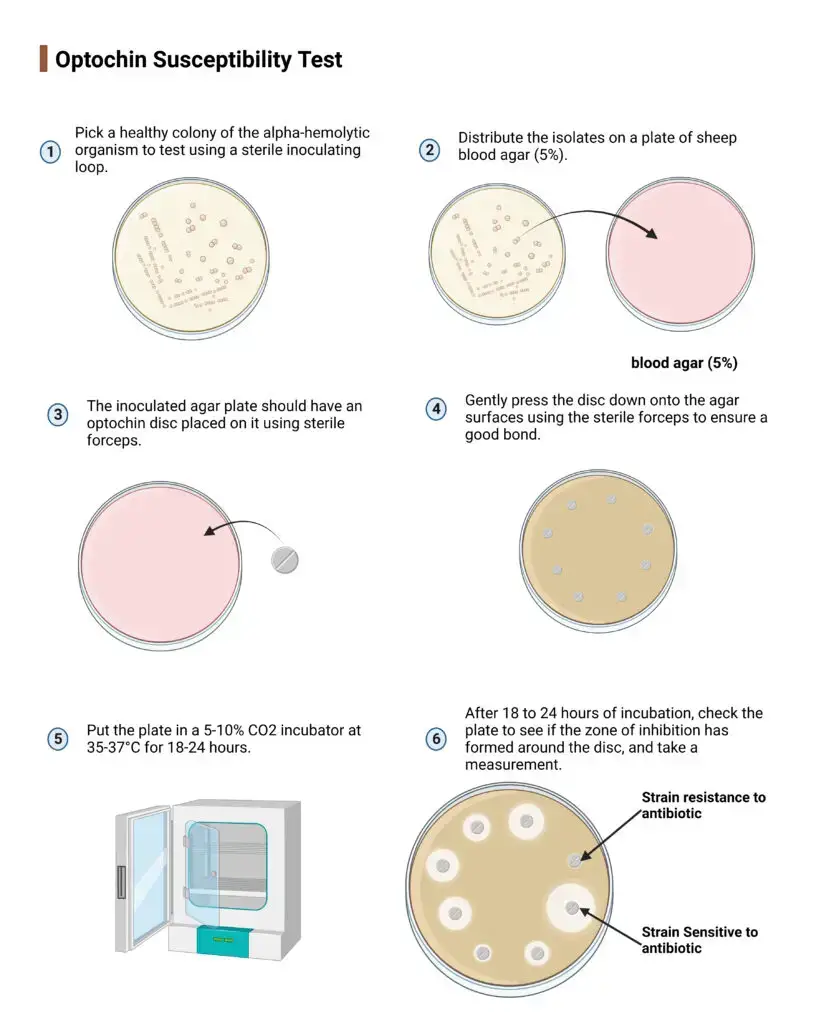
Optochin susceptibility test is a laboratory test used to presumptively identify Streptococcus pneumoniae and differentiate it from other alpha hemolytic streptococci like viridans group. It is a disk diffusion based test in which optochin (ethylhydrocupreine hydrochloride) is used as chemical agent. The chemical causes rapid lysis in S. pneumoniae cells due to its action on ATP synthase and cell membrane.
In this test a filter paper disk impregnated with optochin (5 µg) is placed on a blood agar plate (5% sheep blood agar) that is inoculated with pure bacterial culture. The plate is incubated at 35 to 37°C in 5% to 10% CO2 for 18 to 24 hours. Optochin diffuses into agar and growth response is observed.
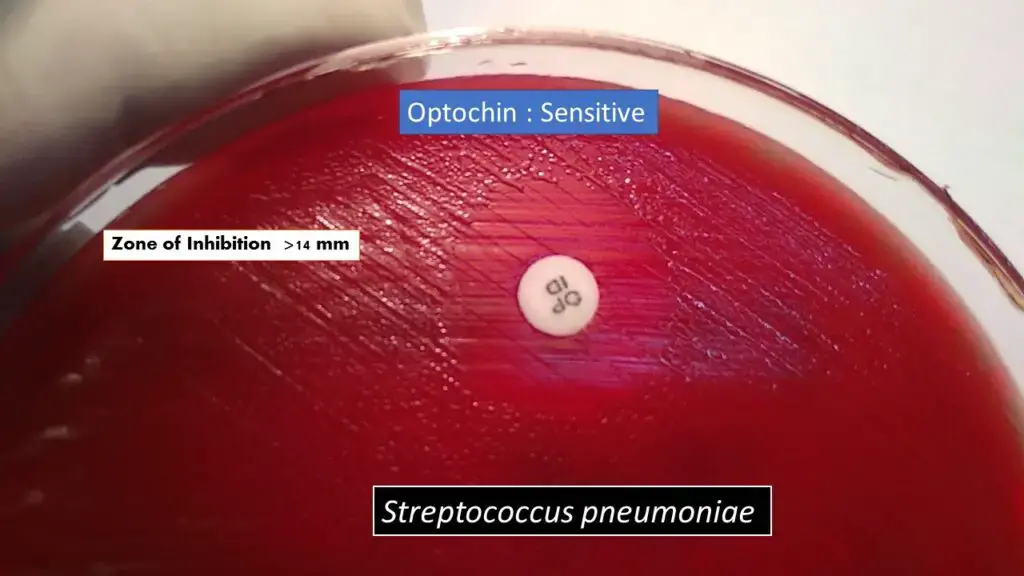
If organism is S. pneumoniae a clear well defined zone of inhibition is formed around the disk and the zone is usually 14 mm or more. Other alpha hemolytic streptococci are resistant so growth is seen up to edge of disk or very small zone is formed. The intermediate zone (6 to 13 mm) are required further confirmatory test like bile solubility test.
Objective of Optochin Susceptibility Test
The objectives are-
- To presumptively identify Streptococcus pneumoniae by using optochin (ethylhydrocupreine hydrochloride) sensitivity.
- To differentiate S. pneumoniae from other alpha hemolytic streptococci mainly viridans group (like S. mitis and S. oralis) which are resistant.
- To determine whether the test organism is susceptible or resistant to optochin by observing growth inhibition and lysis or growth up to disk.
Principle of Optochin Susceptibility Test
Optochin susceptibility test is based on disk diffusion method that is used to differentiate Streptococcus pneumoniae from other alpha hemolytic viridans streptococci. In this test a paper disk impregnated with optochin (ethylhydrocupreine hydrochloride) is placed on blood agar plate inoculated with the organism. The optochin diffuses into the agar around the disk.
Optochin specifically targets the bacterial F0F1 ATP synthase by binding to the interface of a and c subunits in the F0 domain. This binding inhibits rotation of c-ring and blocks proton passage. It causes immediate depletion of cellular energy and metabolic collapse.
Optochin also acts as a surface active agent and it alters surface tension of pneumococcal cell membrane. Due to disruption and loss of membrane potential the endogenous autolytic enzyme LytA is activated early. This enzyme degrades the bacterial cell wall rapidly and cellular lysis is produced.
Because S. pneumoniae is sensitive to optochin a clear zone of growth inhibition is formed around the disk. In contrast other alpha hemolytic streptococci are resistant so growth is seen up to margin of the disk.
Requirements of Optochin Susceptibility Test
The requirements are-
- Culture media– 5% sheep blood agar (Trypticase Soy Agar (TSA) base is recommended to avoid false identification).
- Optochin disk– filter paper disk (about 6 mm) impregnated with optochin (ethylhydrocupreine hydrochloride) 5 µg.
- Turbidity standard– 0.5 McFarland standard (or equivalent) to adjust bacterial suspension.
- Inoculating tools– sterile inoculating loop or straight wire or sterile cotton tipped swab for picking colony and making confluent lawn on plate.
- Sterile forceps– it is used for placing optochin disk aseptically and pressing it gently for proper adherence.
- Incubator– 35 to 37°C with 5% to 10% CO2 enriched environment (as S. pneumoniae grows poorly in ambient air).
- Measuring scale– millimeter ruler or caliper to measure zone of inhibition after incubation.
Procedure of Optochin Susceptibility Test
The procedure is as follows-
- Colony selection– 3 to 4 well isolated alpha hemolytic colonies are selected from 18 to 24 hours pure culture by using sterile inoculating loop.
- Inoculum preparation– the selected colonies are transferred into sterile tube containing saline or broth and turbidity is adjusted to 0.5 McFarland standard.
- Inoculation– sterile cotton swab is used and bacterial suspension is streaked on Trypticase Soy Agar (TSA) plate with 5% sheep blood. The plate is streaked in at least three directions to make uniform confluent lawn.
- Drying– the plate surface is allowed to dry for 5 to 10 minutes before placing disk.
- Disk placement– by using sterile forceps 5 µg optochin disk (6 mm) is placed at center of inoculated agar.
- Securing the disk– the disk is pressed gently with forceps so that it adheres properly and diffusion occurs evenly.
- Incubation– the plate is inverted and incubated at 35 to 37°C for 18 to 24 hours in 5% to 10% CO2 enriched atmosphere.
- Measurement– after incubation the zone of inhibition around disk is observed and full diameter is measured by millimeter ruler or calipers.
Result and Interpretation of Optochin Susceptibility Test
The result and interpretation are-
- Sensitive (Positive)– a clear zone of inhibition of 14 mm or more is seen around 6 mm optochin disk. This is presumptively identified as Streptococcus pneumoniae.
- Intermediate (Questionable)– a zone of inhibition between 7 mm to 13 mm (or 6 to 13 mm) is seen. These isolates are required further confirmatory test like bile solubility test. If organism is bile soluble it is identified as S. pneumoniae.
- Resistant (Negative)– no zone of inhibition is seen or zone is less than 7 mm and growth is present up to margin of disk. It indicates non pneumococcal alpha hemolytic streptococcus like S. mitis or S. oralis (viridans group).
- Atypical (S. pseudopneumoniae)– it shows environment dependent result. In 5% CO2 it appears resistant or intermediate but in ambient air it is sensitive and zone is 14 mm or more.
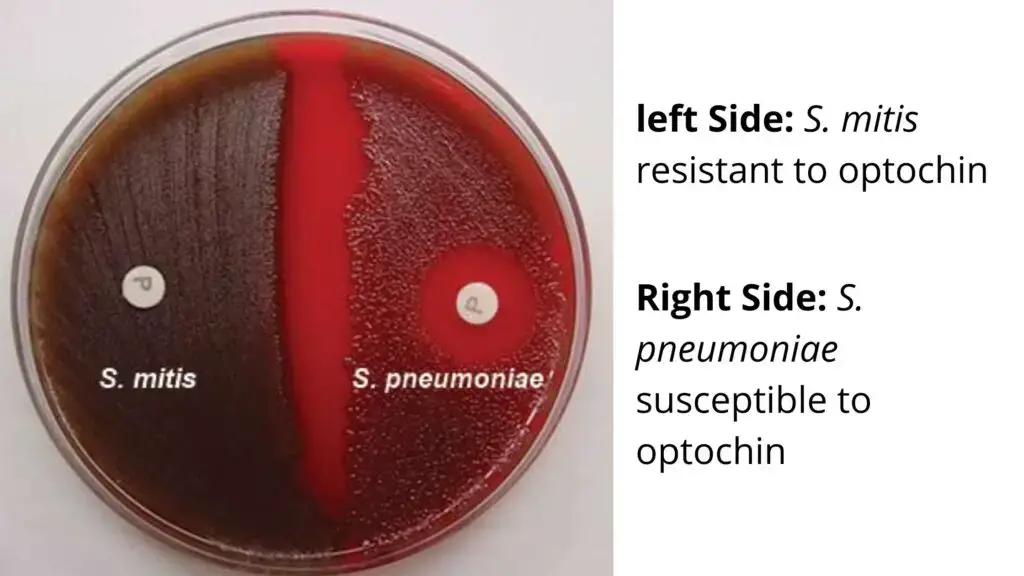
To exclude the possibility that other strains of Streptococcus pneumoniae are present, further tests (bile solubility or serology) should be conducted if the zone of inhibition is less than 14 mm.
Organisms showing Optochin Susceptibility Test result
The organisms are-
Positive result (Optochin sensitive)
- Streptococcus pneumoniae.
Negative result (Optochin resistant)
- Streptococcus mitis.
- Streptococcus oralis.
- Streptococcus sanguinis (formerly S. sanguis).
- Streptococcus mutans.
- Streptococcus salivarius.
- Streptococcus anginosus.
- Streptococcus pyogenes (negative control).
- Enterococcus faecalis (negative control).
- Other non pneumococcal alpha hemolytic viridans group streptococci.
Variable / environment dependent result
- Streptococcus pseudopneumoniae (it appears resistant or intermediate in 5% CO2 but it appears sensitive in ambient air).
Uses of Optochin Susceptibility Test
The uses are-
- It is used for presumptive identification of Streptococcus pneumoniae in bacteriological culture.
- It is used to differentiate optochin sensitive S. pneumoniae from other alpha hemolytic streptococci mainly viridans group which are optochin resistant.
- It is performed in clinical diagnosis to help in diagnosis of pneumococcal infections from clinical specimens.
Advantages of Optochin Susceptibility Test
The advantages are-
- It is simple and easy to perform as it is a disk diffusion method and it is used in routine microbiology laboratory.
- It gives clear visual interpretation as a distinct zone of inhibition is formed in optochin sensitive Streptococcus pneumoniae and other alpha hemolytic streptococci are not showing this type of zone.
- It is less time consuming as compared to bile solubility test.
- It is cost effective and highly accessible test and it is useful in resource limited settings where advanced methods (MALDI-TOF MS or PCR) are not available.
- It gives reliable presumptive identification of S. pneumoniae in most typical clinical isolates.
Limitations of Optochin Susceptibility Test
The limitations are-
- It gives only presumptive identification and it is not confirmatory. Further test like bile solubility test is required for complete identification of Streptococcus pneumoniae.
- Rare optochin resistant pneumococci can be present due to genetic mutations. Some isolates can show mixed population of susceptible and resistant cells.
- Some viridans group streptococci can show optochin sensitivity or slight susceptibility so false positive result can be obtained. Streptococcus pseudopneumoniae also creates confusion as it shows switch type phenotype (resistant in CO2 but sensitive in ambient air).
- Incubation atmosphere is critical and result depends on it. In ambient air some strains of S. pneumoniae grows poorly and in high CO2 concentration (more than 10%) zone size can be reduced so sensitive strain can appear resistant.
- Specific media is required like 5% sheep blood agar on Trypticase Soy Agar (TSA) base. Other media (Columbia or Mueller Hinton agar) can give indeterminate zone and false resistance.
- Inoculum density error can affect zone size. Heavy inoculum reduces zone and light inoculum increases zone giving false sensitivity.
- Agar volume and depth also affects diffusion. Thin agar allows more diffusion and zone can become artificially large in non pneumococcal species.
- Optochin disks can be degraded if expired or stored in warm moist condition. It reduces zone size and false resistant report can be given.
Precautions of Optochin Susceptibility Test
The precautions are-
- Culture media should be used correctly. 5% sheep blood agar is used and it is preferred with Trypticase Soy Agar (TSA) base. Other media can give indeterminate zone and false identification.
- Inoculum density should be standardized to 0.5 McFarland standard. The lawn should be uniform and confluent. Heavy inoculum can reduce zone size (false resistance) and light inoculum can increase zone size (false sensitivity).
- Pure culture should be used. Well isolated colonies are taken and mixed culture should not be used as faulty result is obtained.
- Optochin disk should be placed properly and it should be pressed gently so that firm adherence is obtained and diffusion occurs evenly. If multiple disks are used then they should be placed at least 25 mm apart and away from edge of plate.
- Incubation atmosphere should be controlled. The plates are incubated in 5% to 10% CO2 as Streptococcus pneumoniae grows poorly in ambient air. CO2 above 10% should be avoided as zone size can shrink and sensitive strain can appear resistant.
- While preparing blood agar manually blood should not be added if molten agar temperature is above 45°C and plates should be poured before temperature drops below 40°C to avoid clumps.
- Optochin disks should be stored properly (refrigerated or at -20°C) and it should be protected from moisture and direct light. Expired or degraded disk gives reduced zone and false resistance.
- It is a presumptive test so intermediate or questionable zone (6 to 14 mm) are required confirmatory test like bile solubility test.
- Biohazard safety should be followed. All specimens are treated as infectious. Proper PPE is used and aerosol producing steps are done in safety cabinet and materials are autoclaved before disposal.
References
- Ambizas, E. M., & Patel, P. N. (2011). Drug-induced optic neuropathy. U.S. Pharmacist, 36(4), HS2-HS6.
- Bio-Rad. (2010). Optochin 53852: Presumptive diagnosis of Streptococcus pneumoniae [Package insert].
- Centers for Disease Control and Prevention. (1981). Optochin test results (Image ID 19332) [Photograph]. Public Health Image Library.
- Condalab. (n.d.). Optochin 5 µg Cat. 7073 [Instructions for use].
- Courbon, G. M., Palme, P. R., Mann, L., Richter, A., Imming, P., & Rubinstein, J. L. (2023). Mechanism of mycobacterial ATP synthase inhibition by squaramides and second generation diarylquinolines. The EMBO Journal, 42(15), e113687. https://doi.org/10.15252/embj.2023113687
- Dahal, P. (2023, March 31). Optochin susceptibility test- Principle, procedure, results. Microbe Notes.
- Dalynn Biologicals. (2014). Optochin disks (Catalogue No. DO60) [Package insert].
- Guo, H., Suzuki, T., & Rubinstein, J. L. (2019). Structure of a bacterial ATP synthase. eLife, 8, e43128. https://doi.org/10.7554/eLife.43128
- Hardy Diagnostics. (2020). Instructions for use: HardyDisk™ Optochin differentiation disks.
- HiMedia Laboratories. (2022). Technical data: DD009 Optochin discs.
- Keith, E. (2010). Laboratory and epidemiological characteristics of Streptococcus pseudopneumoniae [Master’s thesis, University of Otago]. OUR Archive.
- Ktari, S., Ben Ayed, N. E. H., Maalej, S., Mnif, B., Rhimi, F., & Hammami, A. (2021). Clinical optochin resistant Streptococcus pneumoniae and Streptococcus pseudopneumoniae strains in Tunisia. The Journal of Infection in Developing Countries, 15(5), 672–677. https://doi.org/10.3855/jidc.13106
- Mackieh, R., Al-Bakkar, N., Kfoury, M., Roufayel, R., Sabatier, J.-M., & Fajloun, Z. (2023). Inhibitors of ATP synthase as new antibacterial candidates. Antibiotics, 12(4), 650. https://doi.org/10.3390/antibiotics12040650
- Marín, M. M. M., Cercenado, E. C., Sánchez-Carrillo, C. S., Ruiz, A. R., Gómez González, Á. G., Rodríguez-Sánchez, B. R., & Bouza, E. B. (2017). Accurate differentiation of Streptococcus pneumoniae from other species within the Streptococcus mitis group by peak analysis using MALDI-TOF MS. Frontiers in Microbiology, 8, Article 698. https://doi.org/10.3389/fmicb.2017.00698
- Martín-Galiano, A. J., Balsalobre, L., Fenoll, A., & de la Campa, A. G. (2003). Genetic characterization of optochin-susceptible viridans group streptococci. Antimicrobial Agents and Chemotherapy, 47(10), 3187–3194. https://doi.org/10.1128/AAC.47.10.3187-3194.2003
- MedChemExpress. (n.d.). Ethylhydrocupreine hydrochloride (Optochin hydrochloride).
- Mhimdi, S., Meftah, K., Bouafsoun, A., & Smaoui, H. (2023). Accurate identification of Streptococcus pseudopneumoniae and other mitis group Streptococci identified as atypical Streptococcus pneumoniae in Tunisian pediatric population. Acta Microbiologica et Immunologica Hungarica, 70(4), 325–330. https://doi.org/10.1556/030.2023.02165
- Microbe Canvas. (n.d.). Optochin test.
- MicrobiologyInfo.com. (n.d.). Optochin susceptibility test for the identification of Streptococcus pneumoniae.
- National Center for Advancing Translational Sciences. (n.d.). Ethylhydrocupreine hydrochloride. Inxight Drugs.
- Neupane, P., Bhuju, S., Thapa, N., & Bhattarai, H. K. (2019). ATP synthase: Structure, function and inhibition. Biomolecular Concepts, 10(1), 1-10. https://doi.org/10.1515/bmc-2019-0001
- Nix, I. D., Idelevich, E. A., Schlattmann, A., Sparbier, K., Kostrzewa, M., & Becker, K. (2021). MALDI-TOF mass spectrometry-based optochin susceptibility testing for differentiation of Streptococcus pneumoniae from other Streptococcus mitis group streptococci. Microorganisms, 9(10), 2010. https://doi.org/10.3390/microorganisms9102010
- Nunes, S., Sá-Leão, R., & de Lencastre, H. (2008). Optochin resistance among Streptococcus pneumoniae strains colonizing healthy children in Portugal. Journal of Clinical Microbiology, 46(1), 321–324. https://doi.org/10.1128/JCM.02097-07
- Pikis, A., Campos, J. M., Rodriguez, W. J., & Keith, J. M. (2001). Optochin resistance in Streptococcus pneumoniae: Mechanism, significance, and clinical implications. The Journal of Infectious Diseases, 184(5), 582–590. https://doi.org/10.1086/322803
- Pokhrel, P. (2015, October 20). Optochin susceptibility test for Streptococcus pneumoniae. Microbiology Notes.
- PubMed Central (PMC). (n.d.). A case of optic nerve atrophy with severe disc cupping after methanol poisoning.
- PubMed Central (PMC). (n.d.). Accuracy of phenotypic methods for identification of Streptococcus pneumoniae isolates included in surveillance programs.
- PubMed Central (PMC). (n.d.). Antibiotic susceptibility patterns of viridans group streptococci isolates in the United States from 2010 to 2020.
- PubMed Central (PMC). (n.d.). Characteristics of Streptococcus pseudopneumoniae isolated from purulent sputum samples.
- PubMed Central (PMC). (n.d.). Ethambutol-induced optic neuritis and vision loss: A case report.
- PubMed Central (PMC). (n.d.). Identification of Streptococcus pneumoniae: Development of a standardized protocol for optochin susceptibility testing using total lab automation.
- PubMed Central (PMC). (n.d.). Inhibition of Escherichia coli ATP synthase by amphibian antimicrobial peptides.
- PubMed Central (PMC). (n.d.). On the mechanisms of lysis triggered by perturbations of bacterial cell wall biosynthesis.
- PubMed Central (PMC). (n.d.). Optochin revisited: Defining the optimal type of blood agar for presumptive identification of Streptococcus pneumoniae.
- PubMed Central (PMC). (n.d.). Streptococcus pseudopneumoniae: An emerging respiratory tract pathogen.
- Sarathy, J. P., Gruber, G., & Dick, T. (2019). Re-understanding the mechanisms of action of the anti-mycobacterial drug bedaquiline. Antibiotics, 8(4), 261. https://doi.org/10.3390/antibiotics8040261
- Shahinas, D., Thornton, C. S., Tamber, G. S., Arya, G., Wong, A., Jamieson, F. B., Ma, J. H., Alexander, D. C., Low, D. E., & Pillai, D. R. (2013). Comparative genomic analyses of Streptococcus pseudopneumoniae provide insight into virulence and commensalism dynamics. PLOS ONE, 8(6), e65670. https://doi.org/10.1371/journal.pone.0065670
- Taylor & Francis. (n.d.). Optochin – Knowledge and references.
- Technical monograph on the optochin susceptibility test: Molecular mechanisms, clinical protocols, and diagnostic challenges in pneumococcal identification. (n.d.).
- UK Health Security Agency. (2025). UK Standards for Microbiology Investigations: Optochin test (Test Procedures | TP 25 | Issue 4.1).
- Vohrnová, S., Kozáková, J., & Honskus, M. (2025). Comparison of Streptococcus pneumoniae isolates occurring in optochin-susceptible and optochin-resistant variants by analyzing whole-genome sequencing data. Microbiology Spectrum, 13(4), e01939-24. https://doi.org/10.1128/spectrum.01939-24
- VUMIE. (2022, June 12). Optochin susceptibility test. Virtual Microbiology Lab Simulator Software.
- Wikipedia contributors. (2025). Optochin. In Wikipedia, The Free Encyclopedia.
- Yahiaoui, R. Y. (2020). Streptococcus pneumoniae carriage and effect of vaccination on prevalence, serotypes and antibiotic resistance [Doctoral thesis, Erasmus University Rotterdam]. EUR Research Information Portal.