Transmission Electron Microscope (TEM) is an advanced analytical instrument used to visualize the internal structure of specimens. It uses highly energetic beam of electrons instead of light.
It works under high vacuum condition. The electron gun produces electron beam. This beam is then focused and guided by a series of electromagnetic lenses.
The specimen used in TEM must be very thin. It is generally less than 100 nanometer thick. This is because the electrons have to transmit or pass directly through the specimen for formation of image.
When the electrons pass through the sample, some electrons pass through it clearly. Some electrons are scattered or blocked by the dense region of the material. Due to this difference a shadow like contrast is produced.
The transmitted electrons are magnified and projected on fluorescent viewing screen or digital detector. Thus a highly detailed 2D image of the internal part of the specimen is formed.
TEM has very high magnification and resolving power. This is because electrons have much shorter wavelength than visible light. It can magnify the specimen more than 50 million times and can resolve structures smaller than single atom, less than 50 picometers.
Transmission Electron Microscope is used to study crystal structures, cellular machinery, viruses and individual columns of atoms. It is an important instrument in nanotechnology, material science and biology.
Working principle of Transmission Electron Microscope
Transmission Electron Microscope is based on the transmission of high energy electron beam through a very thin specimen. It uses electrons instead of visible light to illuminate the sample.
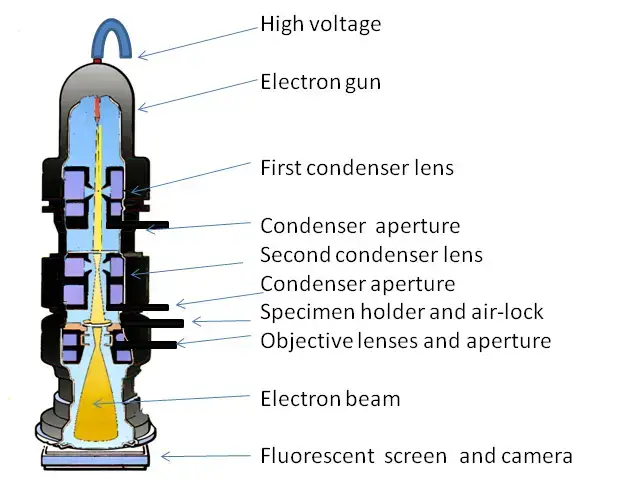
The process starts from the top of the microscope column. In this part, electron gun produces a beam of electrons. These electrons are accelerated with the help of high voltage power source.
The whole system is kept in high vacuum condition. This is because electrons are easily scattered when they collide with air molecules. So air is removed from the column for proper movement of electrons.
The electron beam then passes down through the column. It is guided and focused by a series of electromagnetic lenses. The condenser lens first focuses the beam directly on the specimen.
The specimen used in TEM must be extremely thin. It is generally less than 100 nanometer thick. This makes the specimen electron transparent and allows electrons to pass through it.
When the beam enters the specimen, the electrons interact with the atoms of the specimen. Some electrons are scattered or absorbed by thicker or denser parts. Some electrons pass straight through the thin parts of the specimen.
The transmitted electrons are collected by the objective lens. This lens forms the first image of the specimen. Then the image is further magnified by intermediate lens and projector lens.
Finally, the magnified electron image falls on a detector. It may be a fluorescent viewing screen or a digital camera. Thus a highly detailed 2D image of the internal structure of the specimen is formed.
In this image, the areas where electrons are blocked or scattered appear darker. The areas where more electrons pass through appear brighter. This difference produces contrast in the TEM image.
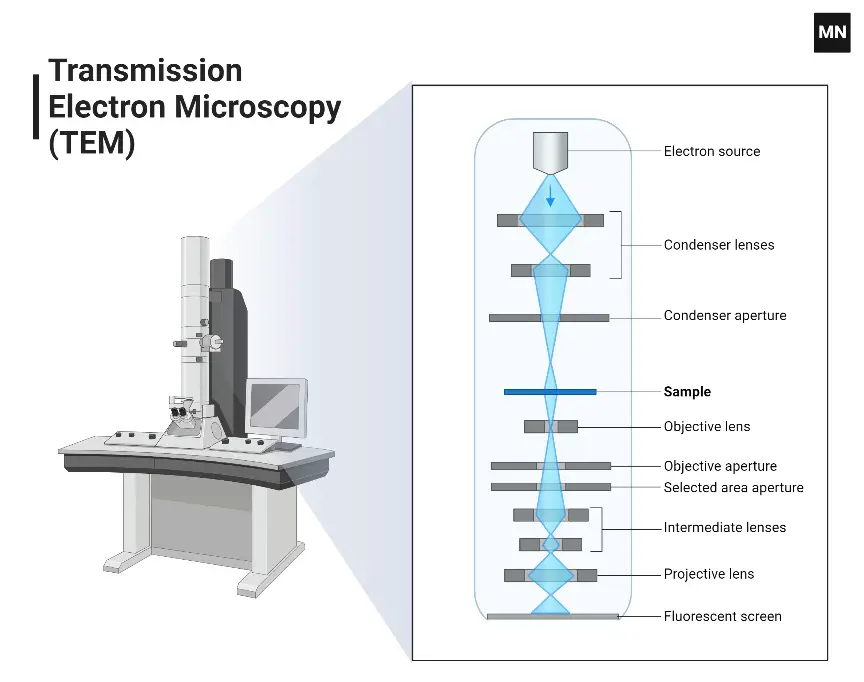
Parts of A Transmission Electron Microscope
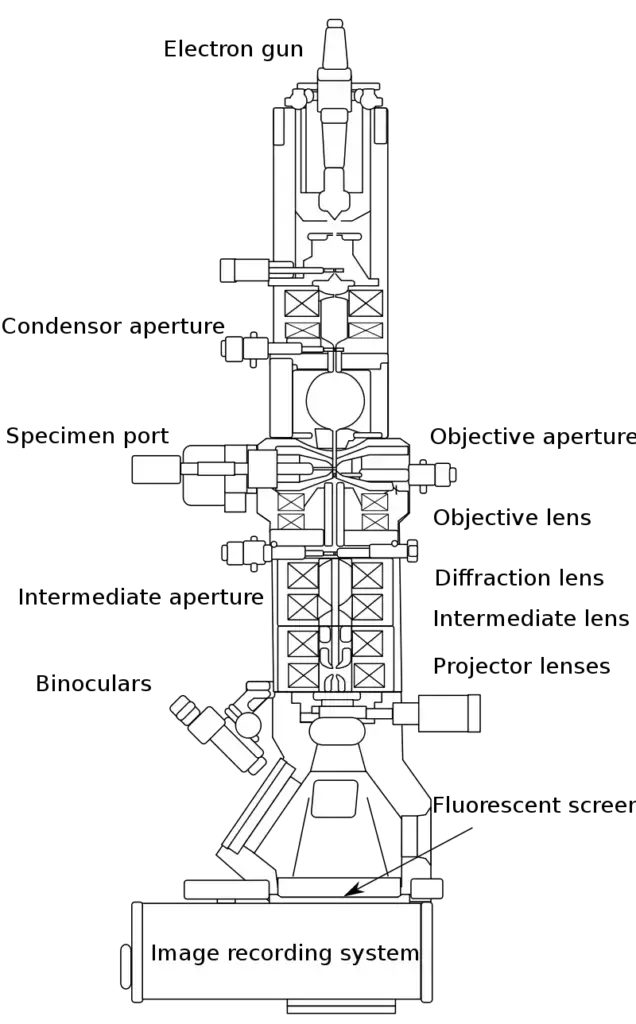
The following are the important parts of Transmission Electron Microscope-
- High voltage source- It provides electric field potential to accelerate the electron beam through the microscope column.
- Vacuum system– It removes air from the column and maintains high or ultra-high vacuum. It prevents the electrons from colliding with gas molecules and also protects the electron gun.
- Electron gun- It is present at the top of the column and produces a stable and bright beam of electrons. It may be tungsten filament, LaB6 crystal or field emission gun.
- Wehnelt cylinder and extraction anode- These are associated with electron gun and helps in focusing and controlling the emitted electrons.
- Electromagnetic lenses– These are copper coils around soft iron pole pieces which produce magnetic fields to focus and guide the electron beam.
- Condenser lenses– These are located below the electron gun. They control the illumination area and convergence angle of the electron beam on the specimen.
- Objective lens- It is the most important lens of TEM. It forms the first intermediate image and primary diffraction pattern.
- Intermediate and projector lenses– These lenses magnify the image or diffraction pattern and project it on the final screen or camera.
- Apertures– These are thin metallic plates with circular holes. They block off-axis or scattered electrons and helps in contrast control.
- Condenser aperture– It controls the size and quality of the electron beam reaching the specimen.
- Objective aperture– It blocks scattered electrons after the specimen and improves image contrast.
- Selected area diffraction aperture- It selects a specific area of the specimen for diffraction study.
- Specimen chamber– It is the part where the specimen is inserted into the objective lens gap.
- Stage– It holds the specimen and allows movement, rotation and tilting of the sample.
- Specimen holder- It holds the thin specimen in stable position. It may be side-entry or top-entry holder.
- Detectors and image capture system- These are located at the bottom of the column and record images, diffraction patterns and analytical signals.
- Fluorescent or phosphor screen- It is a retractable viewing screen used for direct visual observation by the operator.
- Digital cameras– These are used to record high resolution digital images. CCD, CMOS and direct electron detectors are used.
- Spectrometers– These are analytical detectors used for elemental and chemical analysis. EDS or EDX and EELS are commonly used.
- Main control panel, computer and software- These are used to control stage, magnification, lens currents, apertures and image acquisition.
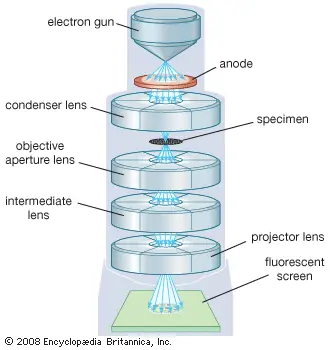
How does the transmission electron microscope work?
The working of Transmission Electron Microscope takes place in the following steps-
- Vacuum generation- The whole microscope column is kept under high vacuum condition. This prevents the electron beam from colliding with air or gas molecules.
- Electron beam generation- The electron gun present at the top of the column produces a bright stream of electrons. These electrons are then accelerated downwards by high voltage power source.
- Beam focusing- The electron beam travels down through the column. Electromagnetic condenser lenses and apertures guide, shape and focus the beam on the specimen.
- Specimen preparation- The specimen used in TEM must be extremely thin. It is generally less than 100 nanometer thick, so that electrons can pass through it.
- Specimen interaction- The focused beam passes or transmits through the specimen. During this process, the electrons interact with atoms of the specimen.
- Scattering of electrons- Thicker or denser regions of the specimen scatter more electrons. Thin regions allow more electrons to pass through it.
- Initial image formation- The electrons that come out from the lower part of the specimen are collected by the objective lens. This lens forms the first magnified image and primary diffraction pattern.
- Further magnification- The intermediate and projector lenses are present below the objective lens. These lenses further magnify the image millions of times.
- Projection of image- The magnified electron image is projected towards the bottom part of the microscope.
- Detection and display- Finally, the projected electrons strike on fluorescent viewing screen or digital detector. CCD or CMOS camera captures the electron signal and converts it into a detailed 2D image.
- Image observation- The final image is viewed on the screen or computer. It shows the internal structure of the specimen with high detail.
Sample Preparation for TEM
The sample preparation for Transmission Electron Microscope is different for biological samples and hard material samples. It is done to make the specimen very thin and electron transparent.
A. Biological and Organic Sample Preparation
- Fixation- The sample is preserved to maintain its natural structure. It also stabilizes the molecules from damage during other steps.
- Primary fixation- Glutaraldehyde is used for primary fixation. It cross-links the proteins and keeps the cellular structure stable.
- Secondary fixation- Osmium tetroxide is used after primary fixation. It preserves lipids and also gives initial contrast to the sample.
- Cryo-fixation- In this method, the sample is rapidly frozen. It immobilizes the sample in non-crystalline ice without chemical changes.
- Dehydration- Water is removed from the sample because TEM works under high vacuum. If water remains, the sample may collapse.
- Ethanol treatment- The tissue is passed through increasing concentration of ethanol. This gradually replaces water from the sample.
- Transitional solvent- Acetone is used after ethanol treatment. It helps to prepare the sample for embedding.
- Embedding- The dehydrated sample is infiltrated with liquid resin. Spurr’s resin or epoxy/acrylic resin is used as supporting matrix.
- Polymerization- The sample is placed in a mold and resin is hardened. Heat or UV light is used for hardening the resin.
- Sectioning- The hardened resin block is cut into very thin sections. The sections must be thinner than 100 nanometer.
- Ultramicrotomy- Ultramicrotome is used for cutting the sections. Glass knife or diamond knife is used in this process.
- Grid collection- The ultrathin sections are carefully collected on a metal grid. This grid is used for observation under TEM.
- Contrasting or staining- Biological samples do not scatter electrons properly because they are made of light elements. So contrast is poor without staining.
- Heavy metal staining- Uranyl acetate and lead citrate are commonly used. These heavy metal salts bind with cellular structures and increase electron scattering.
- Image contrast- Due to heavy metal staining, a clear image is formed. The cellular parts become more visible under TEM.
B. Inorganic and Materials Sample Preparation
- Mechanical thinning- The hard sample like metal, ceramic or semiconductor is first ground and polished. It is made about 20 to 50 micrometer thick.
- Dimpling- In this step, a central depression is made in the sample. It helps in thinning the central part of the specimen.
- Final thinning- The mechanically thinned sample is further made very thin. The final thickness should be less than 100 nanometer.
- Electropolishing- This method is used for conductive metals. Electric current and chemical electrolyte are used to thin the centre of the sample.
- Hole formation- During electropolishing, a microscopic hole appears in the thinned region. The area near this hole becomes electron transparent.
- Ion beam thinning- Argon ion beam is used in this method. It removes or sputters the material from the sample at a glancing angle.
- Electron transparency- Ion beam thinning is continued until the sample becomes thin enough for electrons to pass through it.
- Focused ion beam milling- A focused beam of gallium or xenon ions is used. It cuts a specific ultra-thin cross section from the bulk sample.
- Lamella preparation- The thin section made by FIB is called lamella. It is taken from the selected region of interest for TEM study.
General Operating Procedure of transmission electron microscope
The general operating procedure of Transmission Electron Microscope is as follows-
- Sample insertion- The specimen grid is first placed into a specimen holder. Then the holder is inserted into the microscope column through airlock system.
- Vacuum pump-down- The airlock is evacuated by internal vacuum pumps. This maintains the high or ultra-high vacuum condition required for movement of electron beam.
- Instrument alignment- The electron beam is aligned so that it can travel straight down the column. This is important for proper formation of image.
- Condenser lens centering- The condenser lens is centered during initial alignment. It helps to bring the electron beam in correct position.
- Condenser stigmation correction- The shape of the beam is corrected by condenser lens stigmation. This makes the beam more proper for illumination.
- Setting the eucentric height- The Z-height of the specimen is adjusted by using goniometer stage. This brings the sample to the eucentric plane.
- Eucentric plane adjustment- At the eucentric plane, the objective lens works at constant and proper current. It also helps to reduce lens aberrations.
- Low magnification observation- The observation is first started at low magnification. Usually 100x to 1000x magnification is used for scanning the grid.
- Location of specimen area- The operator scans the grid and finds the specimen section. Then the required area of interest is selected for further study.
- Aperture selection- Objective aperture is inserted into the beam path. It is a thin metallic plate with holes.
- Aperture alignment- The aperture is aligned to select unscattered or diffracted electron beams. It helps in selecting the imaging mode.
- Imaging mode selection- Bright-field or dark-field imaging can be selected by using aperture. It also improves the contrast of the image.
- High magnification- After selecting the area of interest, the magnification is increased. The image is observed at higher magnification for detailed study.
- Final focusing- The focus is adjusted carefully to make the image sharp. Objective lens is mainly used for this focusing.
- Objective stigmator adjustment- Objective lens stigmators are adjusted to correct astigmatism. This makes the electron probe more circular and gives clear image.
- Image capture- The final magnified image or diffraction pattern is projected on fluorescent viewing screen. It may also be captured digitally.
- Digital recording- CCD, CMOS or direct electron camera is used for recording the image. The captured image is then used for analysis.
Application of Transmission Electron Microscope
The following are the important applications of Transmission Electron Microscope-
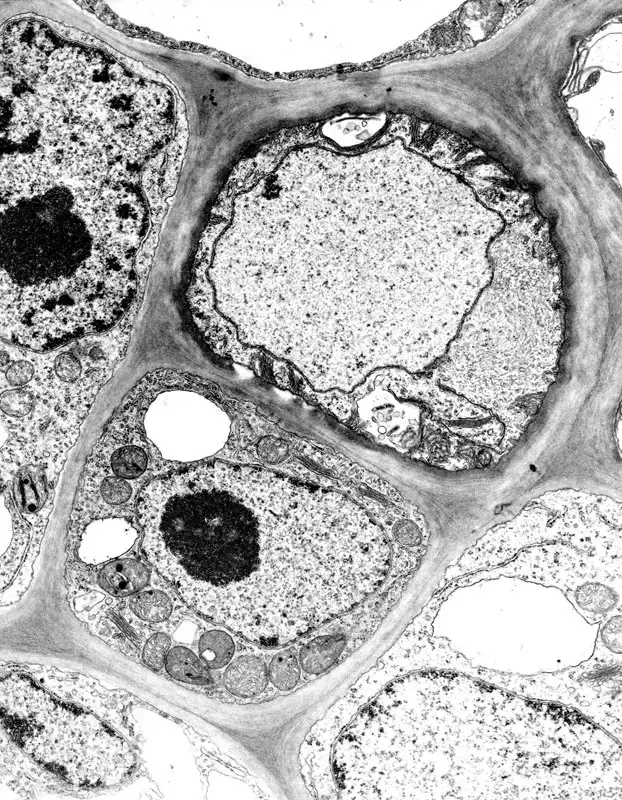
- Life sciences and Biology- It is used to visualize internal structure of biological tissues, cells and sub-cellular organelles.
- Cell structure study- TEM helps in study of proteins and macromolecular assemblies at near atomic resolution.
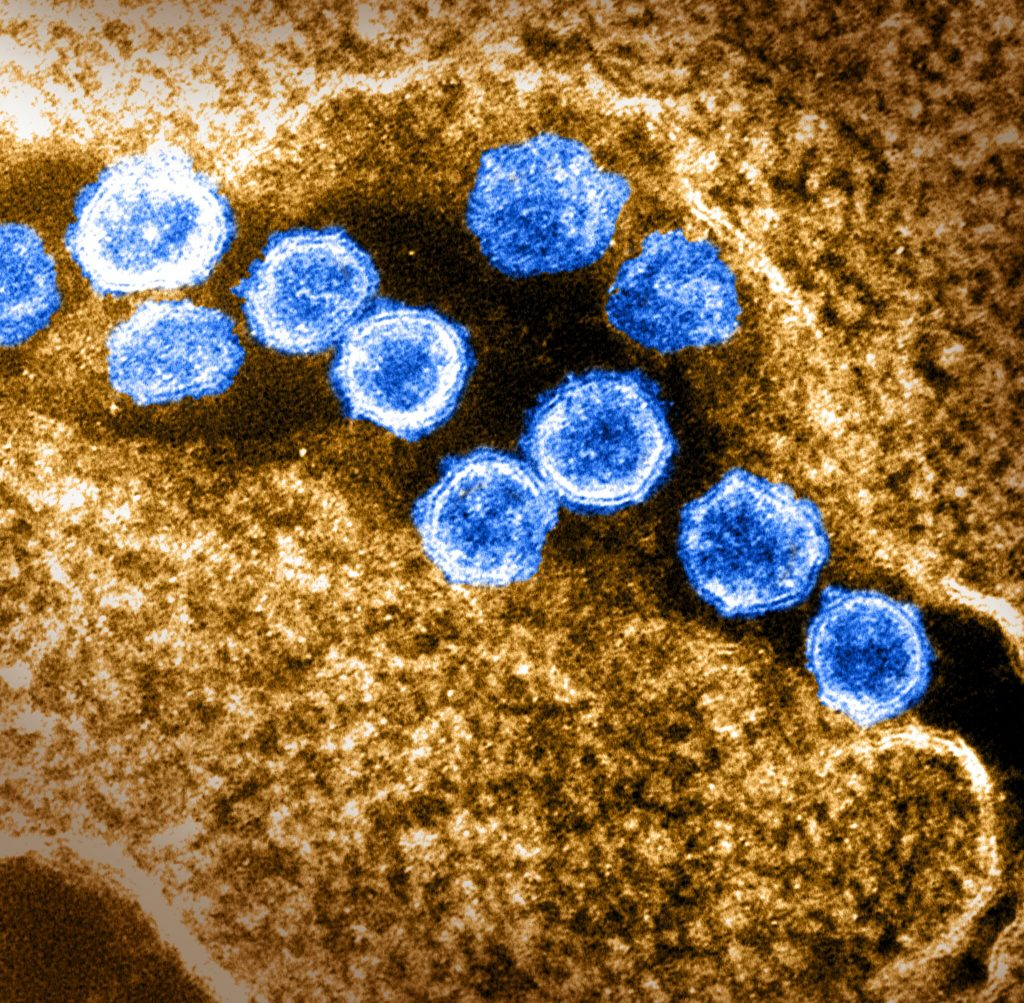
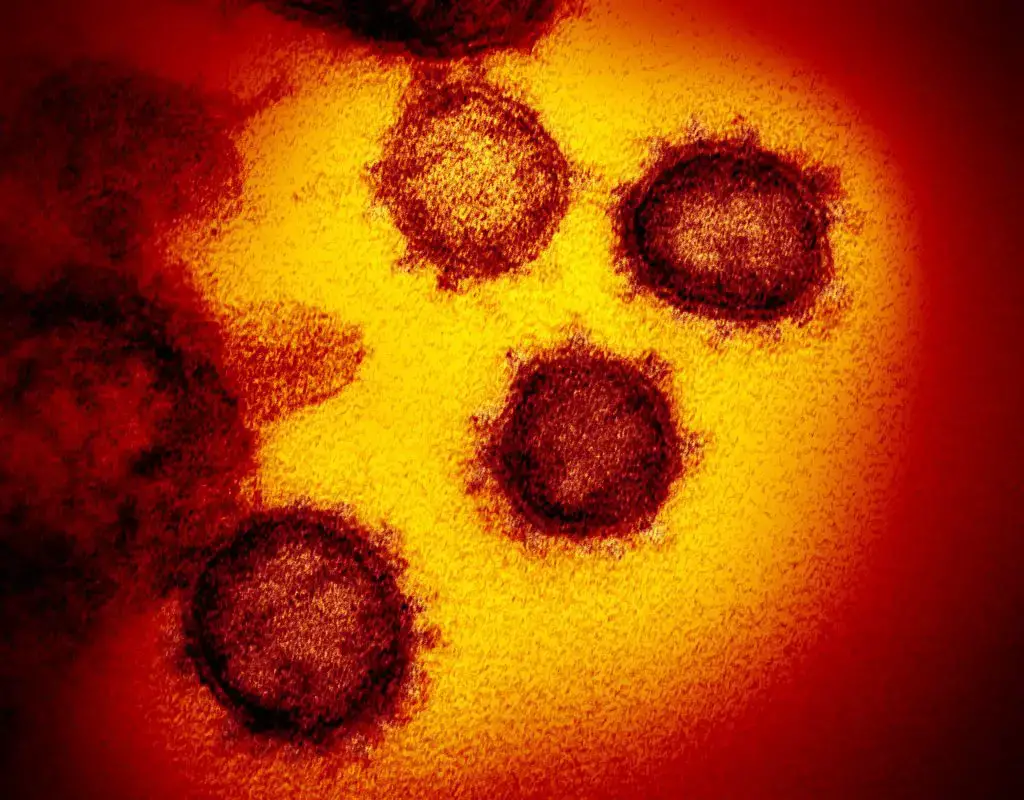
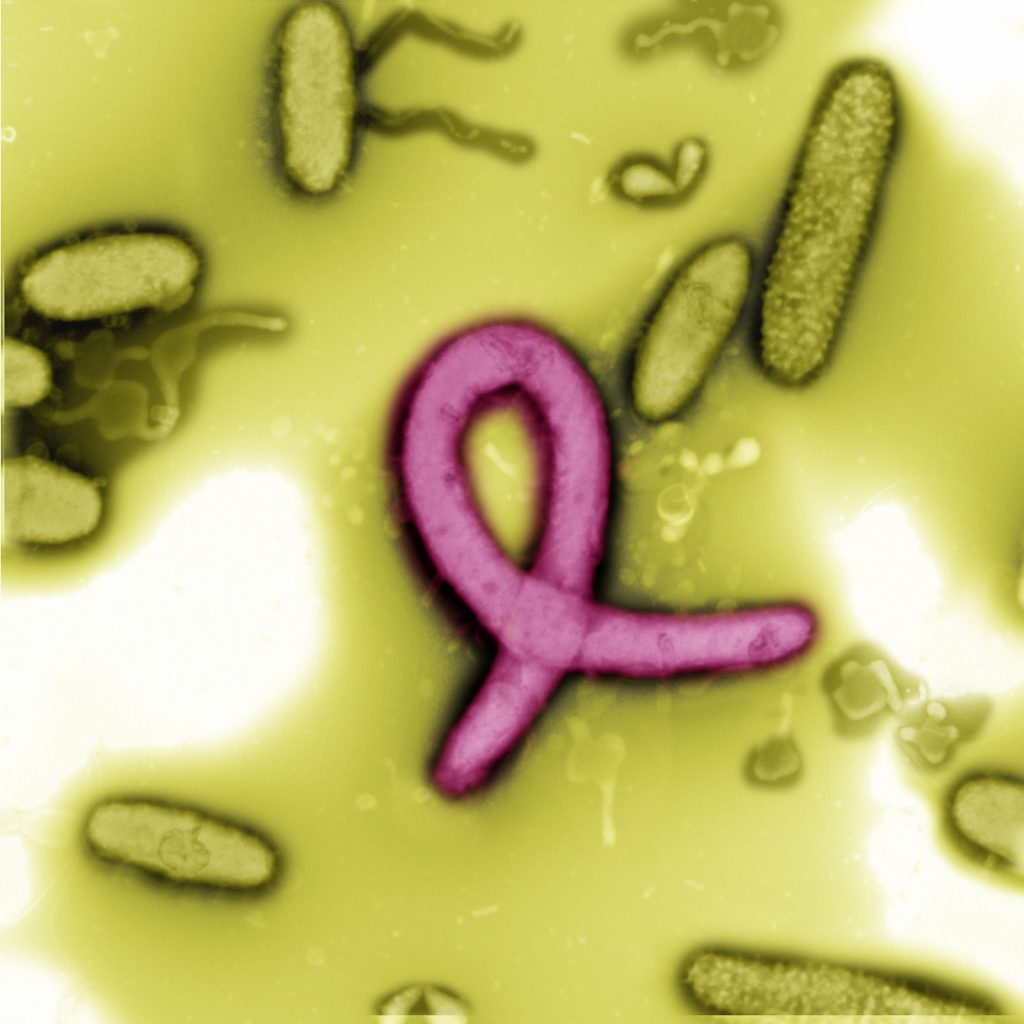
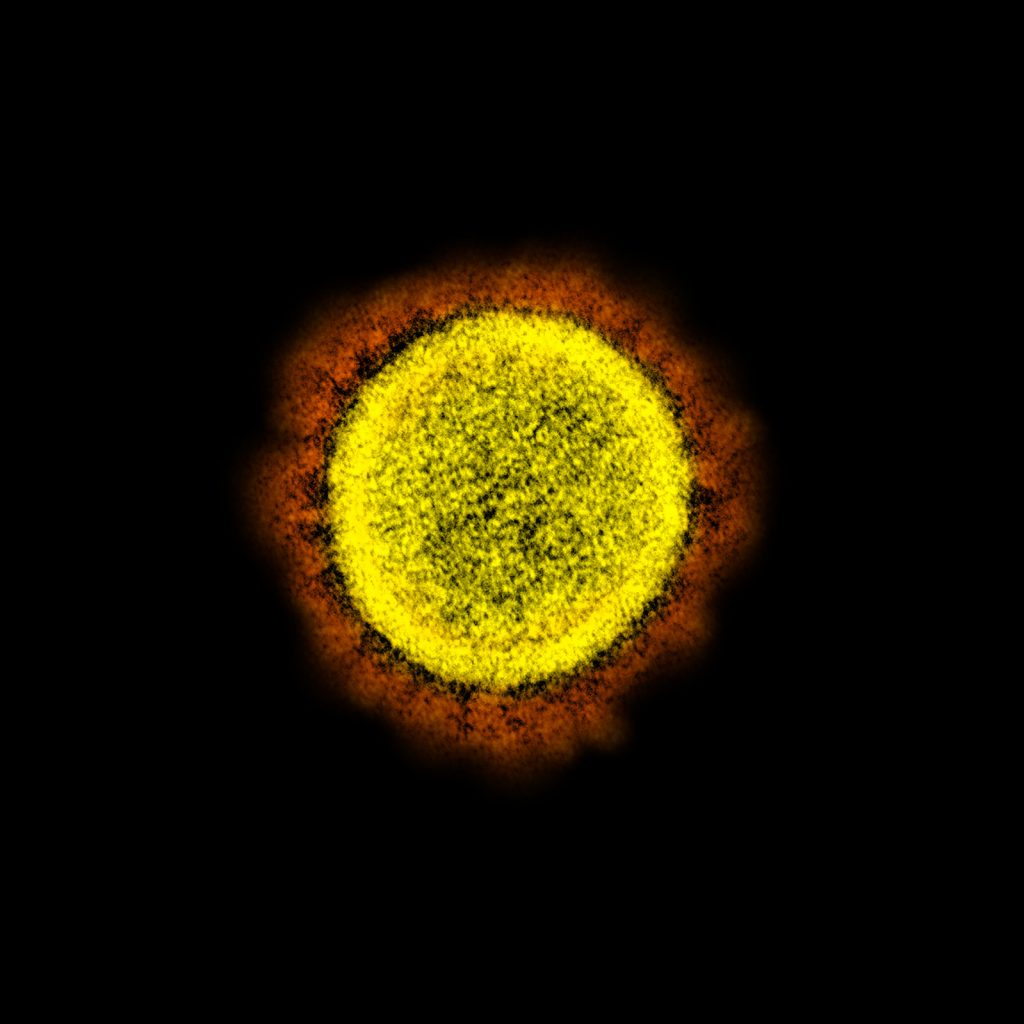
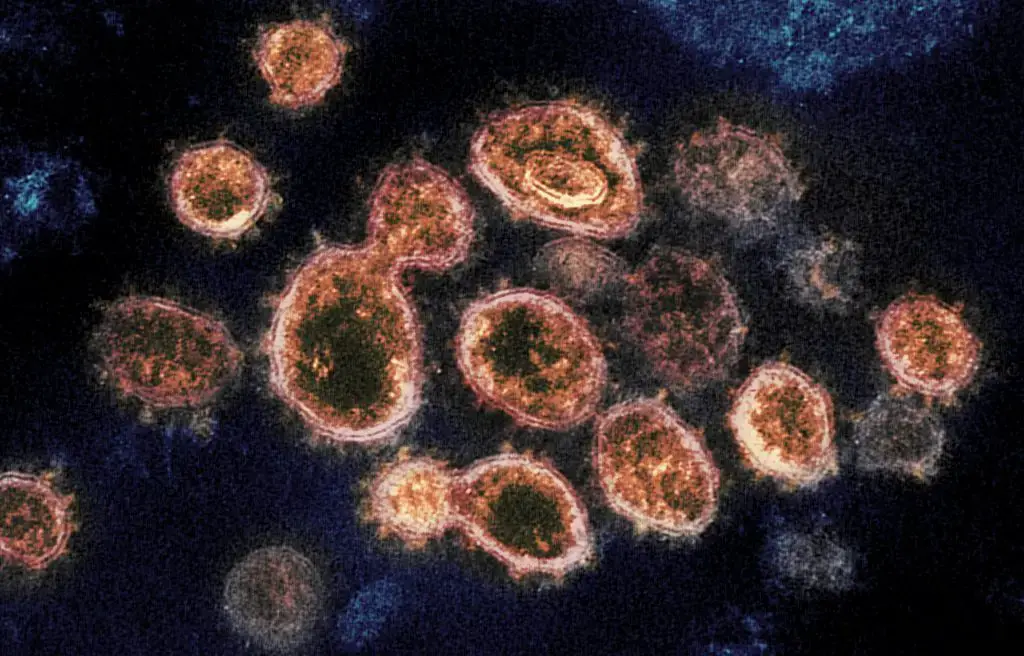
- Virology- It is used for studying the detailed structure of viruses such as SARS-CoV-2.
- Disease research- Cancer cells and diseased cells can be studied by TEM, which helps in medical research.
- Drug and vaccine development- It gives structural information which is useful in drug discovery and vaccine development.
- Materials science- It is used to analyze crystal structure, morphology and stress states of materials.
- Metallurgy- Metals, alloys and ceramics are studied by TEM for finding defects, dislocations and grain boundaries.
- Semiconductor engineering- TEM is used in checking microchips and semiconductor devices.
- Atomic layer analysis- It helps to observe layer growth and thickness of atomic layers.
- Failure analysis- The root causes of electrical failures in semiconductor devices can be identified.
- Nanotechnology- Nanomaterials like quantum dots, nanowires and nanoparticles are examined by TEM.
- Catalysis research- Structural changes in catalytic materials are studied by this microscope.
- Energy research- Battery materials and energy related materials can be observed, sometimes by in-situ method.
- Chemical analysis- TEM with EDX and EELS is used for finding chemical composition of the specimen.
- Elemental analysis- Electronic properties and bonding states of internal structure can also be determined.
- Environmental science- Water samples and environmental specimens are examined for nanoscale contaminants.
- Pollution study- It is used to identify and characterize very small pollutants.
- Paleontology- Ancient micro-fossils can be studied at very high resolution.
- Palynology- Spores and pollen are observed in detailed structure by using TEM.
Advantages of Transmission Electron Microscope (TEM)
The following are the important advantages of Transmission Electron Microscope-
- High magnification- TEM gives very high magnification. It can magnify the specimen more than 50 million times.
- High resolution- It has very high resolving power due to shorter wavelength of electrons.
- Sub-nanometer observation- TEM can show very small structures at sub-nanometer level. It can resolve about 0.1 nanometer or even less than 50 picometers.
- Atomic level study- Individual columns of atoms can be visualized by TEM.
- Internal structure study- The electron beam passes through the specimen. So internal architecture of the specimen can be observed.
- Cellular details- It is useful for studying internal morphology, cellular compartments and intracellular structures.
- 2D image formation- TEM gives a detailed 2D projection image of the internal part of specimen.
- Crystal structure analysis- TEM is used to identify crystal structures and symmetry of materials.
- Diffraction study- Electron diffraction patterns can be collected from very small nanometer-sized regions.
- Defect observation- Structural defects like dislocations and grain boundaries can be detected by TEM.
- Chemical analysis- TEM with EDS or EDX helps to know the elemental composition of the specimen.
- Electronic analysis- EELS with TEM can show oxidation states, chemical bonding and electronic properties at nanoscale level.
- Different imaging modes- TEM can be used in bright-field, dark-field, phase contrast and STEM modes.
- Better contrast study- Dark-field imaging helps in compositional and crystallographic contrast.
- High resolution TEM- Phase contrast or HRTEM helps to observe fine details of atomic structure.
- 3D image study- TEM can form three dimensional structure by electron tomography.
- Tilt series imaging- Images are taken from different angles and then reconstructed to form 3D representation.
- Wide use- TEM is useful in biology, material science, nanotechnology and chemical analysis.
Limitations of Transmission Electron Microscope (TEM)
The following are the important limitations of Transmission Electron Microscope-
- Complex sample preparation- The sample must be made very thin, generally less than 100 nanometer. This makes the preparation process difficult and time consuming.
- Artificial changes- During preparation, the sample structure may be changed. Sometimes artificial defects may also be produced.
- Difficult image interpretation- TEM image is a 2D projection of a 3D structure. So actual three dimensional shape is difficult to understand.
- Small field of view- TEM shows only a very small area at a time. In high resolution image, the area may be about 100 nm².
- Poor representation of whole sample- As only a small region is observed, it may not represent the whole bulk sample properly.
- Electron beam damage- The high energy electron beam can damage sensitive specimens.
- Damage of soft samples- Biological samples, polymers and soft materials may degrade, melt or destroy under electron beam.
- High vacuum requirement- TEM needs high vacuum condition for proper movement of electron beam.
- Living sample cannot be observed- Due to high vacuum, living biological specimens cannot be studied in their natural condition.
- Natural state problem- Some materials cannot be observed in their normal ambient state.
- High cost- TEM is very costly instrument. Its operation and maintenance cost is also high.
- Large size- TEM instruments are very large. They may require a full room for installation.
- More costly than SEM- TEM is generally more expensive than scanning electron microscope.
- Skilled operator needed- Operation of TEM is complex and needs trained person.
- Alignment difficulty- The user must know proper optical alignment of electron beam and lenses.
- Artifact problem- Wrong sample preparation may introduce artifacts in the image.
- Result interpretation problem- Accurate interpretation of TEM result needs experience and training.
Differences between a SEM and a TEM
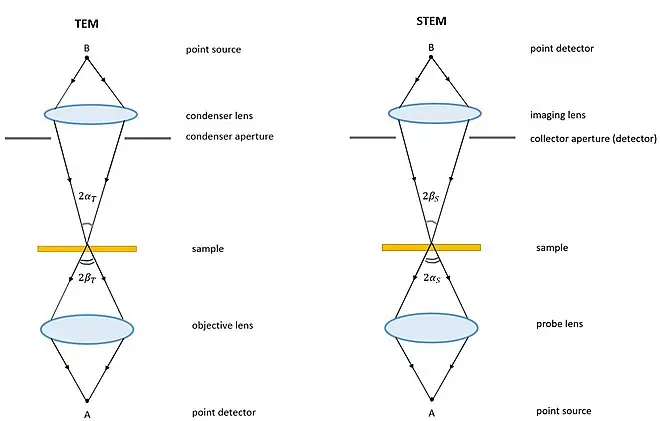
The following are the important differences between SEM and TEM-
- Electron interaction- SEM uses scattered electrons or secondary electrons for image formation. TEM uses electrons which pass directly through the sample.
- Image type- SEM gives 3D like image of the surface of the specimen. TEM gives 2D projection image of the internal structure.
- Surface and internal view- SEM is mainly used for surface topography and composition. TEM is used for internal structure of the specimen.
- Resolution- TEM has higher resolution than SEM. TEM can resolve less than 50 picometers, while SEM resolution is about 0.5 to 2 nanometer.
- Magnification- TEM gives higher magnification. It can magnify more than 50 million times, but SEM generally magnifies upto 1 to 2 million times.
- Accelerating voltage- TEM works at higher voltage, generally 60 to 300 kV. SEM works at lower voltage, generally 1 to 30 kV.
- Sample thickness- SEM can observe bulk samples of any thickness which can fit in the chamber. TEM needs very thin sample, generally less than 100 to 150 nanometer.
- Sample preparation- SEM sample preparation is comparatively simple and fast. TEM sample preparation is complex, tedious and laborious.
- User requirement- TEM needs specially trained users for sample preparation and operation. SEM is comparatively easier to use.
- Field of view- SEM has larger field of view. TEM shows very small area of the specimen at one time.
- Depth of field- SEM has higher depth of field. TEM has comparatively low depth of field.
- Spectroscopy- Both SEM and TEM can be used with EDX. But EELS is mainly compatible with TEM.
- Cost- SEM is generally less costly than TEM. TEM is more expensive to buy, operate and maintain.
- Size- SEM may be smaller and even desktop models are available. TEM is large and usually requires a full room.
- Speed- SEM gives faster time-to-image. TEM is slower because of complex preparation and operation.
Differences between SEM and TEM Table
| Basis | SEM | TEM |
|---|---|---|
| Full form | SEM means Scanning Electron Microscope. | TEM means Transmission Electron Microscope. |
| Electron interaction | It uses scattered electrons or secondary electrons for image formation. | It uses electrons which transmit directly through the sample. |
| Image type | It gives 3D like image of the specimen surface. | It gives 2D projection image of internal structure. |
| Main observation | Surface topography and composition are observed. | Internal structure of the specimen is observed. |
| Resolution | Resolution is about 0.5 to 2 nanometer. | Resolution is very high, less than 50 picometers. |
| Magnification | It magnifies about 1 to 2 million times. | It can magnify more than 50 million times. |
| Accelerating voltage | It works at low voltage, generally 1 to 30 kV. | It works at high voltage, generally 60 to 300 kV. |
| Sample thickness | Bulk samples can be observed if it fits in the chamber. | Very thin sample is needed, generally less than 100 to 150 nanometer. |
| Sample preparation | Sample preparation is simple and fast. | Sample preparation is complex, tedious and laborious. |
| Field of view | It has large field of view. | It shows very small area at one time. |
| Depth of field | Depth of field is high. | Depth of field is comparatively low. |
| Spectroscopy | It can be used with EDX. | It can be used with EDX and EELS. |
| Cost | It is comparatively less costly. | It is more costly to buy, operate and maintain. |
| Size | It may be small and desktop models are also available. | It is large and usually needs full room. |
| Speed | Time-to-image is faster. | It is slower due to complex preparation and operation. |
Transmission Electron Microscope Definition, Parts, Working Principle, Applications, Advantages – Video
Transmission electron microscope images
References
- A Method for Obtaining Serial Ultrathin Sections of Microorganisms in Transmission Electron Microscopy. (n.d.). PubMed Central (PMC).
- Aberration-corrected transmission electron microscopy. (2026, March 21). In Wikipedia.
- Artifacts – TEM. (n.d.). MyScope, Microscopy Australia.
- Atomic Resolution: NCSU’s Aberration Corrected S/TEM. (2013, January 10). Analytical Instrumentation Facility (AIF), NC State University.
- Dumančić, E., Vojta, L., & Fulgosi, H. (2023). Beginners guide to sample preparation techniques for transmission electron microscopy. Periodicum Biologorum, 125(1–2), 123–131.
- CCD vs CMOS: A Review of Sensor Technology. (n.d.). CMOS Sensor Inc.
- Hata, D. M., Brewer, E. V., & Louwagie, N. J. (n.d.). Chapter 4: Rough Vacuum Regime. In Introduction to Vacuum Technology. Milne Publishing.
- CMOS vs. CCD Detectors: What’s Best for Your Spectroscopy Setup? (2025, October 26). Evolve Sensing.
- Charging Contamination. (n.d.). Hitachi High Technologies America, Inc.
- Comprehensive Analysis of the Transmission Electron Microscope: Principles, Architecture, and Methodologies. (n.d.).
- Liao, Y. (2006). Dark-field (BF) Imaging in TEM. Practical Electron Microscopy and Database.
- Detectors for transmission electron microscopy. (2025, December 12). In Wikipedia.
- Electron Microscopy | TEM vs SEM. (n.d.). Thermo Fisher Scientific.
- Electron detectors – CRYO. (n.d.). MyScope, Microscopy Australia.
- Electron diffraction and dark-field TEM for structural analysis of 2D van der Waals materials. (n.d.). PubMed Central (PMC).
- How to Cut Thin Sections Using Ultramicrotomy. (n.d.). CN Tech.
- Image artefacts and trouble-shooting – SEM. (n.d.). MyScope, Microscopy Australia.
- Introducing TEM components – TEM. (n.d.). MyScope, Microscopy Australia.
- Introduction to TEM – TEM. (n.d.). MyScope, Microscopy Australia.
- Investigating the TEM Microscope: Principles, Advancements, and Applications. (n.d.). JEOL USA Blog.
- Mitigation of artifacts in imaging biosamples with optical scanning transmission electron microscopy. (n.d.). PubMed Central (PMC).
- University of Illinois Grainger College of Engineering. (2024, February 28). Reimagining electron microscopy: Bringing high-end resolution to lower-cost microscopes. ScienceDaily.
- Marya. (2026, April 18). SEM Artifacts: Understanding Common Issues. Element-Pi.
- Güneş, H. (2024, October 23). SEM vs TEM: How to choose the most suitable electron microscopy technique for your application. Measurlabs.
- SEM vs TEM: Microscopy Compared. (n.d.). Technology Networks.
- Specimen Preparation – Electron Microscopy. (n.d.). School of the Biological Sciences, University of Cambridge.
- Davey, R. (2021, February 3). TEM Imaging Modes. AZoLifeSciences.
- TEM Sample preparation. (n.d.). Office of Research and Partnerships (ORP) Core Facilities, The University of Arizona.
- Gaston, B., & Le, H. (2022, August 21). TEM: Bright field versus dark field. Chemistry LibreTexts.
- Gaston, B., & Le, H. (2022, August 21). TEM: Bright field versus dark field. Chemistry LibreTexts.
- Transmission Electron Microscopy. (n.d.). Nanoscience Instruments.
- Transmission electron microscopy. (2026, March 26). In Wikipedia.
- Liao, Y. (2006). Turbomolecular Pumps. Practical Electron Microscopy and Database.
- Types of pumps – SEM. (n.d.). MyScope, Microscopy Australia.
- Moon, G. (2024, December 25). Understanding Electron Diffraction in TEM. Deep Block.
- What is TEM/STEM Standard and Aberration Corrected? (n.d.). ASU Core Research Facilities, Arizona State University.
- What’s the Difference Between SEM & TEM? (n.d.). Nanoscience Instruments.
- Where the tiny becomes mighty: light vs electron microscopy. (n.d.). Abberior.
- Working with ion getter pumps: everything you need to know. (2019, July 5). Edwards Vacuum.
- Direct electron detector | Glossary. (n.d.). JEOL Ltd.