What is Cell Cycle?
Cell cycle is the ordered sequence of events by which a cell grows and divides to form two genetically identical daughter cells. It is the process that ensures continuity of life by producing new cells from pre existing cells. The cell cycle is a carefully regulated process and it occurs in a definite series of steps.
It is divided into two major phases which are interphase and mitotic phase. Interphase is the longest phase of the cell cycle in which the cell prepares itself for division. It is further divided into G₁ phase, S phase and G₂ phase. In G₁ phase the cell increases in size and synthesizes RNA and proteins which are necessary for growth. In S phase the replication of DNA takes place and two identical copies of genetic material is formed. In G₂ phase the cell continues to grow and proteins required for spindle formation is synthesized. Thus interphase is the preparatory stage of cell division.
After completion of interphase the cell enters into mitotic phase. It is the division phase in which the nucleus and cytoplasm are divided. During mitosis the duplicated chromosomes are arranged at the equatorial plate and then separated towards opposite poles of the cell. The nuclear division is followed by cytokinesis where the cytoplasm is divided and two separate daughter cells is formed. Some cells after G₁ phase enter into a resting stage known as G₀ phase. This is referred to as quiescent stage where the cell remains metabolically active but does not divide. Thus cell cycle is the basic process of growth and reproduction in living organisms.
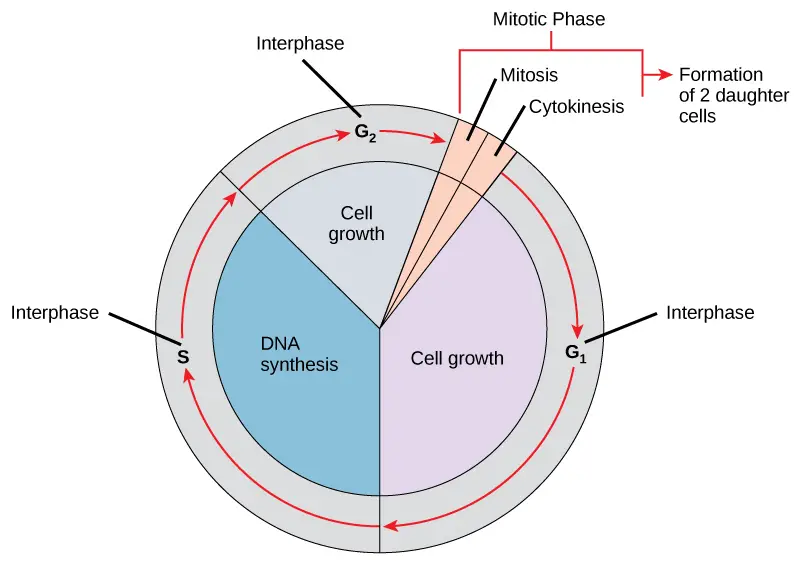
Definition of Cell Cycle
The cell cycle is a series of sequential events that occur in a cell, leading to its growth, DNA replication, and division into two daughter cells. It consists of interphase, where the cell prepares for division, and the M phase, which includes mitosis and cytokinesis. This process is crucial for growth, development, and tissue repair in organisms.
Phases of Cell Cycle
Cell cycle is a series of events in which cell grows and cell division is occurred. It is divided into two main phase- Interphase and Mitotic phase.
- Interphase- It is the period where cell grows and prepares for cell division. It consist of three distinct stages.
- G1 Phase (First Gap)- In this phase cell accumulates the building blocks of chromosomal DNA and proteins. Energy reserves needed for replication is gathered.
- S Phase (Synthesis of DNA)- DNA replication is occurred in this phase. Identical pairs of DNA molecules is formed and it is referred to as sister chromatids. Centrosome is also duplicated to help in chromosome movement.
- G2 Phase (Second Gap)- In this phase energy stores is replenished. Proteins necessary for manipulating chromosomes is synthesized. Certain organelles is duplicated and cell continues to grow.
- Mitotic Phase- It is a multistep process in which duplicated chromosomes and cytoplasm is separated to create two identical daughter cells. It is broken into two parts-
- Mitosis (Nuclear Division)- It is the division of cell nucleus. It occur in five sub-stages.
- Prophase- Chromosomes condense and become visible. Nuclear envelope starts to break down and mitotic spindle begins to form.
- Prometaphase- Nuclear envelope fully disappears. Spindle microtubules is attached to chromosomes by kinetochores (protein complexes).
- Metaphase- All chromosomes align in middle of cell at metaphase plate.
- Anaphase- Sister chromatids separate at centromere and it is pulled rapidly towards opposite poles of cell.
- Telophase- Chromosomes reach at opposite poles and start to decondense. New nuclear envelopes is formed around each set of chromosomes.
- Cytokinesis- It is the physical separation of cytoplasmic components into two completely separate daughter cells.
- Mitosis (Nuclear Division)- It is the division of cell nucleus. It occur in five sub-stages.
- G0 Phase- It is a quiescent resting stage outside the active cell cycle. Cells in this phase are not preparing to divide. It may remain here temporarily until triggered by an external signal or it may stay in G0 permanently.
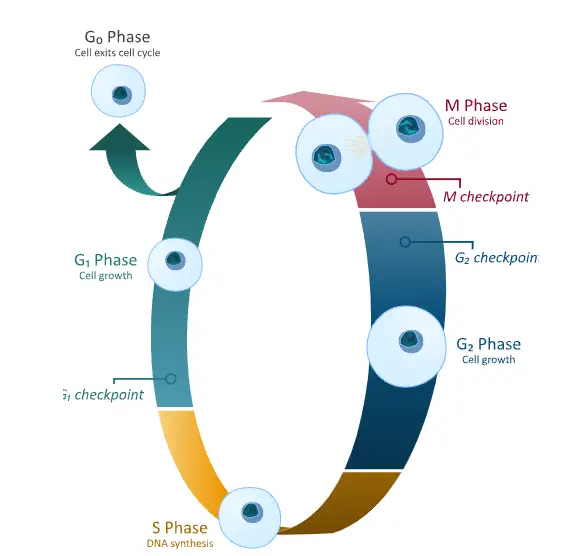
1. Interphase
Interphase is the longest phase of cell cycle and it accounts for at least 91% of total time. In this stage cell performs its normal functions. Cell growth is occurred and preparation for cell division is done. It proceed in three sequential steps- G1 phase, S phase and G2 phase.
- Step 1- G1 Phase (First Gap)
- In this step cell grows in size and it is highly active at biochemical level even though little physical change is visible under microscope.
- The building blocks of chromosomal DNA and associated proteins is accumulated in this phase.
- Necessary energy reserves required for DNA replication is stockpiled by cell.
- Step 2- S Phase (Synthesis of DNA)
- In this step nuclear DNA replication is occurred. Two identical copies of each chromosome is formed and it is referred to as sister chromatids. These sister chromatids are firmly attached at centromere.
- During this phase nuclear DNA remains in loose semi-condensed chromatin form and discrete chromosomes are not formed.
- Centrosome is duplicated in this step and two centrosomes is formed. These centrosomes later give rise to mitotic spindle for orchestrating chromosome movement.
- Step 3- G2 Phase (Second Gap)
- In this step further growth is occurred and energy stores is replenished rapidly.
- Specific proteins necessary for manipulating and moving chromosomes is synthesized.
- Some cellular organelles are duplicated and cytoskeleton is dismantled to provide structural resources for mitotic spindle.
- DNA damage is checked by cell and repair is done to prevent genetic errors from being passed on. Final preparations is completed before entering into mitosis.
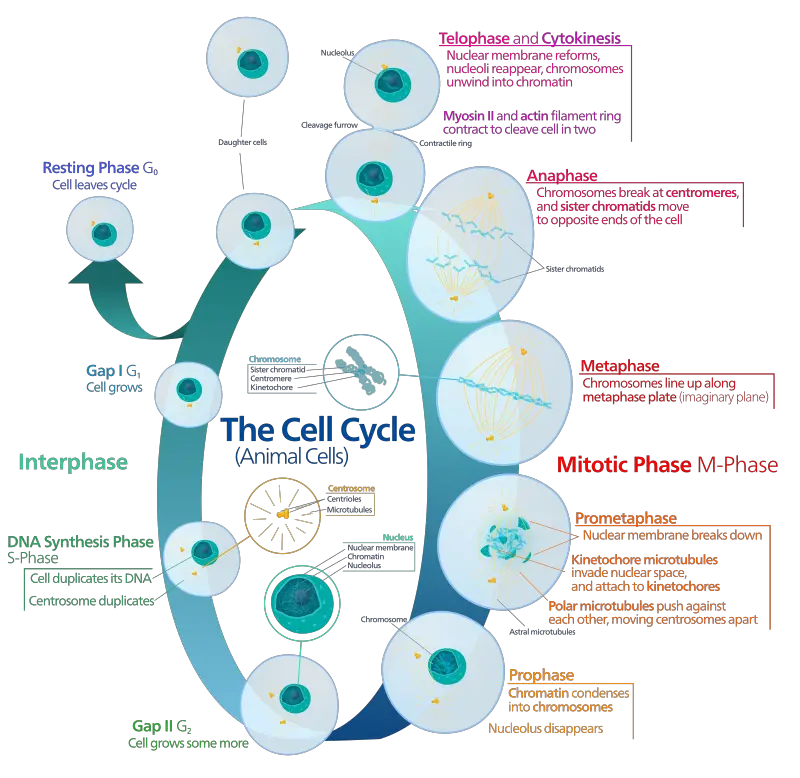
2. Mitotic Phase
Mitotic phase is the phase in which duplicated chromosomes and cytoplasm is separated to form two identical daughter cells. It is divided into two main part- Karyokinesis (Mitosis or Nuclear division) and Cytokinesis.
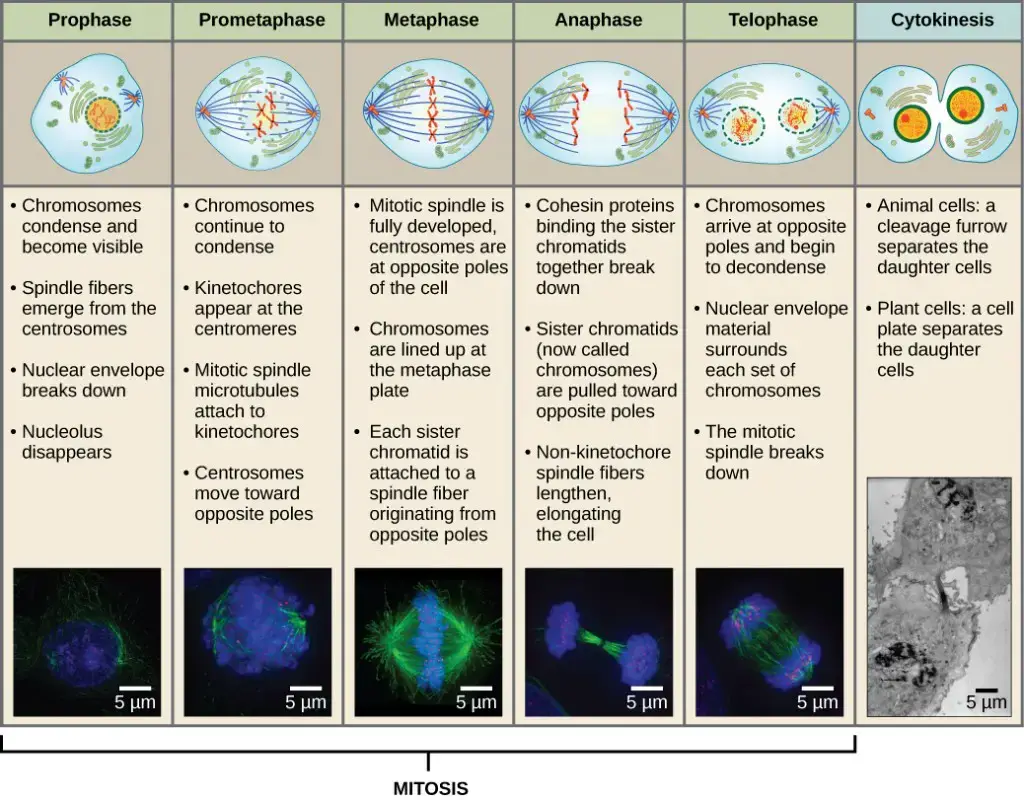
- Karyokinesis (Mitosis or Nuclear Division)
- It is the first main portion of mitotic phase where nucleus divides. It occur in five sequential stages.
- Stages are as follows- Prophase, Prometaphase, Metaphase, Anaphase and Telophase.
- Step I- Prophase
- Nuclear envelope begins to break down into small vesicles and nucleolus disappears.
- Centrosomes migrate to opposite poles of cell and spindle formation is started by extending microtubules.
- Sister chromatids coil tightly and it becomes visible under microscope.
- Step II- Prometaphase
- Nuclear envelope remnants fully disappears and mitotic spindle continues to develop.
- In this step kinetochore (protein structure) is formed at centromere of each sister chromatid.
- Spindle microtubules is attached firmly to kinetochores.
- Step III- Metaphase
- Chromosomes align tightly at the center of cell and it is referred to as metaphase plate (equatorial plane).
- Chromosomes are maximally condensed and sister chromatids remain attached by cohesin proteins.
- Step IV- Anaphase
- Cohesin proteins degrade and sister chromatids separate at centromere.
- Each separated chromatid is now considered as individual chromosome and it is pulled rapidly towards opposite poles by spindle microtubules.
- Cell elongation is started and it becomes visible.
- Step V- Telophase
- Chromosomes reach at opposite poles and it starts to decondense into loose chromatin form.
- Mitotic spindle breaks down.
- New nuclear envelopes is formed around each set of chromosomes.
- Cytokinesis
- It is the final stage of mitotic phase in which cytoplasmic components is separated into two completely separate daughter cells.
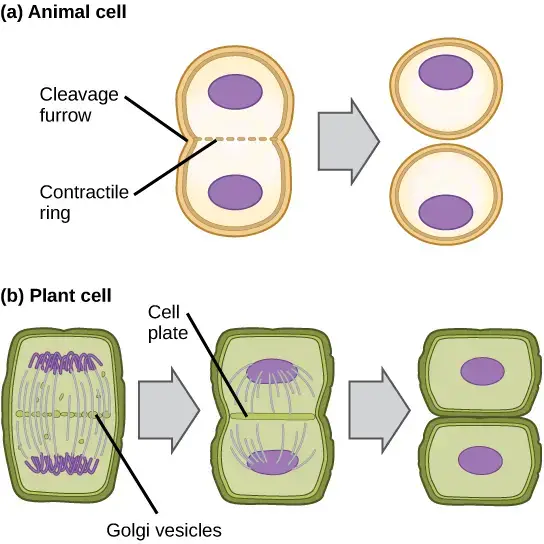
- In animal cells
- Contractile ring of actin filaments is formed just inside plasma membrane.
- It pulls the cell equator inward and cleavage furrow is formed.
- Cleavage furrow deepens and cell is pinched into two halves forming two daughter cells.
- In plant cells
- Cleavage furrow cannot be formed due to rigid cell wall.
- Golgi vesicles transport materials to center of cell and cell plate is formed.
- Cell plate enlarges from inside out and it merges with existing cell wall.
- New wall is formed and two daughter cells is separated.
3. G0 Phase
G0 phase is a quiescent resting stage outside the active cell cycle. It is referred to as resting phase because cell is not dividing and it is not preparing to divide. But cell is still active and normal functions is continued.
- Step 1- Entry into G0 phase
- Cells enters into G0 phase after completing mitosis when cell no longer need to divide.
- Cell may also exit from G1 phase and enter into G0 when environment lacks necessary nutrients and energy.
- In this step if G1 checkpoint is failed due to DNA damage then cell cycle is stopped and G0 phase is entered.
- Step 2- Characteristics of G0 phase
- It is the phase where cell has exited the active cell cycle. Cell division is not occurring.
- Even though it is called resting stage but maintenance activities is done.
- Regulatory functions and specialized physiological functions are performed by cell in this phase.
- Step 3- Molecular maintenance of G0 state
- G0 state is maintained by molecular mechanisms.
- APC/C-Cdh1 complex level remains high and cyclins is degraded continuously for preventing unscheduled re-entry into cell cycle.
- Repressor proteins like p27 and DREAM complex is involved for locking the cell in non-proliferative state.
- Step 4- Temporary (Reversible) G0 phase
- In some cells quiescence is temporary. Stem cells and liver cells are example.
- Cells may remain in G0 for days to years.
- When external signals like growth factors is given then cell can re-enter into active cell cycle at G1 phase.
- Step 5- Permanent (Irreversible) G0 phase
- Many fully differentiated cells exit the cell cycle and it remains in G0 permanently.
- Mature cardiac muscle cells and nerve cells (neurons) are example.
- Because replication is not occurring these cells no longer require cellular checkpoints.
- Step 6- Senescent G0 phase
- Cells can be forced into irreversible G0 due to ageing or severe cellular damage.
- This state is referred to as senescence.
- In this condition division is halted permanently to avoid formation of more damaged cells.
- Cell remains in G0 rather than undergoing apoptosis (programmed cell death).
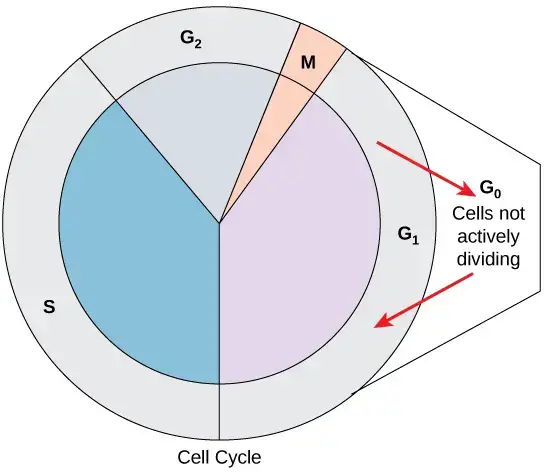
Cell cycle checkpoints
Cell cycle checkpoints are internal control points in which cell cycle progression is monitored. It ensures that damaged cell is not allowed to divide. These checkpoints are mainly present at G1, G2 and M phase.
- G1 checkpoint (Restriction point)
- It occur near the end of G1 phase.
- It acts as the point where cell commits irreversibly for cell division process.
- In this checkpoint conditions for division is assessed.
- Adequate cell size is checked.
- Energy reserves is checked.
- Genomic DNA damage is also checked.
- If requirements are not met then cycle is halted for repair.
- If cell cannot proceed then it may enter into inactive G0 phase.
- G2 checkpoint
- It occur at transition between G2 phase and mitotic (M) phase.
- Cell size and protein reserves is evaluated.
- Most critical role is to ensure that chromosomes are accurately and entirely replicated.
- DNA damage is checked in this checkpoint.
- If DNA damage or incomplete replication is detected then cell cycle halts.
- Replication completion or damaged DNA repair is attempted before entering into mitosis.
- M checkpoint (Spindle checkpoint)
- It occur near the end of metaphase stage during mitosis.
- It determines whether all sister chromatids are correctly attached to spindle microtubules.
- Progression to anaphase is prevented until this checkpoint is passed.
- Kinetochores of every sister chromatid pair must be firmly anchored to spindle fibres.
- Spindle fibres should arise from opposite poles of cell for proper separation.
G1 Checkpoint (Restriction Point)
- G1 checkpoint is referred to as restriction point in mammalian cells and START in yeast. It is the critical point where cell commits irreversibly for cell division process.
- After passing this restriction point progression through rest of cell cycle becomes independent of external growth factors.
- In this checkpoint cell acts as strict quality control monitor and favorable conditions are checked before entry into S phase.
- Genomic DNA damage is checked strictly so mutation is not passed to daughter cells.
- Cell size is checked and it ensures that cell has grown to adequate size for division.
- Energy reserves (ATP) is verified and raw materials required for DNA replication is checked.
- Presence of necessary external signals is confirmed. Growth promoting hormones or factors is needed for allowing progression.
- Transition past this point is controlled by positive regulators and negative regulators. Positive regulators are cyclins and Cdks. Negative regulators are tumor suppressor proteins.
- If DNA damage is detected then p53 halts the cycle for DNA repair. If damage is irreparable then apoptosis (programmed cell death) is triggered.
- p53 increases production of p21 and p21 binds to Cdk/cyclin complexes so activity is inhibited and cell cycle is stopped.
- Retinoblastoma protein (Rb) monitors cell size mainly and it prevents progression by binding to E2F transcription factors.
- When growth signals are sufficient Cyclin D-Cdk4/6 complex phosphorylate Rb and it becomes inactive. Rb releases E2F and genes required for S phase is activated.
- If requirements are not met then entry into S phase is barred. Cell cycle may halt temporarily for remedy like repairing damaged DNA or cell exits active cell cycle and it enters into inactive resting G0 phase.
G2 Checkpoint
- It occur at the end of G2 phase and at the entry point of mitotic (M) phase.
- It is a checkpoint in which cell is not allowed to enter into mitosis when genome is compromised.
- It checks whether DNA replication is completed properly. All chromosomes should be completely and accurately replicated.
- DNA damage is checked in this point. Damage like double strand breaks occurring during S phase is detected.
- Cell size is evaluated. Protein reserves is also checked for supporting two daughter cells.
- If incomplete replication or DNA damage is present then cell cycle is halted and time is given for completing replication or repairing DNA.
- Arrest is maintained by preventing activation of MPF (Maturation promoting factor) and CDKs.
- Sensor kinases like ATM/ATR detects damage and it activates Chk1 and Chk2. These inhibit Cdc25 phosphatases and CDK activation is stopped.
- p53 is involved as major regulator. It halts the cycle for repair and if damage is irreparable then apoptosis (programmed cell death) is triggered.
- Wee1 and Myt1 proteins may be stabilized and CDKs is inhibited for keeping cell arrested in G2.
- When all conditions are satisfied and DNA is verified as undamaged then CDKs signal the cell to proceed into mitosis.
M Checkpoint (Spindle checkpoint)
- Restriction point refers to G1 checkpoint. M checkpoint is referred to as spindle checkpoint.
- It occur near the end of metaphase stage of mitosis (karyokinesis).
- It is the checkpoint in which attachment of sister chromatids with spindle microtubules is assessed.
- It ensures that kinetochores of each pair of sister chromatids are firmly anchored to spindle fibres.
- Spindle fibres should arise from opposite poles of cell and at least two spindle fibres attachment is required.
- This checkpoint is important because separation of sister chromatids in anaphase is an irreversible step.
- If chromosomes are not properly attached then separation of duplicated chromosomes is prevented.
- Cell cycle is halted until favourable conditions are met and proper attachment is achieved.
G0 Checkpoint
- G0 checkpoint is a separate assessment stage in cell cycle.
- Purpose of this checkpoint is to assess the cells for level of maturity.
- If cells are found to be not ready to divide then it does not pass this checkpoint.
- These cells are discarded from division process.
- Restriction point is G1 checkpoint and it is present near the end of G1 phase.
- In restriction point cell commits irreversibly for cell division process.
- In this checkpoint conditions favourable for division is checked like cell size energy reserves and genomic DNA damage.
- External influences like growth factors plays a major role for carrying the cell past this checkpoint.
- If requirements are not met then cell is barred from progressing into S phase where DNA replication occurs.
- When restriction point is failed then cell may halt the cycle for remedy like DNA repair or it exits into inactive G0 phase and it waits for further signals.
Cell Cycle Regulation
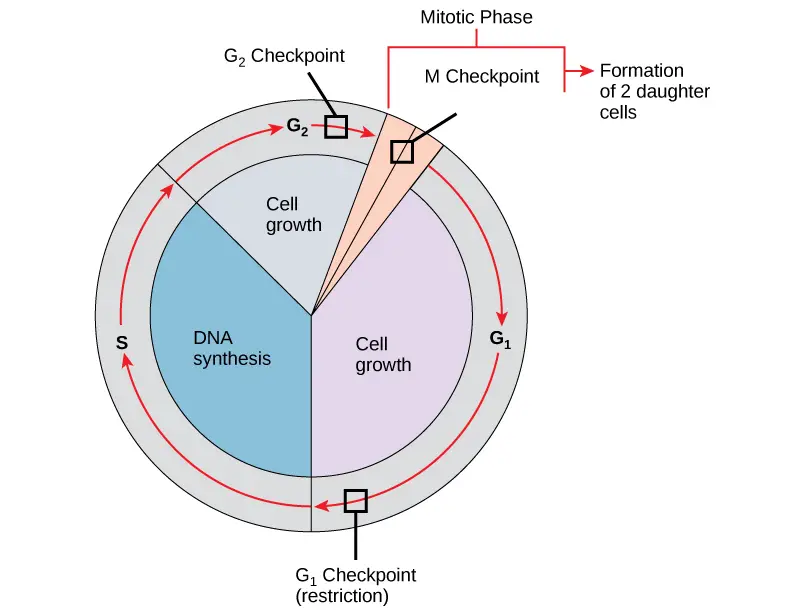
Cell cycle regulation is the control of cell division. It is regulated by external factors and internal checkpoints. The regulation is done to prevent damaged cell from dividing.
- External regulation
- Initiation or inhibition of cell division is triggered by external factors.
- Growth promoting hormones like human growth hormone is involved.
- Cell crowding also affects the division.
- Overall cell size is also a factor for starting or stopping cell division.
- Internal checkpoints
- Cell cycle is monitored by internal control mechanisms. These checkpoints operate at three main points.
- It prevents compromised cells from dividing.
- G1 checkpoint (Restriction point)
- It determines whether conditions are favorable for division.
- Cell size is checked in this point.
- Energy reserves is assessed.
- Genomic DNA damage is checked before cell commits to DNA replication in S phase.
- G2 checkpoint
- Entry into mitotic phase is blocked until all chromosomes are completely and accurately replicated.
- DNA damage is also checked in this point.
- M checkpoint (Spindle checkpoint)
- It occur near end of metaphase.
- It confirms that all sister chromatids are correctly attached to spindle microtubules.
- Separation in anaphase is not allowed until this checkpoint is passed.
- Positive regulators (Advancing the cycle)
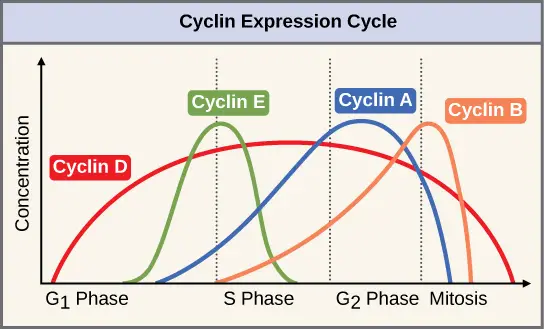
- Cyclins and Cyclin dependent kinases (Cdks) are the main protein groups which push the cell to next phase.
- Cyclin levels fluctuate throughout the cell cycle.
- Cyclins bind tightly to Cdks and phosphorylation is occurred. Active Cdk/cyclin complex is formed.
- These complexes phosphorylate specific target proteins and cell is advanced past checkpoints.
- Different complexes regulate different transitions. Cyclin D-CDK4/6 regulates G1 phase transition and Cyclin B-CDK1 regulates entry into mitosis.
- Negative regulators (Halting the cycle)
- Tumor suppressor proteins act as roadblocks for halting the cell cycle mainly at G1 checkpoint.
- Rb p53 and p21 are the important tumor suppressor proteins.
- Cell cycle is halted until DNA damage and other problematic conditions are resolved.
- p53
- It is a multifunctional protein which monitors DNA damage.
- If damage is found then cell cycle is halted and repair enzymes is recruited.
- If damage is irreparable then apoptosis (programmed cell death) is triggered to prevent duplication of damaged chromosomes.
- p21
- When p53 level rises it triggers production of p21.
- p21 enforces the cell cycle halt by binding directly to Cdk/cyclin complexes.
- Activity of Cdk/cyclin complex is inhibited.
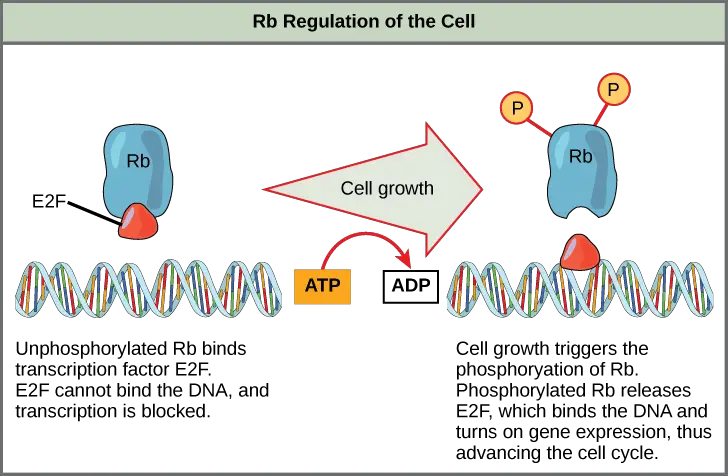
- Retinoblastoma protein (Rb)
- Rb monitors cell size mainly.
- In active state it binds to E2F transcription factors and it blocks them from turning on genes needed for G1/S transition.
- As cell grows to adequate size Rb is phosphorylated and it becomes inactive.
- E2F is released and next phase of cell cycle is promoted.
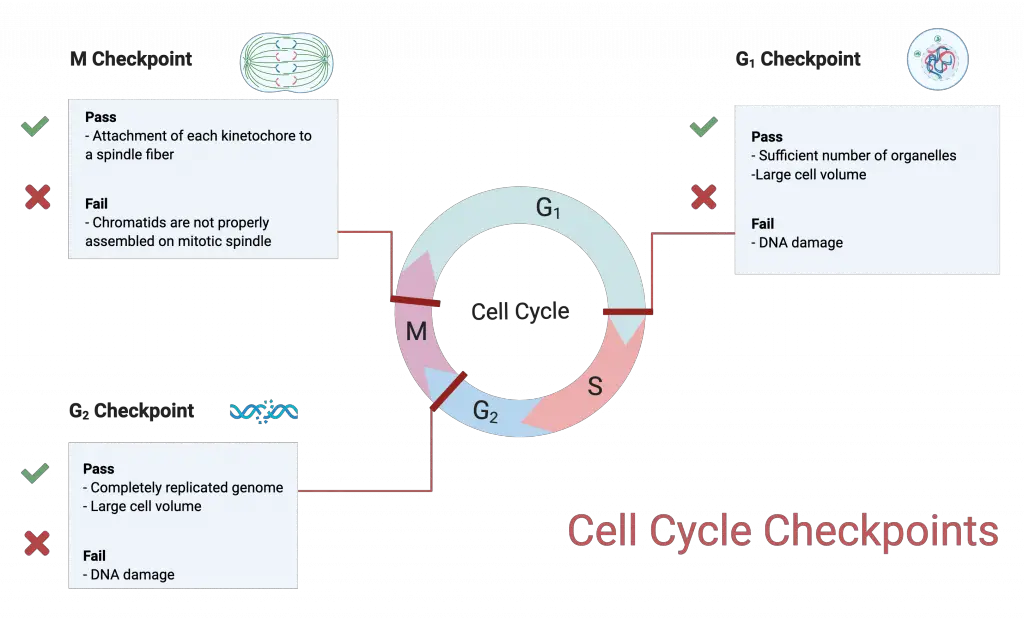
Regulator Molecules of the Cell Cycle
Cell cycle is regulated by regulator molecules. These molecules are responsible for advancing or halting the cell cycle. Regulation is done to ensure proper DNA replication and proper cell division.
Regulator molecules are mainly divided into-
- Positive regulators (Advancing the cell cycle)
- Negative regulators (Halting the cell cycle)
- Additional key regulatory complexes
Positive regulators (Advancing the cell cycle)
- Cyclins
- Cyclins are regulatory proteins and its concentration fluctuates throughout the cell cycle.
- It acts as the main regulatory subunit which pushes the cell to next phase.
- Cyclin binds to specific Cdk and active cyclin-Cdk complex is formed.
- After passing a checkpoint cyclins are degraded by cytoplasmic enzymes so signal is not continued.
- In this way progression is controlled and phase specific control is maintained.
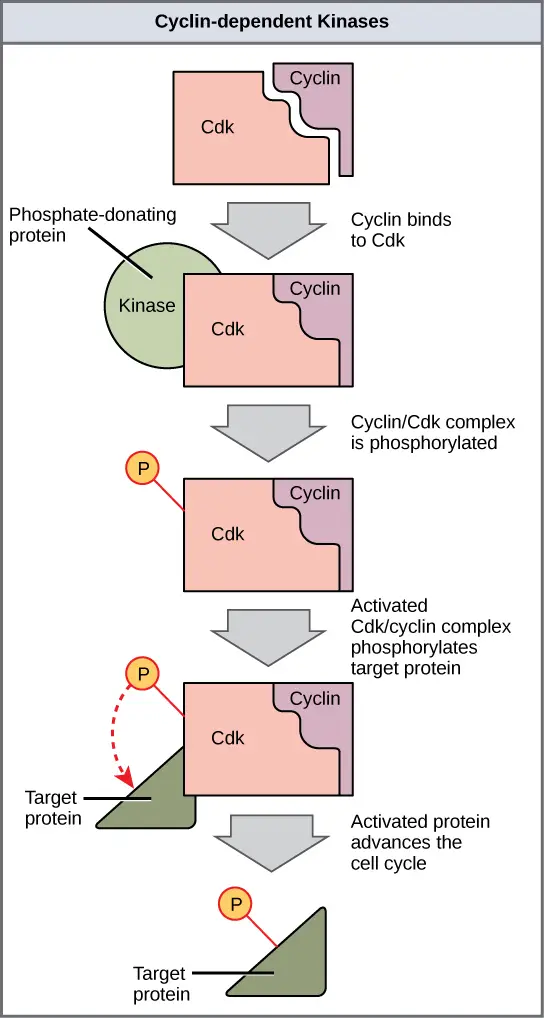
- Cyclin dependent kinases (Cdks)
- Cdks are protein kinases and it remains relatively stable in cell.
- Cdks become active only when it is tightly bound with a specific cyclin and phosphorylation is done in proper site.
- Once active Cdk phosphorylates downstream target proteins and cell is advanced past checkpoints.
- Different cyclin-Cdk complex controls different transition like G1/S transition or entry into mitosis.
- E2F transcription factors
- E2F are proteins which turn on genes required for cell cycle progression.
- It is mainly required for G1/S transition genes.
- E2F remains inactive when it is bound to Rb protein.
- When Rb is inactivated E2F is released and it becomes active.
- After activation expression of S phase genes is promoted and DNA replication related proteins are produced.
- Cdc25 phosphatases
- Cdc25 are enzymes which activate Cdk complexes by removing inhibitory phosphate group.
- For example Cyclin B-Cdk1 complex is activated when inhibitory phosphate group is removed from residues like Threonine-14 (T14) and Tyrosine-15 (Y15).
- This dephosphorylation triggers progression into mitosis.
- When Cdc25 is active CDKs remain active and checkpoint arrest is released.
Negative regulators (Halting the cell cycle)
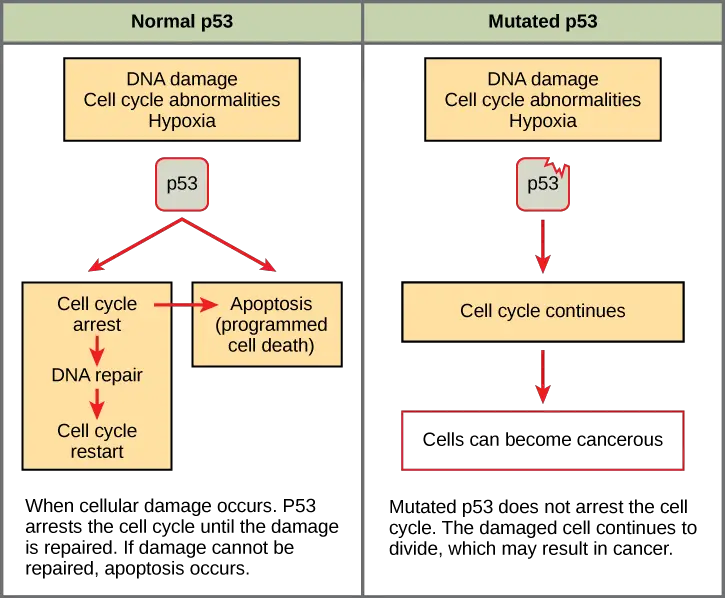
- p53
- p53 is a tumor suppressor protein and it monitors damaged DNA mainly during G1 phase.
- If DNA damage is detected p53 halts the cell cycle and repair enzymes are recruited.
- p53 triggers production of p21 and arrest is maintained.
- If DNA cannot be repaired then apoptosis (programmed cell death) is initiated so damaged genome is not passed.
- p21 (CDKN1A)
- p21 is a Cdk inhibitor and it is triggered by p53.
- It enforces cell cycle arrest by binding directly with Cdk/cyclin complexes.
- For example Cyclin E-Cdk2 complex is inhibited by p21 and entry into S phase is prevented.
- When p21 binds CDK activity is blocked and target protein phosphorylation is not done.
- Retinoblastoma protein (Rb)
- Rb is a tumor suppressor and it monitors cell size mainly.
- It restrains cell proliferation by binding with E2F transcription factors.
- When Rb is active E2F is repressed and genes required for G1/S transition is not expressed.
- When Cyclin/Cdk complexes are active Rb is phosphorylated and it becomes inactive.
- Inactivated Rb releases E2F and cell proceeds to next phase.
- Other Cdk inhibitors (p27 p16)
- These molecules block cycle progression until conditions are favourable.
- p27 binds with Cyclin E-Cdk2 and activation is prevented.
- When Cyclin E-Cdk2 is blocked S phase entry is stopped.
- p16 disrupts Cyclin D-CDK4/6 complexes and G1 progression is inhibited.
- In this way these inhibitors act as brakes in cell cycle.
- WEE1 and MYT1
- WEE1 and MYT1 are kinases and it place inhibitory phosphorylation on CDKs.
- It phosphorylates Cdk1 on residues T14 and Y15 and CDK remains inactive.
- This holds the cell cycle in check and premature entry into mitosis is prevented.
- When inhibition is removed by Cdc25 then CDK is activated and progression is allowed.
- CHK1 and CHK2
- CHK1 and CHK2 are checkpoint effector kinases.
- It is activated by ATM/ATR in response to DNA damage.
- These kinases phosphorylate CDC25 and CDC25 is ubiquitinated and degraded.
- When CDC25 is degraded CDKs remain inactive and cell cycle is halted.
- In this way DNA repair time is provided and damaged DNA is not carried into mitosis.
Additional key regulatory complexes
- APC/C (Anaphase promoting complex or Cyclosome)
- APC/C is a multi-subunit E3 ubiquitin ligase.
- It targets specific cell cycle proteins like cyclins and securin for destruction by proteasome.
- Degradation of securin is important for sister chromatid separation.
- Degradation of cyclins is important for exit from mitosis and resetting the cycle.
- Cdc20 and Cdh1
- Cdc20 and Cdh1 are coactivator subunits which binds to APC/C.
- It dictates substrate specificity of APC/C.
- Cdc20 activates APC/C at metaphase to anaphase transition.
- Cdh1 activates APC/C in late mitosis and G1 phase.
- Cdh1 prevents unscheduled S phase entry by keeping cyclin level low.
- BubR1 and Mad2 (Spindle checkpoint proteins)
- BubR1 and Mad2 forms Mitotic checkpoint complex (MCC).
- It binds to Cdc20 and APC/C activation is inhibited.
- This inhibition continues until all chromosomes are properly aligned and attached to spindle.
- When attachment is proper inhibition is removed and APC/C is activated so chromosome separation is allowed.
Cancer and the Cell Cycle
Cancer is characterized by uncontrolled cell growth and cell division. It is caused due to series of genetic mutations which disrupt normal regulatory mechanisms of cell cycle. Positive regulators and negative regulators are involved in maintaining cellular homeostasis. When these regulatory system fails normal cell is transformed into cancerous cell.
- Cell cycle control mechanisms
- Cell cycle is regulated by checkpoints mainly during G1 S and G2 phase.
- These checkpoints ensure that cell progress only when conditions are optimal and preparations are completed.
- Errors can occur during these processes mainly during DNA replication in S phase.
- Even when checkpoints function correctly some replication errors (mutations) may escape repair and it is passed to daughter cells.
- Role of gene mutations
- All cancers originate from mutations which produce faulty proteins involved in cell reproduction.
- Mutations may affect proto-oncogenes and proto-oncogenes when mutated it becomes oncogenes.
- These mutations can produce less functional or non-functional proteins and cell cycle completion may be affected.
- In some cases mutations increase activity of positive regulators and checkpoints are bypassed.
- For example mutation can allow Cdks to be activated without cyclin and cell progresses prematurely through cycle.
- Proto-oncogenes and oncogenes
- Proto-oncogenes code for proteins which positively regulate cell cycle.
- When proto-oncogenes are mutated into oncogenes cell cycle progression rate is increased.
- Alteration in DNA sequence can produce proteins which push the cycle forward even if conditions are not met.
- If atypical daughter cells continues dividing then more mutations are accumulated and cancer risk is increased.
- Tumor suppressor genes
- Tumor suppressor genes regulate cell cycle by coding negative regulatory proteins.
- Rb p53 and p21 are important tumor suppressor proteins.
- These proteins act as brakes and uncontrolled division is prevented until conditions are met.
- Mutation in these genes compromise their function and it acts like malfunctioning brakes.
- More than half of human tumors show mutation in p53 gene. p53 detects DNA damage and repair is initiated or apoptosis is triggered when damage is irreparable.
- Consequences of p53 mutation
- Defective p53 fails to identify DNA errors and damaged genetic material is propagated through divisions.
- Even if some p53 function remains it may not signal repair enzymes effectively and damaged DNA persists.
- Production of p21 may also decrease and barrier against Cdk activation is lost.
- Cells bypass G1 checkpoint and it enters into S phase under suboptimal conditions.
- Daughter cells inherit mutated p53 gene and other oncogenes and inactivated tumor suppressor genes may also be present.
- Tumor growth and accumulation of mutations
- When daughter cells divide without regulatory control additional mutations are acquired.
- Accumulation of mutations leads to rapid growth of tumors because restriction of division is dismantled.
- Cancer represents culmination of genetic mutations affecting positive and negative regulatory pathways and balance of cell cycle regulation is disturbed.
The Fate of Checkpoint Dysfunction in Human Disease
Checkpoint dysfunction is involved in pathogenesis of human disease. It is mainly linked with cancer. Ability of cell to monitor DNA damage and cellular abnormality is important for maintaining genomic integrity. When checkpoint fails consequence can range from cell death to reprogramming of cell cycle and uncontrolled cell proliferation is occurred.
- Importance of checkpoint in genomic integrity
- DNA damage response is required for preventing mutation.
- Checkpoint arrest is done to stop division of compromised cell.
- When these control is lost then damaged genome is allowed to pass into next generation of cells.
- Impact of p53 dysfunction
- p53 is one of the most frequently mutated gene in cancer.
- Loss of p53 disrupts regulation of CDKs by p21 and G1 checkpoint maintenance is affected.
- In absence of functional p53 cell cannot arrest properly in response to DNA damage.
- Unregulated entry into S phase is occurred and damaged DNA is replicated.
- p53 also directs cells towards apoptosis (programmed cell death) or senescence (permanent cell cycle arrest).
- Due to p53 dysfunction cancer development is promoted and responsiveness of cancer cells to therapy is also affected.
- Role of Chk1 pathway
- In many cancer cells with defective p53 increased reliance on Chk1 pathway is seen.
- Chk1 is required for cell viability under DNA damage condition.
- Chk1 is up-regulated in various cancers and it suggests therapeutic targeting.
- Inhibition of Chk1 and its substrate Wee1 is explored as treatment strategy.
- These pathways are crucial for controlling cell cycle progression and maintaining genome integrity.
- Consequences of checkpoint loss
- In model organisms like fission yeast absence of Chk1 shows major defect mainly when external DNA damage is present or DNA repair deficiency is present.
- Without adequate checkpoint activation cells enter into mitosis with damaged or fragmented chromosomes.
- Catastrophic outcome is produced like cell death or severe chromosomal instability.
- In mammals Chk1 pathway is critical in early embryonic development.
- Disruption of this pathway can be lethal in certain tissues showing dependency of rapid cell division on checkpoint function.
- Aneuploidy and tumorigenesis
- Many solid tumors show aneuploid karyotype (abnormal number of chromosomes).
- Loss or rearrangement of chromosomes can cause tumor suppressor gene loss and oncogene activation.
- Mutations in spindle checkpoint genes are rare in many human cancers but effects can be profound.
- Even modest disruption in mitotic checkpoint function can cause severe chromosomal abnormalities and tumorigenesis is promoted.
- Balance of cell cycle regulation
- Relationship between DNA damage response and cell cycle checkpoint is important for maintaining genomic integrity.
- High DNA damage usually induces cell death. But when checkpoints are dysfunctional cell continues to divide despite severe genomic alteration.
- Dynamics of spindle microtubules becomes critical during mitosis.
- Abnormal spindle function causes chromosomal missegregation and genomic instability is increased further.
Importance of Cell cycle
- Cell cycle is important for growth and development of multicellular organisms. It allows organism to develop from single fertilized egg (zygote) into mature organism. Increase in total number of cells is done by this process.
- It provides cell replacement. New cells are produced to replace aged dead or sloughed off cells. Skin cells lining of digestive tract and red blood cells are replaced by this way.
- It is important for tissue repair and healing. In injuries like scrapes burns and broken bones cell division is occurred and new tissue is formed for healing.
- It maintains genetic stability and continuity. DNA replication is done precisely and division ensures that daughter cells receives identical copy of genetic material. Correct number of chromosomes is maintained.
- It is important for reproduction in single celled organisms. Cell division cycle is the only method of reproduction in many unicellular forms.
- It forms the basis of asexual reproduction in some multicellular organisms. Budding in hydra and vegetative propagation in plants are example.
- It helps in regeneration. Some organisms can regrow lost body parts like starfish regenerating a lost arm.
- It is important in medical and scientific advancements. Understanding of cell cycle is needed for cancer research because cancer is uncontrolled cell division. It is also used in developing stem cell therapies for replacing damaged organs and tissues.
References
- Alfieri, C., Chang, L., Zhang, Z., Yang, J., Maslen, S., Skehel, M., & Barford, D. (2016). Molecular basis of APC/C regulation by the spindle assembly checkpoint. Nature, 536(7617), 431–436. https://doi.org/10.1038/nature19083
- Biddle, K., & Chin, G. (n.d.). Cytokinesis in Animals and Plants | Definition & Example. Study.com.
- Boundless. (2024). 10.3B: Regulation of the Cell Cycle at Internal Checkpoints. In General Biology (Boundless). Biology LibreTexts.
- Brunelli, B. (n.d.). Cell Cycle (Mitosis). In Introduction to Living Systems. CalState-Pressbooks Network.
- Carter, S. (n.d.). Introduction to Cell Cycle Checkpoints. In Biology for Majors I. Lumen Learning.
- Chen, C., Piano, V., Alex, A., Han, S. J. Y., Huis in ‘t Veld, P. J., Roy, B., Fergle, D., Musacchio, A., & Joglekar, A. P. (2023). The structural flexibility of MAD1 facilitates the assembly of the Mitotic Checkpoint Complex. Nature Communications, 14, 1529. https://doi.org/10.1038/s41467-023-37235-z
- Coller, H. A. (2019). Regulation of Cell Cycle Entry and Exit: A Single Cell Perspective. Comprehensive Physiology, 10(1), 317–344. https://doi.org/10.1002/cphy.c190014
- Diaz-Martinez, L. A., Tian, W., Li, B., Warrington, R., Jia, L., Brautigam, C. A., Luo, X., & Yu, H. (2015). The Cdc20-binding Phe Box of the Spindle Checkpoint Protein BubR1 Maintains the Mitotic Checkpoint Complex During Mitosis. Journal of Biological Chemistry, 290(4), 2431–2443. https://doi.org/10.1074/jbc.M114.616490
- Engeland, K. (2022). Cell cycle regulation: p53-p21-RB signaling. Cell Death & Differentiation, 29(5), 946–960. https://doi.org/10.1038/s41418-022-00988-z
- Fang, G. (2002). Checkpoint Protein BubR1 Acts Synergistically with Mad2 to Inhibit Anaphase-promoting Complex. Molecular Biology of the Cell, 13(3), 755–766. https://doi.org/10.1091/mbc.01-09-0437
- Fong, C. S., Mazo, G., Das, T., Goodman, J., Kim, M., O’Rourke, B. P., Izquierdo, D., & Tsou, M.-F. B. (2016). 53BP1 and USP28 mediate p53-dependent cell cycle arrest in response to centrosome loss and prolonged mitosis. eLife, 5, e16270. https://doi.org/10.7554/eLife.16270
- Gallegos, H., & Potts, E. (n.d.). G0 Phase of the Cell Cycle | Overview & Importance. Study.com.
- Huang, M., Liu, C., Shao, Y., Zhou, S., Hu, G., Yin, S., Pu, W., & Yu, H. (2022). Anti-tumor pharmacology of natural products targeting mitosis. Cancer Biology & Medicine, 19(6), 774–801. https://doi.org/10.20892/j.issn.2095-3941.2022.0006
- Kalucka, J., Missiaen, R., Georgiadou, M., Schoors, S., Lange, C., De Bock, K., Dewerchin, M., & Carmeliet, P. (2015). Metabolic control of the cell cycle. Cell Cycle, 14(21), 3379–3388. https://doi.org/10.1080/15384101.2015.1090068
- Keighley, N. (2020). The Fight Against Cancer. In Miraculous Medicines and the Chemistry of Drug Design. Taylor & Francis.
- Kernan, J., Bonacci, T., & Emanuele, M. J. (2018). Who guards the guardian? Mechanisms that restrain APC/C during the cell cycle. Biochimica et Biophysica Acta (BBA) – Molecular Cell Research, 1865(12), 1924–1933. https://doi.org/10.1016/j.bbamcr.2018.09.011
- Kishimoto, T. (2015). Entry into mitosis: a solution to the decades-long enigma of MPF. Chromosoma, 124(4), 417–428. https://doi.org/10.1007/s00412-015-0508-y
- Kishimoto, T. (2018). MPF-based meiotic cell cycle control: Half a century of lessons from starfish oocytes. Proceedings of the Japan Academy, Series B, Physical and Biological Sciences, 94(4), 180–203. https://doi.org/10.2183/pjab.94.013
- Kousholt, A. N., Menzel, T., & Sørensen, C. S. (2012). Pathways for Genome Integrity in G2 Phase of the Cell Cycle. Biomolecules, 2(4), 579–607. https://doi.org/10.3390/biom2040579
- Leacock, S. W. (2024). 1.4: Mitosis. In Genetics Textbook. Biology LibreTexts.
- Meitinger, F., Belal, H., Davis, R. L., Martinez, M. B., Shiau, A. K., Oegema, K., & Desai, A. (2024). Control of cell proliferation by memories of mitosis. Science, 383(6690), 1441. https://doi.org/10.1126/science.add9528
- Mercadante, A. A., & Kasi, A. (2023). Genetics, Cancer Cell Cycle Phases. In StatPearls. StatPearls Publishing.
- Overlack, K., Primorac, I., Vleugel, M., Krenn, V., Maffini, S., Hoffmann, I., Kops, G. J. P., & Musacchio, A. (2015). A molecular basis for the differential roles of Bub1 and BubR1 in the spindle assembly checkpoint. eLife, 4, e05269. https://doi.org/10.7554/eLife.05269
- Pediangco, C. A., & Pediangco, J. A. (n.d.). Advanced Applications of Mitosis and Meiosis. Scribd.
- Robb, A. (n.d.). Cell Division: Medical Advancements & Technology. Study.com.
- Sionov, R. V., Hayon, I. L., & Haupt, Y. (2013). The Regulation of p53 Growth Suppression. In Madame Curie Bioscience Database. Landes Bioscience.
- Stracker, T. H. (2024). Regulation of p53 by the mitotic surveillance/stopwatch pathway: implications in neurodevelopment and cancer. Frontiers in Cell and Developmental Biology, 12, 1451274. https://doi.org/10.3389/fcell.2024.1451274
- University of Southern Queensland. (n.d.). Cell Cycle Checkpoints. In Book 1: Biosciences for Health Professionals.
- Uzbekov, R. E., & Prigent, C. (2022). A Journey through Time on the Discovery of Cell Cycle Regulation. Cells.
- Wikipedia contributors. (2025). Cell cycle checkpoint. Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (2025). Maturation promoting factor. Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (2025). Mitosis. Wikipedia, The Free Encyclopedia.
- Wikipedia contributors. (2026). Cell cycle. Wikipedia, The Free Encyclopedia.
- Xie, G. (2024). Applications of Cell Cycle: Phases of Interphase, Mitosis, and Meiosis. Journal of Cell Science & Therapy, 15(3), 453. https://doi.org/10.35248/2157-7013.24.15.453
- Yang, Z., Mogre, S., Jun, H., He, R., Chaudhary, S. G., Bhattarai, U. R., Sui, S. J. H., Matulonis, U. A., Lazo, S., Shetty, A., Cameron, A., Nguyen, Q.-D., & Hill, S. J. (2025). Functional Profiling of p53 and RB Cell Cycle Regulatory Proficiency Suggests Mechanism-Driven Molecular Stratification in Endometrial Carcinoma. Cancer Research Communications, 5(4), 719–742. https://doi.org/10.1158/2767-9764.CRC-24-0028
- Zhang, Y., van der Zee, L., & Barberis, M. (2023). Two-way communication between cell cycle and metabolism in budding yeast: what do we know? Frontiers in Microbiology, 14, 1187304. https://doi.org/10.3389/fmicb.2023.1187304
- Zhou, Y., Nakajima, R., Shirasawa, M., Fikriyanti, M., Zhao, L., Iwanaga, R., Bradford, A. P., Kurayoshi, K., Araki, K., & Ohtani, K. (2023). Expanding Roles of the E2F-RB-p53 Pathway in Tumor Suppression. Biology, 12(12), 1511. https://doi.org/10.3390/biology12121511