Giemsa stain is a polychromatic nucleic acid stain that is used mainly for demonstrating different blood cells and parasites in stained blood films. It is named after the German chemist Gustav Giemsa. It belongs to a group of stains known as Romanowsky stains.
These are neutral stains made from a mixture of oxidized methylene blue, azure, and eosin Y. It is performed on an air-dried smear which is then fixed with methanol. It is the process that is applied widely in hematology and parasitology where the cells of blood and bone marrow are differentiated, and parasites like Plasmodium are demonstrated on the stained smear.
Giemsa stain is prepared as a combination of the basic dye methylene blue together with the acid dyes eosin and azure B. This mixture binds with phosphate groups of DNA, especially in the regions rich in adenine–thymine. The acidic nuclear components get stained in dark shades while the cytoplasmic parts appear in paler tones.
In stained blood smears, erythrocytes appear pink in colour, platelets appear light pink, lymphocyte cytoplasm gets sky-blue appearance and monocyte cytoplasm appears pale blue, while the chromatin of leukocytes becomes magenta. This is referred to as the characteristic Romanowsky effect which helps in identifying cells and parasites clearly on the slide.
Objectives of Giemsa stain
The main purpose of this staining method is to stain the nuclear material and study the detailed morphology of the nucleus.
Principle of Giemsa stain
The principle of Giemsa stain is based on differential staining, and it is the process that depends on the interaction of both acidic and basic dyes present in the stain mixture. It belongs to the Romanowsky group of neutral stains where methylene blue, azure B, and eosin Y together form the coloured reaction. In this method the basic dyes carry positive charge and these are attracted toward the acidic components of the cell, mainly the phosphate groups of DNA and RNA. These nuclear materials are strongly acidic and therefore the nucleus becomes stained in deep blue or purple shades. The acidic dye eosin carries negative charge and it is attracted toward the basic cytoplasmic proteins which then appear in pink to red colour on the stained smear.
It is the process in which the combination of acidic and basic dyes produces a wide range of colour tones known as the Romanowsky effect. The nucleus shows a characteristic purple appearance because the DNA acts as a polyanionic material that helps in the binding of the basic dyes in very close position and this produces a changed colour tone on the smear.
The stain requires buffering at around pH 6.8–7.2 because the ionic conditions must be controlled for proper binding of the dyes to the cell structures. When the correct pH is maintained the dyes get ionized properly and the staining becomes clear and reproducible. This principle allows Giemsa stain to differentiate blood cells and to demonstrate parasites like malaria on the prepared slides.
Requirement for Giemsa staining
- General Requirements
- The stain should contain the mixture of Eosin Y, Methylene blue and Azure B which form the Romanowsky-type stain.
- It is diluted in buffered water (phosphate buffer) and the pH is usually kept around 6.8–7.2.
- It is the working solution that must be freshly prepared because the dye components gets degraded.
- The stock solution mostly has glycerol which act as stabilizing agent.
- The working stain is prepared by diluting stock in buffer mostly in 1:10 or 1:20 ratio.
- Specimen Preparation and Fixation Requirements
- For thin smears, the smear is immediately fixed with absolute methanol.
- Methanol helps in stabilizing nuclear matrix and maintaining morphology.
- Fixation usually needs around 1–3 minutes for thin films.
- For thick films, the smear is only air dried and methanol is not used.
- In thick film preparation, avoiding methanol is required so that RBC lysis occur during staining.
- For tissue sections, paraffin sections are deparaffinized and rehydrated before staining.
- Staining Time Requirements
- Standard staining requires around 20–30 minutes using 1:10 dilution.
- Some procedures especially gastric biopsies may require 30–45 minutes.
- Rapid method using 10% stain usually needs 8–10 minutes.
- Slow method using 3% stain needs around 45–60 minutes.
- Ultrafast methods can complete staining in less than 5 minutes.
- Requirements for G-Banding (Chromosome Analysis)
- Cells are arrested at metaphase using Colcemid.
- Hypotonic treatment (0.075 M KCl) is used so that chromosomes spread.
- Slides are baked at high temperature for some hours.
- Trypsin is used shortly to partially digest proteins which help in forming banding pattern.
- After trypsinization slides are stained with diluted Giemsa.
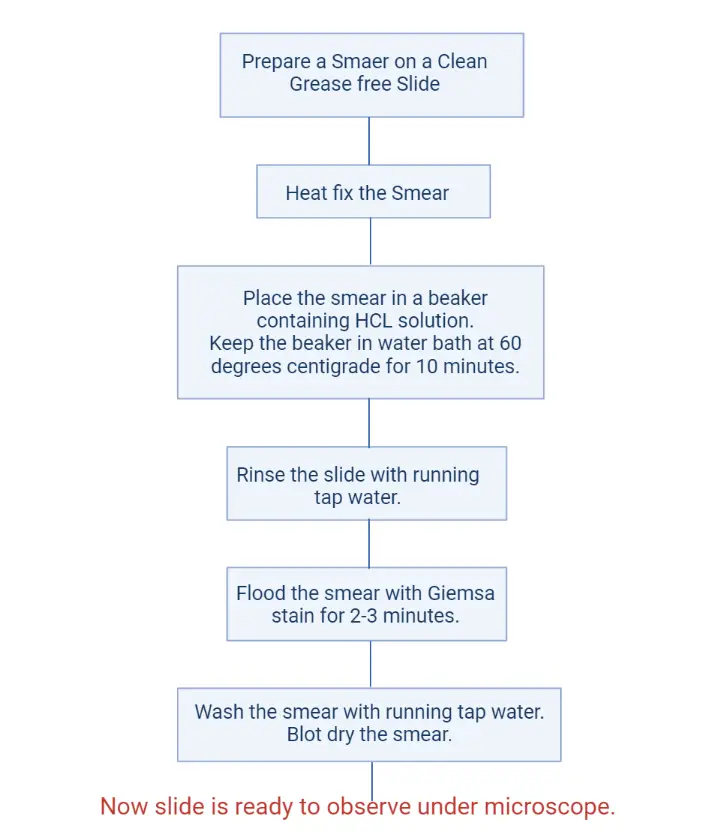
Procedure of Giemsa staining
Giemsa staining is the process used for staining different types of cells and tissues. It is prepared in buffered water (pH 6.8–7.2) and it is applied in various diagnostic procedures.

I. Histology (Paraffin-Embedded Tissue Sections)
This procedure is used for identifying microorganisms like H. pylori in gastric biopsy sections.
Steps
- Preparation– Tissue sections are deparaffinized and then rehydrated in descending alcohol series. It is the initial step for removing wax from paraffin sections.
- Working Solution– Giemsa working solution is prepared in pH 6.8 buffer.
- Staining– The sections are immersed in the working stain. Staining time is about 30–45 minutes (some sections were stained for 25 minutes at room temperature).
- Microscopy– The stained slides are examined microscopically for organism detection.
II. Cytology (Thin Blood Films / General Smears)
This is referred to as the staining method used for peripheral blood and bone marrow aspirates to preserve cell architecture.
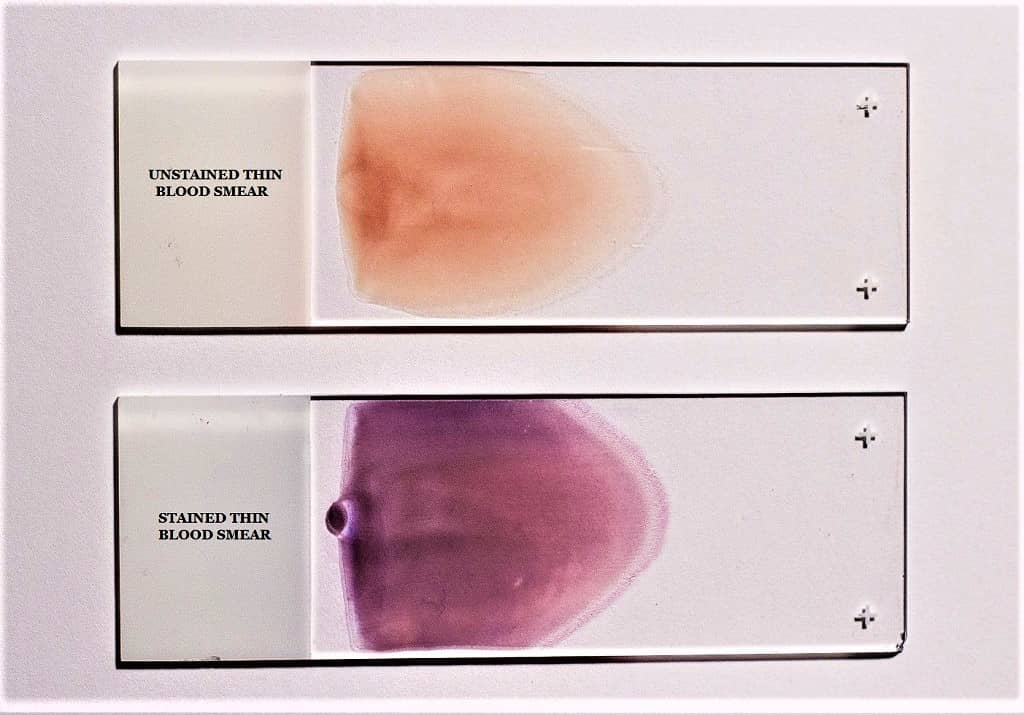
Steps
- Smear Preparation– A thin smear is prepared on a clean slide and allowed to air dry.
- Fixation– The smear is fixed in absolute methanol for 1–3 minutes.
- Working Solution– A 1:10 diluted Giemsa stain is prepared in phosphate buffer (pH 7.2).
- Staining -The fixed slide is immersed in stain-buffer mixture for 20–30 minutes. It is the step where staining intensity is adjusted by time.
- Rinsing -The smear is gently rinsed with buffered water. For Coplin jar method, the smear is dipped in buffer for about 5–30 seconds.
- Drying– The slide is air-dried completely.
- Microscopy– Observation is done using immersion oil.
III. Rapid Cytology (Modified Ultrafast Giemsa – MUFG)
This is the rapid method used mainly during onsite evaluation of FNAC samples.
Steps
- Smear Preparation and Fixation– Air-dried smear is fixed in methanol for 1 minute.
- Staining– Undiluted and filtered Giemsa stock solution is placed directly on the smear. Four to five drops are used and then a coverslip (22 × 40 mm) is placed for spreading. Contact time is 3 minutes.
- Washing and Drying– The coverslip is removed and washed gently. The slide is rinsed with buffered water and air dried.
- Microscopy– The slide is mounted for immediate examination. Total procedure time is less than 5 minutes.
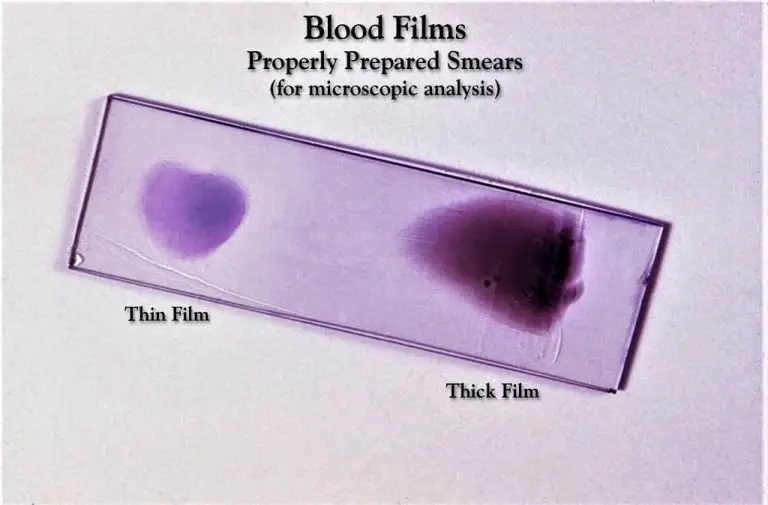
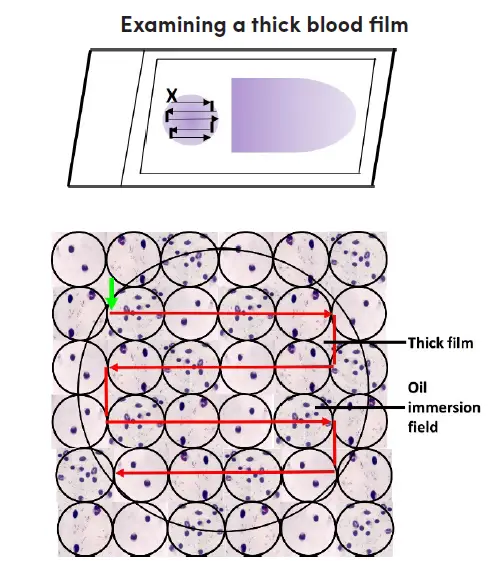
IV. Parasitology (Thick Blood Films for Malaria)
This process is used when there is a requirement for detecting parasites by concentrating cellular components.

Steps
- Smear Preparation– A thick smear is prepared and air-dried completely. The thick film is not fixed because RBCs must hemolyze during staining.
- Working Solution– Working stain is prepared in distilled water buffered to pH 7.2. Rapid method uses 10% stain and slow method uses 3% stain.
- Staining – Rapid (10%)– Staining time is 8–10 minutes. Stain is poured gently until the film is covered.
- Staining – Slow (3%)– Staining time is 45–60 minutes. Slides are placed back to back in a trough and stain is added slowly.
- Rinsing– Buffered water is added dropwise to flush the stain. The stain is never poured directly off the slide.
- Drying– Slides are placed film-side downwards on rack for drying.
V. Cytogenetics (G-Banding Method)
It is the technique used for banding of chromosomes during karyotype analysis.
Steps
- Preparation and Baking– Prepared metaphase slides are baked at 72–75°C for about 3 hours.
- Trypsin Digestion– Slides are placed in 0.025% trypsin solution (pH 7.2–7.4). Digestion lasts about 1 minute depending on banding quality.
- Rinsing– Slides are rinsed in 0.9% NaCl. Some methods use HBSS and HBSS with serum for inactivation.
- Staining– Slides are stained with Giemsa solution diluted in acetone or Gurr buffer. Staining time is 5–10 minutes.
- Final Step– Slides are rinsed, dried and then examined for chromosome banding.
Result of Giemsa staining
Giemsa stain produces a differential coloration of cells and tissues. It is the process where basic dyes (Methylene blue, Azure B) and the acidic dye (Eosin Y) act together. The Romanowsky effect gives the purple tone of chromatin.
Nuclei and other basophilic structures stain blue to purple due to binding of dyes with DNA and RNA. Cytoplasm and eosinophilic structures is stained pink or red because Eosin binds with alkaline proteins.
Staining quality is controlled by the pH of buffer (pH 6.8–7.2).
A. Hematology and Cytology Smears
These are the smears of blood and bone marrow which show clear cellular morphology after staining.
Some main results are–
- Erythrocytes – Pink or pink-tan appearance. At low pH they are more red and at pH 7.2 they become blue-gray.
- Platelets – Light blue cytoplasm with pale pink to red-purple granules.
- Leukocyte Nuclei – Purple or bluish-purple color because dyes bind with nuclear DNA.
- Lymphocytes – Dark blue nucleus and light blue cytoplasm.
- Monocytes – Purple nucleus with pink or gray-blue cytoplasm and some azure granules.
- Neutrophils – Purple nucleus, pale pink cytoplasm and fine lavender granules.
- Eosinophils – Dark blue nucleus and bright red or reddish-orange granules.
- Basophils – Dark blue nucleus and violet-black granules with strong metachromasia.
In cytology smears, nuclear details are crisp and lymphoglandular bodies are clearly seen.
B. Infectious Disease Detection
This stain is used for identifying different microorganisms.
Some important observations are–
- H. pylori – Bacteria appear blue to dark blue in light blue background. It is the most reliable method for detecting the organism and shows high sensitivity and specificity.
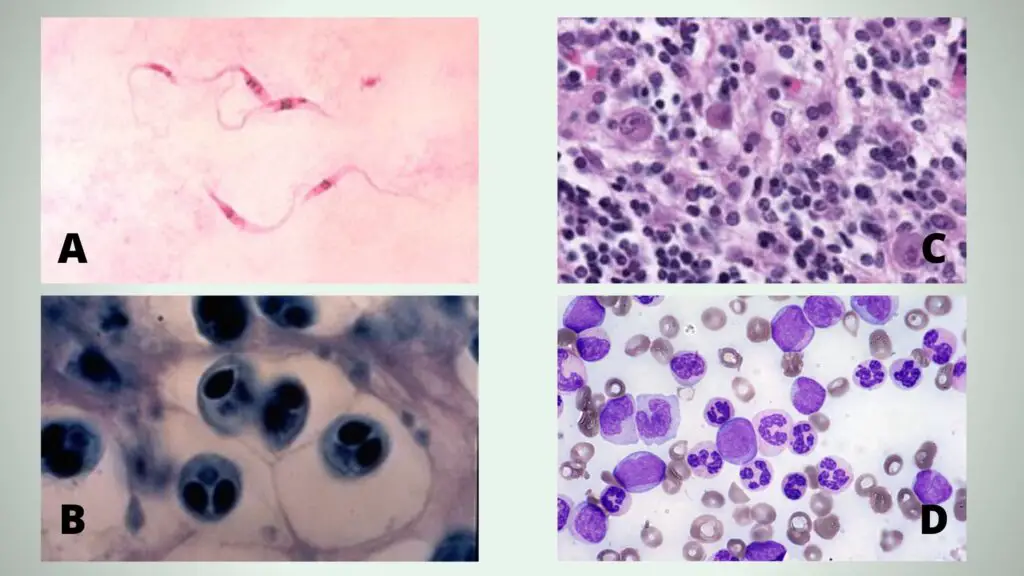
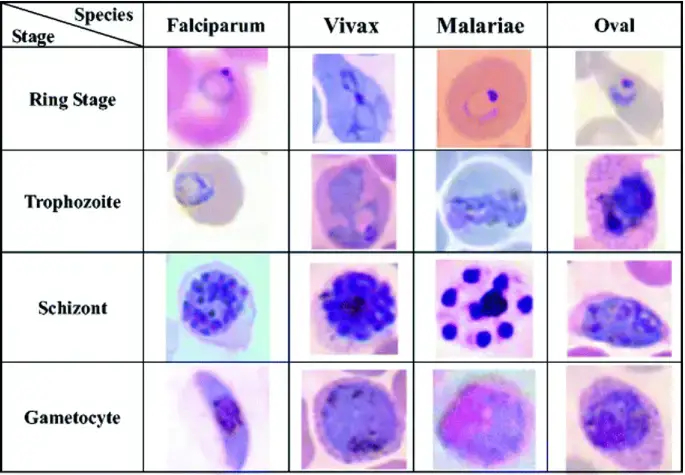
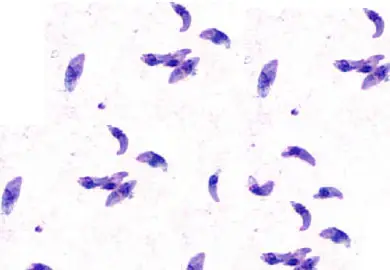
- Plasmodium spp. (Malaria) – Parasite cytoplasm stains blue and nucleus stains red to purple. Schuffner’s dots appear red.
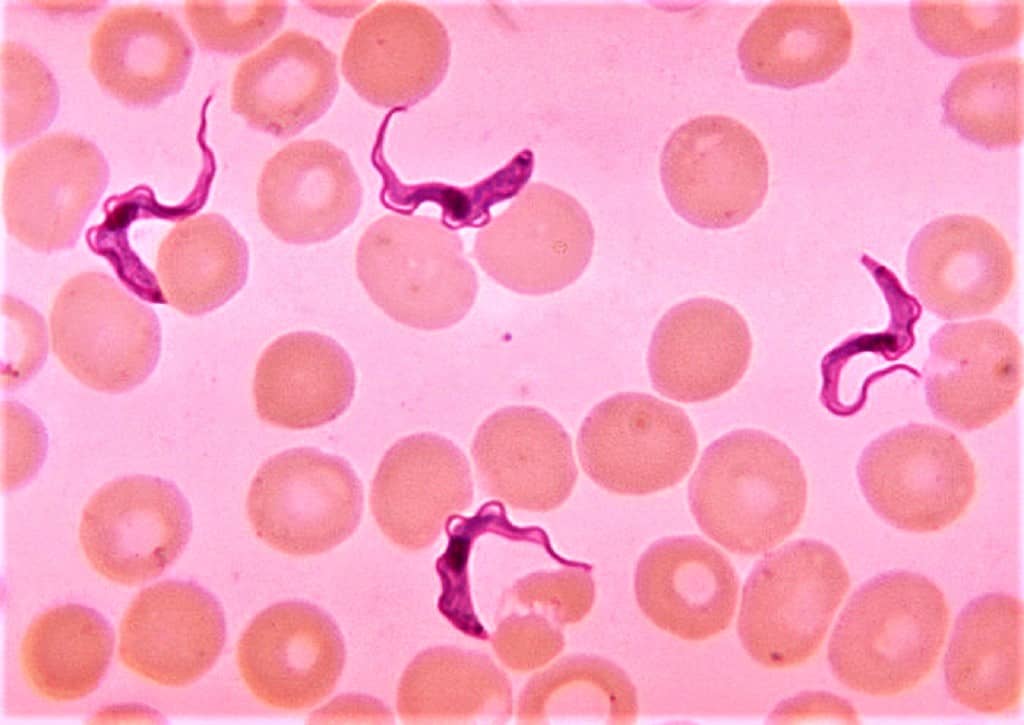
- Trypanosomes – The elongated flagellated forms stain blue with purple nucleus.
- Leishmania – Small oval amastigotes appear within macrophages having blue/violet nucleus and cytoplasm.
- Other microorganisms – Chlamydia inclusion bodies, Borrelia, Yersinia, Histoplasma and Pneumocystis cysts are also demonstrated.
C. Cytogenetics (G-Banding Method)
This is the technique where chromosomes are stained after partial digestion. Chromosomes appear purple with alternate light and dark bands.
Key results are–
- Dark bands (G-positive) – Stain dark and are AT-rich regions. These are heterochromatin areas.
- Light bands (G-negative) – Stain lightly and are GC-rich regions. These are euchromatin areas.
- Banding pattern – Each chromosome shows a fixed banding pattern and this helps in detecting chromosomal abnormalities like deletions, translocations and aneuploidy.
D. Combination Stains
Giemsa is sometimes used with other Romanowsky stains.
Some main features are–
- May-Grünwald Giemsa (MGG/Pappenheim) – Used for FNAC slides and helps in studying tumor cell morphology.
- Wright-Giemsa – Produces brighter reddish-purple granules compared to Wright stain alone.
Images of Giemsa staining
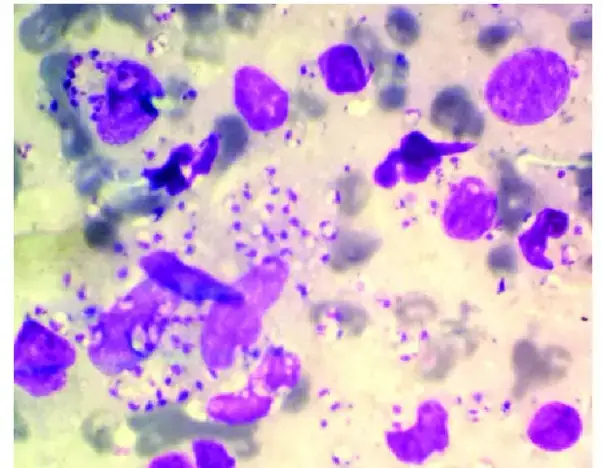
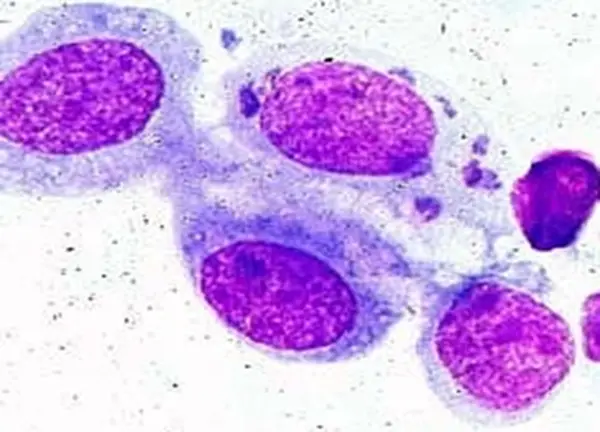
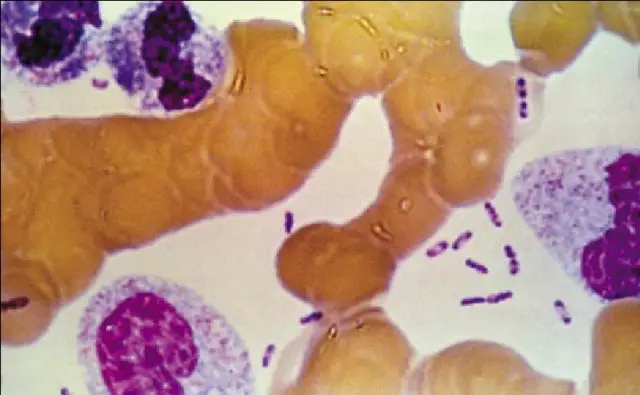
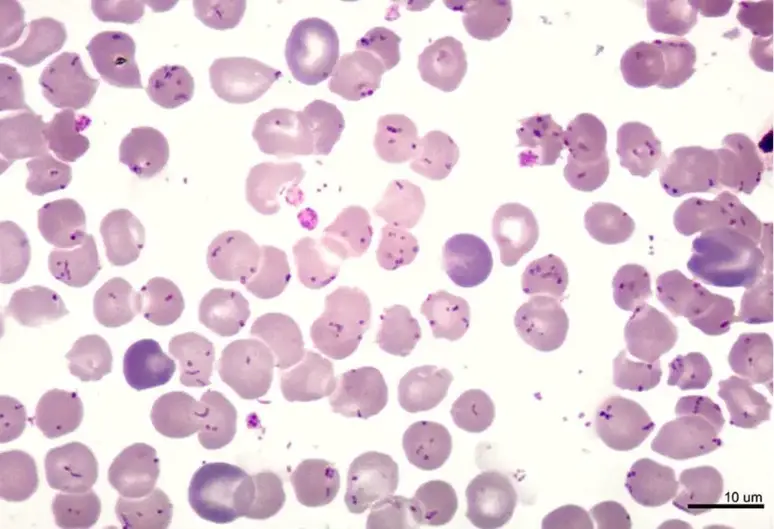
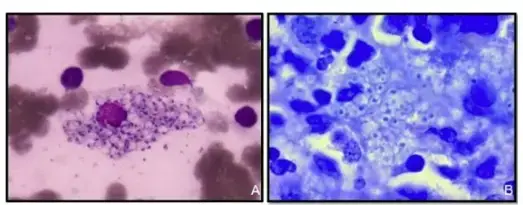
Uses of Giemsa staining
- It is used for detection of malaria parasites in thin and thick blood films.
- It is used for identifying other blood and tissue parasites like Trypanosomes and Leishmania.
- It is used to detect Helicobacter pylori in gastric biopsy sections as the organism is stained distinctly.
- It is used to visualize different microorganisms including spirochetes, Chlamydia trachomatis inclusion bodies, Borrelia spp., Yersinia pestis, and some fungal forms.
- It is used for staining peripheral blood smears and bone marrow samples to differentiate RBCs, WBCs, and platelets.
- It is used in cytology for FNAB samples, lymph node and tumor touch preparations, urine sediments, sputum and other body fluids.
- It is used in rapid onsite evaluation (ROSE) where modified ultrafast Giemsa helps to check sample adequacy.
- It is used in histology for staining tissue sections showing chromatin and cytoplasmic details.
- It is used in cytogenetics for G-banding of metaphase chromosomes to visualize chromosome bands.
- It is used for detecting chromosomal abnormalities like translocations, deletions, aneuploidy and other structural defects.
- It is used for chromatin pattern study as dark bands correspond to AT-rich regions and light bands correspond to GC-rich active regions.
- It is used in gene mapping where banding pattern helps to locate specific loci on chromosomes.
Advantages of Giemsa staining
- It is highly reliable for detection of H. pylori in gastric biopsy sections because the organism is stained clearly against the tissue background.
- It shows high accuracy for H. pylori identification as studies reported very high sensitivity and specificity values.
- It gives sharp contrast where the bacteria appear thin and blue to dark blue which helps in easy recognition.
- It is the gold standard stain for malaria parasite detection in thick and thin blood films.
- It helps in differentiating various stages of the parasite as the cytoplasm is stained blue and the nucleus appears red to purple.
- It allows concentration of parasites in thick films because RBCs are lysed during staining.
- It is useful for broad pathogen detection like Trypanosomes, Leishmania, Treponema pallidum, Histoplasma, Pneumocystis cysts and Chlamydia inclusion bodies.
- It provides detailed nuclear and cytoplasmic morphology of RBCs, WBCs, platelets and parasites in blood and bone marrow samples.
- It is a versatile polychromatic stain used in hematology, histology, cytology, bacteriology and cytogenetics.
- It produces differential staining because of the Romanowsky effect where nuclei stain purple and cytoplasm stains pink to red.
- It shows metachromasia which helps in observing fine chromatin patterns for cellular differentiation.
- It is important in cytogenetics for G-banding to visualize chromosome band patterns.
- It helps in identifying chromosomal abnormalities like translocations, deletions and aneuploidy.
- It can be adapted into rapid staining methods like Modified Ultrafast Giemsa which gives quick results in a few minutes.
- It is cost-effective and cheaper compared to many advanced staining methods.
- It is considered a low-maintenance stain as it is easy to prepare and use in routine laboratories.
Limitation of Giemsa staining
- It is not suitable for general histopathology because it does not show overall tissue architecture like H&E stain.
- It shows reduced sensitivity for detecting H. pylori after treatment with proton pump inhibitors or antibiotics.
- It may give very low detection in post-therapy cases where bacterial density is decreased.
- It can be misinterpreted when there is low organism load, staining artifacts or faded slides.
- It has limited resolution in cytogenetics as small chromosomal changes below few megabases cannot be identified.
- It requires freshly prepared working solution because the dye component gets degraded on exposure to air and water.
- It is considered time-consuming as classical staining needs around 20–45 minutes.
- It needs experience for consistent results because staining intensity depends on pH, reagent age and timing.
- It shows non-uniform staining when smears are thick or contain excessive cellular material.
- It gives poor quality in hemorrhagic or very thick smears where patchy under staining or heavy background staining is seen.
- It requires a thin and even smear preparation for proper microscopic observation.
- Alkhamiss, A. S. (2020). Evaluation of better staining method among hematoxylin and eosin, Giemsa and periodic acid Schiff-Alcian blue for the detection of Helicobacter pylori in gastric biopsies. Malaysian Journal of Medical Sciences, 27(5), 53–61.
- Alwahaibi, N., Al Mamari, A.-M., Al Aamri, A., Al Sulimani, Y., & Al Balushi, A. (2025). Assessing the diagnostic accuracy of routine hematoxylin and eosin, Alcian Blue/Periodic Acid-Schiff, and Giemsa stains in the detection of Helicobacter pylori in gastric biopsies (Version 1). F1000Research, 14(1148).
- Azer Scientific. (n.d.). Giemsa Stain Protocol (for visualization of blood microorganisms) (Cat. No. ES915, ES915-16).
- Creative Bioarray. (2025). Chromosome G Banding Technology Protocol.
- Deepthi, B., Prayaga, A. K., & Rukmangadha, N. (2022). Comparison of Modified Ultrafast Giemsa Stain with the Standard May Grunwald Giemsa Stain in FNAC of Various Organs. Journal of Cytology, 39(4), 174–179.
- Ethos Biosciences. (2025). Choosing Between Wright’s Stains Or Wright-Giemsa Stains.
- Hardy Diagnostics. (2024). How to do a Wright-Giemsa Stain.
- Howe, B., Umrigar, A., & Tsien, F. (2014). Chromosome preparation from cultured cells. Journal of Visualized Experiments, (83), 50203.
- Jackson, S., Grabis, D., & Manav, C. (2018). Giemsa: The Universal Diagnostic Stain. MilliporeSigma.
- Jackson, S., Grabis, D., & Manav, C. (2018). Giemsa: The Universal Diagnostic Stain (Poster). [Document 347–362]
- Loharamtaweethong, K., & Puripat, N. (2020). Comparison of immunohistochemistry and conventional stains for Helicobacter pylori detection in gastric biopsies of patients receiving proton pump inhibitors. ResearchGate. [Excerpt 129]
- Microbiology Information (MI). (n.d.). Giemsa.
- Mokobi, F. (2022). Giemsa Stain – Principle, Procedure, Results, Interpretation. Microbe Notes.
- NOUL Co., Ltd. (2025). Wright-Giemsa Stain to NGSI – The Future of Blood Staining Methods. NOUL Blog.
- Taylor & Francis. (n.d.). G banding – Knowledge and References.
- Wikipedia contributors. (n.d.). G banding. Wikipedia. Retrieved from [Source 171–176].
- Wikipedia contributors. (n.d.). Metachromasia. Wikipedia. Retrieved from [Source 274–280].
- Wikipedia contributors. (n.d.). Romanowsky stain. Wikipedia. Retrieved from [Source 293–314].
- World Health Organization (WHO). (2016). GIEMSA STAINING OF MALARIA BLOOD FILMS (Malaria Microscopy SOP – MM-SOP-07A, Version 1).