reticulum (ER) and grow by division. |
| Distribution & Presence | Present mainly in animal cells (hundreds per cell); absent in red blood cells. | Found in virtually all eukaryotic cells; variable number based on cell type. |
| Pathways & Outcomes | Lysosomal breakdown yields monomers (amino acids, sugars, fatty acids) that are recycled or used for energy. | Peroxisomal oxidation produces medium-chain fatty acids and detoxified products; supports energy metabolism and lipid synthesis. |
Practice Quiz
What is the primary function of peroxisomes in cells?
a) Protein synthesis
b) DNA replication
c) Breakdown of fatty acids
d) Cell division
[expand title=”Show answer” swaptitle=”Hide answer”]
Answer: c) Breakdown of fatty acids
[/expand]
Which enzyme is responsible for breaking down hydrogen peroxide in peroxisomes?
a) Catalase
b) Peroxidase
c) Lipase
d) Amylase
[expand title=”Show answer” swaptitle=”Hide answer”]
Answer: a) Catalase
[/expand]
Which of the following is NOT synthesized in peroxisomes?
a) Bile acids
b) Plasmalogens
c) Steroid hormones
d) Phospholipids
[expand title=”Show answer” swaptitle=”Hide answer”]
Answer: c) Steroid hormones
[/expand]
Peroxisomes are primarily involved in the metabolism of:
a) Glucose
b) Amino acids
c) Fatty acids
d) Nucleic acids
[expand title=”Show answer” swaptitle=”Hide answer”]
Answer: c) Fatty acids
[/expand]
Which organelle is closely related to peroxisomes in terms of origin?
a) Mitochondria
b) Endoplasmic reticulum
c) Golgi apparatus
d) Lysosomes
[expand title=”Show answer” swaptitle=”Hide answer”]
Answer: b) Endoplasmic reticulum
[/expand]
Peroxisomes are most abundant in which type of cells?
a) Muscle cells
b) Nerve cells
c) Liver cells
d) Red blood cells
[expand title=”Show answer” swaptitle=”Hide answer”]
Answer: c) Liver cells
[/expand]
Which of the following diseases is associated with a defect in peroxisome function?
a) Alzheimer’s disease
b) Zellweger syndrome
c) Parkinson’s disease
d) Cystic fibrosis
[expand title=”Show answer” swaptitle=”Hide answer”]
Answer: b) Zellweger syndrome
[/expand]
Which enzyme is responsible for the initial steps in the synthesis of plasmalogens in peroxisomes?
a) Acyl-CoA oxidase
b) Dihydroxyacetone phosphate acyltransferase
c) Catalase
d) Peroxidase
[expand title=”Show answer” swaptitle=”Hide answer”]
Answer: b) Dihydroxyacetone phosphate acyltransferase
[/expand]
Peroxisomes are involved in the detoxification of which of the following compounds?
a) Alcohol
b) Glucose
c) Amino acids
d) Nucleotides
[expand title=”Show answer” swaptitle=”Hide answer”]
Answer: a) Alcohol
[/expand]
Which of the following is a unique feature of peroxisomes compared to other cellular organelles?
a) They contain DNA
b) They can self-replicate
c) They lack a membrane
d) They are involved in lipid synthesis
[expand title=”Show answer” swaptitle=”Hide answer”]
Answer: b) They can self-replicate
[/expand]
FAQ
What are peroxisomes?
Peroxisomes are small, membrane-bound organelles found in the cytoplasm of most eukaryotic cells. They play a crucial role in lipid metabolism and the detoxification of harmful substances.
How are peroxisomes formed?
Peroxisomes can form by budding off from the endoplasmic reticulum (ER) or through the division of pre-existing peroxisomes.
What is the primary function of peroxisomes?
The primary function of peroxisomes is to break down long-chain fatty acids and produce hydrogen peroxide, which is then converted into water by the enzyme catalase.
Why are peroxisomes important for the liver?
Peroxisomes play a vital role in detoxifying various toxic substances, including alcohol. The liver, being the primary detoxifying organ, has a high concentration of peroxisomes.
Do peroxisomes contain DNA?
Unlike mitochondria and chloroplasts, peroxisomes do not contain DNA. They rely on the nucleus for the synthesis of their proteins.
How do peroxisomes protect cells from oxidative damage?
Peroxisomes contain the enzyme catalase, which breaks down hydrogen peroxide (a harmful byproduct of various metabolic processes) into water and oxygen, thus preventing oxidative damage to the cell.
What is the difference between lysosomes and peroxisomes?
While both are membrane-bound organelles involved in breaking down substances, lysosomes digest cellular waste using enzymes in an acidic environment, whereas peroxisomes focus on lipid metabolism and detoxification processes.
Are peroxisomes found in all cells?
Peroxisomes are found in most eukaryotic cells but are especially abundant in the liver and kidney cells due to their detoxifying roles.
What diseases are associated with peroxisomal dysfunction?
Disorders related to peroxisomal dysfunction include Zellweger syndrome, Adrenoleukodystrophy, and Refsum disease, among others.
Can peroxisomes be seen under a light microscope?
Due to their small size, peroxisomes are typically not visible under a light microscope but can be observed using an electron microscope.
- Van der Klei, I. J. (2014). Peroxisomes. Pathobiology of Human Disease, 108–113. doi:10.1016/b978-0-12-386456-7.01407-6
- Joshi, S., & Subramani, S. (2013). Peroxisomes. Encyclopedia of Biological Chemistry, 425–430. doi:10.1016/b978-0-12-378630-2.00503-x
- Stott, W. T. (1993). Peroxisomes. Handbook of Hazardous Materials, 545–561. doi:10.1016/b978-0-12-189410-8.50051-1
- Gould, S. J. (2013). Peroxisomal Metabolism. Encyclopedia of Biological Chemistry, 413–417. doi:10.1016/b978-0-12-378630-2.00104-3
- Cooper GM. The Cell: A Molecular Approach. 2nd edition. Sunderland (MA): Sinauer Associates; 2000. Peroxisomes. Available from: https://www.ncbi.nlm.nih.gov/books/NBK9930/
- Eckert, J. H., & Erdmann, R. (n.d.). Peroxisome biogenesis. Reviews of Physiology, Biochemistry and Pharmacology, 75–121. doi:10.1007/s10254-003-0007-z