Sulfur Reduction Test (H2S production test) is a biochemical test used to see the ability of bacteria to reduce sulfur containing compounds and liberate hydrogen sulfide gas (H2S). It is used mainly for differentiation of Gram negative enteric bacilli. It helps to separate H2S producers like Salmonella and Proteus from non producers like Shigella and Escherichia coli.
H2S production in bacteria occurs by two common mechanism. In one mechanism, sulfur containing amino acids like L-cysteine is broken down during protein putrefaction and H2S is released (cysteine desulfurase). In another mechanism, during anaerobic respiration, inorganic sulfur compound like thiosulfate is used as terminal electron acceptor and it is reduced to H2S (thiosulfate reductase). H2S is colourless so it is not seen directly.
The test is done on indicator media like SIM medium, TSI agar, Kligler Iron Agar (KIA) or by lead acetate paper strip. These media contains sulfur source (sodium thiosulfate or cysteine rich peptones) and also a metallic salt indicator (ferrous ammonium sulfate or lead acetate). When H2S is produced, it reacts with the metal salt and forms insoluble black precipitate (ferrous sulfide or lead sulfide).
Positive test is shown by black colour or blackening in the medium or strip. Negative test is shown by no blackening. This test is also used in water quality checking for fecal contamination and in industrial area for bacteria related to pipeline corrosion.
Objectives of Sulfur Reduction Test
- To determine whether a microorganism can reduce sulfur containing compounds (sulfur metabolism).
- To detect production of hydrogen sulfide (H2S) gas during growth in the test medium.
- To differentiate and presumptively identify Gram negative enteric bacilli (Enterobacteriaceae) by H2S reaction.
- To distinguish H2S producing organisms like Salmonella, Proteus (also Francisella) from non H2S producers like Shigella and Escherichia coli.
- To detect fecal contamination in water by rapid indication of fecal coliforms or enteric bacteria.
- To identify sulfate reducing bacteria in industrial samples which are involved in pipeline corrosion.
Principle of Sulfur Reduction Test
Principle of Sulfur Reduction Test is based on the ability of bacteria to reduce sulfur containing compounds and produce hydrogen sulfide (H2S) gas. The medium used contains a sulfur source like sodium thiosulfate or cysteine rich compounds. It also contains a metallic indicator like ferrous ammonium sulfate (iron salt) or lead acetate strip.
When the organism reduces the sulfur source, H2S gas is liberated in the medium. This H2S is colourless so it cannot be seen directly. The liberated H2S reacts with the metallic indicator present in the medium or on the strip. As a result, an insoluble black precipitate is formed (ferrous sulfide or lead sulfide).
Formation of black colour or blackening is taken as positive sulfur reduction test. Absence of black precipitate or no blackening is taken as negative test.
Requirements for Sulfur Reduction Test
- Culture media (any one can be used)
SIM medium. TSI agar. Kligler iron agar (KIA). Lead acetate agar. - Sulfur source in medium
Sodium thiosulfate. Cysteine rich peptone or other sulfur containing substrate. - Indicator system
Ferrous ammonium sulfate or other iron salt in medium. Lead acetate paper strip (for paper method). - Test tubes and cotton plug / screw cap tubes
Media is dispensed in test tubes (slant or deep as required). - Inoculating tools
Sterile inoculating needle (for stab in SIM deep). Inoculating loop (for slant). - Incubator
Incubation is done at around 35±2°C. - Heat source
Bunsen burner / spirit lamp for sterilization of wire and mouth of tube. - Media preparation items
Autoclave for sterilization. Weighing balance and glassware for media preparation. - Test organism
Fresh culture (18-24 hours) of the organism to be tested. - Controls
Positive control organism. Salmonella / Proteus (Proteus mirabilis).
Negative control organism. Shigella / Escherichia coli. - Extra requirements for lead acetate paper method
Peptone water or nutrient broth (liquid medium). Tweezers for handling strip. Parafilm or sealing material. Scissor / blade if needed. - Safety items (mainly for lead acetate)
Gloves. Eye protection. Lead acetate is toxic so careful handling is done.
Procedure of Sulfur Reduction Test
Method 1. SIM agar deep method
- Take a fresh (18-24 hours) pure culture of the test organism from solid medium.
- Sterilize a straight inoculating needle and pick a small colony (do not use loop for SIM deep).
- Stab the needle straight in the center of SIM medium up to about 1/2 to 2/3 depth of the tube.
- Withdraw the needle carefully through the same stab line without making side cuts.
- Incubate aerobically at 35-37°C for 18-48 hours keeping cap loose.
- After incubation observe blackening or black precipitate formation in the medium.
Method 2. TSI / KIA slant method
- Take a fresh colony of the test isolate using sterile straight inoculating needle.
- Stab the needle through the center of the medium till the bottom (butt).
- While withdrawing the needle, streak the slant surface in zig-zag manner.
- Incubate aerobically at 35-37°C for 18-24 hours with loose cap.
- Observe the tube for blackening in the medium, mainly in the butt region.
Method 3. Lead acetate paper strip method
- Take a tube/bottle of peptone water or nutrient broth and inoculate with the test organism.
- Place a lead acetate paper strip in the neck of the tube, lower end should hang just above the liquid.
- The strip should not touch the liquid medium (it can inhibit growth).
- Fix the strip by cotton plug or screw cap, and keep it in position.
- Incubate aerobically at 35-37°C for 18-24 hours.
- Observe the lower part of strip for black or brownish black colouration.
Results of Sulfur Reduction Test
Positive result
- Positive test is indicated by formation of black colour or black precipitate (insoluble precipitate).
- In SIM deep, blackening is seen along the stab line or it may spread in the medium.
- In TSI / KIA, blackening is seen in the medium, mainly in the butt region (sometimes it may spread).
- In lead acetate paper method, lower part of the strip turns brownish black to black.
Negative result
- Negative test is indicated by absence of blackening.
- In SIM medium, no black precipitate is formed and medium remains original colour.
- In TSI / KIA, no blackening is formed and medium remains its original colour.
- In lead acetate strip method, strip shows no black colour change.
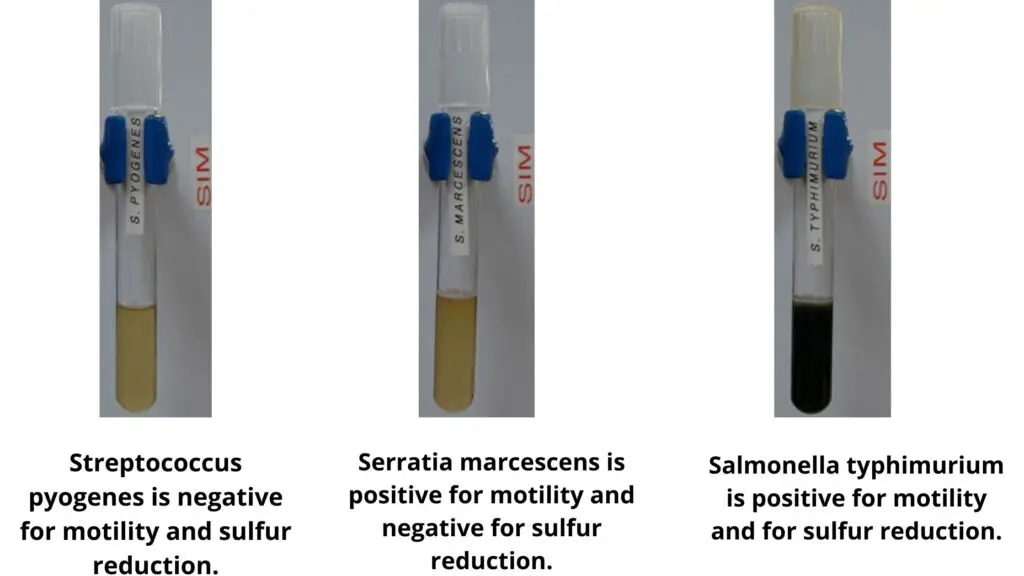
List of Sulfur Reduction Test Results of Some Common Bacteria
Positive (H2S producers)
- Salmonella species (S. Typhi, S. Typhimurium, S. Enteritidis).
- Proteus species (P. mirabilis, P. vulgaris).
- Citrobacter freundii.
- Francisella species.
- Campylobacter species.
- Erysipelothrix species.
- Staphylococcus saprophyticus.
- Bacteroides species.
- Brucella species.
Negative (Non H2S producers)
- Escherichia coli.
- Shigella species (S. flexneri, S. sonnei).
- Morganella morganii.
- Providencia species (P. rettgeri).
- Klebsiella species (K. pneumoniae).
- Enterobacter species.
- Citrobacter diversus.
- Pseudomonas aeruginosa.
- Staphylococcus aureus.
- Neisseria gonorrhoeae.
- Vibrio cholerae.
- Yersinia pestis.
- Lactobacillus species.
Quality Control of Sulfur Reduction Test
Positive control strains
- Proteus mirabilis (ATCC 29906). It gives positive result (blackening).
- Salmonella Typhimurium / Salmonella enterica (ATCC 14028). It gives positive result (blackening).
- Salmonella Enteritidis (ATCC 13076). It gives positive result (blackening).
Negative control strains
- Shigella flexneri (ATCC 12022). It gives negative result (no blackening).
- Escherichia coli (ATCC 25922 or ATCC 8739). It gives negative result (no blackening).
Precautions of Sulfur Reduction Test
- Media tubes should be checked before use. If cracks, dehydration or already blackened medium is seen, it should be discarded. Already dark medium is not used for test.
- Fresh culture (18-24 hours) from solid medium should be used. Broth culture is avoided because it can delay growth or produce turbidity and wrong reading.
- Sterile straight inoculating needle is used for inoculation. Loop is not preferred for SIM deep.
- Stabbing should be done straight in the center and the needle is withdrawn in the same line. Twisting, fanning or side cutting should not be done, it may give false interpretation.
- In SIM medium, stabbing is done only about half to 2/3 depth and glass bottom should not be touched. Irregular stabbing gives irregular growth pattern.
- In TSI / KIA, stab should reach the butt (bottom) properly. H2S reaction is seen well in low oxygen butt zone, so incomplete stab gives error.
- Tubes should be incubated with loose cap for air exchange. Tight cap may affect growth and gives erroneous result.
- In lead acetate paper method, lead acetate strip is handled with gloves and tweezers. Used strips are disposed as hazardous waste.
- Lead acetate strip should be suspended above the liquid and it should not touch the medium. If it touches, growth can be inhibited because lead acetate is toxic.
- In SIM medium, H2S and motility result should be read first before adding Kovac’s reagent for indole. After reagent addition, reading becomes difficult.
Uses of Sulfur Reduction Test
- It is used to detect the ability of microorganism to produce hydrogen sulfide (H2S) gas from sulfur containing compounds.
- It is used for biochemical differentiation and presumptive identification of bacterial isolates, mainly Gram negative enteric bacilli (Enterobacteriaceae).
- It is used to differentiate H2S producing organisms like Salmonella, Proteus, Francisella, Erysipelothrix from non H2S producers like Shigella, Morganella morganii, Providencia rettgeri, Lactobacillus.
- It is also used sometimes in identification of other organisms like Bacteroides and Brucella species.
- It is used in water quality testing to detect fecal coliforms and enteric bacteria, as an indicator of fecal contamination.
- It is used in industrial samples to identify sulfate reducing microorganisms responsible for pipeline corrosion and oil acidification.
- Lead acetate paper method is used to detect trace amount of sulfur or weak H2S producing organisms (sensitive method) and it may be used in some material science or forensic testing.
Advantages of Sulfur Reduction Test
- It is a reliable test for differentiation and identification of Gram negative enteric bacteria. It helps to differentiate H2S producing organisms like Salmonella and Proteus from non producers like Shigella and Escherichia coli.
- Lead acetate paper method is highly sensitive. It can detect very small amount of H2S and it is more sensitive than iron based agar methods, so weak H2S producers can also be detected.
- SIM medium gives good sensitivity among agar media. It shows H2S better than TSI or KIA because interfering carbohydrates are not present in SIM which can suppress H2S formation.
- It is a simple test and results are observed by blackening, so interpretation is easy. Black precipitate formation gives clear positive result.
- Combination media like SIM, TSI and KIA can give more than one information in same tube. Motility, indole, fermentation pattern and H2S can be checked together, so time and media is saved.
- It has wide applications. It is used in clinical lab for diagnosis, in water testing for fecal contamination and in industrial samples for sulfate reducing bacteria causing corrosion.
- In lead acetate method, very small sample is enough for detection (tiny amount of material can be tested).
Limitations of Sulfur Reduction Test
- It is not a confirmatory test alone. Other tests are required for final identification.
- It cannot tell the exact pathway of H2S production (cysteine breakdown or thiosulfate reduction).
- Broth or liquid inoculum can give delayed growth and wrong result. Fresh solid culture is required.
- In TSI, sucrose fermentation can suppress H2S production and false negative may occur.
- In TSI / KIA, stab should reach the butt. If not reached, H2S reaction may not be seen.
- In SIM, heavy black precipitate can mask motility result.
- Wrong stabbing and fanning motion can give false motility in SIM.
- In lead acetate method, strip touching liquid can inhibit growth because lead acetate is toxic.
- Lead acetate paper can be darkened by soot/fumes and misinterpretation can occur, confirmatory step may be needed.
- Lead acetate method gives only H2S detection, no extra data like motility or fermentation.
References
- Advanced microbiological and biochemical analysis of the sulfur reduction test: Principles, diagnostic architectures, and pathophysiological implications. (n.d.).
- Cooper, C. R., Jr. (2018). Triple sugar iron (TSI) test. Youngstown State University.
- Cysteine catabolism and cysteine desulfhydrase (CdsH/STM0458) in Salmonella enterica serovar Typhimurium. (n.d.). PubMed Central (PMC).
- Dahal, P. (2023, October 6). Sulfur reduction test: Principle, procedure, results, uses. Microbe Notes.
- DrChika. (2022, December 28). Triple sugar iron agar (TSIA). Microbiology Class.
- Fusion of a bacterial cysteine desulfurase to redox-sensitive green fluorescent protein produces a highly sensitive cysteine biosensor for monitoring changes in intracellular cysteine. (n.d.). PubMed Central (PMC).
- Gart, E. V., Suchodolski, J. S., Welsh, T. H., Jr., Alaniz, R. C., Randel, R. D., & Lawhon, S. D. (2016). Salmonella Typhimurium and multidirectional communication in the gut. Frontiers in Microbiology, 7, 1827.
- Hardy Diagnostics. (2020). Lead acetate strips – for hydrogen sulfide detection [Instructions for Use].
- Hardy Diagnostics. (2020). SIM (Sulfide, Indole, Motility) medium – for gram negative enteric bacilli [Instructions for Use].
- Hartline, R. (2023, February 18). 1.23: SIM deep tests. Biology LibreTexts.
- HeredityBioAcademy. (n.d.). Sulfer reduction test | Heredity Biosciences | Microbiology [Video]. YouTube.
- KEGG PATHWAY: ko00920 (Sulfur metabolism). (n.d.). Kyoto Encyclopedia of Genes and Genomes.
- Kligler iron agar (KIA) differs from triple-sugar iron agar (TSI) in the:.. (n.d.). Filo.
- Li, H., Wang, J., Li, X., Jia, G., Gan, H., Wang, Y., Ma, Z., Zhu, Z., Shang, X., & Niu, W. (2026). Hydrogen sulfide acts as a sulfur source for iron sulfur cluster biosynthesis in cysteine desulfurase-deficient Escherichia coli under anaerobic conditions. Frontiers in Microbiology, 17, 1759970.
- Mechanisms of H2S production from cysteine and cystine by microorganisms isolated from soil by selective enrichment. (n.d.). PubMed Central (PMC).
- Microscopic methods for identification of sulfate-reducing bacteria from various habitats. (n.d.). PubMed Central (PMC).
- Pathways of cysteine degradation to H2S. Cysteine desulfhydrase is a… (n.d.). ResearchGate.
- Salmonella produces sulfide to compete with Escherichia coli in the gut lumen. (n.d.). PNAS / PubMed / ResearchGate.
- Salmonella Typhimurium strain ATCC14028 requires H2-hydrogenases for growth in the gut, but not at systemic sites. (n.d.). PubMed Central (PMC).
- Sekar. (2008, November 29). Triple sugar iron agar/Kligler iron agar: Evaluating the ability of microorganism to produce acid, gas and H2S. iNVISIBLE YET iNVINCIBLE.
- Selwyn, L. (2017). How to test for sulfur in materials using lead acetate test paper – Canadian Conservation Institute (CCI) Notes 17/5. Government of Canada.
- Sigma-Aldrich Co. LLC. (2013). 06728 Hydrogen sulfide test strips (Lead acetate test strips, H2S test strips) [Product Information].
- Sulfur-Element containing metabolic pathways in human health and crosstalk with the microbiome. (n.d.). PubMed Central (PMC).
- The multifaceted bacterial cysteine desulfurases: From metabolism … (n.d.). PubMed Central (PMC).
- TM Media. (2019, November 8). TBL 024 – Lead acetate paper strips [Product Data Sheet].
- Wang, J., Guo, X., Li, H., Qi, H., Qian, J., Yan, S., Shi, J., & Niu, W. (2019). Hydrogen sulfide from cysteine desulfurase, not 3-mercaptopyruvate sulfurtransferase, contributes to sustaining cell growth and bioenergetics in E. coli under anaerobic conditions. Frontiers in Microbiology, 10, 2357.
- Watson, R. (n.d.). Tests used to identify Gram negative bacteria. Microbiology Lab: MOLB 2210.